Abstract
The global unmet need for kidney replacement therapy means that millions of people die every year as they cannot afford treatment. Peritoneal dialysis (PD) offers comparable survival to haemodialysis and is often more affordable, but one barrier to increasing access is that conventional manufacturing and distribution of PD fluid is costly. Here we report the results from a pilot proof-of-principal study demonstrating for the first time that the Ellen Medical Devices Point-of-Care system can be used by patients to produce sterile PD fluid at the point-of-care. With further development, this low-cost system could offer a solution to the many millions of people around the world who currently cannot afford treatment for kidney failure.
Introduction
Peritoneal dialysis (PD) offers comparable patient survival to haemodialysis 1 and is more affordable in most countries. 2 Despite this, the cost of PD fluid still prohibits access to PD globally, particularly when local manufacturing is not available. 2
To address this unmet need, The Affordable Dialysis Prize was announced at the World Congress of Nephrology in March 2015 as the search for a new, affordable way of providing dialysis as safely and effectively as standard therapy but at a fraction of the cost. In 2016, Vincent Garvey’s Ellen Medical Devices Point-of-Care (EM-POC) affordable peritoneal dialysis system was announced as the winner. 3
The EM-POC system (Figure 1) comprises two components. The first is a lightweight, portable pure water distiller capable of producing sterile medical grade water. The second component is a plastic bag containing a proprietary concentrate of salts and sugars. Sterile water produced by the distiller is used to reconstitute conventional PD fluid at the point-of-care in the patient’s home. The freshly made PD fluid has been shown to be sterile and endotoxin free in extensive laboratory testing and meets the British Pharmacopoeia definition. A bag fill takes approximately 90 min. The patient is present for a few minutes at the start to connect the bag and a few minutes at the end to disconnect when it is filled. When complete, the bag can be used to perform PD in the conventional way. Although 2 L bags were used for this study, the system can be readily adapted to fill larger bags for nocturnal cycler dialysis.
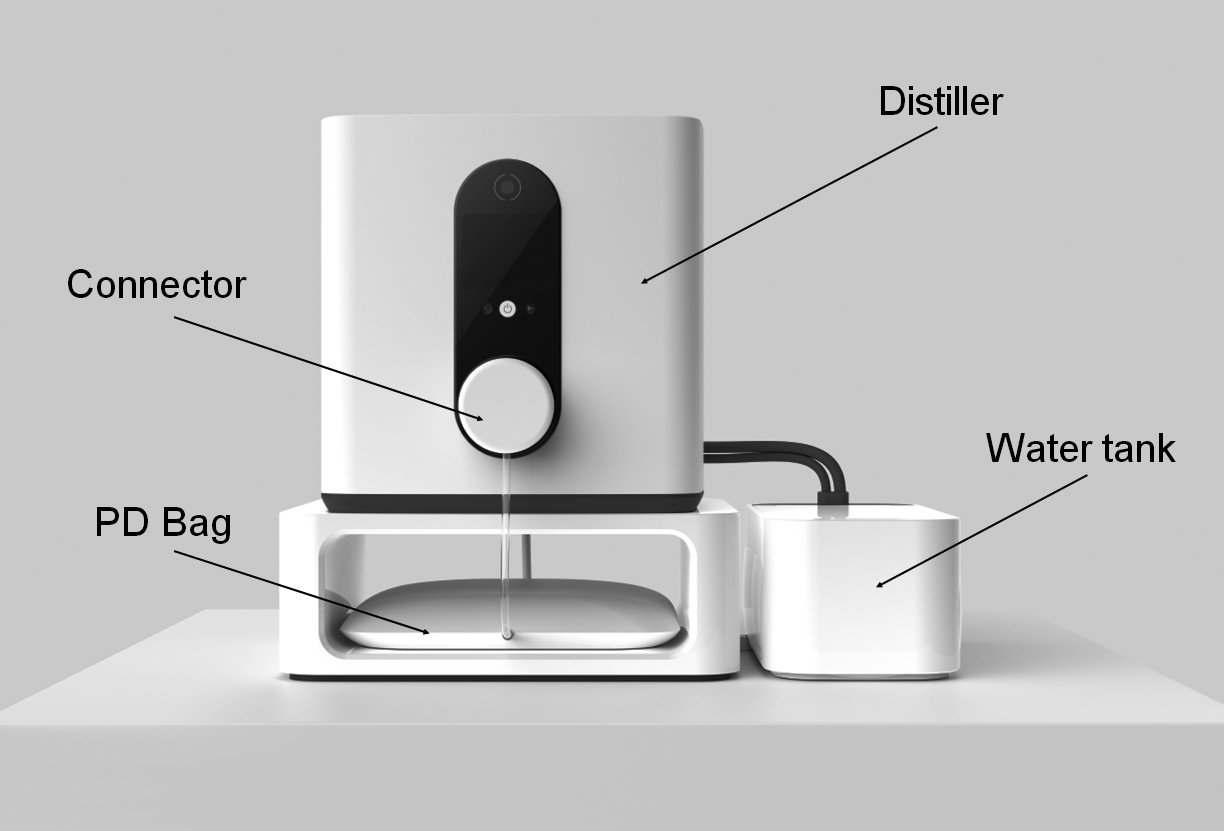
Annotated concept image of the EM-POC affordable dialysis system final design.
Having previously demonstrated the EM-POC PD fluid to be sterile and endotoxin free in benchtop testing, this study sought to confirm that patients can use the EM-POC system to produce sterile PD fluid in their homes. The Point-of-Care Peritoneal Dialysis System Early Evaluation Study (POC-PDEE) was a pilot proof-of-principal study comprising three inter-linked components: (1) human factors usability assessments, (2) an open label interventional study to assess whether patients could use the EM-POC system to generate PD fluid meeting sterility and chemical composition requirements for PD fluid and (3) semi-structured interviews to assess patient perspectives regarding the EM-POC system. We present the results of the sterility testing of the PD fluid from this novel point-of-care dialysis system. The usability and patient perspective data will be published at a later date.
Methods
Study outline
The study commenced in June 2022 and planned to enrol five participants from two hospitals in Sydney, Australia. An EM-POC prototype was installed in the home of each participant who was trained to use the system for three days by an experienced PD nurse prior to study commencement. Each participant then used the EM-POC system independently to fill PD bags which were sent for microbiological culture, endotoxin measurement and chemical composition testing. Reconstituted PD fluid was not used for dialysis by any participant. Potable Sydney tap water was used as input water to the distiller, and participant homes required a reliable supply of electricity. Two identical EM-POC prototypes were used for the study.
The study was designed and supervised by an international multidisciplinary steering committee, including a patient with first-hand experience of PD. The study received local institutional ethics approval (CH62/6/2020-149) and was registered prospectively with the Australian New Zealand Clinical Trials Registry (ACTRN12622000078752). The intention was to enrol five participants, but the study was terminated early for commercial reasons. No safety concerns were identified. Following study completion, chemical composition analyses were deemed unreliable by the steering committee due to suspected laboratory error and are therefore not reported.
Participants
Participants aged between 18 and 80 years, treated with PD for a minimum of 6 months and able to be trained to use the EM-POC system were eligible for inclusion. Patients with any condition or anticipated complication, procedure or kidney replacement therapy (KRT) modality change which would prevent compliance with study protocols were deemed ineligible. All participants provided written informed consent.
Study procedures
Participants used EM-POC prototypes to fill four PD bags/day for 5 days. A study nurse visited the participants each day to collect the filled PD fluid bags, which were collected within 24 h of reconstitution, and to assess for serious unexpected adverse events (SUAEs) or unexpected serious adverse device effects (USADEs).
Microbiological samples were cultured for 5 days for aerobic, anaerobic and yeast microorganisms. Presence of endotoxin was confirmed using Limulus amebocyte lysate test methods. Microbiological and endotoxin testing were conducted at an independent National Association of Testing Authorities (NATA)-accredited laboratory. Reconstituted PD fluid bag volume was assessed by weight, measured by the study nurse, minus the pre-filled bag weight.
Outcomes
Primary: Reconstituted PD bags were tested for the presence of microorganisms and endotoxin (number and proportion of bags with detected microbial growth or endotoxin concentration >0.03 EU/mL are reported).
Secondary: The volume of delivered PD fluid for each reconstituted PD bag. Adverse events or device malfunctions which created risk for the patient were recorded.
Statistics
Results are reported as counts and percentages for categorical outcomes and median with interquartile ranges (IQR) for non-normally distributed continuous outcomes.
Results
Five participants were screened, and three were subsequently enrolled in the study (Figure 2). Participants were aged 49, 63 and 73 years with a median PD experience of 20 months (IQR: 6–29). One participant was female. All 60 scheduled bag fills were completed and submitted for testing.

Flow diagram of participant enrolment to the POC-PDEE.
Primary outcome
Microbiological testing: All 60 samples tested were sterile (0 colony forming units (CFU)/200 mL) for aerobic, anaerobic and yeast cultures (Table 1).
Endotoxin testing: 2 of 60 samples (3%) had evidence of endotoxin detected. One sample with an endotoxin concentration confirmed between 0.05 EU/mL and 0.25 EU/ml. The second sample was reported as >0.03 EU/mL with the laboratory unable to retest this sample further (Table 1).
Results of microbiological and endotoxin testing of EM-POC peritoneal dialysis fluid samples.
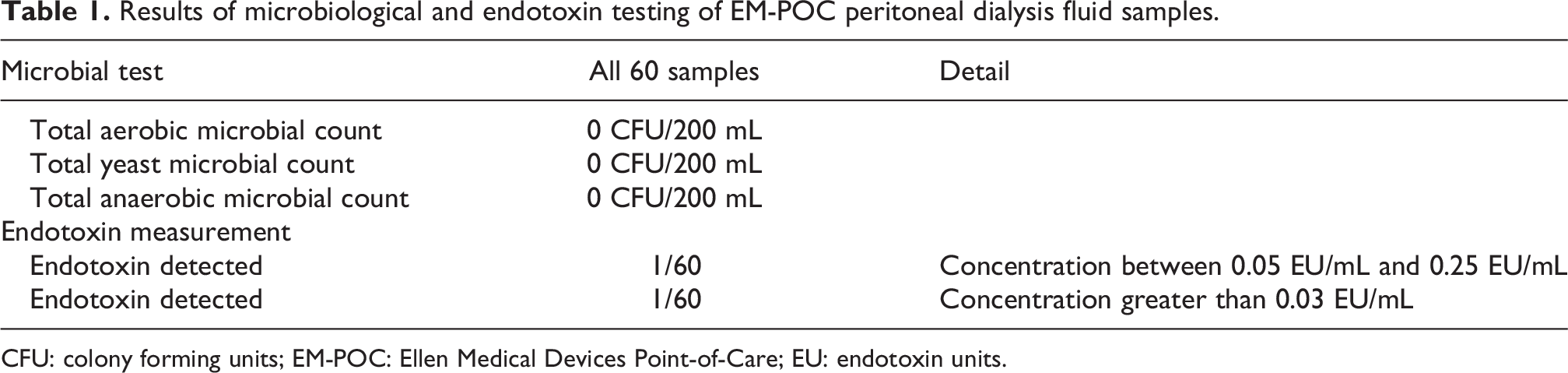
CFU: colony forming units; EM-POC: Ellen Medical Devices Point-of-Care; EU: endotoxin units.
Secondary outcomes
The median volume of reconstituted PD fluid in each filled bag was 2190 mL (IQR: 2184–2195). The median difference between the reconstituted volume and the intended volume of 2200 mL was 11 mL (IQR: 5–16).
No SUAEs or USADEs were reported. One reconstituted bag overfilled as the bag was left resting on the ledge of the trolley rather than hanging freely during filling.
Discussion
There were no detectable microorganisms in any of the 60 bags of PD fluid produced by participants using the EM-POC system, confirming that patients can produce sterile PD fluid at the point-of-care, and participants reported the system as simple and easy to use.
PD-related infection is associated with both mortality and transfer to haemodialysis 4 and is a critically important outcome to patients, clinicians and policymakers. 5 Fear of possible contamination of PD fluid has been described as a deterrent to clinicians preparing PD fluid at the bedside, limiting treatment options when commercially manufactured fluid is unavailable. 6 Results from this study provide reassurance regarding the sterility of EM-POC PD fluid produced at the point-of-care.
Presence of endotoxin in PD fluid can result in sterile peritonitis, 7,8 and attributable outbreaks have been reported. 7,9 Endotoxin was detected in two samples from this study. Extensive benchtop testing of output water from EM-POC prototypes has previously demonstrated total organic carbon and conductivity levels meeting the criteria for sterile medical grade water suitable for injection. The absence of endotoxin and microbiological contamination in both output water and reconstituted PD fluid has previously been further confirmed using NATA laboratory testing of samples. It is generally recognised that distillation achieves a 3–6 log reduction in endotoxin concentration from feed water levels, 10,11 and although endotoxin concentration of the input water was not quantified in this study, previous assessments of Sydney tap water in our laboratory have shown it to be >0.25 EU/mL with consistent complete removal by the EM-POC system. Other potential sources of the positive endotoxin results include contamination of the bags during manufacture, which for this proof-of-concept study were completed under experimental manufacturing arrangements, and contamination between reconstitution of the PD fluid and testing. The positive endotoxin results in this study came from different prototypes used by different participants, with no detected endotoxin in other samples tested from bags filled before or after from either prototype and are therefore believed to represent contamination rather than insufficient clearance of endotoxin. Further larger-scale testing is required. Future testing of the EM-POC system in different settings, particularly low resource environments where the quality of source water may vary, will also be important, including correlation between input and output water.
Although cost-effectiveness was not the primary purpose of this study, it is clear that the cost of transporting PD bags from a central manufacturing site – often in a different country – to patients’ homes is a major contributor to the cost of PD. A standard treatment regimen of four 2 L bags a day equates to around 3 tonnes of fluid per patient year compared with a year’s supply of EM-POC PD bags weighing around 230 kg. The savings in shipping could be substantial.
The study has some limitations. Testing was performed on a relatively small number of PD bag fills to minimise inconvenience to each participant, and a small number of participants were recruited for this pilot study. The study was terminated early for commercial reasons, and the planned chemical composition testing of the PD fluid was unreliable and therefore not reported. Despite these limitations, this study confirms for the first time that patients can use the EM-POC system to produce sterile PD fluid at the point-of-care. Large-scale manufacturing and testing are now required, but this represents important progress towards increasing access to PD globally, particularly in low-resource settings.
Footnotes
Acknowledgements
The authors thank the Sydney Local Health Department Peritoneal Dialysis Unit for their support and also the patients who took part in the study.
Author contributions
Research conceptualisation: BT, SD, VG and JK; data analysis: BT; supervision or mentorship: SD and JK. All authors contributed to study design, data interpretation and reviewed the manuscript prior to submission.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BT, JB, VG, AB, NK and JK are employees of Ellen Medical Devices on secondment from the George Institute for Global Health. BT, VG, VP, MJ, MG and JK are employees or hold appointments at the George Institute for Global Health which is the majority owner of Ellen Medical Devices. These relationships are considered to be synergies rather than conflicts. SD receives research funding from Baxter HealthCare, lecture fees from Baxter and Fresenius Medical Care, Advisory Board Fees from Ellen Medical and is president of EuroPD. MJ is responsible for research projects that have received funding from Amgen, Baxter, CSL, Dimerix, Eli Lilly, Gambro and MSD; has received fees for Advisory, Steering Committee and/or Scientific Presentations from Akebia, Amgen, Astra Zeneca, Baxter, Bayer, Boehringer Ingelheim, Cesas Linx, Chinook, CSL, Janssen, Medscape, MSD, Roche and Vifor; with any consultancy, honoraria or travel support paid to her institution.
Ethical approval
Sydney Local Health District Human Research Ethics Committee
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Ellen Medical Devices.
Informed consent to participate
All participants provided written informed consent to participate.
Informed consent to publish
All participants provided written informed consent to publish.
Trial registration
Australian New Zealand Clinical Trials Registry (ACTRN12622000078752).
