Abstract
Background
Acute kidney injury (AKI) is common in critically ill neonates, and peritoneal dialysis (PD) can be a lifesaving option. In China, however, much of the equipment for PD in neonates is not available. We describe results with a novel system for PD, which has been developed locally to improve access to therapy and care for critically ill neonates requiring PD in China.
Methods
The system comprises a 14-gauge single-lumen central venous catheter serving as a PD catheter, inserted by Seldinger technique, with an adapted twin bag PD system. Ten neonates with AKI were treated using the novel PD system.
Results
The 10 patients ranged in age from 1 day to 22 days, with bodyweights between 700 g and 3,300 g. Average time to renal function recovery was between 14 and 96 hours. Complications related to the novel PD system included leak (n = 1), catheter displacement (n = 1), and catheter obstruction (n = 1). There were no complications related to insertion, no cases of peritonitis or exit-site infection, and no subsequent hernias. A comparison of costs indicated that the novel PD system is less expensive than conventional systems involving open insertion of Tenckhoff catheters.
Conclusions
Peritoneal dialysis using the novel PD system is simple, safe, and effective for suitable neonates with AKI in China.
Neonates with AKI can be treated with a range of renal replacement therapy (RRT) modalities, including intermittent hemodialysis (HD), continuous RRT, and peritoneal dialysis (PD). For many reasons, the application of these techniques is not straightforward, however. In general, there is less cumulative clinical experience of RRT in neonates, and there is a small base of evidence with which to define best practices and guide decisions around starting and stopping, etc. Of relevance to this article, one of the most common difficulties is the limited availability of suitable equipment, such as low-volume extra-corporeal circuits and correctly sized hemodialyzers.
Peritoneal dialysis is not infrequently utilized in this population, since it avoids the need for anticoagulation or invasive vascular access, and provides excellent hemodynamic stability (6–8). Importantly for resource-constrained environments, PD is operationally simple and relatively inexpensive. However, there are 3 major challenges that need to be overcome when using PD in neonates: obtaining suitable peritoneal catheters, placing peritoneal access in a non-traumatic manner, and accurately adminstering and measuring the volumes of exchanges. We describe a novel PD system for neonates with AKI that addresses these needs as they exist in China. We report our initial experience, and compare our results with those of others who have published on similar systems in the literature.
Methods
We analyzed prospectively-collected data of all patients who underwent treatment with the novel system from 2011 to 2015 in our unit. Data were sourced from a single-center Excel database and were enriched with further data extracted from hospitalization clinical records. The patients’ demographic data (age, gender, weight), clinical history (cause of AKI, PD prescription, duration of PD therapy), peri-procedural details, and post-procedural outcomes (complications related to PD, laboratory results, requirement for vasopressors and mechanical ventilation, and clinical outcome) were evaluated.
Intraperitoneal bleeding was diagnosed if the dialysate was bloody after the first 3 exchanges. Catheter blockage was diagnosed if the dialysate flow was sluggish or absent at any time. An exit-site leak was diagnosed with the appearance of any fluid around the PD catheter exit site identified as dialysate. Peritonitis was diagnosed with an effluent cell count with white blood cells (WBC) more than 100/μL, with at least 50% polymorphonuclear neutrophilic cells.
We have reported our experience in line with the standard “preferred reporting of case series in surgery” (PROCESS) reporting guideline (9). This also includes the reporting of adverse surgical events in detail in accordance with the Clavien-Dindo Classification (10). The study received full ethics approval from the hospital institutional review board, and the parents of all children in the study provided informed consent.
Study Setting and Participants
The study was set in Xiangya Hospital, Changsha, Hunan Province, China. It is a 3,000-bed hospital which provides all secondary and tertiary services in the city. The nephrology department provides usual dialysis and kidney transplant care, but also inpatient RRT services to all AKI patients in the hospital. The study was conducted in sequential patients who were treated with the novel PD system.
Patients were recruited into the study if they were neonates (< 28 days old), with AKI as defined by Kidney Disease: Improving Global Outcomes (urinary volume < 0.5 mL/kg/h for more than 6 hours, or an increase in serum creatinine equal to or greater than an absolute value of 26.5 μmol/L or 1.5 times baseline value), and suitable for PD therapy. Those with major trauma, burns, or abdominal/abdominal wall infection were excluded from the study on the basis of being unsuitable for PD. Patients were followed up until discharge from hospital.
Pre-Intervention Considerations
All patients were treated in the pediatric and neonatal intensive care unit. Mechanical ventilation and vasopressor support were available and utilized for patients with indications such as acute respiratory distress syndrome (ARDS) and hypotension. All patients were treated with broad spectrum antibiotics as appropriate. In our unit, the usual indications for RRT in AKI are threatening metabolic abnormalities (e.g. refractory acidosis) and fluid overload; RRT is not initiated for AKI per se in the absence of clinical indications.
Description of the Intervention and Post-Intervention Considerations
The novel PD system is illustrated in Figures 1 and 2. The peritoneal catheter is a single-lumen 14-gauge central venous catheter (Royal Forria Medical Equipment Co. Ltd., Zhuhai, Guangdong, China), which is 20 cm in total length, with a 2.2-mm inner diameter and 2.8-mm outer diameter, with 1 side hole and another hole at the tip of the catheter. The catheter is connected to 23-way taps. One port of the 3-way tap is connected to a graduated syringe and PD solution, while another port is connected with a graduated drainage bag. This creates a Y-connector set for the twin-bag PD system. The new device is subject to a national patent in China (patent number: 2014 2 0035268.4).
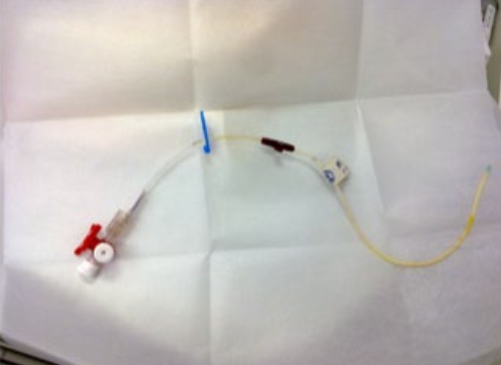
The 14-gauge central venous catheter serving as peritoneal access in the novel PD system. PD = peritoneal dialysis.
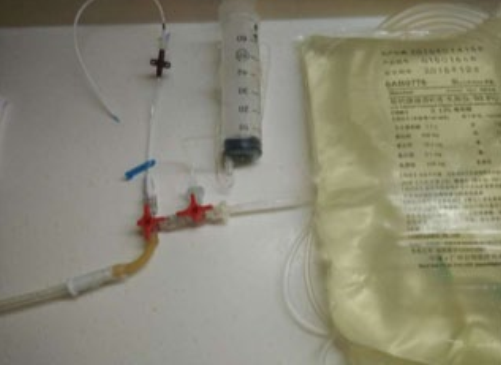
Connectology of the closed 2-bag system in the novel PD system. PD = peritoneal dialysis.
The catheter is inserted by modified Seldinger technique. The exit site of the catheter is marked 1 cm lateral to the umbilicus on the left. After preparation of the abdominal skin with antispetic, the exit site is anesthetized by 2 mL of 1% lidocaine. Antibiotic prophylaxis is administered in patients who are not already on suitable antibiotics. The puncture needle is slowly introduced with negative pressure until penetrating into the peritoneal cavity. Peritoneal entry is determined clinically by a “pop” and loss of resistance, or aspiration of ascites, and ultrasound guidance is typically not required. A “J” tip guide-wire (0.81 mm x 60 cm) provided in the central venous catheter insertion pack is then introduced through the puncture needle, and the needle is then removed, after which the tract is dilated using dilators before threading the catheter over the guide wire. The length of catheter in the peritoneal cavity depends on the vertical length between the umbilicus and pubic symphysis. After the catheter is positioned, 0.9% saline 15 – 20 mL is flushed into the peritoneal cavity to check patency and correct positioning of the catheter, with re-siting if necessary. An X ray is taken to confirm the location of the catheter (see Figure 3).
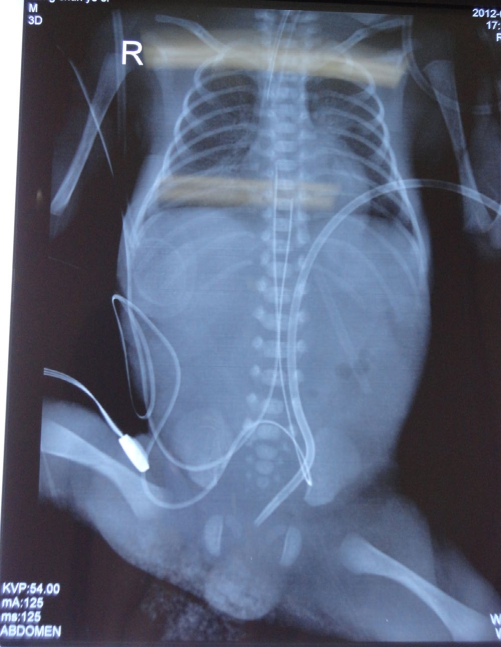
Example of a post-procedural X ray after PD catheter insertion using the novel PD system. PD = peritoneal dialysis.
In our center, there is only 1 operator responsible for the insertion of PD catheters in neonates (XA), and all the catheters inserted prior to this reported experience were done so using an open technique. At present, there are no China-wide credentialing criteria for operators performing this procedure: competency is based on local heuristics, and a common and informal requirement that the operator has at least 5 successful procedures behind them.
Once peritoneal access is satisfactory, PD is administered using the 23-way taps. These taps control the inflow and outflow of PD fluid, with volume titration of exchanges measured precisely and administered by syringe, according to prescription. Drainage of the PD fluid is by gravity. The PD fluid used for the system is Dianeal PD4 (Baxter China Ltd, Shanghai, China), and the prescription and management of PD performed according to local and international guidelines (8,11). The performance of the PD catheter is assessed frequently by examining the volume and speed of drainage, and other examinations when necessary (e.g. X rays for suspected catheter displacement, etc.). Once PD is no longer required, the catheter is removed by simple withdrawal and appropriate dressing of the exit site.
Results
Participants
Ten patients were recruited for the study, with the first patient being recruited on 10 August 2011 and the last on 14 October 2015. Over this period, no patient received PD using a Tenckhoff catheter inserted by open surgery, and all PD was performed using the novel PD system. In addition, no changes were made to standard operating procedures for catheter insertion or implementation of dialysis with the system.
Main Results
Descriptive demographic and clinical data of the 10 patients are shown in Table 1. There were no procedural or peri-procedural complications. Post-procedural complications included 1 case each of a leak, catheter displacement, and catheter obstruction. There were no cases of peritonitis, exit-site infection, or hernia at discharge, and all patients had a satisfactory renal recovery. These are detailed in Table 2. All studied patients survived to hospital discharge.
Descriptive Demographic and Clinical Data
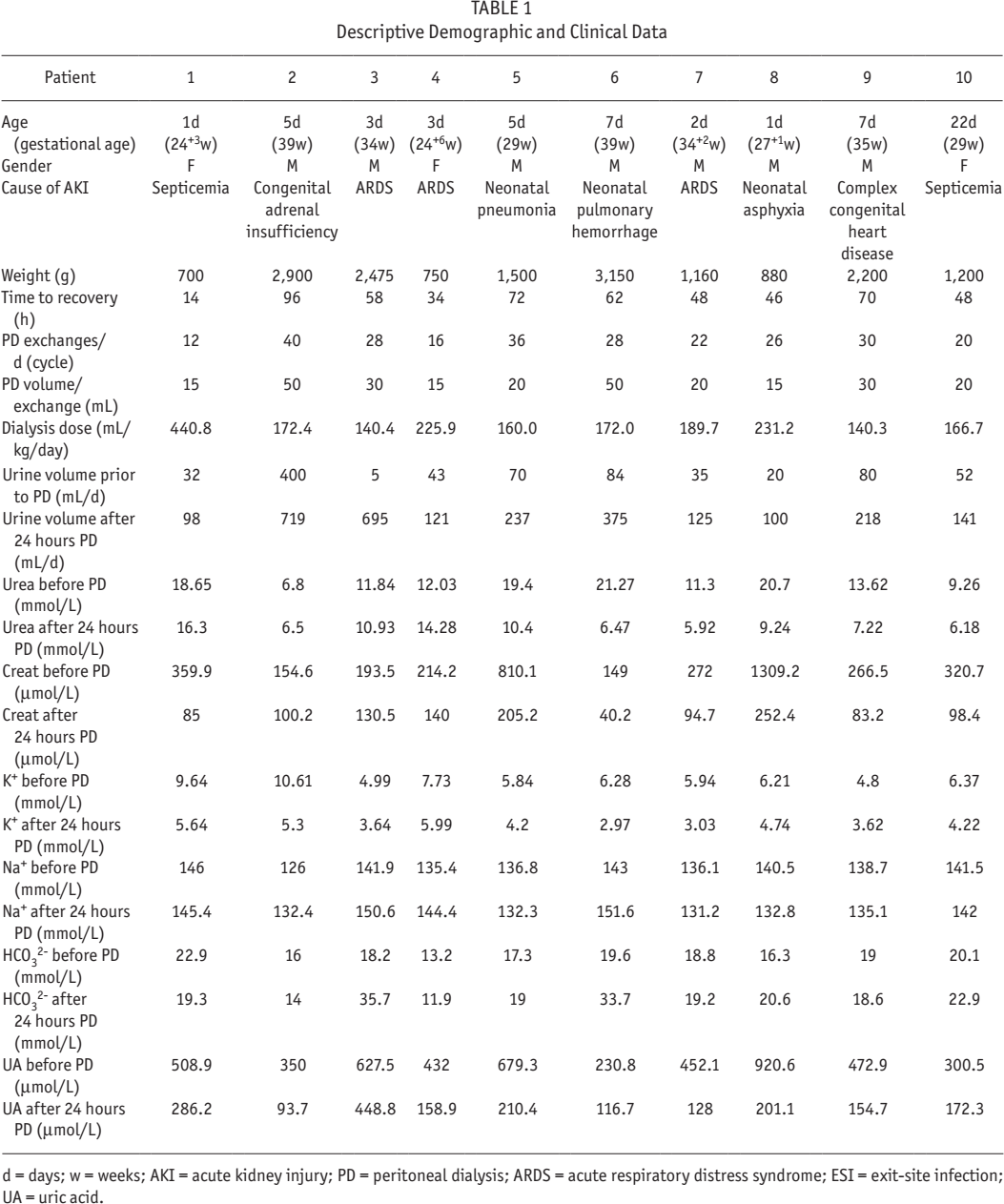
d = days; w = weeks; AKI = acute kidney injury; PD = peritoneal dialysis; ARDS = acute respiratory distress syndrome; ESI = exit-site infection; UA = uric acid.
Description and Classification of Complications

ESI = exit-site infection.
Clavien-Dindo Classification of Surgical Complications: Grade I–Any deviation from the normal postoperative course without the need for pharmacological treatment or surgical, endoscopic, and radiological interventions. Grade II–Requiring pharmacological treatment with drug other than such allowed for Grade I complications. Grade III–Requiring surgical, endoscopic, or radiological intervention. Grade IIIa–Intervention not under general anaesthesia. Grade IIIb–Intervention under general anaesthesia. Grade IV–Life-threatening complication. Grade IVa–Single organ dysfunction. Grade IVb–Multiorgan dysfunction. Grade V–Death of a patient.
Discussion
In a prior study, we reported outcomes in our center for infants with AKI following an operation for complex congenital heart disease. These infants were treated with PD therapy, using a modified Tenckhoff catheter and open insertion. In these patients, we reported excellent outcomes with a total mortality rate of 11%—an illustration that PD can be an effective and safe treatment of AKI (12). Our ongoing positive experience of PD therapy, however, has been marred by the difficulties we experienced with using available equipment and techniques in China, which are described below.
In China, and the rest of the world, the traditional Tenckhoff catheter is the most widely used device, even for neonates (13,14). These catheters have a large-diameter lumen and side holes that allow uninterrupted dialysate flow and good clearance, as well as double cuffs with subcutaneous tunneling that reduce peritonitis and leaks (15). In China, however, the smallest available Tenckhoff catheters are 30 cm in length, with 12-cm abdominal segments, 8 cm of which has side holes. The Tenckhoff catheters that are specifically for neonates and small infants (such as are reported by Kara et al. [16] and Mok et al. [17]) are not available in China. Therefore, the catheters that are available in China have to be adapted. The vertical distance between the umbilicus and the symphysis pubis in neonates and infants with low bodyweight is only 3 – 6 cm. The available Tenckhoff catheters are therefore trimmed before insertion, which reduces the number of side holes and therefore dialysate flow, as well as making the tip rough and potentially harmful to viscera.
Tenckhoff catheters, especially if they have been modified, are usually inserted by open surgery that requires skin and peritoneal incision, purse-string suturing, and subcutaneous tunneling. This can constitute a “second-strike” on critically ill neonates with AKI or multi-organ failure. Any surgery in neonates is challenging and can cause clinicians to hesitate, inappropriately delaying the initiation of dialysis (18). The repositioning or removal of Tenckhoff catheters requires a second surgical procedure. Even under the best of conditions, there is a high rate of complications related to PD access in neonates. In several recent studies, the incidence of catheter-related complications was 20% – 60%, with catheter malfunction and leakage being the most common problems (16,17,19,20).
There is, therefore, a pressing need for PD systems with improved safety and efficacy in neonates. In China, with its particular limitation in equipment availability, this need is even more pressing. Our study showed that PD with an improvised PD system rapidly corrected the biochemical disruption characterizing AKI and enabled satisfactory renal recovery—all patients were recovered or in the recovery phase by 14 to 96 hours. Importantly, the system was simple and safe to insert and operate, and allowed early institution of PD in an effective manner in children presenting with AKI to our service. The improvised PD system in our study includes a mechanism for easy and accurate administration of exchanges, which may be only between 10 – 100 mL in volume in these neonates. Anecdotally, we felt that the system allowed accurate and easier fluid balance.
Conclusion
Our study is certainly not the first to use an improvised device for PD in neonates, although it is the first experience in China. Improvised devices were obviously the only option before the widespread availability of dedicated silicone catheters. In a selection of the literature, clinicians have used central venous catheters or devices derived from them (21–23), intravenous cannulae (18,23,24), pleural catheters (25), adaptations of rigid PD catheters (26,27), and general purpose drains (23,28,29) (see Table 3). They all report a reasonable experience, although note is made of procedural death due to massive intraperitoneal hemorrhage from vascular injury from a stylet (23), and a generally higher rate of dialysate leak and catheter blockage when intravenous cannulae are used (21). We acknowledge that our experience is limited, but we feel that it is encouraging enough to motivate a larger multicenter exploration of the effectiveness of the system in perhaps a pragmatic clinical trial. The principles of our novel PD system could be enhanced by further testing and device engineering, and the system is of course not restricted to only neonates with AKI, but also those who need blood purification for other reasons such as those with inherited metabolic diseases, hyperammonemia, etc.
Reported Experience of PD in Neonates Using Improvised Devices
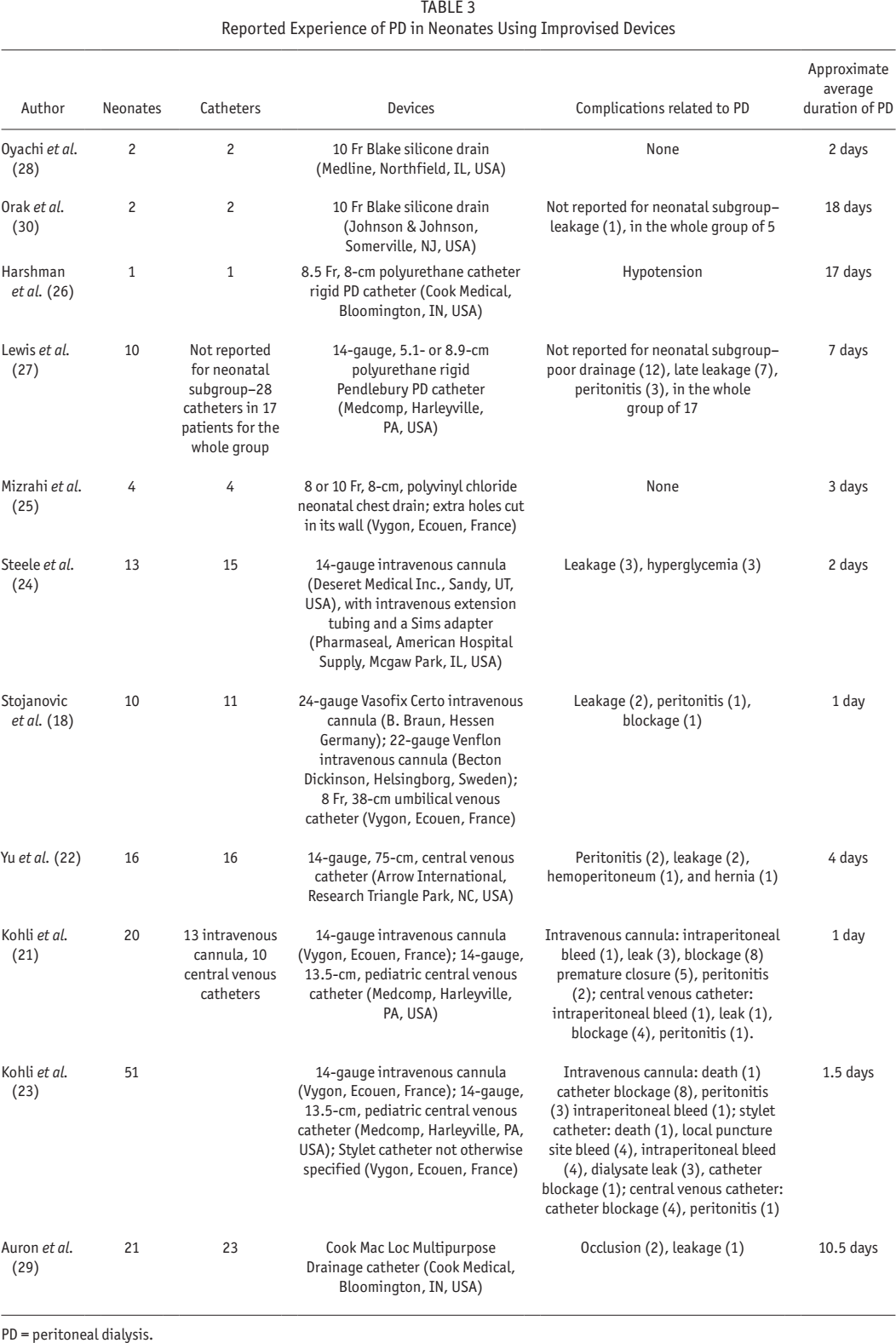
PD = peritoneal dialysis.
In summary, we have shown the feasibility and advantages of a novel PD system for neonates, which provides suitable peritoneal access, the means to non-traumatic insertion and removal, accurate administration and measurement of PD exchanges, and low cost (Table 4). The availability of the system could improve access to therapy and care in China, as an increasing number of neonates and small infants with complex diseases are presenting to renal physicians and pediatricians for care.
Local Costs of Key Equipment Used for PD for AKI in Neonates
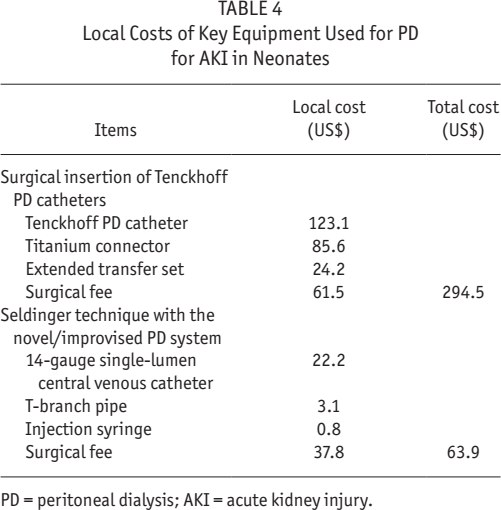
PD = peritoneal dialysis; AKI = acute kidney injury.
Footnotes
Acknowledgments
We gratefully acknowledge all patients, families, and physicians who contributed significantly to this study. The research reported here was funded by a grant from the Science and Technology Department of Hunan Province (No:2014FJ3148), the Renal Research Grant of Baxter China (No. CHN-RENAL-IIS-2012–005), and the Patent “A new PD device for infants” (patent number: 2014 2 0035268).
The results presented in this paper have not been published previously in whole or part, except in abstract format. MRM and FT are full-time employees of Baxter Healthcare. MRM is also an adjunct Associate Professor of the University of Auckland (New Zealand) and a part-time employee of Counties Manukau Health (New Zealand) as a clinical nephrologist.
