Abstract
Chronic neuropathic pain represents a clinically challenging state with a poor response to current treatment options. Long-term management of chronic pain is often associated with the development of tolerance, addiction, and other side effects, reducing the therapeutic value of treatment. Alternative strategies based on cell therapy and gene manipulation, balancing the inhibitory and excitatory events in the spinal cord, may provide sustained pain relief in the long term. Transplantation of GABAergic cells has been successfully used to enhance inhibition and to restore physiological spinal pain processing. However, since the underlying mechanism of chronic pain development involves changes in several pain-signaling pathways, it is essential to develop an approach that targets several components of pain signaling. Recombinant cell therapy offers the possibility to deliver additional analgesic substances to the restricted area in the nervous system. The current study explores the analgesic potential of genetically modified rat embryonic GABAergic cells releasing a peptidergic NMDA receptor antagonist, Serine1-histogranin (SHG). Overactivation of glutamate NMDA receptors contributes to the hyperexcitability of spinal neurons observed in chronic pain models. Our approach allows us to simultaneously target spinal hyperexcitability and reduced inhibitory processes. Transplantable cells were transduced by viral vectors encoding either one or six copies of SHG cDNAs. The analgesic potential of recombinant cells after their intraspinal transplantation was evaluated in a model of peripheral nerve injury. Enhanced reduction of hypersensitivity to thermal and mechanical stimuli was observed in animals treated by recombinant cells compared to the nonrecombinant group. The recombinant peptide was detected in the spinal tissue, suggesting its successful production by transplanted cells. Our results demonstrate the feasibility of using recombinant cells releasing adjunct analgesic peptides in the therapy of neuropathic pain.
Introduction
Chronic neuropathic pain induced by peripheral nerve injury represents a clinically challenging disease with low efficacy of current treatment options. Long-term pharmacological intervention often leads to the development of tolerance and addiction, further reducing the therapeutic potential of treatment. The insufficient treatment is also caused by the multifactorial characteristic of neuropathic pain, where overlapping mechanisms contribute to the development of chronic pain. Conventional pharmacotherapy has limited options to specifically and locally target altered pain signaling, especially for chronic interventions. Therefore, alternative approaches intended to produce long-term or permanent alterations in pain transmission, such as gene therapy or cell transplantation, may be particularly suitable for patients suffering from chronic, disabling pain. As the dysfunctional signaling in the spinal pain processing sites due to reduced GABAergic inhibition is one of the mechanisms underlying chronic pain development (9,11,20,31,38,52,54), various attempts to restore GABAergic signaling have been experimentally evaluated. Intraspinal transplantation of GABAergic cells, either engineered (12,13,15) or embryonic (35,42), showed a positive impact on reducing pain-like behavior in animal models. Transplanted cells may serve as local minipumps, balancing the level of neurotransmitters in the area of transplant and close vicinity and may also support endogenous cells by releasing neuroprotective agents. However, the analgesic effect of such an approach seems to be limited by the capacity of the transplanted cells to counterbalance pathological changes in spinal pain processing and also by the survival of the transplanted cells. Enhancement may be achieved by engineering of recombinant cells that produce complementary analgesic substances, thus targeting several dysfunctional pain pathways at the same time. Such an approach is supported by pharmacological observations using a multitargeting approach, whereby enhanced analgesia is achieved by a subthreshold amount of drugs in proper combinations (24–26). Simultaneous targeting of different pain pathways by analgesic peptides delivered via gene therapy using viral vectors also yielded promising results for chronic pain management (33,34,43). The aim of this study was to therefore evaluate the potential of a novel approach using gene engineering and generation of recombinant GABAergic cells releasing Serine1-histogranin (SHG) as an additional analgesic peptide. SHG has been previously shown to exhibit an N-methyl-D-aspartate (NMDA) receptor antagonist activity (21,22,25–27,30). As NMDA signaling is another key event in the pathology of neuropathic pain, we evaluated the analgesic abilities of recombinant GABAergic cell-producing SHG as a proof of concept for the cell-based multitargeting approach for the management of chronic pain.
Materials and Methods
Animals
Male Sprague–Dawley (SD) rats were used as a model of peripheral nerve injury by implantation of intrathecal catheters and intraspinal injections (140–160 g at the time of the first surgery); pregnant female SD rats were used for E14 embryo harvesting (Harlan Lab, Indianapolis, IN, USA). Animals were housed two per cage with free access to food and water and a 12-h light/dark cycle. Experimental procedures were reviewed and approved by the University of Miami Institutional Animal Care and Use Committee (IACUC) and followed the recommendations of the Guide for the Care and Use of Laboratory Animals (National Research Council). All surgical procedures were conducted under 2.5% isoflurane/O2 (South-medic, Barrie, Ontario, Canada) anesthesia using aseptic conditions.
Peripheral Neuropathic Pain Model
Chronic constriction injury (CCI) of the sciatic nerve was used to induce peripheral neuropathic pain (4). The common sciatic nerve was exposed on the right side at the midthigh level using aseptic surgical techniques. Four 4-0 chromic gut ligatures spaced about 1 mm apart were loosely tied around the sciatic nerve proximal to the trifurcation. The wound was closed in layers, and animals were left to recover in heated cages (total n = 80; six animals failed to develop pain-like behavior at 1 week post-CCI and were excluded for subsequent experiments; ns for individual experiments and treatment groups appear in the figure legends).
Intrathecal Catheter Implantation
Indwelling catheters (7.5–8 cm; ReCathCo, Allison Park, PA, USA) were inserted through a slit in the atlantooccipital membrane down the intrathecal space and secured to the neck muscles with sutures similar to previously described methods (23,58). Rats were allowed to recover for at least 3 days following intrathecal surgery prior to use in experiments. A 48-h washout period was included between drug dosing. SHG peptide (21st Century Biochemicals, MA, USA) was dissolved in saline and injected in 5-μl volumes in concentrations of 0.3 μg, 1 μg, 3 μg, and 10 μg, followed by a 5-μl flush with saline. The total number of animals used for insertion of the catheter was 30; due to mortality (two rats) and postsurgical complications, such as clogged catheters and partial paralysis (four rats), 24 rats were used for the behavioral evaluation, with 12 rats in the CCI saline group and 12 rats in the CCI transplant (CCI NPC) group.
Engineering of Recombinant Neuronal Progenitor Cells (NPCs)
Recombinant lentiviral and adeno-associated virus (AAV) particles encoding SHG were generated by the Viral Vector Core at the Miami Project. We have previously generated lenti-SHG constructs, which resulted in successful production of SHG peptide in a variety of cell types (18). To increase the antinociceptive potential of the construct in latter studies, we engineered SHG multimers to generate up to six copies of the SHG peptide (34,36). The AAV 2/8 hybrid vector was chosen for engineering of recombinant viral particles based on our screenings of different AAV serotypes as the most efficient serotype for transduction of neuronal cells. This vector has also been shown by other studies to have five- to 100-fold higher efficiency for transgene delivery compared to AAV2 serotypes in cardiac muscle (5), skeletal muscle (56), the brain (7), the lungs (40), and the liver (19).
E14 fetal neocortical tissue (cortical lobes and underlying lateral ganglionic eminences) from SD rats was microdissected in Hank's balanced salt solution (HBSS; Gibco, Waltham, MA, USA) and mechanically triturated to obtain single-cell suspensions. Cells were plated at a concentration of 5 ° 105 cells/ml in Stemline neural stem cell (NSC) medium (Sigma-Aldrich, St. Louis, MO, USA) containing 10 ng/ml of human recombinant basic fibroblast growth factor (FGF-2; Sigma-Aldrich) and incubated for 5–7 days with media changes after 2–3 days. Twenty-four hours prior to transplantation, FGF-2 was withdrawn from the media in order to initiate pre-differentiation into GABAergic progenitors, as we have found this environment to be favorable for GABAergic differentiation (17). For transduction with viral vectors, NPCs were harvested and prepared as detailed above. Twenty-four hours after harvesting, cells were pelleted at 143 ° g for 5 min, and 10 μl of purified virus (1 ° 106 TU/ml for lentivirus, 1 ° 109 TU/ml for AAV) prepared by the Miami Project Viral Vector Core was added to a minimal volume of culture media for 4–5 h at 37°C. After washing the pellet to remove unbound virus, cells were plated with an appropriate amount of culture media.
Immunocytochemical Evaluations
The standard immunocytochemical protocols were used to characterize the phenotype of cultured NPCs. Cells were plated in poly-L-ornithine/fibronectin-coated Lab-Tek (Nunc; Sigma-Aldrich) chambers at a concentration of 5 ° 105/well and incubated at 37°C for 2–3 days. After that, cells were fixed with 4% paraformaldehyde (PFA; Sigma-Aldrich), washed, and incubated in 5% normal goat serum (Gibco) for 2 h. Primary antibodies anti-GABA (1:200; Sigma-Aldrich), anti-SHG (1:100; 21st Century Biochemicals) and anti-monomeric red fluorescent protein (mRFP, 1:200; Abcam, Cambridge, MA, USA) were added for overnight incubation, followed by washes and incubation with appropriate secondary antibodies (anti-mouse and anti-rabbit Alexa Fluor 488 and 597, 1:250; Invitrogen, Waltham, MA, USA). 4’,6-Diamidino-2-phenylindole (DAPI, 1:400; Invitrogen, Waltham, MA, USA) was used for nuclear staining. After a final wash, the upper structure of the chamber was carefully removed, and slides were air dried and coverslipped (Vectashield; Vector Labs, Burlingame, CA, USA). To estimate the transduction rate and to compare differentiation of NPCs into a GABAergic proneuronal phenotype, at least two images of areas with clearly identified single cells per well were labeled with GABA, SHG, and mRFP were captured by confocal microscopy (Spectral Confocal Microscope Fluoview 1000; Olympus, Center Valley, PA, USA) and analyzed by ImageTool software [University of Texas Health Science Center at San Antonio (UTHSCSA), San Antonio, TX, USA] by manual counting based on the overlapping of single-colored images and colocalization of markers. The experiment was designed to have at least two chambers stained with the same combination of antibodies, thus having at least four images per count.
Intraspinal NPC Transplantation
At 1 week post-CCI, animals with clear signs of pain-like behavior (n = 42) received intraspinal injections of either NPCs, recombinant NPCs, or equal volumes of saline solution (Teknova, Hollister, CA, USA). NPC transplantation was done at the lumbar enlargement. Prior to transplantation, cells were pelleted at a concentration of 50,000/μl. Under anesthesia, rats were placed in a stereotaxic holder with spinal clamps (David Kopf Instruments, Tujunga, CA, USA), and a laminectomy was performed to expose the L3–L4 spinal cord level. Intraspinal injection was done at L3–L4 using a pulled glass microneedle (diameter ~50 μm) attached to a Hamilton syringe (Hamilton, Reno, NV, USA). The needle was placed 0.5 mm from the central vein (right side), and 2 μl of cell suspension or saline was injected at 0.5 μl/min with a digital microinjector (Stoelting, Wood Dale, IL, USA) at a depth 0.5 mm from the dorsal lumbar spinal surface. The needle was left in place for another 3 min to prevent backflow. Muscles were sutured in layers, overlying skin was closed with staples, and animals were transferred to heated cages for recovery. All transplanted rats received cyclosporine A (CyA) [intraperitoneal (IP), 10 mg/kg; Bedford Labs, Bedford, OH, USA] daily, starting 1 day prior to the transplantation.
Behavioral Evaluations
Rats were tested weekly for up to 5 weeks post-CCI using a battery of innocuous and noxious stimuli. Tests for tactile and cold allodynia were performed on the same day, followed by thermal and mechanical hyperalgesia tests the day after. For drug evaluation experiments, rats were tested every 30 min up to 120 min following intrathecal injections with 48 h of washout periods between drugs/tests. Behavioral responses were recorded by observers blinded to the experimental treatments.
Tactile Allodynia
The threshold response to an innocuous mechanical stimulus was measured with calibrated von Frey hairs ranging from 0.25 to 15 g. Animals were placed beneath an inverted clear plastic cage on an elevated wire mesh floor, and filaments were applied to the plantar skin of the hindpaw sequentially with increasing or decreasing force based on negative or positive responses, respectively (10). A brisk hindpaw withdrawal together with at least one other pain-related behavior, such as turning the head toward the stimulus, licking or shaking the paw, was considered as a positive response in order to incorporate involvement of nociceptive processing at the supraspinal level (1).
Mechanical Hyperalgesia
Mechanical hyperalgesia was evaluated using the Randall–Selitto method (49). A pressure apparatus (Ugo Basile, Gemonio, Italy) was used to assess the pressure threshold of hindpaws. An increasing force was applied to the hindpaw, and the force inducing paw withdrawal was recorded. The test was repeated three times on each paw with at least a 5-min delay between tests on the same paw. Differences in scores were calculated by subtracting withdrawal thresholds on the intact side from the injured side; thus, negative difference scores are indicative of mechanical hyperalgesia.
Cold Allodynia
Sensitivity to a nonnoxious cooling stimulus was evaluated using acetone (VWR, Radnor, PA, USA). Acetone (100 μl) was dropped onto the plantar hindpaws, and responses were recorded. Acetone was applied five times, with about 1–2 min between applications. Hindpaw lifting following acetone application was usually accompanied by supraspinal responses (head turning and licking of the affected paw or shaking of the paw). The total number of positive lifting responses out of five was converted to a percent response frequency.
Heat Hyperalgesia
The response to a noxious thermal stimulus was evaluated using the Hargreaves test (28). Rats were placed beneath an inverted clear plastic cage on an elevated glass floor, and a radiant heat source beneath the glass was aimed at the plantar hindpaw, which activates a timer. Withdrawal latencies between the activation of the heat source and the hindpaw withdrawal from the glass were recorded. To avoid tissue damage in the absence of a withdrawal, the cutoff latency was set at 20 s. Testing was alternated on both hindpaws for three trials at least 30 s apart, and the average values were used for statistical analyses.
Neurochemical Analysis of NPC Transplants
To evaluate the production of the recombinant peptide, spinal cord samples were dissected from animals at 2 weeks posttransplantation (n = 2 for saline, NPC and 1SHG NPC; n = 3 for 6SHG NPC groups, respectively), at the peak of the behavioral effect. The spinal lumbar region around the transplant site was dissected and frozen on dry ice. Samples were homogenized in RIPA lysis buffer (Santa Cruz Biotechnology, Dallas, TX, USA), and the protein concentration was estimated by a bicinchoninic acid assay (BCA) method (Thermo Fisher Scientific, Waltham, MA, USA). The fluorescence-linked immunosorbent assay (FLISA) method was used to quantify the amount of SHG in spinal cord samples. Sample homogenates (100 μl) were loaded into 96-well plates (Thermo Fisher Scientific, Rochester, NY, USA) in capture buffer according to the manufacturer's protocol (Odyssey FLISA; LI-COR Biosciences, Lincoln, NE, USA) and incubated overnight at 4°C, followed by incubation in the blocking buffer for 3 h at room temperature and anti-SHG primary antibody (1:100; 21st Century Biochemicals) for 18 h at 4°C. Plates were then washed and incubated in secondary antibody (IRDye goat anti-rabbit) for 2 h, washed, and absorbance was measured by the Odyssey Infrared Imager (LI-COR). SHG peptide at different concentrations was used to calculate the standard curve and the relative amount of SHG in samples. Glyceraldehyde-3-phosphate dehydrogenase (GADPH) was used as a loading control. Data were analyzed by Graph Prism polygnomial analysis (GraphPad Software, La Jolla, CA, USA).
Immunohistochemical Analysis of NPC Transplant
To confirm the presence of the recombinant cells in the spinal cord, three animals from the 6SHG NPC group were used for immunohistochemical analysis at 2 weeks posttransplantation. Animals were deeply anesthetized and intracardially perfused with 0.9% saline followed by 4% paraformaldehyde in 0.1 M phosphate buffer. Spinal cords were removed and postfixed for 12 h in the same fixative and transferred to 25% sucrose for cryoprotection. Cryostat sections, either free floating or slide mounted, were prepared and processed according to standard immunohistochemical protocols. Sections were incubated in 5% normal goat serum for 2 h followed by overnight or 48 h incubation with primary antibodies as above. After washing, sections were incubated in the appropriate secondary antibodies, washed, and coverslipped. The primary antibodies used were rabbit anti-GABA (1:200; Sigma-Aldrich), mouse anti-NeuN (1:200; EMD Millipore, Billerica, MA, USA), and rabbit anti-SHG (1:1,000; 21st Century Biochemicals). Secondary antibodies used were goat anti-rabbit and goat anti-mouse Alexa Fluor 488 (1:250; Invitrogen), goat anti-rabbit and goat anti-mouse Alexa Fluor 594 (1:250; Invitrogen), and DAPI (Invitrogen). Images were analyzed by confocal microscopy (Spectral Confocal Microscope Fluoview 1000; Olympus) and Stereoinvestigator (MBF Bioscience, Williston, VT, USA). Five to seven 40-μm sections (at least 160 μm apart) of spinal cords with visible transplanted cells were evaluated. The area with transplanted cells was outlined at low magnification, and mRFP-, SHG-, GABA-, and NeuN-positive cells were manually counted in each slide at 20° magnification. From these, the estimated number of cells per animal labeled by certain markers and colocalization of markers were calculated.
Statistical Analysis
Data are expressed as mean ± SEM. For statistical evaluation of behavior, all animals were included in the NPC groups if at least one GABA or DAPI cell was found in the transplant site within the lumbar dorsal horn, as nondetectable transplants may be the result of misplacement or nonviability (6). Using these criteria, the transplant success rate was approximately 80%. To compare the effect of treatments over time, a two-way ANOVA for repeated measures was done, followed by the Student– Newman–Keuls post hoc test. Relative optical density in FLISA tests were evaluated by t-tests. Statistical analyses were performed using SigmaStat (Systat Software, San Jose, CA, USA). Statistical significance was taken with values of p < 0.05 and p < 0.01.
Results
Intrathecal Injection of SHG in CCI Model
To test the hypothesis that intrathecal (IT) injection of SHG may provide additional analgesic effects in animals with transplanted GABAergic NPCs, SHG (0.3 μg, 1 μg, 3 μg, or 10 μg) or saline was IT injected into CCI animals transplanted with NPCs and to control, nontransplanted CCI animals (Fig. 1). IT injections were administered 2 weeks following transplantation (corresponding to 3 weeks post-CCI). At this time point, animals in both groups had significantly reduced paw withdrawal threshold compared to baseline values (p < 0.01). However, nerve injury-induced tactile allodynia was partially attenuated in animals with NPC transplants compared with control (intraspinal saline) animals (Fig. 1, indicated as higher pre-IT SHG injection paw withdrawal thresholds in CCI NPC vs. CCI saline). The IT injection of the vehicle did not alter paw withdrawal thresholds in either transplant group over the 120-min observation period. However, IT injection of SHG reduced tactile allodynia in both NPC and control groups. Significant overall differences between NPC-transplanted and control groups were observed for all IT treatments, F(1, 8) = 13.3, p = 0.006 for saline; F(1, 8) = 14.9, p = 0.005 for 0.3 μg SHG; F(1, 8) = 1.3, p = 0.036 for 1 μg SHG; F(1, 8) = 11.2, p = 0.01 for 3 μg SHG; F(1, 8) = 13.4, p = 0.006 for 10 μg SHG. In the saline-injected animals, the peak response to SHG was generally observed between 60 and 120 min postinjection. The highest dose (10 μg) elicited a mild analgesic response at 30 min postinjection, with a peak at 60 min (p = 0.01 vs. saline and p = 0.025 vs. 0.3 μg) and remained at this level for 120 min. The other doses showed a slower rise and peaked at different times: 1 μg at 60 min (p < 0.001 vs. saline), 3 μg at 90 min (p < 0.001 vs. saline), 0.3 μg at 120 min (p = 0.003 vs. saline). Overall, increasing doses of SHG in control CCI animals produced only modest antinociceptive effects without statistically significant differences between the analgesic effects of most doses. The effect appeared to plateau in the 1- to 3-μg dose range, with no further beneficial effects observed at higher doses, an effect observed in our previous studies as well (22,53).

IT injection of SHG peptide in CCI rats treated with saline or NPCs. Saline (CCI saline) or GABAergic progenitor cells (CCI NPCs) were intraspinally injected at 1 week post-CCI. SHG was IT injected 2 weeks posttransplantation at various concentrations. Saline was used as the vehicle. *p < 0.01 versus vehicle; n = 6/dose.
In the NPC-transplanted animals, dose-related increases in peak paw withdrawal values were observed, which approached the 15-g maximum cutoff responses in some cases, similar to intact noninjured animals. The peak response was observed at 60 min postinjection with paw withdrawal threshold (PWT) level reflecting the SHG dose, and significant differences were observed between the lowest and the highest dose (10 μg, p < 0.001 vs. saline, p = 0.005 vs. 0.3 μg; 3 μg, p < 0.001 vs. saline; 1 μg, p = 0.006 vs. saline). At 90 min postinjection, significant differences compared to saline were observed for 0.3 μg (p = 0.009), 3 μg (p = 0.03), and 10 μg (p = 0.035). The highest dose of SHG remained at significantly elevated levels from the control at the 120-min point as well (p = 0.026).
Evaluation of Cultured NPCs
The fluorescent signal of the mRFP reporter gene was initially used to confirm transduction of NPCs with the SHG construct. The mRFP signal was detected in the majority of cultured cells. Image analysis estimated a 79.3 ± 9.8% transduction rate of all DAPI-positive objects. To evaluate the expression of SHG in mRFP-positive cells, the colocalization of mRFP signals with the custom-made SHG antibody was determined (Fig. 2A–D). Quantitative evaluation showed that 90 ± 12.3% of mRFP-positive cells colocalized with the SHG signal. An orthogonal view of Figure 2D with fluorescent profiles on the respective axes (inset D’) shows several objects with overlapping signals from blue (DAPI), green (SHG), and red (mRFP) channels, confirming colocalization of these markers. In order to enhance the signals from transduced cells for further analysis, a red secondary fluorescent antibody was used to detect SHG in addition to the red fluorescence emitted by mRFP in the immunostaining for colocalization between SHG and GABA. Analysis showed that 72 ± 13.6% of GABA-positive cells colocalize with SHG, although numerous SHG-positive cells did not show a GABAergic profile (Fig. 2E–H). Figure 2H shows a cluster with nearly all GABA-positive cells containing SHG/mRFP. The presence of both GABA and SHG signals in the same cells was confirmed by confocal analysis. Figure 2H inset shows a fluorescent profile through a cell soma colocalization of GABA and SHG/mRFP. In nontransduced cells (Fig. 2I–L), no mRFP/SHG signal was detected. This is confirmed by orthogonal profile analysis showing signals in the green (GABA) and blue (DAPI) channels but no signaling in the red channel. No differences in the transduction rate between the single and multi-SHG constructs were apparent.

Immunohistochemical identification of SHG produced by transduced cells. Colocalization of immunofluorescent staining for SHG (B) with mRFP signal (C) was observed in most of the transduced cells (D). GABAergic phenotype (F) of SHG-transduced cells (G) was detected in a subpopulation of recombinant cells (H). No SHG/mRFP signal was observed in nontransduced cells (I–L). DAPI (A, E, I) was used for nuclear staining. Orthogonal profiles in (D), (H), and (L) show colocalization of signals for different antibodies in transduced cells.
Effects of Recombinant NPC Transplant on Peripheral Neuropathic Pain
As the in vitro results indicated successful transduction of NPCs and generation of SHG peptide, the antinociceptive properties of transplanted recombinant GABAergic NPCs were evaluated in the CCI model of peripheral nerve injury-induced pain. NPCs were intraspinally injected at 1 week post-CCI injury when pain-related behavior was fully developed. Nontransduced cells served as controls. We chose nontransduced cells over the cells transduced with control viral particles, as the current study was based on previous findings in our laboratory using nontransduced NPCs. However, in a small pilot study, natural nontransduced NPC transplants were compared with NPCs transduced with AAV-green fluorescent protein (GFP). Evaluation of tactile and cold allodynia did not show any significant differences between transduced and nontransduced transplants (data not shown).
As transplanted recombinant NPCs could alter spinal nociceptive processing at multiple targets, a comprehensive evaluation of pain-related behavioral changes, including tactile allodynia and mechanical hyperalgesia, and heat hyperalgesia and cold allodynia, was done. Results showed that transplantation of recombinant cells significantly improved mechanical and heat hyperalgesia and cold allodynia, with modest effects on tactile allodynia (Fig. 3).

The analgesic effects recombinant and nonrecombinant GABAergic NPCs in CCI animals. Animals transplanted with nonrecombinant GABAergic NPCs, recombinant 1SHG and 6SHG GABAergic NPCs, or saline injections at 1 week following CCI. *p < 0.05 versus saline-injected animals; #p < 0.05 versus nonrecombinant GABAergic NPCs. n = 6 saline; n = 6 NPC; n = 10 1SHG; n = 8 6SHG.
The paw withdrawal threshold in the tactile allodynia test was significantly higher in CCI animals with recombinant 1SHG and 6SHG transplants at 2 weeks postinjury (1 week posttransplantation) compared to nonrecombinant transplants (p < 0.001 for 1SHG and 6SHG) and compared to saline controls (p = 0.001 for 1SHG; p = 0.002 for 6SHG). In the following weeks, 6SHG-transplanted animals demonstrated attenuation of tactile allodynia, while the 1SHG-transplanted group was not significantly different from controls, except for 5 weeks post-CCI (4 weeks posttransplantation), when both recombinant transplants reached modest but statistically significant antiallodynia compared to the saline control and the nonrecombinant NPC group [p = 0.001 vs. saline and p = 0.017 vs. NPC; overall F(3, 5) = 13.2] (Fig. 3A). In this analysis, the effects of the nonrecombinant NPC transplants on tactile allodynia did not reach statistical significance compared with saline controls (p > 0.05), similar to our previous observations showing lack of significant effects of NPC transplants alone on tactile allodynia in the CCI model (35).
Mechanical hyperalgesia was partially reduced in animals receiving NPC transplants (Fig. 3B). The addition of 1SHG provided a modestly significant improvement at 1 week posttransplantation [p = 0.01 vs. saline and p = 0.02 vs. nonrecombinant NPC group; overall F(3, 5) = 4.2]. The effect of the enhanced transplant with 6SHG was not significantly different from the 1SHG. At later time points, the responses of all transplant groups were similar but still significantly higher than the saline group up to 5 weeks post-CCI (p < 0.05). Cold allodynia was reduced similarly in all the recombinant and nonrecombinant groups with significantly reduced responses compared with the saline control group [p < 0.05 vs. saline; overall F(3, 5) = 1.8] (Fig. 3C). Effects of the transplants on cold allodynia were robust, reaching nearly preinjury baseline levels (near 0%) by 5 weeks post-CCI in NPC-transplanted animals; thus, further improvements by addition of SHG were likely not detectable (floor effect). The most robust improvement in pain-related behavior was observed for heat hyperalgesia (Fig. 3D). Recombinant SHG cells completely reversed the hyperalgesic responses to noxious heat during the first week posttransplantation [p = 0.002 vs. saline and p < 0.05 vs. NPC; overall F(3, 5) = 6.1]. Nonrecombinant NPCs also improved heat hypersensitivity posttransplantation (p = 0.045 vs. saline). Attenuation of heat hyperalgesia was maintained through the 5-week post-CCI follow-up, with significantly enhanced analgesia in the 6SHG transplant group compared to the control group (p < 0.01 vs. saline at 3 weeks and 4 weeks), with no reemergence of heat hyperalgesia at any time point following transplantation. An antinociceptive effect of 1SHG and nonrecombinant cells was also observed, statistically different from saline controls through 3 weeks post-CCI. However, a partial spontaneous improvement and waning hyperalgesia at 4–5 weeks was observed in control CCI animals, similar to previous findings (35); this spontaneous recovery limits longer term evaluation and comparisons in this model.
Immunohistochemical Analysis and Quantification of the Transplant
Immunohistochemical staining of the spinal cord of CCI rats (n = 2) showed a reduction of GABAergic cells on the ipsilateral side in agreement with the main hypothesis of dysfunctional GABAergic signaling as an underlying mechanism in the development of neuropathic pain (Fig. 4). NPCs were therefore transplanted into the ipsilateral spinal cord. Successful transplants were localized in the dorsal horn of the lumbar spinal cord (Fig. 5). Figure 5A shows an example of a transplant placed unilaterally in a CCI animal. In graft regions, DAPI-positive cells were densely packed and circumscribed compared with the host dorsal horn cellular distribution. Transplanted recombinant cells were identified in the spinal dorsal horn based on the red fluorescent signal of mRFP and/or positive staining for SHG peptide. Some transplanted cells were detected in the deeper dorsal horn areas where they may have migrated. No SHG cells were observed on the contralateral side of the spinal cord. The rostrocaudal extent of the graft was approximately 0.5 mm from the injection site. An example of the estimated transduced cells (mRFP) in the total transplant area is shown in Figure 5B. In the delineated region, ~360 DAPI profiles were counted, with ~40 red profiles; thus, approximately 10% was transduced within this transplant. Immunohistochemical staining showed differentiation of transplanted cells into the GABAergic phenotype by detecting colocalization of SHG/mRFP signal with GABA (Fig. 5D). Quantification of transplanted cell phenotypes in CCI animals revealed about 11.5% of GABA profiles within the transplant area (out of all DAPI objects presented) and 6.3% of SHG/mRFP profiles (average DAPI n = 363.5 ± 85; GABA n = 42.7 ± 8.3; SHG/mRFP n = 23.8 ± 5). Colocalization between GABA and SHG/mRFP signals was detected in about 20% of GABAergic objects. Colocalization of SHG with neuronal marker NeuN (Fig. 5E) was detected in 11.7 ± 5.4% of NeuN cells located in the transplant area.

The reduction of GABA immunofluorescence in the spinal dorsal horn of CCI animals. Neuronal staining (NeuN, green) at the contralateral (A) and ipsilateral (A’) sides shows no difference in the signal, while reduction in GABA signal (GABA, red, B, B’) has been observed. Merged images (C) and (C’) show rectangular areas in (B) and (B’).

Immunofluorescent detection of transplanted cells in the spinal cord at 2 weeks posttransplantation. (A) Transplants (outlined area, asterisk) were localized unilaterally in the dorsal horn within the laminae L II–L I V. (B) For quantification, the area of the transplant was overlaid with grid; DAPI- and mRFP-positive profiles were estimated in each grid square and used to calculate the total of mRFP profiles per treatment group. (C) At 4 weeks posttransplantation, the amount of detected cells was lower but still visible. (D) Colocalization of mRFP signal with GABA immunostaining demonstrated differentiation and survival of transplanted progenitors in the host tissue. (E) Colocalization of neuronal staining NeuN and SHG suggest differentiation of transplanted SHG cells into neuronal phenotype. Orthogonal profiles in (D–E) show localization of mRFP and SHG signals, with red peaks for mRFP shifted next to green peaks for GABA, while overlaying of red and green peaks detected for SHG/NeuN. DAPI was used as nuclear staining.
FLISA Analysis of the Transplant
The presence of SHG in the spinal cord tissue of CCI animals transplanted with recombinant or control cells was evaluated by FLISA (Fig. 6). Since SHG is a synthetic peptide not normally produced in the mammalian spinal cord, its presence is indicative of successful production by engineered transplanted cells. The SHG peptide loaded in different concentrations allowed us to quantify the relative amount of SHG in spinal tissue; GADPH staining was used to normalize the signal among samples. Samples from animals with transplants of 6SHG NPCs, 1SHG NPCs, and NPCs and saline were loaded and compared for the presence of SHG. Quantification of the positive signal showed the highest level of SHG in samples from 6SHG NPC animals compared to 1SHG NPC animals (Fig. 6B). The ratio of the amount of SHG between 6SHG NPC (0.052 ± 0.01 mg/ml) and 1SHG NPC (0.014 ± 0.004 mg/ml) samples (1:3.7) reflects the differences in the type of plasmid used for cell transfection. Further analysis of samples from the 6SHG NPC group from the ipsilateral and the contralateral sides showed the presence of SHG mainly on the ipsilateral side of the transplant (Fig. 6C). Quantification showed significant differences between the amount of SHG detected on the contralateral and ipsilateral sides, with 0.018 ± 0.008 mg/ml and 0.035 ± 0.01 mg/ml SHG, respectively (p < 0.05; t-test) (Fig. 6D). Since no SHG or mRFP cells were found immunocytochemically on the contralateral side when grafts were placed unilaterally, the contralateral presence of SHG in limited, but detectable, amounts may be due to peptide diffusion or neuronal projections to the contralateral dorsal horn.
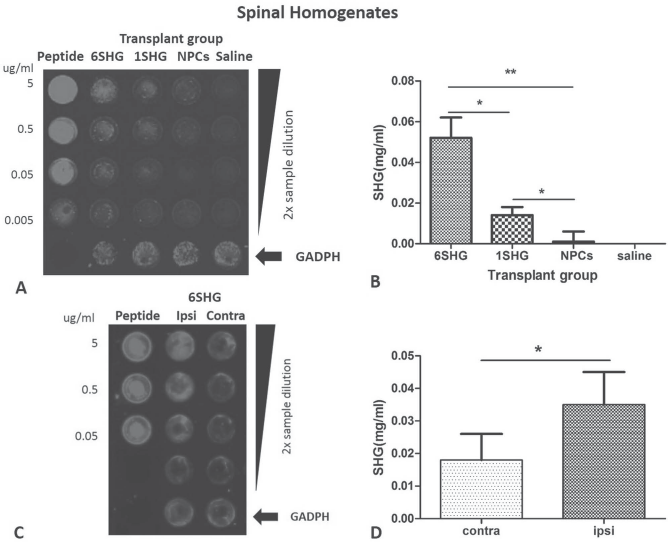
FLISA analysis of SHG in the spinal cord tissue homogenates. (A) Enhanced SHG signal in the samples from 6SHG NPC transplants compared to 1SHG NPCs. Background staining in the NPC group and negative signal in saline confirm specificity of the SHG antibody. (B) Estimated amount of SHG in the samples from different groups. (C) Differences in the SHG signal between contralateral and ipsilateral sides demonstrate the production of SHG in the transplanted (ipsilateral) area. (D) Estimated amount of SHG detected in the tissue. Differences were detected between the ipsilateral and contralateral sides. Authentic SHG peptide at known concentration was used as a standard; GADPH was used to normalize data. *p < 0.05; **p < 0.01.
Discussion
Our study demonstrated an enhanced analgesic effect of transplanted recombinant GABAergic NPCs that produced additional analgesic peptides with NMDA antagonist activity in a model of peripheral nerve injury-induced pain. Our results support the hypothesis that dysfunctional GABAergic and glutamatergic signaling contribute to the development of chronic pain and that the simultaneous targeting of multiple pain pathways may provide a better analgesic outcome compared to the single-target approach.
Pain associated with injury to the peripheral or central nervous system is characterized by the hyperexcitability of spinal neurons. Although the exact mechanisms are not fully known, numerous studies point to the disruption of GABA signaling as one of the key events in the development of chronic pain. Reduced inhibitory control of neurons in the spinal pain processing site leads to overactivation of neurons in response to afferent signals. Those are subsequently transmitted to higher nerve centers and miscoded as high threshold or pain signals, resulting in hyperalgesia and allodynia as physiological outcomes. The exact reason for reduced GABAergic signaling is still under debate. Originally, proposed mechanisms of GABAergic cell death have been misleading, as no significant reduction of spinal dorsal horn neurons have been observed in animals despite their pain-related behavior (45–47). However, numerous studies demonstrated a reduction in GABA signaling using histological, biochemical, and electrophysiological methods. In addition, experimental treatment focused on increasing the level of GABA in the spinal cord or on enhancement of GABA signaling and showed beneficial outcomes in several models of pain. Transplantation of GABAergic cells or engineered cells producing GABA has been demonstrated as an effective way to reduce neuropathic pain symptoms (6,12,14,29,35,42,55,57). The findings in our lab suggest that the positive effects of neural progenitor grafts are likely mediated, at least in part, by GABA produced by transplanted cells in the dorsal horn, as both antinociceptive effects and attenuation of dorsal horn hyperexcitability observed in animals with NPC transplants were reduced by GABAergic antagonists (32,35). However, the contribution of endogenous GABAergic neurons cannot be ruled out. In fact, several studies suggest that neurotrophic substances released by progenitor cell transplants may have neuroprotective effects on surrounding cells and possibly rescue some of the dysfunctional GABAergic interneurons. The analgesic effect observed in transplanted animals may therefore be mediated by both transplanted and endogenous cells; however, the presence of the transplant is essential for the analgesic outcome. Our previous study showed that pharmacological inhibition of GABA receptors increased electrophysiological hyperexcitability and reduced analgesia more significantly in rats transplanted with GABAergic cells compared to control animals, further suggesting the active role of the transplant in the observed effect (35).
Although promising, NPC therapy shows limitations regarding its efficacy for long-term treatment. However, as the cells may serve as local minipumps, the concept of cell therapy gives us an opportunity to deliver additional therapeutic components via recombinant cells and thus enhance the efficacy of such an approach. Our previous research with adrenal chromaffin cells used as cell therapy for chronic pain revealed the presence of a peptide with NMDA antagonist activity within the cocktail of neuroactive substances released by an adrenal transplant. Histogranin and its synthetic derivate SHG display a potent analgesic effect after IT injection and reduce neuronal hyperexcitability after iontophoretic application in animal pain models (22,27,30). The activation of NMDA receptors initiates hyperexcitability of spinal neurons that cause abnormal responses to peripheral stimuli (3,8,16,4) and is considered as another key element in the pathophysiology of chronic pain. NMDA receptor antagonists have been shown to attenuate injury-induced hyperalgesia in animal models; however, the analgesic effect varies based on the pain model and the route of administration (25). Moreover, clinical use of NMDA antagonists is compromised by their narrow therapeutic window and adverse side effects such as motor weakness and hallucinations (37). Previous studies in our lab showed that the novel NMDA antagonist SHG can reduce pain symptoms without motor deficits and may be a promising adjunct in pain therapies (22,27,30,4,53). The possibility of dual targeting of inhibitory and excitatory signaling by a single intervention, the transplantation of recombinant cells releasing GABA and SHG, was the main motivation for this study.
As a proof of concept to explore the potential enhanced antinociceptive effects of SHG in combination with GABAergic NPC transplant, IT injections of the authentic SHG peptide (synthetized and purchased from 21st Century Biochemicals) in animals were evaluated. Results showed enhanced analgesia in animals treated by the combination of the GABAergic transplant and IT SHG peptide. Since this initial screen revealed promising antinociceptive enhancement of NPC transplants, SHG-producing NPCs were generated for transplantation. Using multiple outcome measures, findings suggested antinociceptive enhancement of the antiallodynic and antihyperalgesic effects of 1SHG NPC transplant. However, these promising improvements were modest and transient in most outcome measures, perhaps in part due to the dynamic nature of peripheral and central plasticity following the CCI injury and recovery. Multimeric 6SHG constructs resulted in more stable attenuation of tactile allodynia and stable and robust reversal of heat hyperalgesia following peripheral nerve injury. These results are in agreement with our previous observation of enhanced analgesic efficacy of multi-SHG constructs compared to the single SHG (33,34). It has been suggested that the variability of the analgesic efficacy of the SHG construct observed in different behavioral tests may be related to its physiological properties, such as modulation of other receptors, including NK1 and dopamine (27,30,39,4). Also, several studies pointed to a greater role of NMDA receptors in tactile allodynia than other modalities (48). Changes in other tests over time may indicate reduced release of SHG from cells, low survival potency of transplants, or spontaneous recovery of animals. Moreover, several studies suggest that activation of GABA receptors may attenuate the activity of NMDA receptors (41). With GABAergic neuronal progenitors we may target this pathway; therefore, the effect of SHG released by these cells may be less than completely additive in comparison with the effect of nonrecombinant NPCs.
The ability of recombinant cells to produce SHG has been demonstrated by FLISA and by immunohistochemistry. However, the immunohistochemical staining revealed relatively low amounts of recombinant cells in the area of transplant compared to the total amount of injected cells. Similar observations have been described in other studies (6,35,42). Nevertheless, behavioral observations showed that the analgesic effect may be achieved in spite of low transplant survival within the host nerve tissue. Similarly, studies focused on neurodegenerative diseases report therapeutic effects of transplanted NPCs with less than 10% of cells producing desired therapeutic substances (2,50).
This study demonstrated the feasibility of recombinant cell transplantation in the management of chronic pain. The proposed concept may be useful in the therapy of other disorders of the nervous system as well. Several clinical trials using stem cell therapies are currently ongoing. Equipping these cells with additional features allowing them to locally deliver therapeutic peptides using recombinant cell strategies may enhance the therapeutic potential of cell-based treatments.
Footnotes
Acknowledgment
Supported in part by the Craig H. Neilson Foundation 190926 (S.J.), R01 NS51667 (J.S.), and the Florida Department of Health. The authors thank Dr. Aldric Hama and David A. Collante for their excellent intellectual and technical assistance, Alexandra Lanjewar for technical assistance with the preparation of the manuscript, and Drs. Melissa Carballossa-Gonzales and Beata Frydel, Miami Project Image Core, for their assistance with image analysis. The authors declare no conflicts of interest.
