Abstract
Cell replacement therapy is a promising avenue into the investigation and treatment of Parkinson's disease (PD), and in some cases, significant long-term motor improvements have been demonstrated. The main source of donor tissue is the human fetal ventral mesencephalon (FVM), which consists of a mixed neuronal population, and its heterogeneity likely contributes to the inconsistent outcome observed in clinical trials. Therefore, detailed knowledge about the neuronal subpopulations in the VM seems essential for successful cell transplantation. Interestingly, it has been reported that some tyrosine hydroxylase-positive (TH+) neurons in the VM of adult rats and in cultured midbrain-derived neuroblasts coexpress additional neurotransmitters. Thus, the present study investigated, by means of colocalization analyses, the possible expression of GABA or serotonin in TH+ neurons. For that purpose, both fetal rat and human dissociated, organotypic and neurosphere FVM cultures as well as an animal model of PD were investigated. In dissociated rat FVM cultures, approximately 30% of the TH+ neurons coexpressed serotonin, while no colocalization with GABA was observed. Interestingly, coexpression of TH and serotonin was found to be dependent on the time in culture, the plating density, and the exposure to neurotrophic factors, that is, higher cell densities and treatment with brain-derived neurotrophic factor resulted in a significantly reduced coexpression rate. Notably, even though approximately 30% of the dopaminergic neurons in the donor tissue coexpressed serotonin, no colocalization could be detected in grafts 1 month after intrastriatal transplantation into hemiparkinsonian rats. In conclusion, a significant and susceptible subpopulation of dopaminergic neurons in FVM tissues coexpresses serotonin. This might have potential implications for the future selection and handling of cells prior to transplantation in PD.
Keywords
Introduction
Increasing efforts have been made in the field of dopaminergic cell replacement as a therapeutic strategy for Parkinson's disease (PD). At present, the fetal ventral mesencephalic (FVM) tissue is still considered the “gold standard” for cell transplantation approaches in PD. Despite encouraging long-term clinical improvements that have been reported for several patients up to 20 years after fetal VM transplantation 1 , this technique is far from being considered an established clinical intervention. Indeed, the controversial outcome of the different studies addressing the efficacy of fetal midbrain dopaminergic cell transplantation for PD patients emphasized the necessity to define standardized tissue selection, collection, storage, and implantation procedures2-11. In particular, a detailed cellular characterization of the graft tissue and more knowledge on the development of the different neuronal subpopulations present in these tissues are essential for a safe and effective outcome of cell transplantation for PD patients. Indeed, it is known that FVM tissue grafts consist of a mixture of different neuronal cell types. Beside tyrosine hydroxylase-positive (TH+) dopaminergic neurons, serotonin, γ-aminobutyric acid (GABA), enkephalin, and substance P-containing neurons also have been shown to be present in FVM tissue grafts12-16. In fact, it has been suggested that such heterogeneity likely accounts for the variable outcome seen among groups of grafted patients17-19. The content of dopaminergic and serotonergic cells assumes a special relevance in the framework of cell transplantation, as the first has the potential to antagonize and the latter has the capacity to promote the development of graft-induced dyskinesia20-22. Thus, improvement of current cell transplantation protocols relies also on interventions that might concomitantly optimize the yield of dopaminergic neurons and reduce the level of serotonergic neurons in grafts for transplantation23,24. Despite the known importance of graft cell content, a detailed characterization of the tissue grafts employed in clinical and preclinical settings is still missing. Above all, the phenotypic signature of the neurons and the mechanisms that might steer their fate remain to be defined. Thus, a broader definition of the expression profile of critical phenotypic markers might provide insights into the field of transplantation. Hence, the present study aimed at assessing potential coexpression of dopaminergic neurons with GABAergic and, especially, serotonergic markers in FVM tissue of rats and humans.
Materials and Methods
Animals
Pregnant Wistar rats were purchased from Janvier Labs (Le Genest-Saint-Isle, France) and housed at 12-h light-dark cycle with food and water ad libitum. All experiments were carried out in the light phase and in accordance with the guidelines of the Animal Research Ethics Committee of the Canton Bern, Switzerland, and the University of Bern Animal Care and Use Committee, Switzerland (Study Nos. 89/05 and 6/12).
Human Tissue
All experiments were carried out in accordance with the ethics committee of the Medical Faculty of the University of Bern and the ethics committee of the State Bern, Switzerland (Study No. 181/07). Written consent was given by the women seeking abortion.
Preparation and Culture of Rat Organotypic Fetal Ventral Mesencephalic Cultures
Cell cultures from embryonic day 14 (E14) VM were prepared as described previously 25 . In brief, pregnant Wistar rats were deeply anesthetized using gas inhalation (4.5%-5% isoflurane, 75% N2O, 20% O2) followed by an intraperitoneal (IP) injection of a mix of ketamine (Narketan; 120 mg/kg; Vetoquinol, Ittigen, Switzerland) and xylazine (Xylapan; 20 mg/kg; Vetoquinol). The fetuses were removed by cesarean section, and the FVM was isolated under a stereoscopic microscope. For preparation of organotypic cultures, that is, free-floating roller tube (FFRT) cultures, the FVM explants (2 mm × 1.5 mm × 1 mm in size) were cut into four equally sized quadrants and transferred into gas-permeable conical plastic tubes (BD Falcon, Allschwil, Switzerland) supplied with 1 ml of culture medium. The medium consisted of 55% Dulbecco's modified Eagle's medium (DMEM; Thermo Fisher Scientific, Reinach, Switzerland), 32.5% Hank's balanced salt solution (HBSS; Thermo Fisher Scientific), 1.5% glucose, 10% fetal calf serum (FCS; GIBCO), 1% 0.01 M 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES; Merck, Zug, Switzerland), and 1% antibiotics-antimycotics (streptomycin, 10,000 μg/ml; penicillin, 10,000 U/ml; and amphotericin B, 25 μg/ml; Thermo Fisher Scientific). The tubes were placed in a rotating roller drum (1 rev/min) placed in an incubator at 37°C in a 5% CO2 atmosphere25,26. The medium was changed after 2 days in vitro (DIV) and then every other day. At DIV8, the four FVM FFRT cultures originally derived from one fetal brain were pooled, washed twice in 0.1 M phosphate-buffered saline (PBS; pH 7.4), and either fixed in 4% paraformaldehyde (PFA; Merck) in PBS for 1 h and processed for histology or used for transplantation, as described below. Cultures for histology were cut at 20 μm on a cryostat and mounted on Superfrost glass slides (Thermo Fisher Scientific).
Preparation and Culture of Rat Neurospheres
For the preparation of neurospheres, the FVM explants were cut into equally large pieces, mechanically dissociated, and transferred into uncoated six-well plates. For the first 7 days, the cultures were placed in 2 ml of medium consisting of DMEM/F12 with
Preparation and Culture of Rat Dissociated Fetal Ventral Mesencephalic Cultures
The FVM explants were mechanically dissociated as previously described
25
and plated at different densities into wells with precoated poly-
Preparation and Culture of Human Organotypic Fetal Ventral Mesencephalic Cultures
The embryonic age of the specimen was 9.1 to 10 weeks after conception (n = 4) for the analyses as determined by the method of Evtouchenko et al. 27 . The FFRT cultures were prepared from FVM derived from aborted human fetuses using the protocol described in detail previously28,29. In brief, the FVM tissue was dissected out and subdivided into equally sized pieces. Each piece was then placed into conical tubes supplied with 1 ml of medium and then transferred in a slowly (1 rev/min) rotating roller drum placed in an incubator at 37°C in a 5% CO2 atmosphere. The medium was identical to the one used for rat FVM FFRT cultures as described above and was changed every second day. At DIV8, the cultures from one embryo were pooled, washed twice in ice-cold HBSS, and fixed for 1 h in 4% PFA in PBS. Cultures were cut on a cryostat at 20 μm and mounted on Superfrost glass slides and used for histology, as described below.
Preparation and Culture of Human Neurospheres
The human FVM neurospheres were prepared from FVM derived from aborted human fetuses, as described above. The embryonic age of the specimen (n = 4) was 7.4, 8.0, 10.3, and 10.5 weeks after conception. The FVM tissue was cut into small equally large pieces, mechanically dissociated, and transferred into uncoated six-well plates. For the first 7 days, the cultures were placed in 2 ml of medium consisting of DMEM/F12 with
Hemiparkinsonian Rats, Transplantation Surgery, and Perfusion
Female Wistar rats (Janvier Labs), weighing 180-220 g, were anesthetized with gas inhalation (4.5%-5% isoflurane, 75% N2O, 20% O2) followed by an IP injection of a mixture of ketamine (75 mg/kg) and xylazine (5 mg/kg) and a subcutaneous (SC) injection of buprenorphine (0.5 mg/kg; Reckitt Benckiser, Wallisellen, Switzerland) 30 min before the surgical intervention. Subsequently, the rats were placed in a stereoscopic frame (Kopf instruments, Tujunga, CA, USA) on a heating pad. The lesions were performed as previously described30,31. In brief, animals received an injection of 4 μl of 6-hydroxydopamine (6-OHDA, 32 mM; Sigma-Aldrich Chemie, Buchs, Switzerland) into the right striatum through a small burr hole created in the skull. The injection was performed over 6 min using a 10-μl Hamilton (Reno, NV, USA) microsyringe. The following coordinates in relation to the bregma (Paxinos Watson rat brain atlas) were used: 1.0 mm posterior, 3.0 mm lateral, and 5.0 mm ventral to the dura; the incisor bar was set at 0.0 mm.
Five months after the 6-OHDA lesions, rats were anesthetized as described above. The rats were mounted on a stereoscopic frame on a heating pad, and each rat received a transplant consisting of four FVM cultures (corresponding to one FVM) grown for 8 DIV. The four FVM cultures were loaded on a 20-gauge spinal needle (Nihon Unisys, Tokyo, Japan) and injected into the right striatum of the rats as previously described 26 . The following coordinates in relation to the bregma (Paxinos Watson rat brain atlas) were used: 1.0 mm posterior, 2.7 mm lateral, and 4.5 mm ventral to the dura; the incisor bar was set at 0.0 mm. After injection, the needle was slowly retracted (1 mm/min).
One month after the transplantation, the animals were anesthetized using gas inhalation (4.5%-5% isoflurane, 75% N2O, 20% O2) followed by an IP injection of a mixture of ketamine (75 mg/kg) and xylazine (5 mg/kg). Just prior to opening the thorax, the animals received an IP injection of fentanyl (0.005 mg/kg; Janssen-Cilag, Zug, Switzerland). The rats were transcardially perfused with ice-cold 0.1 M PBS (pH 7.4) containing heparin (1,000 I.E./100 ml; NOVO Nordisk, Zürich, Switzerland) followed by 4% PFA in 0.1 M PBS. Immediately thereafter, the brains were removed and placed in the same fixative overnight and then placed in a cryoprotective of 10% sucrose-PBS solution for at least 48 h30,31. The brains were cut on a cryostat in 30-μm-thick sections and mounted on Superfrost™ (Thermo Fisher Scientific) slides30,31.
Immunohistochemistry
Dissociated cell cultures, neurospheres, FVM FFRT sections, and brain sections were washed in PBS and preincubated for 1 h in PBS containing 0.1% Triton X-100 (Sigma-Aldrich Chemie) and 10% horse serum (Thermo Fisher Scientific). After a brief wash in PBS, they were incubated overnight at 4°C with primary antibodies [mouse monoclonal anti-TH, 1:1,000 (Millipore, Zug, Switzerland); rabbit polyclonal anti-TH, 1:1,000 (Pel Freez Biologicals, Rogers, AR, USA); mouse monoclonal anti-5-hydroxytryptamine (5HT), 1:100 (Agilent Technologies, Basel, Switzerland); rabbit polyclonal anti-GABA, 1:5,000 (Sigma-Aldrich Chemie); guinea pig polyclonal anti-serotonin transporter (SERT), 1:2,000 (Thermo Fisher Scientific); mouse monoclonal anti-tryptophan hydroxylase (TrpH), 1:100 (Thermo Fischer Scientific)] diluted in PBS containing 0.1% Triton X-100 and 2.5% horse serum. Subsequent to washes in PBS, the cultures and slides were incubated for 2 h with fluorescently labeled antibodies (Alexa Fluor donkey anti-mouse 488 nm; Alexa Fluor donkey anti-rabbit 594 nm; Alexa Fluor goat anti-mouse 532 nm; Alexa Fluor donkey anti-guinea-pig 488 nm; 1:250; Molecular Probes, Reinach, Switzerland) and a fluorescent dye for detection of the cell nuclei (Hoechst 33352; 1:10,000; Invitrogen). Cultures and sections were then washed in PBS and mounted in 0.1 M PBS containing 50% glycerol.
Histological Analyses
All quantitative analyses were performed by a researcher blinded to the treatment groups. Immunoreactive cells in dissociated cultures were counted in 10% of the glass slide surface in a randomized way using a 10× magnification, as previously described 25 . Only cells with a distinct immunoreactivity and a clear neuronal shape were counted under a microscope (Olympus DP72; Olympus Schweiz AG, Volketswil, Switzerland) equipped with a motorized stage that was connected to a digital camera (Olympus Schweiz AG).
To assess the colocalization of different markers, six randomly chosen visual fields in each culture were taken, and the ones containing both phenotypes [4.8 ± 0.1 fields per culture; mean ± standard error of the mean (SEM); lower 95% confidence interval (CI): 4.3, upper 95% CI: 5.3] were analyzed at a magnification of 40x using the same microscope (culture time experiments: n = 12-14, three independent experiments; plating density experiments: n = 6-17, three independent experiments; BDNF treatment experiments: n = 8-12, two independent experiments; rat midbrain neurospheres: n = 2 embryos; human FVM FFRT cultures: n = 4 embryos; human midbrain neurospheres: n = 4 embryos; FVM FFRT transplants: n = 4).
Statistical Evaluation
Statistical analysis of cell densities was performed using a commercially available software package (Graph Pad Prism 6, La Jolla, CA, USA). To compare group means of several groups, one-way analysis of variance (ANOVA) followed by Bonferroni's multiple comparison test was used. Statistical significance of two groups only was assessed by two-tailed unpaired t-test, and the statistical significance level was set at p ≤ 0.05. Data are presented as mean ± SEM.
Results
Expression of Tyrosine Hydroxylase and Serotonin in Organotypic E14 Rat Ventral Mesencephalic Cultures and in Rat Midbrain Neurospheres
We first explored the presence of serotonergic neurons in rat organotypic FVM FFRT cultures that are typically employed as graft tissue in animal models of cell transplantation for PD26,31. Notably, we have observed that a subpopulation of 5HT immunoreactive (-ir) neurons colocalized with the dopaminergic marker TH in cultures grown for 8 days (Fig. 1A). Likewise, we detected 5HT+ cells colocalizing with TH in neurons sprouting from rat FVM neurosphere cultures subjected to differentiation (Fig. 1B).

Coexpression of serotonin and tyrosine hydroxylase in rat ventral mesencephalic organotypic cultures and in rat neurospheres. Representative microphotographs showing double immunofluorescence stainings for the serotonergic marker 5-hydroxytryptamine (5HT; green) and the dopaminergic marker tyrosine hydroxylase (TH; red) in rat organotypic ventral mesencephalic free-floating roller tube cultures grown for 8 days in vitro (A) and neurospheres cultured for 7 days under expansion (proliferative) conditions and 14 days under differentiation conditions (B). Note the colocalization of 5HT and TH (arrowheads). Scale bars: 200 μm (A), 100 μm (B), 20 μm (insets).
Expression of Tyrosine Hydroxylase, Serotonergic Markers, and GABA in Dissociated E14 Rat Ventral Mesencephalic Cultures
These findings prompted us for a detailed characterization of the phenotypic nature of these TH-ir neurons. For this purpose, we assessed the expression of neuronal markers in dissociated E14 rat FVM cultures. In line with the results obtained for the organotypic FVM FFRT cultures, we observed that a subpopulation of TH-ir neurons also expressed 5HT. In contrast, no coexpression of GABA in TH-ir neurons was detected (Fig. 2). Notably, the finding that all 5HT-ir cells also expressed the rate-limiting enzyme TrpH as well as the SERT confirmed the serotonergic classification of these neurons. Consistent with the results described above, a subpopulation of the TrpH and SERT+ neurons also expressed TH (Fig. 3).
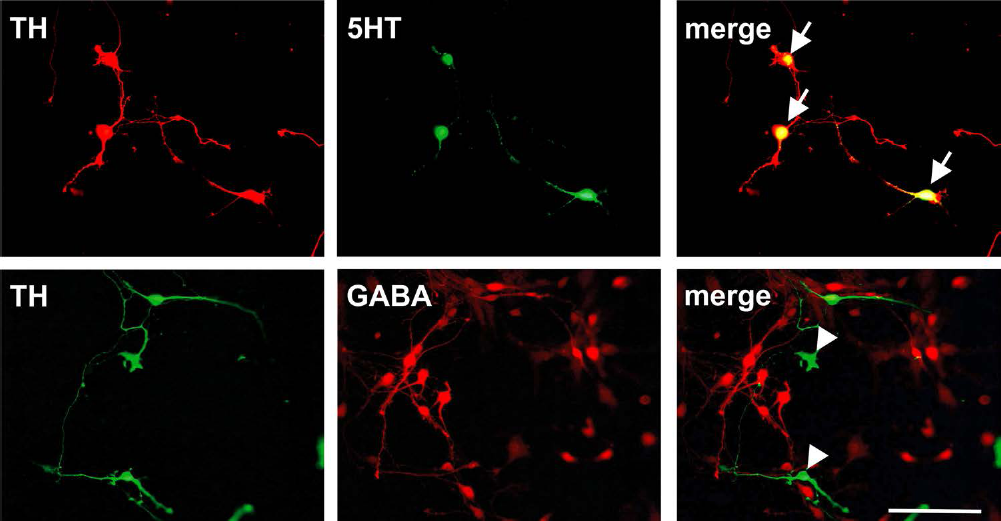
Coexpression of tyrosine hydroxylase and serotonin in rat ventral mesencephalic dissociated cultures and lack of colocalization with γ-aminobutyric acid (GABA). Representative microphotographs illustrating double immunofluorescence stainings for the dopaminergic marker [tyrosine hydroxylase (TH); red upper row and green lower row] and the serotonin [5-hydroxytryptamine (5HT); green) or the GABAergic marker (GABA; red) in rat dissociated ventral mesencephalic cultures grown for 8 days in vitro. Note the colocalization of TH and 5HT (arrows) but not with GABA (arrowheads). Scale bar: 100 mm.

Coexpression of different serotonergic markers with tyrosine hydroxylase in rat ventral mesencephalic dissociated cultures. Representative microphotographs showing double immunofluorescence stainings for the serotonergic marker 5-hydroxytryptamine (5HT; red) and tryptophan hydroxylase (TrpH; green; row A) and TrpH (green) with the dopaminergic marker tyrosine hydroxylase (TH; red; row C) as well as double immunofluorescence stainings for 5HT (red) and the serotonin transporter (SERT; green; row B) and SERT (green) with TH (red; row D). Stainings were performed in rat dissociated ventral mesencephalic cultures grown for 8 days in vitro. Note that all TrpH and SERT+ cells also expressed 5HT, and some colocalized with TH (arrowheads) but not all (arrow). Scale bar: 100 μm.
Effects of Culture Time on the Colocalization Rate of TH+ and 5HT+ Neurons in Dissociated E14 Rat Ventral Mesencephalic Cultures
We initially examined the TH and 5HT expression profile in our dissociated FVM cultures depending on the time in culture. The density of the whole TH-ir cell population was found to steadily decline from DIV2 to DIV8 (15.7 ± 1.0, 13.5 ± 0.7, and 10.8 ± 1.0 cells/mm2 for DIV2, DIV5, and DIV8, respectively) (Fig. 4A), resulting in a significant reduction in TH-ir cell density at DIV8 (p < 0.0001 vs. DIV2) (Fig. 4A). In contrast, the number of 5HT-ir neurons displayed a transient but significant increase at DIV5 (5.5 ± 0.4, 8.1 ± 0.6, and 4.9 ± 0.4 cells/mm2 for DIV2, DIV5, and DIV8, respectively; p = 0.03 for DIV5 vs. DIV8) (Fig. 4A). When we analyzed cell densities of the subpopulation of the TH-ir neurons not expressing 5HT, we detected a significant drop already at DIV5 (13.2 ± 1.0, 8.6 ± 0.5, and 8.3 ± 0.8 cells/mm2 for DIV2, DIV5, and DIV8, respectively; p = 0.0002 and p < 0.0001 for DIV5 and DIV8 vs. DIV2) (Fig. 4B). Interestingly, the density of 5HT-ir cells negative for TH did not vary throughout the culture period (3.0 ± 0.5, 3.2 ± 0.5, and 2.4 ± 0.4 cells/mm2 for DIV2, DIV5, and DIV8, respectively) (Fig. 4B).
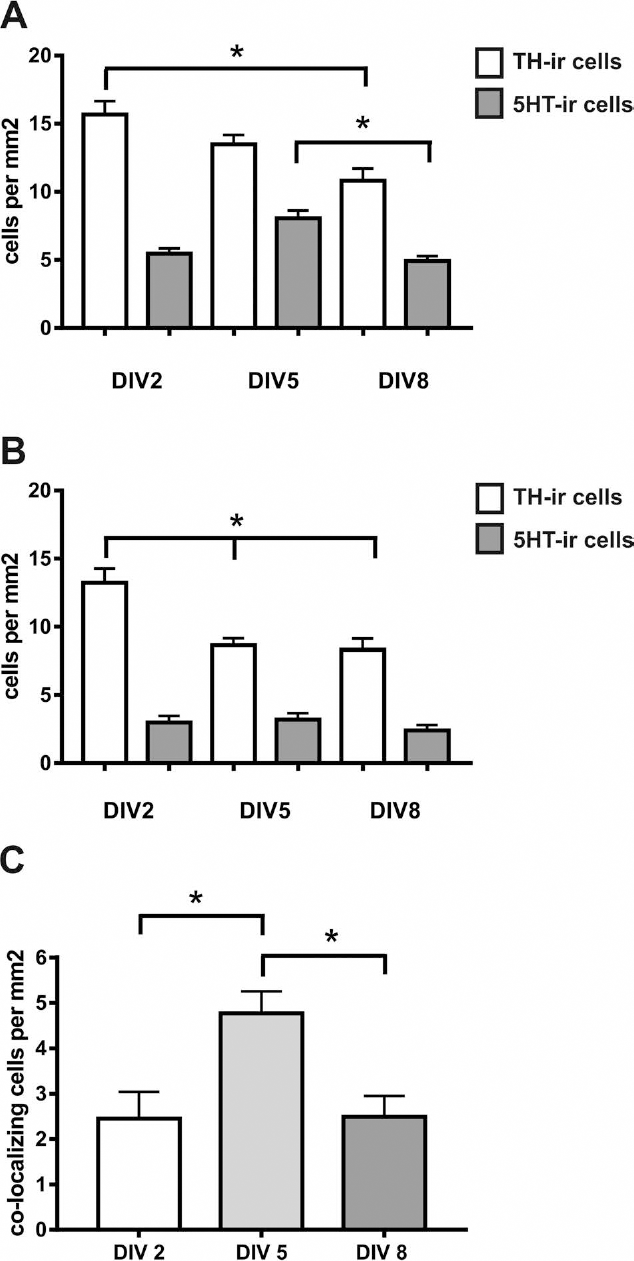
Quantitative analysis of the effect of time in culture on the cell densities of tyrosine hydroxylase and serotonin+ neurons. The cell densities of tyrosine hydroxylase immunoreactive (TH-ir) and 5-hydroxytryptamine immunoreactive (5HT-ir) neurons (A), of TH-ir and 5HT-ir neurons without coexpression of the other marker (B), and of colocalizing neurons (C) were measured at day in vitro (DIV) 2, DIV5, and DIV8 in rat ventral mesencephalic dissociated cultures with plating densities of 0.5 embryos per well. Note the decline of TH-ir cells over time as well as the peak of colocalizing cells at DIV5. Data are expressed as cells/mm2 and given as mean + standard error of the mean (SEM); n = 12-14 from three independent experiments. *p <0.05.
As a consequence of these observations, the densities of cells expressing both TH and 5HT almost doubled from DIV2 to DIV5 (2.5 ± 0.6 and 4.8 ± 0.5 cells/mm2, respectively; p = 0.01 vs. DIV2) and returned to the initial level at DIV8 (2.5 ± 0.5 cells/mm2; p = 0.009 vs. DIV5) (Fig. 4C). When we analyzed the percentage of all TH-ir cells that coexpress 5HT, we detected a significant increase at DIV5 and DIV8 compared to DIV2 (12.9 ± 2.5%, 36.5 ± 3.9%, and 30.3 ± 5.8% at DIV2, DIV5, and DIV8, respectively; p = 0.003 and p = 0.03 for DIV5 and DIV8 vs. DIV2) (Fig. 5A). A similar trend for a higher percentage at DIV5 compared to DIV2 was observed for the 5HT-ir cells coexpressing TH, but the differences did not reach statistical significance (47.4 ± 10.4%, 63.8 ± 6.1%, and 47.6 ± 8.2% at DIV2, DIV5, and DIV8, respectively) (Fig. 5B).

Quantitative analysis of the effects of time in culture on the colocalization rate of tyrosine hydroxylase and serotonin. The colocalization rate of tyrosine hydroxylase-positive (TH+) cells also expressing 5-hydroxytryptamine (5HT) (A) and of 5HT+ cells also expressing TH (B) is presented over the period of 2, 5, and 8 days in vitro (DIV) in rat ventral mesencephalic dissociated cultures at 0.5 embryos per well plating densities. Note the increase in TH+ cells colocalizing with 5HT at DIV5 and DIV8 (A), whereas the colocalization rate of 5HT+ cells also expressing TH remains consistently high (B). Data are expressed as percentage of TH+ or 5HT+ cells, respectively, and given as mean + standard error of the mean (SEM); n = 12-14 from three independent experiments. *p < 0.05 versus DIV2.
Effects of Plating Density on the Colocalization Rate of TH+ Neurons with 5HT and 5HT+ Neurons with TH in Dissociated E14 Rat Ventral Mesencephalic Cultures
We next reasoned whether the plating density might influence the TH-ir and 5HT-ir colocalization rates. Accordingly, dissociated FVM cell preparations were plated at densities corresponding to 0.25, 0.5, 0.8, and 1.25 embryos per well (e) and analyzed at DIV8. In this set of experiments, the percentage of TH-ir cells coexpressing 5HT progressively declined with increasing plating density (53.3 ± 9.0%, 35.0 ± 5.7%, 8.8 ± 4.6%, and 0.9 ± 0.9% for 0.25e, 0.5e, 0.8e, and 1.25e, respectively; p = 0.0001 and p = 0.0002 for 0.5e and 0.8e vs. 0.25e, p = 0.002 and p = 0.003 for 0.8e and 1.25e vs. 0.5e) (Fig. 6A). Similarly, the percentage of 5HT-ir neurons coexpressing TH was significantly lower at higher plating densities compared to low cell densities (74.6 ± 8.9%, 52.7 ± 7.3%, 14.0 ± 5.5%, and 4.8 ± 4.8% for 0.25e, 0.5e, 0.8e, and 1.25e, respectively; p < 0.0001 for 0.5e and 0.8e vs. 0.25e; p = 0.0002 and p = 0.0006 for 0.8e and 1.25e vs. 0.5e) (Fig. 6B).

Quantitative analysis of the effects of cell densities on colocalization rate of tyrosine hydroxylase and serotonin. Effects of plating densities on the colocalization rate of tyrosine hydroxylase-positive (TH+) cells coexpressing 5-hydroxytryptamine (5HT) (A) or 5HT+ cells coexpressing TH (B) in rat dissociated ventral mesencephalic cultures grown for 8 days in vitro at 0.25, 0.5, 0.8, and 1.25 embryos (e) per well density. Note the density-dependent decline for both colocalization rates (A, B). Data are expressed as percentage of TH+ or 5HT+ cells, respectively, and given as mean + standard error of the mean (SEM); n = 6-17 from three independent experiments. *p < 0.05 versus 0.25e, #p < 0.05 versus 0.5e.
Effects of BDNF Treatment on Colocalization Rate of TH+ Neurons with 5HT and 5HT+ Neurons with TH in Dissociated E14 Rat Ventral Mesencephalic Cultures
Based on these results, we sought to verify the hypothesis that neurotrophic factors, such as BDNF, might influence the TH and 5HT expression pattern in the dissociated FVM cultures. Overall, in the control cultures, the percentages of TH-ir neurons coexpressing 5HT and of 5HT-ir neurons coexpressing TH were consistent with the results described above (Fig. 5). At DIV2 (Fig. 7A and B) and DIV5 (Fig. 7C and D), FVM cultures grown in the presence of BDNF did not show substantial differences in the colocalization rates compared to controls. In contrast, supplementation of BDNF for a prolonged time period, that is, DIV8, resulted in a significant reduction of TH-ir neurons coexpressing 5HT compared to controls (12.2 ± 3.5% vs. 25.3 ± 4.2%; p = 0.03) (Fig. 7E) and 5HT-ir neurons coexpressing TH (23.1 ± 6.6% vs. 47.2 ± 7.4%; p = 0.03) (Fig. 7F).

Quantitative analysis of the effects of BDNF treatment on colocalization rate of tyrosine hydroxylase and serotonin. Effects of brain-derived neurotrophic factor (BDNF) treatment on the colocalization rate of tyrosine hydroxylase-positive (TH+) cells coexpressing 5-hydroxytryptamine (5HT) (A, C, E) or 5HT+ cells coexpressing TH (B, D, F) in rat dissociated ventral mesencephalic cultures grown for 2 (A, B), 5 (C, D), and 8 (E, F) days in vitro at plating densities of 0.5 embryos per well. Note that BDNF treatment reduced the colocalization rate of both TH+ cells expressing also 5HT and 5HT+ cells coexpressing TH at day in vitro 8. Moreover, fewer TH+ cells coexpress 5HT than 5HT+ neurons express TH. Data are expressed as percentage of TH+ or 5HT+ cells, respectively, and given as mean + standard error of the mean (SEM); n = 8-12 from two independent experiments. *p < 0.05 versus corresponding controls.
Expression of Tyrosine Hydroxylase and Serotonin in Organotypic Human Ventral Mesencephalic Cultures and in Human Midbrain Neurospheres
In order to investigate whether the observed colocalization of 5HT in TH-ir neurons derived from rat fetal tissues is conserved from rats to humans, we performed FVM FFRT and neurosphere cultures of human embryonic origin. Indeed, the immunohistochemical analyses of FVM FFRT cultures and differentiated neurospheres evidenced a distinct coexpression of 5HT as well as TrpH in a subpopulation of TH-ir neurons (Fig. 8A-C).
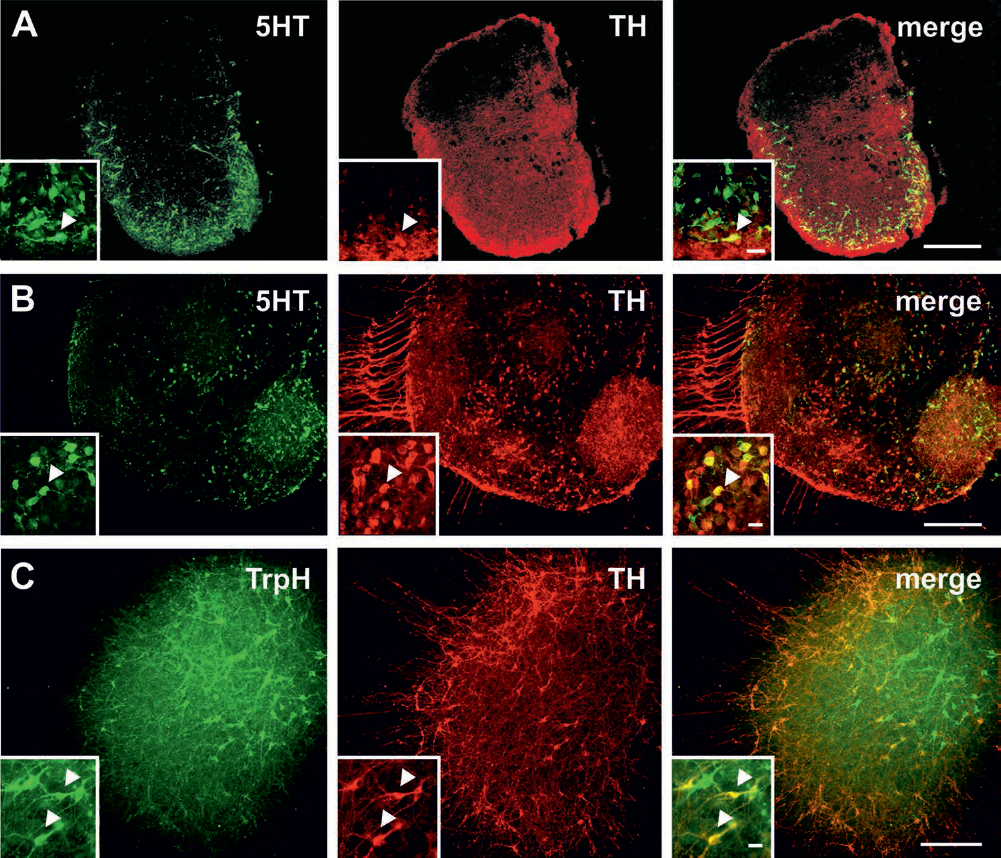
Coexpression of serotonin and tyrosine hydroxylase in human ventral mesencephalic organotypic cultures and in human neurospheres. Representative microphotographs of double immunofluorescence stainings for the serotonergic marker 5-hydroxytryptamine (5HT; green) and the dopaminergic marker tyrosine hydroxylase (TH; red) in human organotypic ventral mesencephalic free-floating roller tube cultures grown for 8 days in vitro (A) and human neurospheres cultured for 7 days under expansion (proliferative) conditions and 14 days under differentiation conditions (B). Note the colocalization of 5HT and TH (arrowheads). Moreover, double immunofluorescence stainings for the serotonergic marker tryptophan hydroxylase (TrpH; green) and the dopaminergic marker TH (red) in human neurosphere cultures demonstrated colocalization of the two markers (arrow head) (C). Scale bars: 200 μm (A, C), 100 μm (B), 20 μm (insets).
Expression of TH+ and 5HT+ Neurons in Transplanted Rat Organotypic Ventral Mesencephalic Grafts
Finally, we assessed for the presence of 5HT-ir neurons in intrastriatal FVM tissue grafts in the 6-OHDA animal model of PD. While the immunohistochemical analyses of the transplants revealed the presence of numerous TH-ir neurons as well as 5HT-ir neurons, no coexpression of 5HT was detected in any of the TH+ neurons in the grafts 1 month after transplantation (Fig. 9).

No colocalization of serotonin and tyrosine hydroxylase in grafts of rat organotypic ventral mesencephalic tissue. Representative microphotographs of double immunofluorescence staining for the serotonergic marker 5-hydroxytryptamine (5HT; green, arrows) and the dopaminergic marker tyrosine hydroxylase (TH; red, arrowheads) in rat ventral mesencephalic free-floating roller tube grafts transplanted into the striatum of 6-hydroxydopamine (6-OHDA)-lesioned rats. The immunohistochemical analysis was conducted 1 month after the transplantation. Note that none of the TH+ neurons coexpresses 5HT. Scale bar: 50 μm.
Discussion
Improving our knowledge about the tissue employed for transplantation in PD is of critical importance for the success of this intervention. Despite the advances in neurobiology, a deep characterization of FVM tissue is still missing. The present work provides important insights into the content of FVM cultures of rat and human origin by demonstrating that dopaminergic neurons coexpress the specific serotonergic markers 5HT, TrpH, and SERT but fail to display colocalization with GABA. Furthermore, we observed that the colocalization of TH and 5HT is influenced by culture conditions such as the plating density, the time in culture, and the treatment with BDNF.
The well-established notion that FVM tissue consists of a mosaic of different cell populations including serotonergic cells has significant relevance in transplantation medicine. This is because the presence of serotonergic cells in the graft has been associated with drawbacks of FVM transplantation, such as graft-induced dyskinesias20-22. Moreover, it has been described that the relationship of the densities between serotonergic neurons and dopaminergic neurons in the grafts plays an important role for long-term efficacy of FVM transplants20-22. Hence, there are increasing efforts carried out to obtain a defined population of dopaminergic cells such as by phenotypic sorting from dissociated mesencephalic tissue23,24 or through strategies of somatic cell reprogramming 32 . The results of the present study, however, point to the idea that in E14 FVM, the dopaminergic and serotonergic cell populations cannot be univocally distinguished. The dual dopaminergic/serotonergic identity of a pool of TH-ir neurons is supported by the coexpression of TH with TrpH and SERT. The presence of this partially overlapping phenotype and its dynamic changes in culture indicate that at this developmental stage, the cells of FVM tissue display still a high degree of plasticity. These findings thus implicate that the fate of a fraction of the mesencephalic cells could eventually be steered in order to increase the pool of transplantable dopaminergic cells. Our results are in line with the close interconnection of the dopaminergic and serotonergic cell ontogenesis, during which they reside rostral and caudal to the midbrain-hindbrain boundary, respectively. For their development, both cell populations depend closely on the secretion of specific factors to initiate their proper phenotype33,34.
In the present work, we have not addressed whether at the time of the analyses the cells were terminally differentiated or still responsive to fate determinants. However, the effects of time in culture and plating density on the colocalization rate strongly suggest that indeed the phenotype of FVM neurons is still influenced by environmental factors. While the progressive decrease in the number of TH+ neurons in dissociated cultures over time is in line with previous reports35-37, the high rate of dopaminergic cells coexpressing 5HT reaching approximately 30% at DIV5-DIV8 is a novel and noteworthy finding. Currently, the reason of a transient increase in 5HT expression in dopaminergic cells at DIV5 is not known. In contrast to culture time, cell density negatively affected the coexpression rate of TH+ and 5HT+ cells. This observation is likely associated with enhanced survival of TH+ cells with increasing cell density in combination with a steady number of 5HT+ cells. This is in line with previously published reports, demonstrating that higher plating densities result in a less pronounced decline in dopaminergic neurons after 8 days in culture 38 and induce an increase in TH mRNA, TH protein, and dopamine release 39 . Although in the framework of cell transplantation these results suggest that culturing mesencephalic cells at high density prior to grafting would be optimal for a superior yield of dopaminergic neurons, likely these advantages cannot be fully exploited given the difficulties to bring such cultured cells in suspension. Notably, however, we provide further evidence of the benefit of graft pretreatment with neurotrophic factors, which goes beyond an augmented number of TH+ cells and improved graft function in animal models of PD26,40,41. Indeed, the observed significantly reduced number of TH+ cells coexpressing 5HT after BDNF treatment may hint to a possible decrease in serotonergic cell-dependent side effects after grafting. That different growth factors can influence the phenotype of dopaminergic and serotonergic neurons is also highlighted by a study demonstrating that the fate of neural progenitors in FVM aggregates changed toward a dopaminergic instead of a serotonergic phenotype when fibroblast growth factor 4 (FGF-4) was lacking 34 .
The lack of any colocalization of serotonergic and dopaminergic markers in grafts of rat organotypic FVM tissue 1 month after transplantation in the striatum of parkinsonian rats is another important finding of the present work. Given earlier observations that most implanted embryonic dopamine neurons die within 1 to 2 weeks after transplantation7,42-45, one may speculate that a specific vulnerability of the TH/5HT-ir cell subpopulation might account for the loss of colocalization of these markers in the graft. Alternatively, the absence of TH/5HT-ir cells in the graft might be the result of neuronal-type fate decision processes and/or commitment to a specific neuronal fate program elicited by in vivo differentiating cues. In this context, an intriguing aspect of the neuron-type specification involves the neurotransmitter switch, which occurs also in the adult brain as a result of the orchestrated interaction between intrinsic genetic determinants by trophic factors and signals delivered by morphogenetic proteins, neurotransmitters, and local electric activity 46 . Accordingly, it has been reported that serotonergic neurons derived from progenitors in the raphe nucleus might be induced to switch to a cholinergic phenotype by ciliary neurotrophic factor (CNTF) and leukemia inhibitory factor (LIF) 47 . Further studies are needed to evaluate the underlying dynamics of fate determination in the colocalizing cells in the graft over broader time periods after transplantation. Analysis in the first weeks after transplantation might provide relevant information as this is the time period in which most of the grafted cells die and the fibers grow out into the host tissue 44 . Moreover, it would be of high interest to investigate the coexpression of TH and 5HT in grafts of postmortem PD patients, as their host brain environment might have a critical effect, different to the one in the rat brain. In this respect, it is important to note that the present study is based particularly on the analysis of phenotypic traits. A more comprehensive investigation involving additional neuronal parameters would help to classify the identity of the cells in our cultures.
In conclusion, our findings illustrate that a subpopulation of cultured FVM dopaminergic neurons expresses serotonergic markers. Notably, we provide in vitro evidence of the TH and 5HT coexpression independent of the culture system analyzed, that is, being detectable in dissociated cells, explants, and neurospheres undergoing differentiation. Although we have not yet explored how to govern the differentiation programs of these cells, the present work may have an impact on studies investigating the generation of dopaminergic neurons from neuronal precursors/progenitors and highlights the potential of neurotrophic factors on emerging neuronal phenotypes in the developing midbrain.
Footnotes
Acknowledgments
The expert technical assistance from Darja Büschi and Susanne Wälchli is gratefully acknowledged. This research was supported by the Swiss Parkinson Foundation, the HANELA Foundation, and the Swiss National Science Foundation (Nos. 3100A0-112529 and 31003A-135565). All authors declare no conflicts of interest.
