Abstract
Oligodendrocytes play a crucial role in creating the myelin sheath that is an important component in neural transmission. In an animal model of transient cerebral ischemia, application of oligodendrocyte progenitor cells (OPCs) has not yet been reported. In this study, the effects of F3.Olig2 transplantation on memory and cognitive dysfunction were investigated in the aged gerbil in which ischemic stroke was induced. To investigate the possible mechanisms underlying repair, changes in the expression of myelin basic protein (MBP), oligodendrocyte-specific protein (OSP), and brain-derived neurotrophic factor (BDNF) were examined. Experimental ischemic stroke was induced by occlusion of bilateral common carotid arteries in aged gerbils. Gerbils (n = 31 per group) were randomly divided into three groups: (1) vehicle sham group, (2) vehicle ischemia group, and (3) F3.Olig2 ischemia group. After 1, 3, and 7 days of ischemia–reperfusion (I-R), saline or F3.Olig2 cells (1 × 10 6 cells in 100 μl) were injected into the gerbils intravenously. The gerbils were sacrificed 10 days after I-R for identification of grafted F3.Olig2 cells, and 15 and 30 days after I-R for tissue analysis after conducting passive avoidance and novel object recognition test. Injected F3.Olig2 cells and MBP, OSP, and BDNF were detected by specific antibodies using immunohistochemistry and/or Western blots. Memory and cognition were significantly increased in the F3.Olig2 ischemia group compared with the vehicle ischemia group. In the F3.Olig2 ischemia group, the neurons were not protected from ischemic damage; however, MBP, OSP, and BDNF expressions were significantly increased. Our results show that injection of F3.Olig2 cells significantly improved impaired memory and cognition, which might be related to increased MBP expression via increasing OSP and BDNF expression in the aged gerbil hippocampus following transient cerebral ischemia.
Keywords
Introduction
Cerebral ischemia is the second leading cause of death, and the number of stroke patients over the age of 65 is expected to double between 2005 and 2030 (8,11). Cerebral ischemia is caused by the deprivation of oxygen supply, and it leads to degeneration of specific vulnerable neurons in the hippocampus (35,40,48). In addition, the white matter in the brain is known to be vulnerable to ischemic insult as ischemia causes oligodendrocyte death, myelin damage, and axonal dysfunction, which are the major causes of long-term cognitive impairment (17,24,45).
Recently, stem cell-based therapies have been proposed to promote and restore functional recovery following ischemic stroke (5,18,30,41). Many researchers have reported that transplanted neural stem cells (NSCs) or progenitor cells could migrate toward the ischemic lesion and differentiate into various types of neurons and glia (22,32). However, because of a low survival rate of transplanted NSCs (4), genetically modified stem cells have been developed as one of the strategies for improving the therapeutic efficacy of stem cells such as human NSCs overexpressing brain-derived neurotrophic factor (BDNF) and Akt1 (36,37).
Oligodendrocytes are a type of glial cell in the central nervous system (CNS) that play a crucial role in creating the myelin sheath that is an important component in neural transmission (16). F3.Olig2 cells are a type of human NSCs that overexpresses Olig2 and are generated by the retroviral transduction of an immortalized human NSC line called HB1.F3 (2). Olig2-transfected NSCs play an important role in inducing the development of mature oligodendrocytes and the remyelination of oligodendrocytes in the corpus callosum of an animal model in which there is demyelination (15). On the basis of these properties of Olig2 cells, the transfection of Olig2 cells can be an efficient application to stimulate the differentiation of NSCs into functional oligodendrocytes for remyelination.
During brain repair processes after ischemia, oligodendrogenesis in the gray and white matter is required to form myelin sheaths for sprouting axons (54,60). To promote remyelination after ischemia, it could be possible to stimulate proliferation of endogenous oligodendrocyte progenitor cells (OPCs) via transplantation of exogenous OPCs because existing mature or injured oligodendrocytes do not make new myelin sheaths (16). A few studies regarding the application of OPCs to animal models induced by spinal cord injury have been reported (51); to the best of our knowledge, however, no studies regarding the application of OPCs to an animal model of transient cerebral ischemia have been reported yet.
Therefore, in this study, we investigated the effects of intravenous transplantation of F3.Olig2 cells on memory and cognitive function in the hippocampus of the aged gerbil, in which the transplantation of human NSCs was previously studied (28), after transient cerebral ischemia. We also investigated expressions of myelin basic protein (MBP), oligodendrocyte-specific protein (OSP), and BDNF to elucidate the mechanism of F3.Olig2 in cognitive improvement.
Materials and Methods
Cell Line and Cell Culture
The stable clonal human neural stem cell (hNSC) line HB1.F3 was generated by the retroviral transduction of primary fetal hNSCs with an avian v-myc cell cycle regulatory gene as we previously reported (2). The F3.Olig2 line was generated by the retroviral transduction of an immortalized human NSC line called HB1.F3. Briefly, Olig2 cDNA was ligated into multiple cloning sites of the retroviral vector pLHCX (Clontech, Mountain View, CA, USA). The PA317 amphotropic packaging cell line was infected with the recombinant retroviral vector, and the supernatants from the packaging cells were added to the HB1.F3 cells.
Experimental Animals
Aged male Mongolian gerbils (Meriones unguiculatus, 22 to 24 months) weighing 80 to 90 g were supplied by the Experimental Animal Center of Kangwon National University (Chuncheon, South Korea). The procedures for animal handling and care adhered to guidelines that are in compliance with the current international laws and policies [see (27)], and protocols were approved by the Institutional Animal Care and Use Committee (IACUC) at Kangwon National University (approval No. KW-140409-1). All experiments were conducted to minimize the number of animals used and avoid animal suffering.
Induction of Transient Cerebral Ischemia
As previously described (1), in brief, the animals were anesthetized with a mixture of 2.5% isoflurane (Baxter, Deerfield, IL, USA) in 33% oxygen and 67% nitrous oxide. Bilateral common carotid arteries were occluded for 5 min using nontraumatic aneurysm clips (Yasargil FE 723K; Aesculap, Tuttlingen, Germany). Body (rectal) temperature under normothermic (37 ± 0.5°C) conditions was monitored with a rectal temperature probe (TR-100; Fine Science Tools, Foster City, CA, USA) and maintained using a thermometric blanket before, during, and after the surgery until the animals completely recovered from anesthesia. Sham-operated animals were subjected to the same surgical procedures except the occlusion of the bilateral common carotid arteries.
F3.Olig2 Cell Transplantation
The animals (n = 31 per group) were randomly divided into three groups: (1) vehicle sham group, which received sham operation and was treated with saline; (2) vehicle ischemia group, which received ischemia operation and was treated with saline after ischemia–reperfusion (I-R); and (3) F3.Olig2 ischemia group, which received ischemia operation and was treated with dosage (1 × 10 6 ) of F3.Olig2 cells after I-R. One, 3, or 7 days after I-R, saline, or F3.Olig2, cells in 100 μl of saline were transplanted into the assigned gerbils through the jugular vein, and the gerbils were sacrificed 10 (n = 3 per group for identification of grafted F3.Olig2), 15 (n = 14 per group for tissue analysis), and 30 (n = 14 per group for tissue analysis) days after I-R.
Passive Avoidance Test
As previously described (23), short-term memory was evaluated by assessing the latency of the passive avoidance test, which was performed with an apparatus that consisted of two compartments (light and dark) with a grid floor (GEM 392; San Diego Instruments, San Diego, CA, USA). The test was divided into a training session and a test session. In the training session, the door between the two compartments was kept open, and the gerbils were allowed to explore the environments in the two dark compartments for 2 min. Then the lights were turned on in one compartment, and the gerbils were allowed to explore the environments in both the light and dark compartments for 2 min. Then again, the gerbils were given an inescapable foot shock (0.3 mA for 3 s) after entering the dark compartment by closing the door between the two compartments. The test session was performed 20 min after the training session using the same paradigm but without applying the foot shock, and the interval between the start of the test session and the gerbil's entry into the dark compartment was defined as the latency time of the passive avoidance test. The passive avoidance test was performed 1 day before I-R, and 15 and 30 days after I-R in all groups. All behavioral tests were performed during daylight, and investigators who were blinded to the experimental conditions scored the latency during the test session, and when the animals did not enter the dark room within 180 s, the latency was recorded as 180 s.
Novel Object Recognition Test
Cognitive ability was evaluated using a novel object recognition test. As previously described (9,50,53), gerbils were placed in the cage (41 cm × 41 cm × 41 cm) individually for 10 min to acclimate to the testing environment. In the training trial (familiarization phase), the gerbils were presented with a pair of identical objects that had been placed in opposite corners for 5 min. In the test trial (test phase, performed 24 h later), one of the familiar objects was changed for another one (novel object), and the animals were left in the cage for 5 min. The exploration time for the familiar or novel object during the test phase was recorded. Investigators who were blinded to the experimental conditions scored the time spent exploring each object. The exploration of the objects was considered when a gerbil showed any investigative behavior (head orientation or sniffing occurring) or entered an area within 1 cm around the object. The results were represented as a memory discrimination index (MDI). MDI = (N - F)/(N + F), where N is the time spent exploring the new located object, and F is the time spent exploring the familiar located object. Higher MDI is considered to reflect greater memory ability.
Tissue Processing for Histology
The animals were anesthetized with sodium pentobarbital [40 mg/kg, intraperitoneally (IP); JW Pharmaceutical Co., Ltd., Seoul, South Korea] and perfused transcardially with 0.1 M phosphate-buffered saline (PBS; pH 7.4; Sigma-Aldrich, St. Louis, MO, USA) followed by 4% paraformaldehyde (PFA) in 0.1 M phosphate buffer (PB; pH 7.4). The brain, heart, liver, lung, kidney, and spleen were removed and postfixed with the same solution for 6 h. The brain tissues were cryoprotected and serially sectioned on a cryostat (Leica, Wetzlar, Germany) into 30-μm coronal sections, and they were then collected into six-well plates containing PBS. The other organs were cut sagittally, embedded with paraffin, and cut at a 6-μm thickness with a microtome (Leica).
Double Immunofluorescence for Grafted Cells
For the identification of grafted F3.Olig2 cells in the hippocampal parenchyma or the other organs, the tissues 3 days after the last F3.Oli2 treatment (n = 3 in each group) were examined. The sections were incubated in the mixed solution of primary antibody of mouse anti-human nuclei (Cat. No. MAB1281; 1:100; Chemicon International, Temecula, CA, USA) for F3.Olig2 cells and rabbit anti-GLUT-1 (Cat. No. Ab14683; 1:100; Abcam Cambridge, UK) for microvessels overnight at 4°C. The sections were then incubated with the mixed secondary antibodies of fluorescein isothiocyanate (FITC)-conjugated anti-mouse IgG (Cat. No. 715-095-151; 1:300; Jackson Immunoresearch, West Grove, PA, USA) and Alexa Fluor 546-conjugated anti-rabbit IgG (Cat. No. A-11035; 1:300; Thermo Fisher Scientific, Waltham, MA, USA) for 1 h and followed by 4′,6-diamidino-2-phenylindole (DAPI; Cat. No. D9542; 1: 1,000; Sigma-Aldrich) nuclear counterstaining for 1 h. A negative control test was carried out using preimmune serum instead of the primary antibody to establish the specificity of the immunostaining. The negative control resulted in the absence of immunoreactivity in all structures. The stained sections were then examined under an Olympus fluorescence microscope (Olympus Korea Co. Ltd., Seoul, South Korea).
Fluoro-Jade B (F-J B) Histofluorescence Staining
As previously described (10), F-J B (a high-affinity fluorescent marker for the localization of neuronal degeneration) histofluorescence staining was conducted to investigate the effect of grafted F3.Olig2 cells on neuronal damage/death in the hippocampus after ischemic insult. In brief, the sections were first immersed in a solution containing 1% sodium hydroxide (Sigma-Aldrich) in 80% ethanol, transferred to a solution of 0.06% potassium permanganate (Sigma-Aldrich), and then to a 0.0004% F-J B (Histochem, Jefferson, AR, USA) staining solution. The sections were examined using a fluorescent microscope (Olympus Korea Co., Ltd., Seoul, South Korea) with blue (450–490 nm) excitation light and a barrier filter. With this method, neurons that undergo I-R degeneration brightly fluoresce in comparison to the background. Studied tissue sections were selected with 120-μm intervals according to anatomical landmarks corresponding to AP −1.4 to −1.8 mm of the gerbil brain atlas, and cell counts were obtained by averaging the counts from five sections taken from each animal. F-J B-positive cells were taken from the stratum pyramidale in the CA1 region and in the polymorphic layer in the dentate gyrus through an AxioM1 light microscope (Carl Zeiss) equipped with a digital camera (Axiocam; Carl Zeiss) connected to a PC monitor. The mean number of F-J B-positive cells was counted in a 100 × 100-μm square of the stratum pyramidale in the CA1 region and throughout the polymorphic layer in the dentate gyrus. Cell counts were obtained by averaging the total cell numbers from each animal per group.
Immunohistochemistry for MBP, OSP, and BDNF
According to Ahn et al. (1), immunohistochemical staining for MBP, OSP, and BDNF was performed using rabbit anti-MBP (Cat. No. ab40390; 1:1,000; Abcam), rabbit anti-OSP (Cat. No. ab53041; 1:500; Abcam), or rabbit anti-BDNF (Cat. No. ab108319; 1:500; Abcam) as the primary antibodies, and biotinylated goat anti-mouse or goat anti-rabbit IgG (1:200; Vector Laboratories, Burlingame, CA, USA) and streptavidin peroxidase complex (1:200; Vector Laboratories) as the secondary antibodies. A negative control test was carried out using preimmune serum instead of primary antibody to establish the specificity of the immunostaining. The negative control resulted in the absence of immunoreactivity in all structures. Studied tissue sections were selected like the above-mentioned method. OSP-immunoreactive cells were taken from each layer in the CA1 region. The mean number of OSP-immunoreactive cells was counted in 100 × 100-μm squares of each layer. Cell counts were obtained by averaging the total cell numbers from each animal per group.
Double Immunofluorescence for BDNF/GFAP or Iba-1 or NeuN
To examine the cell type containing BDNF immunoreactivity, the sections (after I-R) were processed by double immunofluorescence staining according to our method (1) using rabbit anti-BDNF (Cat. No. ab108319; 1:200; Abcam)/mouse anti-glial fibrillary acidic protein (GFAP) (Cat. No. MAB360; 1:400; Millipore, Darmstadt, Germany) for astrocytes, goat anti-Iba-1 (Cat. No. ab15690; 1:200; Abcam) for microglia/macrophages, or mouse antiNeuN (Cat. No. MAB377; 1:400; Chemicon International) for neurons. The sections were incubated in the mixture of primary antibodies overnight and incubated in a mixture of both Cy3-conjugated goat anti-rabbit IgG (1:200; Jackson ImmunoResearch) and FITC-conjugated goat anti-mouse IgG (1:200; Jackson ImmunoResearch) or rabbit anti-goat IgG (1:200; Jackson ImmunoResearch). A negative control test was carried out using preimmune serum instead of primary antibody to establish the specificity of the immunostaining. The negative control resulted in the absence of immunoreactivity in all structures. The immunoreactions were observed under a confocal microscope (LSM510 META NLO; Carl Zeiss), and immunoreactive structures were determined using ZEN 2009 software (Carl Zeiss).
Western Blot Analysis for MBP, OSP, and BDNF
To examine the influence of grafted F3.Olig2 on MBP, OSP, and BDNF protein levels in the hippocampus 15 or 30 days after transient cerebral ischemia, the animals (n = 7 in each group) were used for Western blot analysis according to our method (1). Rabbit anti-MBP (Cat. No. ab40390; 1:1,000; Abcam), rabbit anti-OSP (Cat. No. ab53041; 1:1,000; Abcam), rabbit anti-BDNF (Cat. No. ab108319; 1:1,000; Abcam), and mouse anti-β-actin (Cat. No. A5441; 1:5,000; Sigma-Aldrich) were used as primary antibodies. The results of Western blot analysis were scanned, and densitometric analysis for the quantification of the bands was done using ImageJ 1.46 [National Institutes of Health (NIH), Bethesda, MD, USA], which was used to count relative optical density (ROD). A ratio of the ROD was calibrated as a percentage, with sham group designated as 100%.
Statistical Analysis
The data shown here represent the means ± standard error of the mean (SEM). The normality test was performed using the Kolmogorov–Smirnov test for testing normal distributions and the Bartlett test for testing identical standard distributions. All data passed normality tests. Differences of the means among the groups were statistically analyzed by analysis of variance (ANOVA) with a post hoc Bonferroni's multiple comparison test in order to elucidate F3.Olig2-related differences among experimental groups. Statistical analysis was performed using GraphPad Instat (Instat Statistics, GraphPad Software, La Jolla, CA, USA). Statistical significance was considered at p < 0.05.
Results
Passive Avoidance Test
In the vehicle sham group, the latency was maintained until 30 days after sham operation (Fig. 1A). One day after I-R, the latency was significantly decreased (p < 0.05) in the vehicle and F3.Olig2 ischemia groups compared with that in the vehicle sham group (Fig. 1A). Fifteen days after I-R, the latency was similar to day 1 after I-R in the vehicle and F3.Olig2 ischemia groups (Fig. 1A). Thirty days after I-R, the decreased latency was maintained in the vehicle ischemia group; however, at this point in time after I-R, the latency in the F3.Olig2 ischemia group was significantly increased (p < 0.05) compared with that observed in the vehicle ischemia group. The latency of the vehicle ischemia group was comparable to that seen in the vehicle sham group (Fig. 1A).

Effects of F3.Olig2 cell transplantation on passive avoidance test (A) and novel object recognition test (B). The latency and memory discrimination index in the F3.Olig2 ischemia group is significantly increased 30 days after I-R compared with that in the vehicle ischemia group (n = 7 per group; ∗p < 0.05, significantly different from the vehicle sham group; †p < 0.05, significantly different from the vehicle ischemia group at 30 days postischemia).
Novel Object Recognition Test
The total exploration time in the vehicle sham group, vehicle ischemia group, and F3.Olig2 ischemia group, at each point in time, was not significantly different (data not shown). One day before I-R, the gerbils showed a clear discrimination in the novel object recognition test, namely, the animals spent more time with the novel object than with the familiar one in all of the groups. In the vehicle sham group, the MDI was maintained until 30 days (Fig. 1B). On day 1 and day 15, after I-R, the ischemia-induced gerbils did not discriminate between the novel and familiar object, and MDI was significantly decreased (p < 0.05) in the vehicle and F3.Olig2 ischemia groups compared with that in the vehicle sham group (Fig. 1B). Thirty days after I-R, MDI was significantly increased (p < 0.05) in the F3.Olig2 ischemia group compared with that in the vehicle sham group (Fig. 1B).
Migration of F3.Olig2
Human nuclei-positive F3.Olig2 cells were hardly observed in the brain parenchyma (Fig. 2A–D), lung, kidney, and spleen (data not shown); however, many human nuclei-positive cells were detected in the liver (Fig. 2E–G).
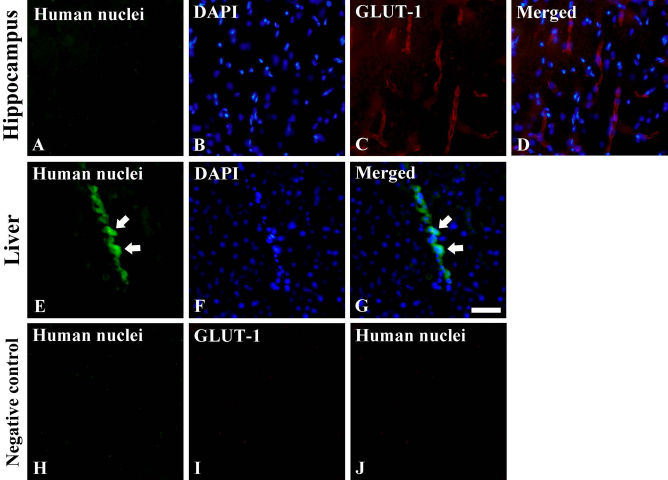
Immunofluorescence staining for the detection of transplanted F3.Olig2 cells in the hippocampus (A–D) and liver (E–G) 10 days after ischemia. Double immunofluorescence staining for human nuclei (green, A)/GLUT-1 (red, C) and DAPI nuclear counter-staining (blue, B) and merged images (D) in the hippocampus revealed that human nuclei-positive F3.Olig2 cells were barely detected in the hippocampus, even in the microvessels (A–D); however, many human nuclei-positive cells are found in the liver (E, white arrows). Negative control results in the absence of immunoreactivity in the hippocampus (H and I) and liver (J). Scale bar: 50 μm.
Effect on Neuronal Damage
In the vehicle sham groups, F-J B-positive cells were not detected in any region of the hippocampus (Fig. 3A1–A3).

F-J B histofluorescence staining in the hippocampus in the vehicle sham (A1–A3), vehicle ischemia (A4–A9), and F3.Olig2 ischemia (A10–A15) groups 15 and 30 days after I-R. In the vehicle ischemia and F3.Olig2 ischemia groups, many F-J B-positive cells (arrows) are detected in the stratum pyramidale (SP) of the CA1 region, and a few F-J B-positive cells were detected in the polymorphic layer (PoL) of the dentate gyrus (DG). GCL, granule cell layer; MoL, molecular layer; SO, stratum oriens; SR, stratum radiatum. Scale bars: 400 μm (left column), 40 μm (middle column), and 100 μm (right column). (B) The mean numbers of F-J B-positive cells in the hippocampal subregions 15 and 30 days after I-R (n = 7 per group). The bars indicate the means ± SEM.
In the vehicle ischemia groups, many F-J B-positive cells were easily detected in the stratum pyramidale of the CA1 region 15 and 30 days after I-R, and a few F-J B-positive cells were found in the polymorphic layer of the dentate gyrus 15 and 30 days after I-R; there was no difference in the mean number of the two groups (Fig. 3A4–A9 and B).
Similar to the vehicle ischemia groups, F-J B-positive cells were detected in the stratum pyramidale of the CA1 region and in the polymorphic layer of the dentate gyrus in the F3.Olig2 ischemia group (Fig. 3A10–A15 and B).
Effect on MBP Expression
MBP Immunoreactivity
In the vehicle sham groups, MBP-immunoreactive fibers were easily found in all the layers; MBP immunoreactivity, in particular, was strong in the stratum lacunosum moleculare of the CA1 region and in the polymorphic layer of the dentate gyrus (Fig 4A1–A4).
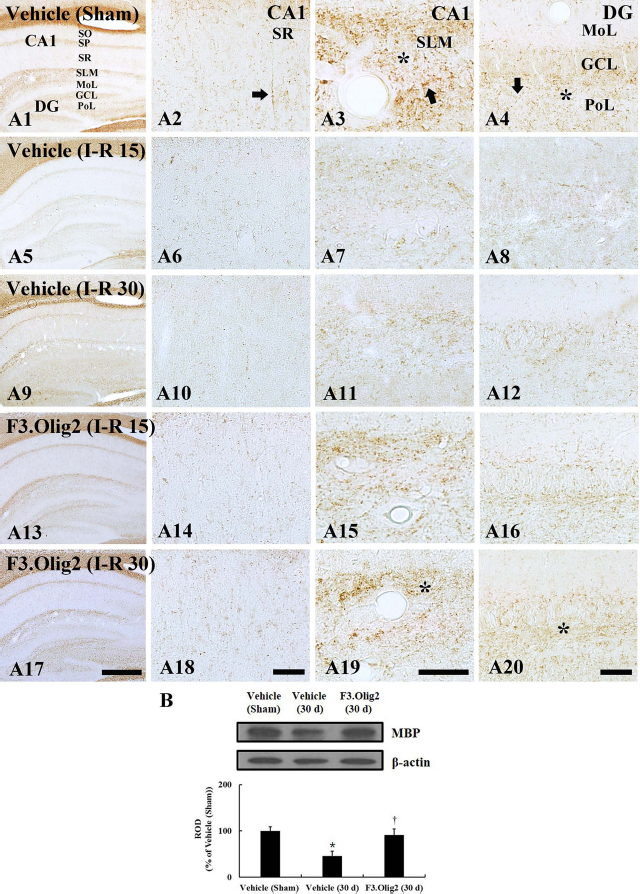
MBP immunohistochemistry in the hippocampus of the vehicle sham (A1–A4), vehicle ischemia (A5–A12), and F3.Olig2 ischemia (A13–A20) groups 15 and 30 days after I-R. In the vehicle sham group, MBP-immunoreactive fibers (arrows) show high MBP immunoreactivity in the stratum lacunosum moleculare (SLM, asterisk) and polymorphic layer (PoL, asterisk). In the vehicle ischemia groups, MBP immunoreactivity is markedly decreased; however, in the F3.Olig2 ischemia group, MBP immunoreactivity is increased, especially in the SLM and PoL at 30 days postischemia. GCL, granule cell layer; MoL, molecular layer; SO, stratum oriens; SP, stratum pyramidale; SR, stratum radiatum. Scale bars: 400 μm (left column), 40 μm (right three columns). (B) Western blot analysis of MBP in the hippocampus 30 days after I-R. Relative optical density is represented as percentage of immunoblot band (n = 7 per group; ∗p < 0.05, significantly different from the vehicle sham group; †p < 0.05, significantly different from the vehicle ischemia group). The bars indicate the means ± SEM.
In the vehicle ischemia groups, MBP immunoreactivity was significantly decreased in all the layers 15 days after I-R (Fig. 4A5–A8). At 30 days postischemia, the distribution pattern of MBP-immunoreactive fibers was similar to that at 15 days postischemia (Fig. 4A9–A12).
Last, in the F3.Olig2 ischemia group, MBP immunoreactivity was apparently increased in all the layers compared with that in the vehicle ischemia groups; however, the MBP immunoreactivity was significantly lower than that in the vehicle sham group. The MBP immunoreactivity at 30 days postischemia was higher than that at 15 days postischemia (Fig. 4A13–A20).
MBP Levels
From the Western blot analysis, we found that the pattern of changes in MBP levels in the hippocampus of all the groups was similar to that observed in the immunohistochemical data (Fig. 4B). MBP levels in the vehicle ischemia groups were significantly lower (p < 0.05) compared with that in the vehicle sham group; there was no difference between the two groups at 15 and 30 days postischemia. In the F3.Olig2 ischemia group, MBP levels were significantly increased (p < 0.05) compared with those in the vehicle ischemia groups; the protein levels on day 30 postischemia were significantly higher than those on day 15 postischemia.
Effect on OSP Expression
OSP Immunoreactivity
In the vehicle sham groups, weak OSP immunoreactivity, which was present in many dot-like processes, was distributed throughout all the layers; in particular, OSP immunoreactivity was high in the stratum lacunosum moleculare of the CA1 region and in the polymorphic layer of the dentate gyrus (Fig. 5A1–A4).

OSP immunohistochemistry in the hippocampus of the vehicle sham (A1–A4), vehicle ischemia (A5–A12), and F3.Olig2 ischemia (A13–A20) groups 15 and 30 days after I-R. In the vehicle sham group, OSP-immunoreactive processes are abundant in the stratum lacunosum moleculare (SLM) and polymorphic layer (PoL). In the vehicle ischemia groups, OSP immunoreactivity is markedly decreased. In the F3.Olig2 ischemia group, OSP immunoreactivity is increased compared with the vehicle ischemia groups; especially, OSP-immunoreactive cells are newly shown at 15 days, not 30 days, postischemia. GCL, granule cell layer; MoL, molecular layer; SO, stratum oriens; SP, stratum pyramidale; SR, stratum radiatum. Scale bars: 400 μm (left column), 40 μm (right three columns). (B) The mean numbers of OSP-immunoreactive cells in the CA1 region 15 days after I-R (n = 7 per group; ∗p < 0.05, significantly different from the vehicle sham group). The bars indicate the means ± SEM. (C) Western blot analysis of OSP in the hippocampus 15 days after I-R. Relative optical density as percentage of immunoblot band is represented (n = 7 per group; ∗p < 0.05, significantly different from the vehicle sham group; †p < 0.05, significantly different from the vehicle ischemia group). The bars indicate the means ± SEM.
On day 15 postischemia, OSP-immunoreactive processes were decreased in the vehicle ischemia group compared with the vehicle sham group in the CA1 region; however, OSP immunoreactivity was increased in the neuropil of the strata pyramidale and lacunosum moleculare. In particular, large OSP-immunoreactive cells were shown in all the layers of the CA1 region and showed weak OSP immunoreactivity (Fig. 5A5–A7 and B). At this point in time, any OSP-immunoreactive cells were not found in the dentate gyrus, although OSP immunoreactivity was increased compared with that of the vehicle sham group (Fig. 5A8). At 30 days postischemia, no OSP-immunoreactive cells were found in the CA1 region and dentate gyrus; however, many OSP-immunoreactive processes were found compared with those at 15 days postischemia (Fig. 5A9–A12).
On day 15 postischemia, the distribution pattern of OSP-immunoreactive structures of the F3.Olig2 ischemia group was similar to that in the vehicle ischemia group; however, numbers of OSP-immunoreactive cells were significantly increased, and their OSP immunoreactivity was strong compared with the vehicle ischemia group (Fig. 5A13–A16 and B). At 30 days postischemia, the distribution of OSP immunoreactivity was similar to that in the vehicle ischemia group; however, OSP immunoreactivity was apparently higher (Fig. 5A17–A20).
OSP Levels
The change pattern of OSP levels in the hippocampus of all the groups was similar to that observed in the immunohistochemical data (Fig. 5C). OSP levels in the vehicle ischemia groups were significantly higher (p < 0.05) than those in the vehicle sham group; there was no difference between the two groups at 15 and 30 days postischemia. In the F3.Olig2 ischemia group, OSP levels were significantly increased (p < 0.05) compared with those in the vehicle ischemia groups; the protein levels on day 15 postischemia were significantly higher than those on day 30 postischemia.
Effect on BDNF Expression
BDNF Immunoreactivity
On day 15 after I-R, weak BDNF immunoreactivity was detected in neurons of the stratum pyramidale of the CA1 region (Fig. 6A1) and in neurons of the granule cell and polymorphic layers of the dentate gyrus in the vehicle sham group (Fig. 6A2). On day 30 after I-R, the distribution patterns of BDNF immunoreactivity was similar to that on day 15 after I-R (Fig. 6A3 and A4).
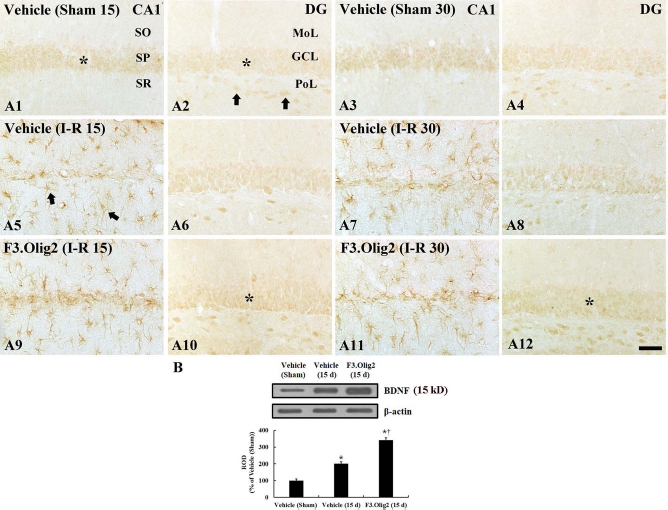
BDNF immunohistochemistry in the hippocampus of the vehicle sham (A1–A4), vehicle ischemia (A5–A8), and F3.Olig2 ischemia (A9–A12) groups 15 and 30 days after I-R. BDNF immunoreactivity is easily shown in neurons (asterisks and arrows) in the vehicle sham group. In the vehicle ischemia groups, BDNF immunoreactivity is not found in the stratum pyramidale (SP); BDNF immunoreactivity is newly expressed in glia-like cells. In the F3.Olig2 ischemia group, the BDNF immunoreactivity is increased in the granule cell layer (GCL, asterisks) compared with the vehicle ischemia group. MoL, molecular layer; PoL, polymorphic layer; SO, stratum oriens; SR, stratum radiatum. Scale bar: 100 μm. (B) Western blot analysis of BDNF in the hippocampus 15 days after I-R. Relative optical density is represented as percentage of immunoblot band (n = 7 per group; ∗p < 0.05, significantly different from the vehicle sham group; †p < 0.05, significantly different from the vehicle ischemia group). The bars indicate the means ± SEM.
On day 15, after I-R in the vehicle ischemia groups, BDNF immunoreactivity was not found in the stratum pyramidale of the CA1 region; however, BDNF immunoreactivity was newly expressed in the nonpyramidal cells in the CA1 region (Fig. 6A5). The BDNF-immunoreactive cells were identified as GFAP-immunoreactive astrocytes (Fig. 7), not Iba-1-immunoreactive microglia/macrophages (data not shown), using double immunofluorescence staining. In the dentate gyrus, the distribution pattern of BDNF immunoreactivity was similar to that in the vehicle sham group (Fig. 6A6): in this group, the BDNF-immunoreactive cells were identified as NeuN-immunoreactive neurons using double immunofluorescence staining (Fig. 7). On day 30, postischemia, the distribution pattern of BDNF immunoreactivity was similar to that on day 15, postischemia (Fig. 6A7 and A8).

Double immunofluorescence staining for BDNF (green, A)/GFAP (red, B) and BDNF (green, D)/NeuN (red, E) and merged images (C, F) in the CA1 region (A–C) and the dentate gyrus (DG; D–F) of the vehicle ischemia group 15 days after ischemia. BDNF-immunoreactive cells in the CA1 region are colocalized within GFAP-immunoreactive astrocytes (arrows), not microglia, and BDNF-immunoreactive cells in the dentate gyrus are localized with NeuN-immunoreactive neurons (arrows). GCL, granule cell layer; MoL, molecular layer; PoL, polymorphic layer; SO, stratum oriens; SR, stratum radiatum. Scale bar: 40 μm.
On day 15 and day 30 postischemia, the distribution pattern of BDNF-immunoreactive structures in the hippocampi in the F3.Olig2 ischemia group was similar to that in the vehicle ischemia groups; however, BDNF immunoreactivity in the dentate gyrus was stronger than that in the vehicle ischemia groups (Fig. 6A9–A12).
BDNF Levels
From the Western blot analysis, we observed that the pattern of changes in BDNF protein levels in the hippocampus of all the groups was similar to that observed in the immunohistochemical data (Fig. 6B). BDNF protein level in the F3.Olig2 ischemia group was significantly increased (p < 0.05) compared with that in the vehicle sham group and vehicle ischemia group (Fig. 6B).
Discussion
The purpose of the present study was to investigate whether the transplantation of F3.Olig2 cells improved memory and cognitive deficits, induced by transient cerebral ischemia, in the aged gerbil. Memory decline is an important problem because many neurological diseases, such as cerebrovascular disease, Alzheimer's disease, and Parkinson's disease, show profound cognitive impairments as well as sensory motor dysfunction (43). It has been known that short-term memory deficits resulted from the loss of CA1 neurons after transient cerebral ischemia would not be reversible (31). In this study, we report for the first time that the intravenous transplantation of F3.Olig2 cells after transient cerebral ischemia produced a significant restoration of short-term memory and cognitive function 30 days after ischemia in the aged gerbil. A previous study reported that the intravenous delivery of mesenchymal stem cells (MSCs) after global cerebral ischemia in the rat elicited the improvement of spatial memory function within 9 days (61). In addition, Yamasaki et al. reported that the transplantation of NSCs in the brain of a mouse model of neuronal loss promoted place memory function within 3 months (58). These and our present findings indicate that transplanted stem cells can help improve a decreased short-term memory induced by neuronal injury.
Many researchers have reported that various types of intravenously transplanted stem cells migrate to ischemic brain regions and differentiate into glia or neurons in animal models (13,29,57). However, in this study we injected F3.Olig2 cells via the jugular vein three times after I-R and did not find any F3.Olig2 cells in the parenchyma of the hippocampus 10 days after I-R, but they were detected in the liver. Recently, it has been reported that the intravenous injection of stem cells has some limitations. A majority of administered stem cells are localized in internal organs, such as the lung, liver, and kidney, and showed an overall 94% signal loss within 1 week (20,47,49). Furthermore, in animal models of focal ischemic stroke, a very small amount of injected stem cells (under 4%) are detected in the injured brains (12,14,39). Previously, Borlongan et al. revealed that intravenously injected human umbilical cord blood cells were not detected in the rat brain parenchyma 3 days after a stroke, even when the cells were cotreated with a blood–brain barrier permeabilizer (mannitol) (7). In addition, it was reported that bone marrow- (21) and adipose-derived MSCs (46) were not observed in the brain after focal ischemia and subcortical ischemic stroke, respectively. On the basis of these results as well as our present finding, it is likely that F3.Olig2 cells could not enter the brain parenchyma induced by transient global cerebral ischemia. To the best of our knowledge, the delivery of stem cells injected intravenously into the injured brain regions in animal models of cerebral ischemia is dependent on the permeability of microvessels for stem cells (59).
Despite the fact that F3.Olig2 cells did not enter hippocampal parenchyma, in our present study short-term memory and cognitive function were obviously improved by the intravenous delivery of F3.Olig2 cells. In this case, it is probable that memory recovery might be correlated with a trophic support (e.g., paracrine effect) to the damaged hippocampus, namely, the treated F3.Olig2 might provide some factors or increase endogenous levels of trophic factors for improving short-term memory and cognitive function after transient cerebral ischemia in the aged gerbil. Therefore, we investigated the effects of F3.Olig2 on neuronal survival, myelination, and neurotrophic factor expression in the ischemic gerbil hippocampus in order to elucidate mechanisms of the transplanted F3.Olig2 cells in increasing memory function after ischemia.
In this study, we found that, in the vehicle ischemia group, pyramidal and polymorphic neurons were dead in the hippocampal formation after 5 min of transient cerebral ischemia. We treated F3.Olig2 cells after I-R and found no neuroprotection in the hippocampus. This finding indicates that intravenously injected F3.Olig2 cells do not influence neuroprotection against transient cerebral ischemic damage.
In addition, F3.Olig2 cell injection in the ischemia group significantly increased expressions of OSP and BDNF on day 15 postischemia. Oligodendrocytes are myelin-forming cells in the CNS, and one oligodendrocyte is able to myelinate multiple axons (52). Oligodendrocytes are known to be highly vulnerable to ischemic insults; ischemic damage to oligodendrocytes and their myelin could lead to impairment of axonal function (16). In addition, it was reported that, in response to demyelination following ischemic injury, new oligodendrocytes were differentiated from OPCs to form myelin sheaths for sprouting axons during the repair process (38) because existing mature or injured oligodendrocytes no longer make new myelin sheaths (54,60).
In our present study, MBP-positive myelinated fibers were significantly decreased in the hippocampus of the vehicle ischemia groups; however, when we treated the ischemia group, MBP-positive myelinated fibers were significantly increased, and their immunoreactivity was strong 30 days after I-R. We suggest that the increased MBP-positive fibers were influenced by increased OSP expression. OSP-positive cells were observed in the vehicle ischemia groups on day 15, but not on day 30, postischemia; however, when F3.Olig2 cells were given to the ischemia group, strong OSP-immunoreactive cells and fibers were significantly increased on day 15 postischemia. These results were consistent with previous studies indicating that adipose-derived MSC administration showed significantly increased oligodendrocyte progenitor cells and restored myelinated axons after subcortical stroke (46) and that F3.Olig2 cell transplantation improved myelination in the white matter of the rat spinal cord following contusive injury (25). On the basis of our findings with previous reports, it is suggested that transient cerebral ischemia induces myelin damage in ischemic regions and provokes remyelination or oligodendrogenesis; therefore, F3.Olig2 cell injection could potentially lead to myelin repair.
On the other hand, in this study, when F3.Oig2 cells were given to the ischemia group, BDNF immunoreactivity was significantly increased in astrocytes, not microglia/macrophages, in the CA1 region and in granule and polymorphic cells in the dentate gyrus after I-R. In addition, BDNF levels were significantly increased in the hippocampus. BDNF is well known as a neurotrophic factor that is observed in various areas or cells of the brain (44) and plays an important role in the proliferation and differentiation of oligodendrocytes to be able to express myelin proteins in the brain (19,55) and to promote the recovery of MBP in the compressed spinal cord (26). Recently, it has been reported that grafted stem cells are sources of trophic factors (7); in particular, Kim et al. demonstrated that F3.Olig2 cells secreted growth factors such as growth differentiation factor (GDF) and platelet-derived growth factor (PDGF) AA and upregulated the BDNF gene (34). Some researchers reported that the intravenous administration of MSCs increased brain BDNF expression after traumatic brain injury in the rat (33,42) and neurotrophic factors in the focal ischemic brain of the rat (56). In addition, it was reported that endogenous BDNF in the hippocampus was required for short-term memory (3) and that neural stem cells improved hippocampal synaptic density and restored cognitive function via BDNF in a transgenic model of Alzheimer's disease (6). On the basis of these reports, our findings indicate that F3.Olig2 cells promote remyelination following oligodendrogenesis in damaged brain areas by increasing endogenous BDNF, which is closely associated with the restoration of neuronal networks in the ischemic hippocampus. However, further studies are needed to understand the related molecular mechanisms associated with F3.Olig2 cellmediated memory recovery after stroke.
In conclusion, the intravenous injection of F3.Olig2 cells did not enter the aged hippocampal parenchyma induced by transient ischemia; however, the injected F3.Olig2 cells significantly improved short-term memory and cognitive function via increasing MBP, OSP, and BDNF expression, which may be related to myelin repair following transient cerebral ischemia. Our findings suggest that even if stem cells do not enter the brain or survive there, as long as the transplanted cells are in the circulatory system they may be sufficient enough to benefit stroke patients for rehabilitation.
Footnotes
Acknowledgments
The authors would like to thank Mr. Seung Uk Lee for his technical help. This research was supported by the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2010-0010580), by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2014R1A2A2A01005307), and by a Priority Research Centers Program grant (NRF-2009-0093812) through the National Research Foundation of Korea funded by the Ministry of Science, ICT and Future Planning. The authors declare no conflicts of interest.
