Abstract
Background
Peritoneal dialysis (PD) is complicated by a high rate of adverse events that might be attributed to cytotoxicity of currently used PD fluids. However, clinical development of novel PD fluids is virtually non-existent, in part due to difficulties in recruiting sufficiently large populations for adequately powered trials. The aim of this study is to understand the potential impact of introducing composite outcomes on clinical trial feasibility in PD.
Methods
A composite outcome “major adverse peritoneal events (MAPE)” was designed to combine clinically relevant complications of PD, such as (1) technical failure (cause-specific for peritonitis and/or insufficient dialysis), (2) peritonitis, and (3) peritoneal membrane deterioration. Incidence rates of individual endpoints were obtained from the literature and expert panel estimations, and population sizes were computed based on Chi-square test for adequately powered confirmatory randomized controlled clinical trials with 2 parallel arms.
Results
Incidence rates for technical failure, peritonitis, and peritoneal membrane deterioration were estimated at 15%, 50%, and 23%, respectively, at 2 years follow-up, with adequate agreement between the literature and expert opinion. Assuming that a given intervention reduces adverse outcomes by 30%, an adequately powered clinical trial needs to recruit up to 1,720 patients when studying individual outcomes. Combining endpoints increases power in simulated trials despite considerable overlap, and the composite outcome MAPE reduces the required population to 202 patients aiming for 80% power.
Conclusion
Introduction of the composite outcome MAPE, covering relevant major adverse peritoneal events, may improve the feasibility of clinical trials to adequately test novel PD fluids.
Keywords
Inherent cytotoxicity of currently used PD fluids is a relevant factor contributing to PD-related clinical complications (9–12). However, despite major investments into pharmaceutical development improving the production process, there has been no successful advancement in PD-fluid development toward improved clinical outcome with reduction of PD-related complications for over 20 years (13). A meta-analysis, compiling almost 2,000 patients from 29 clinical trials, was not able to confirm any significant improvement in individual PD-related outcomes related to technical failure, in particular peritonitis and peritoneal membrane damage, with use of more biocompatible multi-chamber PD fluids (13).
One major obstacle for clinical innovation in PD may be caused by difficulties in conducting adequately powered trials in the PD population (14,15). Low feasibility and thus high risk of inherently expensive PD trials beyond strictly clinical aspects are likely to contribute to the inertia in PD fluid development despite the unsolved medical problem of high rates of PD-related complications.
Composite endpoints may provide a solution to this problem (16). This tool combines different clinical outcomes that are clinically relevant and expected to be beneficially affected by the treatment. In their draft guidance on this matter, the Food and Drug Administration (FDA) suggests that composite endpoints might be used in trials where the incidence rates of the individual components are too low to achieve adequate power and reasonable study size and duration (16). In oncology and cardiovascular research, progression-free survival (PFS) and major adverse cardiac events (MACE) are good examples of combined clinically relevant outcomes that resulted in lower study sizes needed to achieve an adequate power. These are now accepted as pivotal clinical trial endpoints that potentially allow regulatory approval of a new product.
The aim of this study is to develop and test a composite outcome as a novel tool to improve PD trial design and thus feasibility. We introduce a combination of clinically relevant PD-related complications that are likely to be amenable to improvement by novel PD fluids, by combining (1) technical failure (cause-specific for peritonitis and/or insufficient dialysis), (2) peritonitis, and (3) peritoneal membrane deterioration, defined as loss of ultrafiltration and/or changes in peritoneal membrane transport characteristics predictive of inherent ultrafiltration loss. We used data from the literature and from an expert panel to estimate incidence rates of individual outcomes and combinations thereof (termed “major adverse peritoneal events” [MAPE]) to compute minimal populations to obtain meaningful results in PD trials.
Methods
Incidence rates for outcome measures reflecting predefined PD-related complications were estimated from data from the public domain (NCBI PubMed database; https://www.ncbi.nlm.nih.gov/pubmed). In a non-systematic literature search approach, multiple search terms aiming to cover “cause-specific technical failure (due to infection/peritonitis and/or inadequate dialysis),” “infection/peritonitis,” and “peritoneal membrane deterioration” were independently used by 2 authors (AV and CA) to obtain titles and abstracts for screening of potentially relevant studies. Where reports from multicentric registries were available, only these data were used. In case of repeated publications, only the most recent and specific report was used. For the selected studies, full text was retrieved for discussion.
Data on “cause-specific technical failure (due to infection/ peritonitis and/or inadequate dialysis)” and “infection/peritonitis” of PD-related complications were identified in the French peritoneal dialysis registry (RDPLF) and the Australia and New Zealand Dialysis and Transplant Registry (ANZDATA). Incidence rate of the outcome measure “peritoneal membrane deterioration” could not be identified in registry reports and this outcome was therefore individually searched in published clinical reports on repeated peritoneal equilibration tests (PET). The PET assesses time-dependent changes in dialysate volume and solute levels (e.g., creatinine or sodium) during the dwell, given as ultrafiltration, as ratios of dialysate to plasma (D/P) solute levels (e.g., creatinine), and as sodium sieving or free water transport (17 18 19 20 21-22). “Peritoneal membrane deterioration” was defined as ultrafiltration loss, increase of D/P creatinine transport ratio, and/or decrease of sodium dip. These PET variables either directly demonstrate loss of peritoneal ultrafiltration or demonstrate deterioration of peritoneal transport characteristics associated with ultrafiltration loss (17, 21, 23–25). Ambiguous findings were clarified by consensus discussion.
Incidence rates of the 2 major drivers of PD fluid-related complications, i.e., “peritonitis” and “peritoneal membrane deterioration,” were also estimated by independent expert opinions. The expert group was selected on the basis of expertise and reputation (they were invited speakers in at least 2 of the previous 4 Euro PD and International Society for Peritoneal Dialysis [ISPD] meetings), geographical coverage (Europe, US/Canada, and Asia), and availability at the ISPD conference 2018, and included 7 individuals (see Acknowledgments). Standardized expert interviews were conducted as a semi-structured face-to-face meeting in May 2018, covering several topics relevant for planning a PD trial. These questions included, “What percentage of patients would you expect to experience (i) peritonitis and (ii) measurable functional impairment of the peritoneal membrane at your institution within 2 years of follow-up?” Each expert was asked by 1 of the authors (CA) to separately make their estimates; no attempt was made to develop a consensus.
The composite outcome MAPE, combining these 3 individual outcomes, was designed to cover clinically relevant endpoints for PD-related complications that can be assumed to be potentially influenced by PD fluid bio(in)compatibilties. As MAPE combines individual components whose pathophysiological mechanisms and clinical presentations are likely correlated, it can be assumed that these adverse events are overlapping, meaning that some of the patients could experience more than 1 event.
Sample sizes were computed for adequately powered trials based on individual outcome parameters and combinations thereof. Sample size calculations were carried out using a χ2 test for a parallel group design comparing an experimental treatment (E) with a control (C) in binary composite endpoint MAPE with 2-sided significance level α of 5% to achieve power of 80%. MAPE was obtained by adding the event rate reductions of the individual components accounting for the correlation between the outcomes using methodology, as for example in (26). Different overlaps of events were investigated and compared in terms of the required resulting sample size (for details of statistical modeling of MAPE event rates see Supplemental Material).
Results
Cause-specific technical failure has been reported in ANZDATA due to infectious complications, primarily peritonitis, in 6% – 8% (after 1 year) and in 15% – 19% (after 3 years), due to inadequate dialysis in 2% – 3% (after 1 year) and in 7% (after 3 years) (27). Peritonitis has been reported in both ANZDATA and in RDPLF in 42% of patients after 24 months of observation (8,28). Signs of peritoneal membrane deterioration such as ultrafiltration loss and increase of D/P transport ratio have been reported as 7% – 8% and in 16% of patients, respectively '(29–31). According to expert opinion, mean incidence rates for “peritonitis” and “peritoneal membrane damage” were estimated as 50% (range 40% – 60%) and 23% (10% – 35%) per 2-year period, respectively, showing acceptable agreement with the literature-derived data. For further analysis, incidence rates for “technical failure (cause-specific for peritonitis and/or inadequate dialysis),” “peritonitis,” and “peritoneal membrane damage” were estimated at 15%, 50%, and 23% at 2 years’ follow-up, respectively. The resulting sample sizes for a 2-year period could be hence calculated.
Table 1 shows results of scenarios where MAPE composite endpoint reduces required minimal population sizes to achieve 80% power. For example, assuming the aforementioned scenario in which a given intervention reduces adverse outcomes by 30% over a 2-year study period, adequately powered studies to test these effects on the individual outcome parameter with incidence rates from cause-specific technical failure, peritonitis, and peritoneal membrane deterioration need a population of 1,724, 340, or 1,036 patients, respectively. Combining these outcome parameters to the composite outcome MAPE reduces the required population size to approximately 202 patients under the assumption of a 50% overlap of peritoneal membrane deterioration with peritonitis and an overlap of 75% of technical failure with the other 2 components. The latter means that 75% of patients with technical failure will also experience either peritonitis or peritoneal membrane damage.
Sample Size Requirements for Different Endpoints
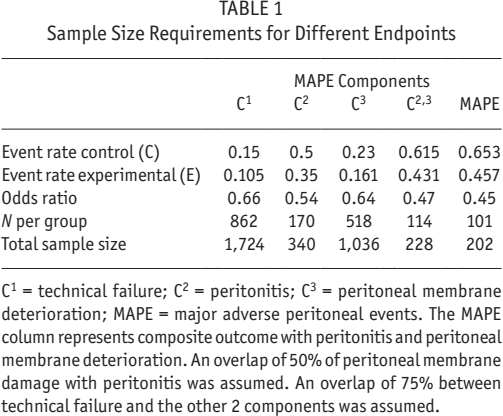
C1 = technical failure; C2 = peritonitis; C3 = peritoneal membrane deterioration; MAPE = major adverse peritoneal events. The MAPE column represents composite outcome with peritonitis and peritoneal membrane deterioration. An overlap of 50% of peritoneal membrane damage with peritonitis was assumed. An overlap of 75% between technical failure and the other 2 components was assumed.
Figure 1 shows the effects of different overlaps on population sizes to achieve a power of 80% with an assumed reduction of adverse events by 30%. It can be seen in the plots that the increase in sample size is smaller if the overlap of technical failure with peritonitis and peritoneal membrane damage is varied and the sample size varies from around 65 to 115 patients per treatment group. It can be seen that the smaller the overlap the smaller the sample size. This happens because the MAPE rate increases if there is no overlap between components, resulting in a lower sample size.
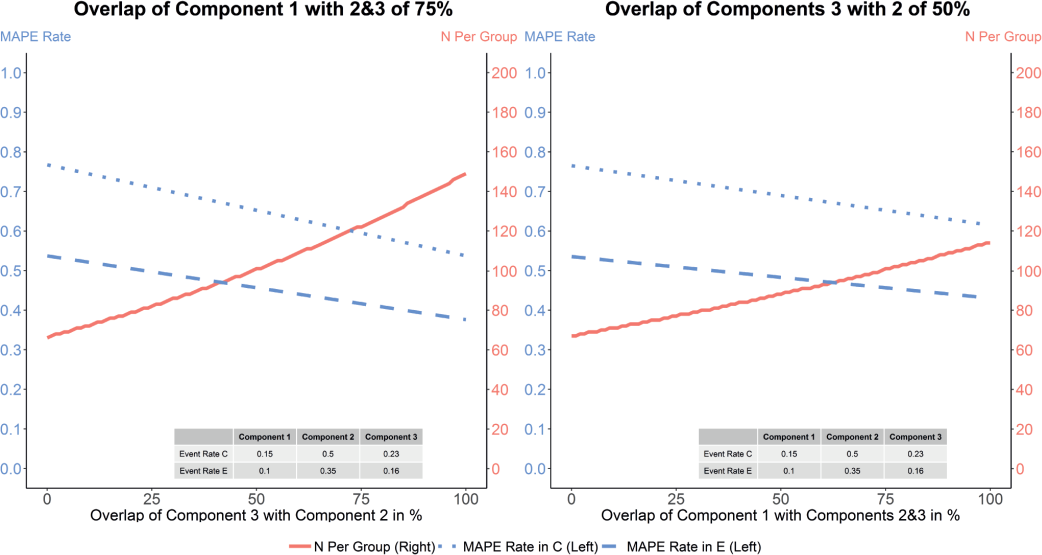
Plots showing MAPE rate and sample size per group for different overlaps between components. The plot on the left corresponds to the results where the overlap of components 3 and 2 (peritoneal membrane damage and peritonitis) is varied from 0 to 100%. The overlap of component 1 (cause-specific technical failure) with them is set to be constant and equal to 75%. The plot on the right-hand side shows results for a fixed overlap between components 3 and 2 of 50%, and the overlap of component 1 (cause-specific technical failure) is varied from 0 to 100%. The y-axis on the left hand side on each plot shows the MAPE rate, whereas the y-axis on the right hand side shows the sample size per treatment group. The red full lines correspond to the sample size for a given overlap, the blue dotted lines to MAPE rate in the control group and blue dashed lines to MAPE rate in the experimental group. C = control; E = experimental; MAPE = major adverse peritoneal events.
Discussion
The effects of novel PD fluids on reducing the risk of PD-related complications have not yet been successfully demonstrated by any a priori defined endpoint in a confirmative clinical trial. These disappointing results have led to different conclusions in the scientific community, including the assumption of only minimal influence of PD fluid bio(in)compatibility on relevant clinical outcomes, resulting in views of the a priori futility of future clinical trials (15,32–34). The 2018 Cochrane meta-analysis, however, clearly demonstrated that almost all trials performed until then were severely underpowered (13). Only very few trials included more than 100 PD patients (13). Two larger PD trials, balANZ and IMPENDIA-EDEN, were terminated early, needed inclusion of additional centers, and/or were synthesized from more than 1 trial, weakening their power to assess moderate but clinically relevant effects (35,36). Thus, novel and better trials are required to provide evidence for innovation in PD fluid composition.
The difficulty of recruiting PD patients into clinical trials, even in multi-country efforts supported by global PD fluid manufacturers, suggests that a number of 200 to 300 patients may represent the “ceiling” of clinical trial feasibility in PD. However, Blake as well as Holmes and Smit suggested that population sizes of at least 1,000 to 1,400 patients are needed to reach an acceptable power of 80% in order to detect clinically relevant improvements in membrane protections by a novel PD fluid candidate (14,15). Likewise, our model shows that a reduction of 30% using the endpoint “peritoneal membrane deterioration” would require a trial of more than 1,000 PD patients. Even focusing on the individual outcome with the highest incidence and the most patient-relevant clinical outcomes, i.e., peritonitis, would still require following at least 340 patients in a 2-year trial, thus exceeding the presumed maximally feasible population.
In this paper, our model demonstrates that the introduction of composite endpoints may successfully address the current limiting factors of PD trials (too low feasibility and high risk of such trials). The composite outcome “major adverse peritoneal events” (MAPE) was constructed by combining “technical failure,” “peritonitis,” and “peritoneal membrane damage,” each representing a PD-related complication corresponding to clinical events relevant at the patient level. The composite outcome MAPE was defined as “occurrence of any of these events” thereby increasing the proportion of patients experiencing a relevant PD outcome (see Figure 2). In a future PD trial, MAPE could be also assessed using other definitions, such as “time to first occurrence of any of the designated component events” or alternately as “total endpoint events,” as each PD patient might have more than 1 event (16).
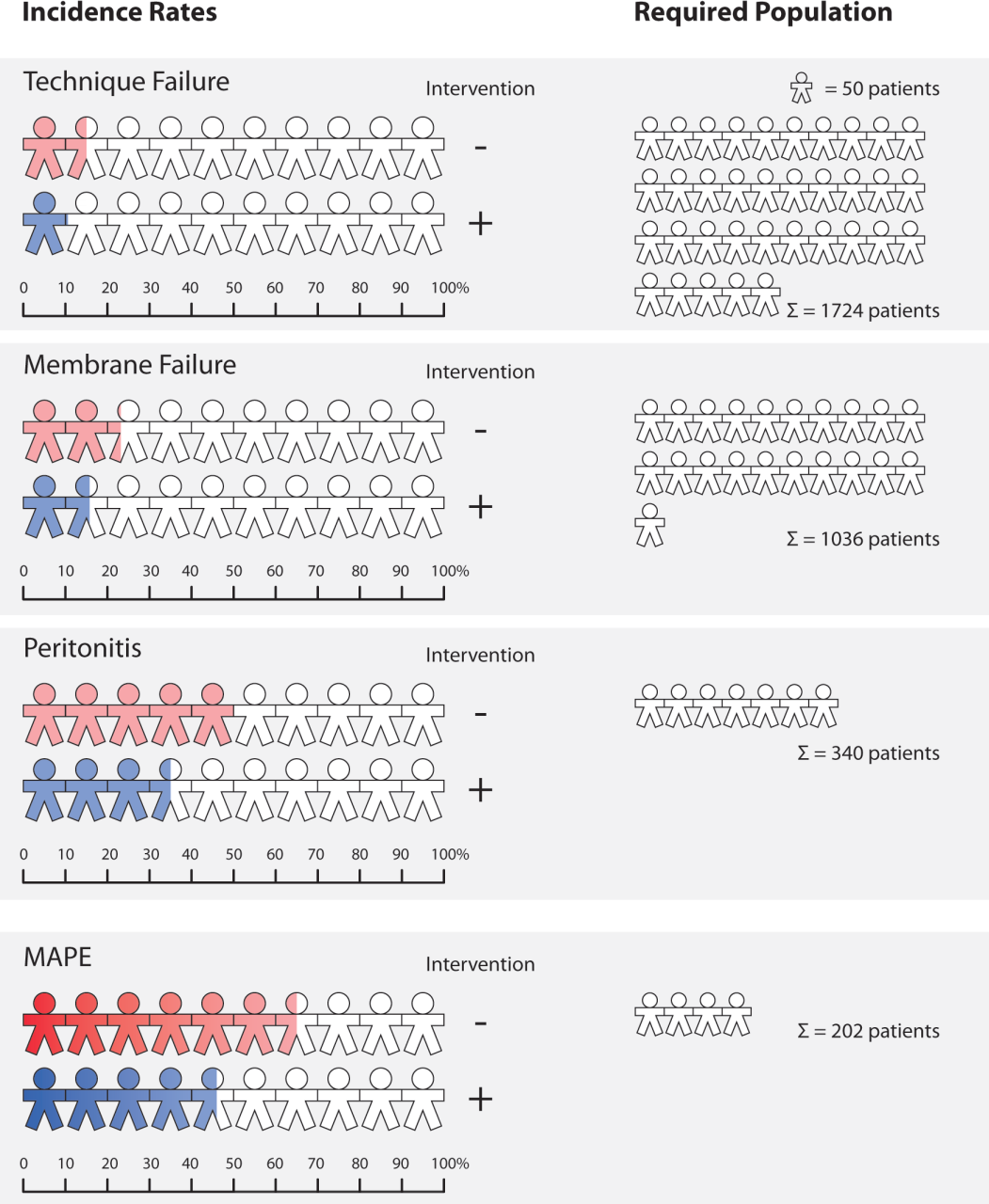
Example of the effect of introducing the composite outcome MAPE on event rate and corresponding requirement of population size for an adequately powered randomized controlled trial. On the left panel, each patient symbol reflects 10% of a given PD patient population. A colored symbol represents the proportion of patients experiencing a predefined event (PD-related complication). On the right panel, each symbol reflects 50 PD patients needed to be randomized to each arm to detect a 30% effect of treatment with 80% power.
The composite outcome MAPE will improve feasibility of clinical trials in PD by reducing the required population size for sufficient power to adequately capture the clinical impact of a novel PD fluid. Figure 2 compares 4 hypothetical trials to show that the combination of 3 relevant PD outcomes increases the power of trials (by increasing event rates) and thereby markedly reduces the required sample size. In contrast to a case when only 1 of the components is used, the resulting required sample size may be up to 6 times smaller. Thus, use of MAPE allows detection of an overall reduction of 30% of clinically relevant PD outcomes in a trial of 202 PD patients with 80% power, resulting in a required population size that should be feasible according to previous trial experiences.
Clinically, the individual outcomes “peritonitis” and “peritoneal membrane deterioration,” in MAPE are regarded as major PD-fluid related complications resulting in cause-specific technical failure of PD (23,24). Importantly, these outcomes have also been ranked as prime candidates for standardized core endpoints relevant for patients by the standardized outcomes in nephrology (SONG) initiative (37 38-39).
Furthermore, the biological rationale for MAPE is based on experimental and clinical evidence of inherent cytotoxicity of currently available PD fluids on respective peritoneal cell populations (23,24): injury to peritoneal leucocytes, the immunological barrier against infectious complications, results in reduced immunocompetence—and an increased risk for peritonitis (23,24,40,41). Injury to mesothelial cells, lining the peritoneal cavity, triggers chronic inflammation and fibrosis of the peritoneal membrane and vasculature— ultimately resulting in deterioration of the peritoneal membrane (23,24). Thus, the biological plausibility of examining the effects of novel PD fluids is satisfied, and any component of MAPE can be expected to be beneficially affected by an appropriately developed novel PD fluid.
Peritonitis is well defined and its diagnosis established in international guidelines (42). Peritonitis tends to cluster within the first 6 months after initiation of PD treatment and is strongly influenced by center effects, resulting in corresponding requirements in trial design, such as adequate stratification procedures (43). As mentioned above, a recent compilation of relevant trials has failed to show a clear impact of the introduction of novel PD fluids on the risk of peritonitis based on underpowered reports in recent literature. In a MAPE-based trial, peritonitis will likely represent the main driver constituting the majority of clinically relevant outcomes.
With regard to peritoneal membrane deterioration, definitions and the modality of its assessment are less established, although the impact is observed clinically and influences patient outcome. Overt ultrafiltration failure presents as over-hydration, typically resulting in cause-specific technical failure, but is preceded by a long phase of challenging fluid management problems requiring frequent prescription changes. In MAPE, membrane-based assessment of ultrafiltration loss is clinically assessed by the PET. Increases in solute transport and/or decreases of sodium sieving and/or ultrafiltration in repeated PET during the course of PD treatment are generally considered to be markers of inherent ultrafiltration failure and thus included in MAPE assessment. Although changes in solute transport status and ultrafiltration are frequently concordant, uncoupling of parameters may also reflect severe morphologic damage of the peritoneal membrane (24,25).
As peritonitis and peritoneal membrane deterioration are relevant causes for technical failure of PD, the latter outcome was also included as a component of MAPE, consistent with current guidelines of the relevant regulatory authorities (16,44). Recent definitions of “technical failure” in PD compile all events that are thought to cause unplanned termination of PD; these definitions also contain causes of PD termination that are less likely to be influenced by PD fluids, such as patient burn-out (although less peritonitis or fewer PD prescription changes could influence this parameter) or catheter dysfunction (45). However, if components of a composite outcome are predictably not influenced by the treatment, the overall effect will be diluted and statistical power reduced: the null hypothesis might fail to be rejected, even if the effects of the other components are in favor of the treatment (16,44). Thus, cause-specific technical failure for “peritonitis” and “inadequate dialysis” has been chosen for inclusion into MAPE, and all-cause technical failure will be separately analyzed.
The introduction of MAPE might be beneficial for clinical trial design (46) by not only increasing the power of a trial, requiring smaller sample sizes, but also by reducing the length and costs of a trial. Fewer patients would be exposed to new and potentially unsafe treatments. The use of composite endpoints, however, may also exhibit disadvantages. A major problem in the use of composite outcomes arises if the intervention has different effects on different components (47). Composite endpoints might be particularly difficult to interpret if the effects observed in the components are not in the same direction, or if the overall effect is driven by only 1 component. This may lead to overestimation of the effects of an intervention (48). Thus, in order to make sense from a clinical perspective, a composite endpoint must be a priori carefully constructed in terms of the underlying biological rationale, and each individual component needs to be a posteriori individually analyzed as a separate endpoint (16,44,48–50). In the case of the use of MAPE as a primary outcome in a PD trial, individual assessment of peritonitis and peritoneal membrane deterioration together with addition of all-cause technical failure (= technical survival) as secondary outcomes should allow global capture of potential effects of novel PD fluids.
In conclusion, introduction of the composite outcome MAPE, covering major PD-related complications, may improve feasibility of future clinical trials in PD. Use of MAPE results in the need for significantly lower population sizes to assess clinically relevant effects of novel PD fluids. Certainly, additional outcomes with harmonized and patient-reported outcome parameters will be needed beyond MAPE to allow evidence-based decision making at the meta level to comprehensively assess the potentially clinically relevant impact of novel PD fluids on the reduction of the currently high rates of patient attrition during PD therapy.
Footnotes
ACKNOWLEDGMENTS
This project has received funding from the European Union's Horizon 2020 research and innovation program under the Marie Sklodowska-Curie grant agreement No 633567. The authors would like to thank the participants in the Expert Group for their time, effort, and expertise. Participants were Simon Davies (Keele University), Eric Goffin (Université catholique de Louvain), Raj Mehrotra (University of Washington), Roberto Pecoits Filho (Pontificia Universidade Catolica do Parana), CC Szeto (The Chinese University of Hong Kong), Isaac Teitelbaum (University of Colorado Hospital), and Martin Wilkie (Sheffield Teaching Hospitals NHS Foundation Trust). Financial support by the Austrian Federal Ministry of Science, Research and Economy and the National Foundation for Research, Technology and Development is gratefully acknowledged.
The authors declare the following interests: AV has received honoraria and travel grants from Baxter and Fresenius (manufacturers of dialysis solutions) unrelated to this study. CA is cofounder of Zytoprotec GmbH, a spin-off of the Medical University Vienna that holds the patent “Carbohydrate-based peritoneal dialysis fluid comprising glutamine residue” (International Publication Number: WO 2008/106702 A1). KK is a former employee of Zytoprotec GmbH; PR is a former employee of Baxter Healthcare. MB, JN, HH, and FK have no conflicts of interest to declare.
