Abstract
Several in vivo studies have found that transplanting mesenchymal stem cells (MSCs) into degenerative intervertebral discs (IVDs) leads to regeneration of disc cells. Since the exact underlying mechanisms are not understood, we investigated the mechanisms of action of MSCs in regeneration of degenerative IVDs via paracrine actions. Human MSCs and degenerative disc cells from the same donor vertebrae were directly or indirectly cocultured. The multidifferentiation potential, cell proliferation, collagen synthesis, and mRNA expression levels were assessed. The proliferation rates of MSCs and degenerative disc cells were higher in the coculture system than in the monolayer cultures or in the conditioned medium of each cell type. During coculturing with nucleus pulposus (NP) cells, mRNA expression of the extracellular matrix (ECM) components aggrecan, versican (VCAN), SOX9, and type II and type VI collagen was significantly increased in MSCs, whereas mRNA expression for type V collagen was increased in MSCs cocultured with annulus fibrosus (AF) cells. In addition, the accumulation of total ECM collagen was greater in cocultured degenerative disc cells than in monocultured cells. During coculturing, MSCs downregulated the expression levels of various proinflammatory cytokine genes in degenerative NP [interleukin-1α (IL-1α), IL-1β, IL-6, and tumor necrosis factor-α (TNF-α)] and AF cells (IL-1α and IL-6), which are involved in the degradation of ECM molecules. In association with the trophic effect of MSCs on degenerative disc cells, upregulation of growth factor mRNA expression was shown in MSCs cocultured with degenerative NP cells [epidermal growth factor (EGF), insulin-like growth factor-1 (IGF-1), osteogenic protein-1 (OP-1), growth and differentiation factor-7 (GDF-7), and transforming growth factor-β (TGF-β)] or degenerative AF cells (IGF-1, OP-1, and GDF-7). In terms of MSC-based clinical approaches to IVD regeneration, implanting MSCs into a degenerative IVD may both stimulate MSC differentiation into an NP- or AF-like phenotype and stimulate the biological activation of degenerative disc cells for self-repair.
Keywords
Introduction
Degeneration of the intervertebral disc (IVD) is a leading cause of morbidity, resulting in substantial pain, disabling spinal symptoms, and increased healthcare costs 1 . Lower back pain is a common symptom that can present as a debilitating health problem, and it is therefore an economically important disorder. Several studies have identified the loss of the IVD extracellular matrix (ECM) as a consequence of disc degeneration to be a major cause of lower back pain 2 .
The IVD is composed of a central, highly hydrated gelatinous nucleus pulposus (NP) surrounded by a more highly organized ring composed predominantly of collagen fibrils called the annulus fibrosus (AF). Within the NP, rounded chondrocyte-like cells are embedded in a disorganized matrix made primarily of type II collagen (COL 2) and aggrecan and supplemented with other proteoglycans (PGs), including versican (VCAN)3,4, and collagens, including type VI collagen (COL 6). Aggrecan is a highly hydrophilic molecule, and the amount of aggrecan in the NP is sufficient for water absorption to generate swelling pressure capable of exerting a significant force even on loaded vertebrae, with this force being resisted by the AF. Vertebral stability depends, in part, on the balance of these two opposing forces.
In a healthy IVD, there is a balance between anabolic and catabolic processes through regulation by mechanical loading, cytokines, enzymes, and endogenous cells5–7. Changes in IVD morphology as well as biochemical changes have been observed 8 during the process of IVD degeneration, which is believed to be caused by aging, trauma, excessive mechanical loading, and other diseases. These changes include increased cellular senescence and death, degradation of ECM molecules and PGs, increased proteinases and inflammatory cytokines, and the production of an acidic pH9,10.
The degeneration of IVDs is thought to be mediated by the abnormal production of proinflammatory cytokines secreted by NP and AF cells, as well as immune cells11–13. These cytokines can cause pathogenic responses in the IVD cells that promote senescence or death14–16. Secreted proinflammatory cytokines associated with IVD degeneration include tumor necrosis factor-α (TNF-α), interleukin-1α (IL-1α), IL-1β, IL-6, IL-17, and various chemokines5–7,17,18.
A decreased number of NP cells seems to be the cause of disc degeneration, and so transplanting autologous cells might be effective at inhibiting further disc degeneration in vivo 19 . However, typically very few endogenous cells can be obtained from the NP tissues for cell expansion 20 , and it has also been found that the NP harvesting procedure itself may cause degenerative diseases 21 . Because mesenchymal stem cells (MSCs) provide several theoretical and practical advantages, due to their extensive renewal potential and multilineage differentiation potential, they have become excellent cells for use in the grafting procedures of cell therapy and IVD regeneration 22 . Additionally, MSCs are available from many autologous sources and are currently the most popular cells for use in stem cell therapies. However, it is not fully understood how the complicated microenvironment regulates the detailed functions of MSCs for IVD regeneration, and most of the recent studies have focused on the outcomes of MSC transplantation rather than the mechanisms of action.
Several studies have found that cells in the NP or AF region may respond to exogenous growth factors with changes in collagen and PG synthesis, including transforming growth factor-β (TGF-β), insulin-like growth factor-1 (IGF-1), bone morphogenetic protein-2 (BMP-2), and BMP-723–28. These effects may be related to increased cell proliferation or to an intrinsic stimulatory effect on transcriptional regulation of matrix protein biosynthesis; however, it is not fully understood how cells of the NP and AF respond differently to these cytokines and how these changes may be regulated.
The bone marrow (BM) used in MSC-based clinical applications is harvested from the iliac crest (IC) in almost all cases. However, morbidity at the donor site (i.e., the IC) in clinical applications is a serious postoperative concern29–31 since it manifests as chronic pain in the donor. One way of circumventing problems associated with additional surgical morbidity at the donor site is to use MSCs harvested from the recipient site itself or adjacent tissue. The vertebral body (VB) contains BM that can be easily collected and used during an operative procedure such as orthopedic surgery. In addition, previous studies found that the regenerative potential was higher for BM stem cells from the VB than those from the IC, with higher concentrations of colony-forming unit fibroblasts and progenitor cells32–34. If cells of the lumbar VB BM could be used for IVD regeneration, this would obviate problems linked to applying surgical interventions to other healthy tissues, and possibly also donor site morbidity at the IC.
The aim of the current study was to identify the mechanisms of action of MSC-mediated regeneration of the degenerative IVDs, particularly via paracrine mechanisms. Therefore, this study investigated changes in the cellular responses of MSCs and degenerative disc cells (NP or AF cells) obtained from the same donor vertebrae using a coculture system.
Materials and Methods
Disc Cell Source and Culture
Human NP and AF cells were isolated from the degenerative lumbar IVD tissue (disc level L4 or L5) of five patients (mean ± SD: 40.60 ± 9.61 years; one female and four males) for the experiments of this study, and representative results from one subject (a 57-year-old male) are reported herein. The donors voluntarily provided written informed consent for their tissue to be used in accordance with guidelines approved by the Institutional Review Board (IRB) of the College of Dentistry at Yonsei University (IRB No. 2-2012-0048). After obtaining informed consent from the donors and local Ethics Committee approval, degenerative lumbar IVD tissue was obtained during surgery for herniation of the lumbar disc. NP and AF tissues were separated and digested in a solution containing 2 mg/ml collagenase (Serva Electrophoresis, Heidelberg, Germany) and 1 mg/ml dispase (Gibco, Thermo Fisher Scientific, Grand Island, NY, USA) in Dulbecco's modified Eagle's medium (DMEM; Gibco) containing antibiotics. The digested tissue/cell suspension was passed through a 100-μm mesh (Becton Dickinson, San Jose, CA, USA) to remove tissue debris, and cells were then pelleted by centrifugation at 380 × g for 5 min. The supernatant was removed, and the cells were resuspended and cultured to confluence in T25 cell culture flasks (Nunc, Thermo Fisher Scientific) with DMEM/nutrient mixture F-12 (Gibco) supplemented with 10% fetal bovine serum (FBS; Gibco) and 20 μg/ml gentamicin (Gibco) at 37°C in a humid atmosphere containing 5% CO2, with the medium changed every 3 days.
MSC Source and Culture
Fresh human BM was harvested from the VB adjacent to the degenerative IVD tissue in the same subject during orthopedic surgery (Naeun Hospital, Suwon, Korea), using the same protocol as in our previous studies35,36. The patients provided informed consent for their tissue to be used in accordance with guidelines approved by the IRB of the College of Dentistry at Yonsei University (IRB No. 2-2012-0048). The mononuclear cell (MNC) fraction was harvested from BM aspirates to isolate MSCs using conventional density gradient centrifugation with a density gradient solution (Histopaque 1077; Sigma-Aldrich, St. Louis, MO, USA). These cells were cultured in T75 cell culture flasks with α-modification of minimum essential medium (α-MEM; Gibco), supplemented with 10% FBS (Gibco), 20 μg/ml gentamicin (Gibco), 100 μM L-ascorbic acid 2-phosphate (Sigma-Aldrich), and 1% GlutaMAX (Gibco) at 37°C in a humid atmosphere containing 5% CO2. Nonadherent cells were discarded after 7 days, and the adherent cells were cultured to confluence, with the medium changed every 3 days.
Coculturing of MSCs and Disc Cells
Six-well culture plates and inserts (Becton Dickinson) were used to coculture MSCs and disc cells. Culture inserts consisted of a polyethylene terephthalate track-etched membrane with 0.4-μm pores at the bottom to prevent the exchange of cellular components. MSCs were seeded into plates/inserts at 1 × 103/cm2, while NP or AF cells were seeded at 3 × 103/cm2. For coculture wells without contact (indirect), MSCs (or disc cells) were seeded onto the tissue culture plastic, whereas disc cells (or MSCs) were seeded onto the upper surface of the membrane of the tissue culture inserts. For cocultures with cell–cell contact (direct), MSCs (or disc cells) were cultured on the reverse side of the membrane of the insert in culture media, and disc cells (MSCs) were cultured on the outer culture plate. Each cell type was separately precultured in each aforementioned medium for 4 h, and the culture insert was set in the culture plate including the other cells. The outer plate and the insert including two types of cells were maintained at 37°C and 5% CO2 in a humidified atmosphere. Cocultured cells were maintained for 7 days, with the medium changed every 3 days.
Morphology of MSCs and Disc Cells
MSCs and disc cells were expanded continuously at 37°C and 5% CO2 in a humidified atmosphere. Cell morphologies were observed using a light microscope (CKX41; Olympus, Tokyo, Japan).
In Vitro Osteogenic, Adipogenic, and Chondrogenic Differentiation
Cells at passage 5 (P5) were seeded into six-well plates (Nunc, Roskilde, Denmark) at 1 × 105 cells/well and cultured until they reached subconfluence. Osteogenic differentiation was induced by culturing in α-MEM containing 10% FBS, 20 μg/ml gentamicin (Gibco), 2 mM GlutaMAX-1 100X, 100 μM L-ascorbic acid (Sigma-Aldrich), 0.01 μM dexamethasone (Sigma-Aldrich), and 1.8 mM potassium phosphate monobasic (Sigma-Aldrich). After 2–3 weeks of induction, the cells were fixed in phosphate-buffered saline (PBS; Gibco) containing 4% paraformaldehyde (PFA; BBC Biochemical, Mt. Vernon, WA, USA) solution, and then stained with Alizarin red stain (Sigma-Aldrich). The culture medium for adipogenic differentiation comprised α-MEM containing 10% FBS, 20 μg/ml gentamicin, 2 mM GlutaMAX-1 100X, 100 μM L-ascorbic acid, 0.5 μM hydrocortisone (Sigma-Aldrich), 10 μg/ml insulin (Sigma-Aldrich), 60 μM indomethacin (Sigma-Aldrich), and 0.5 mM methylisobutylxanthine (Sigma-Aldrich). After 2–3 weeks of induction, the cells were fixed in PBS containing 10% formaldehyde solution, and then stained with Oil red O stain (Sigma-Aldrich).
Chondrogenic differentiation was performed with 4 × 105 cells (at P5 in the pellet culture in 15-ml conical tubes; SPL Life Sciences, Pocheon, Korea) and centrifuged at 530 × g for 5 min. After 2–3 days of incubation with chondrogenic media, the cells had coalesced and formed free-floating cell pellets. The chondrogenic differentiation medium used contained α-MEM supplemented with 20 μg/ml gentamicin, 1% ITS (Becton Dickinson), 0.1 μM dexamethasone (Sigma-Aldrich), 0.172 mM L-ascorbic acid, 40 μg/ml proline (Sigma-Aldrich), and 10 ng/ml TGF-β (R&D Systems, Minneapolis, MN, USA). After 3 weeks of chondrogenic induction, the pellets were fixed in PBS containing 4% formaldehyde solution, embedded in paraffin (Sigma-Aldrich), and then sectioned at a thickness of 5 μm. The sections were immunostained with Alcian blue (Sigma-Aldrich) to enable the visual assessment of chondrogenic differentiation. The total areas of mineralized nodule formation (Alizarin red stain), lipid vacuoles (Oil red O stain), and sulfated PG deposition (Alcian blue stain) were measured using an automated image analysis system (Image-Pro Plus; Media Cybernetics, Rockville, MD, USA).
Expression of Surface Markers Using Fluorescence-Activated Cell Sorting
The characteristics of the cells were investigated further by analyzing the cell surface markers of isolated cells using flow cytometry analysis (fluorescence-activated cell sorting; FACSCalibur; Becton-Dickinson), the procedure for which has been previously described 37 . Briefly, the cells were then incubated with 3% bovine serum albumin (BSA; Sigma-Aldrich) and antibodies against CD14, CD44, CD45 (1:100; Beckman Coulter, Pasadena, CA, USA), CD34, CD90 (1:100; BD Pharmingen, La Jolla, CA, USA), CD73, and CD105 (1:50; Becton-Dickinson) for 1 h at room temperature. The cells were then washed three times with washing buffer composed of PBS with 2% FBS and observed with a flow cytometer.
Measurement of Proliferation
Cell proliferation was determined using the Cell Counting Kit-8 (CCK-8; Dojindo Molecular Technologies, Rockville, MD, USA) 38 . A 96-well plate (Nunc) containing MSCs or disc cells was inoculated with prepackaged CCK-8 solution. The samples were incubated at 37°C in a humid atmosphere containing 5% CO2 for 2 h, and the absorbance of the supernatant at 450 nm was measured by a spectrophotometric device (VersaMax; Molecular Devices, San Jose, CA, USA). Cell counts were determined using a calibration curve.
In Vitro Expression of ECM Collagen Synthesis Assay
Levels of total ECM collagen in the culture were measured using the Sircol Collagen Assay Kit (Biocolor, Carrickfergus, County Antrim, UK) as described previously 39 . In brief, MSCs and disc cells were cocultured without contact for 5 days, and the cells adherent to the insert were used in the ECM collagen assay. Cell supernatants were collected by centrifugation at 260 × g for 5 min, and a 200-μl aliquot was added to the dye reagent in the assay kit. The solution was incubated for 30 min and then centrifuged at 9,300 × g, with the suspensions and pellets then being separated. The remnant pellets were dissolved with alkali reagent, and the same aliquots were plated and their relative absorbance was measured at 540 nm.
Generation of Cell-Conditioned Medium
To generate cell-conditioned medium (CM), MSCs or disc cells were seeded at 5 × 105 cells/well (six-well culture plates) in 0.1% FBS medium and cultured at 37°C and 5% CO2. After 24 h of cell culturing, 0.1% FBS medium was replaced with serum-free medium (without FBS) and cultured for an additional 48 h. The medium was retained and filtered through a 0.2-μm Minisart filter (Sartorius Stedim Biotech, Goettingen, Germany) to remove cell debris.
Proliferation in CM
Cells were seeded at 2 × 103 cells/well (24-well culture plates; Nunc) in culture medium and then replaced in culture medium with control (serum-free medium) or CM after 24 h of cell culturing. These cells were incubated in each condition for 7 days. The cell proliferation was measured using the CCK-8 assay.
Quantitative Real-Time Polymerase Chain Reaction Analysis of Gene Expression
The characteristics of the cells (MSCs and degenerative disc cells) were investigated on the basis of the expression of ECM, proinflammatory cytokines, and growth factor genes using quantitative real-time poly-merase chain reaction (qRT-PCR). Cells were harvested from the MSCs or disc cells cultivated on the insert using TRIzol (Invitrogen, Thermo Fisher Scientific, Carlsbad, CA, USA), and total cell RNA was prepared. The qRT-PCR analysis was performed for several ECM genes (including aggrecan, VCAN, SOX9, COL 2, COL 6, COL 5, and COL 1) and proinflammatory cytokine genes (including IL-1α, IL-1β, IL-6, and TNF-α) according to the aforementioned procedures. In addition, the expression levels of the genes encoding epidermal growth factor (EGF), IGF-1, osteogenic protein-1 (OP-1; also known as BMP-7), growth and differentiation factor-7 (GDF-7; also known as BMP-12), and TGF-β were measured. The primers used for each gene are listed in Table 1.
Gene Primers Used for qRT-PCR

Statistical Analysis
All of the in vitro experiments were performed in triplicate. Data were analyzed using SPSS statistics software (version 20.0; IBM, Armonk, NY, USA). Unpaired t-tests were used to analyze differences between two groups. Multiple comparisons were performed using repeated-measures analysis of variance followed by Scheffé's test for post hoc analysis. Differences were considered statistically significant at p < 0.05.
Results
Characterization of MSCs and Degenerative Disc Cells
The MSCs were established from MNC fractions obtained by the Ficoll density gradient method, and the degenerative disc cells were obtained from surgical degenerative lumbar IVD tissue. Figure 1A shows that the MSCs and degenerative disc cells displayed similar fibroblastic morphologies. The NP cells appeared rounded, and the AF cells appeared thin and elongated, which is consistent with previous reports 40 . Flow cytometry analysis showed that both MSCs and degenerative disc cells were positive for the presence of CD105, CD90, CD73, and CD44, and negative for CD45, CD34, and CD14 (Fig. 1B). The in vitro differentiation model revealed that both cell types also successfully differentiated into osteogenic, adipogenic, and chondrogenic lineages under specific differentiation stimuli (Fig. 1C), although osteogenic and adipogenic differentiation was stronger for MSCs than for degenerative disc cells. We examined ECM-related gene expression levels, which revealed that aggrecan showed higher expression in degenerative disc cells despite the similar expression levels of VCAN. The expression levels of SOX9, COL 2, and COL 6 were higher in NP cells than in AF cells, whereas the expression of COL 5 was higher in AF cells (Fig. 1D). Both NP and AF cells exhibited the characteristics of stem cells, but they differed from those of MSCs.

Characterization of mesenchymal stem cells (MSCs) and disc cells [nucleus pulposus (NP) and annulus fibrosus (AF) cells]. (A) Morphologic observation of MSCs, NP cells, and AF cells used in this study (magnification: 200×, scale bars: 100 μm). (B) Immunophenotype analysis of MSCs, NP cells, and AF cells by flow cytometry analysis. Cultured MSCs, NP cells, and AF cells were incubated with specific monoclonal antibodies against the cell surface marker antigens including CD105, CD90, CD73, CD44, CD45, CD34, and CD14 followed by florescence-conjugated secondary antibodies. Data are representative of three independent experiments. (C) In vitro multipotency of MSCs, NP cells, and AF cells was examined. Osteogenic, adipogenic, and chondrogenic differentiation was determined by Alizarin red, Oil red O (magnification: 200×, scale bars: 100 μm), and Alcian blue stains, respectively (magnification: 100×, scale bars: 200 μm). All the differentiation groups successfully induced specific differentiation responding to specific biologic stimuli, while all the control groups (data not shown) that were cultured in normal growth media did not induce corresponding differentiation. Data are representative of three independent experiments. (D) Relative basal gene expression of aggrecan, VCAN, SOX9, COL 2, COL 5, and COL 6 in MSCs, NP cells, and AF cells was determined in monolayer cell culture. Gene expression was measured by quantitative real-time polymerase chain reaction (qRT-PCR). All the values are normalized to GAPDH (glyceraldehyde-3-phosphate dehydrogenase), which was used as a housekeeping gene. Each experiment was performed at least in triplicate. *p < 0.05, **p < 0.001.
Cell Proliferation Following Coculture
The average numbers of cells per well in the monocultures (MSCs, NP, or AF control) and indirect cocultures (with NP, AF, or MSCs) were as follows: MSC group: 1.46 × 104 cells in the MSC control, 1.66 × 104 cells when coculturing with NP, and 1.69 × 104 cells when coculturing with AF; NP cell group: 3.89 × 103 cells in the NP control, and 4.32 × 103 cells when coculturing with MSCs; AF cell group: 4.70 × 103 cells in the AF control, and 5.05 × 103 cells when coculturing with MSCs.
The proliferation rates of MSCs and disc cells were significantly enhanced in cocultures (p < 0.05) (Fig. 2). The results from the coculture system revealed that MSCs and degenerative disc cells each had beneficial effects on the proliferation of the other cell type.

Cell proliferation in MSCs, NP cells, or AF cells after 7 days of monoculture (control) or coculture. Cell proliferation was determined using a CCK-8 assay kit. MSCs and degenerative disc cell proliferation were slightly enhanced in coculture. (A) MSC, (B) NP cell, and (C) AF cell group. *p < 0.05.
Gene Expression of ECM Following Direct Coculture
The relative expression levels of ECM genes in MSCs and disc cells are shown in Figure 3. When MSCs were directly cocultured with NP cells, MSCs showed a significant increase in aggrecan, VCAN, SOX9, COL 2, and COL 6 mRNA expression after 7 days of coculturing compared with MSCs cultured alone (MSC control). MSCs cocultured with AF cells showed a significant increase in COL 5 mRNA expression. However, mRNA expression of aggrecan, VCAN, and SOX9 slightly decreased (p < 0.05) (Fig. 3A and B). Aggrecan, SOX9, COL 2, and COL 6 gene expression in NP cells was increased following coculturing with MSCs (p < 0.05) (Fig. 3C). In the AF cells, aggrecan, VCAN, and COL 6 expression levels decreased following coculturing with MSCs (Fig. 3D).
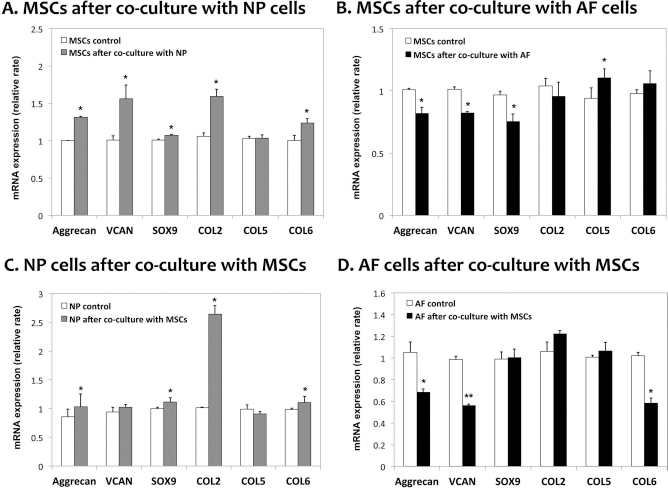
Relative gene expression of extracellular matrix in MSCs, NP cells, or AF cells after 7 days of monoculture (control) or coculture. Gene expression was measured by qRT-PCR. All the values are normalized to GAPDH, which was used as a housekeeping gene. (A) MSCs after coculture with NP cells, (B) MSCs after coculture with AF cells, (C) NP cells after coculture with MSCs, and (D) AF cells after coculture with MSCs. Each experiment was performed at least in triplicate. *p < 0.05, **p < 0.001.
In Vitro Assay of ECM Collagen Synthesis
We examined whether indirect coculturing of MSCs and disc cells affected ECM collagen synthesis. The total amount of ECM collagen in the monoculture (control) or coculture was measured using the Sircol Collagen Assay Kit. More total ECM collagen accumulated in the cocultured disc cells than in the monocultured cells (p < 0.05) (Fig. 4C and D). No significant difference was observed in MSCs cocultured with disc cells (p > 0.05) (Fig. 4A and B).

Insoluble collagen synthesis in MSCs, NP cells, or AF cells after 5 days of monoculture (control) or coculture. Collagen production was determined using a Sircol Assay Kit. (A) MSCs after coculture with NP cells, (B) MSCs after coculture with AF cells, (C) NP cells after coculture with MSCs, and (D) AF cells after coculture with MSCs. *p < 0.05.
Cell Proliferation in CM
To ensure that interaction between MSCs and disc cells resulted in a paracrine effect on cell proliferation, the cells were cultured with MSCs or disc CM. Cell proliferation was measured using the CCK-8 assay. The final cell numbers in NP-CM, AF-CM, or MSC-CM were as follows: MSCs/NP-CM group: 7.3 × 103 cells in controls and 11.41 × 103 cells in NP-CM; MSCs/AF-CM group: 6.5 × 103 cells in controls and 9.2 × cells in AF-CM; NP/MSC-CM group: 2.6 × 103 cells in controls and 3.2 × 103 cells in MSC-CM; AF/MSC-CM group: 2.7 × 103 cells in controls and 3.2 × 103 cells in MSC-CM.
The cells (MSCs or disc cells) cultured in CM showed increased proliferation compared with controls cultured in serum-free medium (p < 0.05) (Fig. 5). These results correspond to those obtained in the cocultures (Fig. 2), which suggests that there is a mutually trophic effect on cell proliferation between MSCs and NP or AF cells. We hypothesize that this trophic effect is able to stimulate MSCs or disc cells for disc regeneration, including enhancement of matrix synthesis, MSC differentiation into an NP- or AF-like phenotype, downregulation of proinflammatory cytokines in disc cells, and upregulation of growth factors in MSCs.

Cell proliferation in MSCs, NP cells, or AF cells after 7 days of MSC, NP, or AF cell-conditioned medium (CM) culture. The cell proliferation was measured by a CCK-8 assay kit. (A) MSCs in NP-CM (MSC/NP-CM group), (B) MSCs in AF-CM (MSC/AF-CM group), (C) NP cells in MSC-CM (NP/MSC-CM group), and (D) AF cells in MSC-CM (AF/MSC-CM group). *p < 0.05.
Proinflammatory Cytokine Gene Expression in Disc Cells Following Coculture
We examined whether the direct coculturing of disc cells and MSCs had an anti-inflammatory effect. The relative expression levels of proinflammatory cytokine genes, including IL-1α, IL-1β, IL-6, and TNF-α, were measured using qRT-PCR. In NP cells directly cocultured with MSCs, IL-1α, IL-1β, and IL-6α mRNA expression levels were significantly decreased, and TNF-α mRNA expression showed the largest decrease (p < 0.05) (Fig. 6A). However, AF cells showed no change in IL-1β and TNF-α mRNA expression levels, while IL-1α and IL-6 mRNA expression levels were slightly decreased (p < 0.05) (Fig. 6B).

Relative gene expression of proinflammatory cytokines in NP or AF cells after 7 days of monoculture (control) or coculture with MSCs. Gene expression was measured by qRT-PCR. All the values are normalized to GAPDH, which was used as a housekeeping gene. (A) NP cells after coculture with MSCs and (B) AF cells after coculture with MSCs. Each experiment was performed at least in triplicate. *p < 0.05.
Growth Factor Gene Expression in MSCs
To confirm the trophic effect of MSCs on disc cells, we measured mRNA expression levels for EGF, IGF-1, OP-1 (BMP-7), GDF-7 (BMP-12), and TGF-β in MSCs after coculturing with NP or AF cells. MSCs were directly cocultured with disc cells, and changes in the expression levels of growth factor genes were measured using the qRT-PCR. MSCs showed significant increases in IGF-1, OP-1, and GDF-7 mRNA expression levels compared with control after coculturing with NP or AF cells. The expression levels of EGF and TGF-β were also increased following coculturing with NP cells (p < 0.05) (Fig. 7).

Relative gene expression of growth factors in MSCs after 7 days of monoculture (control) or coculture with NP or AF cells. Gene expression was measured by qRT-PCR. All the values are normalized to GAPDH, which was used as a housekeeping gene. (A) MSCs after coculture with NP cells and (B) MSCs after coculture with AF cells. Each experiment was performed at least in triplicate. *p < 0.05.
Discussion
Cell-based therapies or tissue engineering for the repair of degenerative IVDs requires a source of viable cells 41 with an NP-like phenotype that can be implanted into the NP of the degenerative disc to produce a functional NP-like matrix. Several studies have found stimulatory effects of human NP cells using coculturing with MSCs. A maintenance function of MSCs in improving the biological and metabolic viabilities of NP cells was identified in the current study. We found that NP cell proliferation, cytokine/growth factor expression, and activation of DNA and PG synthesis were significantly upregulated in a conventional coculture system of MSCs compared with monolayer culturing of NP cells 42 . Previous studies have shown that MSCs can differentiate into cells with an NP-like phenotype43,44, including in experiments transplanting MSCs into IVDs45–49 and coculturing MSCs and IVD cells50–52.
In order to explain the effects on either cell type, several investigators have utilized coculture model systems. We investigated the in vitro interaction between MSCs and degenerative disc cells using an indirect or direct coculture system to verify whether there is active mutual proliferation between MSCs and degenerative disc cells or whether MSCs stimulate degenerative disc cells in terms of enhancing the ECM gene expression. Importantly, we have compared the stimulatory effects between MSCs and NP or AF cells after coculturing. The proliferation rates of MSCs, NP cells, and AF cells were significantly upregulated in the coculture system compared to the monocultures. In addition, these results were confirmed in the culture using each type of CM. This suggests that there is a trophic effect between MSCs and NP or AF cells. Of particular importance is our demonstration that during coculturing with NP cells, the mRNA expression levels of ECM genes in MSCs such as aggrecan, VCAN, SOX9, COL 2, and COL 6 were significantly increased, whereas MSCs exhibited increased mRNA expression of COL 5 in cocultures with AF cells. The aggrecan, VCAN, COL 2, and COL 6 have been reported as NP cell marker genes, and SOX9 is the most commonly analyzed marker of MSC differentiation into chondrocyte-like cells such as NP cells3,53,54. On the other hand, COL 5 was expressed markedly more strongly in AF cells than in NP cells 55 . In the evaluation of the effect of MSCs on degenerative disc cells, the mRNA expression levels of aggrecan, SOX9, COL 2, and COL 6 in NP cells were increased following coculturing with MSCs. In the AF cells, COL 5 expression showed no change following coculturing with MSCs. These results suggest that MSCs acquire an NP- or AF-like phenotype after coculturing and stimulate only NP cells in terms of enhancing the mRNA expression of matrix genes. Thus, implanting MSCs into a degenerative IVD may result in the stimulation of regeneration.
The IVD contains various types of collagen, including COL 2, COL 5, COL 6, and types I, III, IX, XI, XII, and XIV. Collagen is an ECM molecule, as is aggrecan. The continuous structural breakdown of ECM molecules of IVDs results in mechanical instability40,56, which means that the collagen synthesis of NP and AF cells is important for vertebral stability. We used a Sircol collagen assay to assess whether coculturing MSCs and degenerative disc cells affected the synthesis of insoluble collagen. We found that MSCs significantly enhance collagen synthesis of the ECM in NP or AF cells after coculturing with MSCs, whereas MSCs show no change in collagen synthesis of the ECM after coculturing with NP or AF cells. This suggests that during coculturing, MSCs stimulate collagenous ECM regeneration of degenerative disc cells.
Following the initial IVD degeneration, IVD cells upregulate inflammatory cytokines and chemokines, including IL-1α, IL-1β, IL-6, and TNF6,17,18,57. IVD cells express several catabolic molecules in this inflammatory environment, and these enzymes promote the degradation of ECM molecules such as COL 2 and aggrecan, which leads to IVD degeneration and lower back pain 58 . To assess the anti-inflammatory effect during coculturing in degenerative disc cells, we examined the expression levels of the proinflammatory cytokine genes of IL-1α, IL-1β, IL-6, and TNF-α. The expression levels of all four of these genes were significantly downregulated in NP cells, while IL-1α and IL-6 were slightly downregulated in AF cells after coculturing with MSCs. These results show that MSCs inhibit expression of proinflammatory molecules in degenerative disc cells. The decrease in the expression of proinflammatory molecules may inhibit the degradation of ECM molecules. Furthermore, controlling proinflammatory molecules may improve low back pain. However, this effect requires further investigation.
Several studies have shown that MSCs exert a trophic effect on cells in their vicinity by secreting various growth factors and cytokines that have the potential to stimulate the cells 59 . An early study of cytokine-induced changes in disc cell metabolism found changes in cell proliferation and PG synthesis of disc explants when they were cultured in media supplemented with various growth factors. Multiple investigators have contributed to these findings that cells of the NP or AF region may respond to exogenous growth factors with changes in cell proliferation and collagen and PG synthesis, including EGF, IGF-1, TGF-β, and BMP23,60–64. Some of these effects may be related to regeneration of degenerative IVDs. To confirm the trophic effect of MSCs on degenerative disc cells, we measured the mRNA expression levels for EGF, IGF-1, OP-1 (BMP-7), GDF-7 (BMP-12), and TGF-β in MSCs after coculturing with NP or AF cells. Both NP and AF cells significantly increased the mRNA expression levels for IGF-1, OP-1, and GDF-7 in MSCs after coculturing. The expression levels of EGF and TGF-β increased only in NP cells, while that of TGF-β was significantly reduced in MSCs cocultured with AF cells compared to MSCs only. Each of these selective cellular responses might be caused by differences in the environmental effects for NP and AF cells. NP and AF layers have structural differences (e.g., cartilaginous vs. fibrous), and MSCs affected by the cells from these layers might show different response patterns. This increased growth factor gene expression might be the mechanism responsible for the observed increase in cell proliferation, the differentiation of MSCs into an NP- or AF-like phenotype, the decrease in proinflammatory molecule-associated gene expression, and the enhancement of matrix-associated gene expression in NP cells.
Several recent in vivo studies found that MSC implantation into the degenerative IVD leads to regeneration of a disc cell matrix, although the exact mechanisms are not understood38,39,41,65. Our data suggest that implanting MSCs into a degenerative IVD may reduce inflammatory responses and enhance the regenerative capacities of MSCs and NP/AF cells mutually via paracrine effects. In addition, implanted MSCs may lead to enhancement of cell proliferation and collagen synthesis of the ECM and downregulation of proinflammatory cytokine gene expression in degenerative disc cells. Thus, MSCs may be able to stimulate self-repair in degenerative disc cells. Furthermore, our data suggest that these actions in degenerative disc cells during coculturing can be explained by the increase in the growth factor gene expression of MSCs. This further suggests that cocultured MSCs exert trophic effects on degenerative NP or AF cells. In addition, we suggest that different growth factors are involved in the self-repair of degenerative NP or AF cells; therefore, their role in directing MSCs and disc cell behavior in degenerative IVDs requires further investigation.
This study has shown for the first time that MSCs from the VB may be useful in the regeneration of degenerative IVDs via paracrine interaction. The paracrine actions of VB-derived MSCs might enhance the therapeutic efficacy. Additionally, VB-derived MSCs could become a sustainable cell source of MSCs for IVD regeneration in the future, as is currently done for other MSC types, including adipose- and umbilical cord blood-derived MSCs.
Footnotes
Acknowledgments
This research was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Ministry of Science, ICT & Future Planning (Nos. 2012M3A9C6049862 and 2012M 3A9B2052521). The authors declare no conflicts of interest.
