Abstract
To test the hypothesis that the transplantation of adipose precursor cells (APCs) improves nerve regeneration and functional recovery, human APCs were transplanted into the lumen of a nerve guide in a 6-mm unilateral sciatic nerve defect in athymic rats. The three control groups for the study were biodegradable, polycaprolactone-based nerve conduit without APCs, autograft, and empty defect. Behavioral tests were performed every 3 weeks, and the sciatic functional index (SFI) was calculated based on measurements from the hindlimb prints. After 12 weeks, the nerve as well as right and left gastrocnemius muscles were removed and preserved for histological evaluation. Full regeneration of the sciatic nerve occurred on the rats that received the autograft, the guide, and the guide with APCs; no regeneration was observed on any of the rats in which the defect was left untreated (empty defect). APCs survived transplantation for up to 12 weeks in the injured peripheral nerve. No significant colocalization was observed between the immunostaining for glial fibrillary protein and anti-human lamin A/C, implying that the APCs did not differentiate into Schwann cells at the site of injury. In comparison with the rats with untreated defects, a decrease in muscle atrophy was observed on those rats that received the autograft and the guide with cells as indicated by the gastrocnemius muscle weight ratio and the muscle fiber ratio. Significant differences in SFI were observed 3 weeks postinjury between the rats in which the guide was left empty and those that received the guide with APCs; however, these differences were not observed at 12 weeks. The transplantation of APCs promoted the formation of a more robust nerve as evidenced by the results from the cross-sectional area of regenerated nerve, and the transplantation of APCs produced a decrease in muscle atrophy.
Introduction
Injuries to peripheral nerves normally occur as a result of trauma or disease. Nerve guidance tubes have been developed as a replacement for the nerve autograft, which is the gold standard for the treatment of peripheral nerve injuries. Nerve guidance tubes are currently available for short-gap peripheral nerve repair. A variety of prototypes of nerve guidance tubes, based on natural and synthetic polymers such as collagen, fibronectin, chitosan, poly(caprolactone) (PCL), poly(lactic acid) (PLA), and poly(glycolic acid) (PGA), have been evaluated in rat models of nerve injuries (1, 5, 10, 19, 25, 26, 29). Electrically conductive polymers such as polypyrrole have also shown promising results for nerve repair (33). Despite the advances in the development of nerve guidance tubes, commercially available conduits are only suitable to repair defects less than 3 cm in length in humans. Long-gap defects >3 cm can only be treated using the nerve autograft, which has the disadvantage that it involves a secondary surgery and renders a nerve nonfunctional.
Researchers, recognizing that a nerve guidance tube cannot support regeneration over long gaps, have incorporated neurotrophic factors and/or cells into the conduits (27, 31). Neurotrophic factors such as nerve growth factor (NGF), glial cell line-derived neurotrophic factor (GDNF), and brain-derived neurotrophic factor (BDNF) have all been examined with promising results in peripheral nerve repair (7, 8, 13, 22, 30). For example, when immobilized in a gelatintricalcium phosphate (GTG)-based conduit, BDNF incorporation resulted in a significant increase in the gastrocnemius weight ratio, whereas the average axon size was larger on rats that received NGF-treated conduits (7). Fine et al. determined that the delivery of GDNF from polymeric nerve guides led to the formation of a higher number of myelinated fibers in comparison with NGF-releasing guides (13). The neurotrophic factor BDNF appeared to be advantageous for the regeneration and myelination of peripheral nerves in a study by Zhang et al. (32).
As an alternative to neurotrophic factor delivery, postnatal stem cells have shown promising results for the treatment of peripheral nerve injuries. As such, recent effort has been focused on the potential application of stem and/or progenitor cells to promote nerve repair. Mesenchymal stem cells (MSCs), bone marrow stromal cells, skin precursor cells (SKPs), and hair follicle stem cells have been transplanted into the injured peripheral nerve in various animal models (2, 3, 6, 11, 17, 18). Improvements in nerve regeneration were observed by the transplantation of MSCs predifferentiated into Schwann cells into a defect containing a muscle graft and also when MSCs were transplanted within an allogenic nerve graft (17, 18). The in situ differentiation of hair follicle stem cells into GFAP+ Schwann cells was recently documented in a severed sciatic nerve (2, 3). Furthermore, Miller et al. isolated a population of SKPs able to differentiate in vitro into p75+/s100+ Schwann cells (23).
Adipose tissue has been shown to be an abundant source of postnatal precursor cells (12). Adipose precursor cells (APCs) are relatively easy to isolate from fat tissue and can be isolated in sufficient amounts to be injected immediately postisolation, which render APCs attractive for the development of an autologous transplantation therapy for nerve repair. Adipose precursor cells are shown to be multipotent, as demonstrated by the ability of these cells to differentiate in vitro into chondrogenic, osteogenic, adipogenic, and myogenic cell types (12, 24, 34, 35). Despite all of the effort that has been devoted to the in vitro differentiation of APCs towards the neuroectodermal lineage, it is still unclear whether true, functional differentiation occurs (4, 20). Furthermore, the in situ differentiation of APCs in the injured peripheral nerve in the rat model has yet to be documented. In the present study, we demonstrate that APCs survive for up to 12 weeks when transplanted into the injured peripheral nerve. The transplantation of APCs induced the formation of a thicker nerve and produced a modest decrease in muscle atrophy without the requirement of cell differentiation.
Materials and Methods
Materials
Chemicals were obtained from Sigma-Aldrich unless otherwise specified. DMEM/F12, penicillin/streptomycin, and FBS were obtained from Gibco (Invitrogen Corporation, Carlsbad, CA).
Human Adipose Tissue
Samples of adipose tissue were obtained from females undergoing elective abdominal reduction surgery. All women, ages ranging from 38 to 58 years, were healthy according to clinical examination and laboratory tests. The University of Pittsburgh Institutional Review Board (IRB) approved the procedure of collecting the samples of discarded adipose tissue.
Isolation and Characterization of APCs Obtained From Human Adipose Tissue
APCs were isolated from human subcutaneous, abdominal adipose tissue. The sample of adipose tissue was washed with sterile PBS and transferred into a centrifuge tube containing collagenase (1 mg/ml) prepared in PBS. The tissue was finely minced using sterile scissors and incubated in the collagenase for 45 min. The digested tissue was filtered through sterile gauze and the filtrate was centrifuged at 1000 rpm for 10 min. The supernatant was discarded and the pellet washed in erythrocyte lysis buffer (pH 7.4). The final solution was centrifuged at 1000 rpm for 10 min and the pellet resuspended in DMEM/F12 with 10% FBS and 1% penicillin/streptomycin. The final cell suspension was plated and cultured up to passage 5.
Prior to transplantation, APCs were labeled with a PKH26 red fluorescent cell membrane label according to the manufacturer's instructions (Sigma-Aldrich, USA). Briefly, APCs were trypsinized, centrifuged to remove the serum, and resuspended in Diluent C. Immediately after a 4 × 10−6 molar PKH26 dye solution was added to the cell suspension and was immediately mixed. After 5-min incubation at 25°C, the labeling process was stopped by adding serum. The cell suspension was centrifuged and washed three times using PBS. PKH26-labeled APCs are shown in Figure 1B.
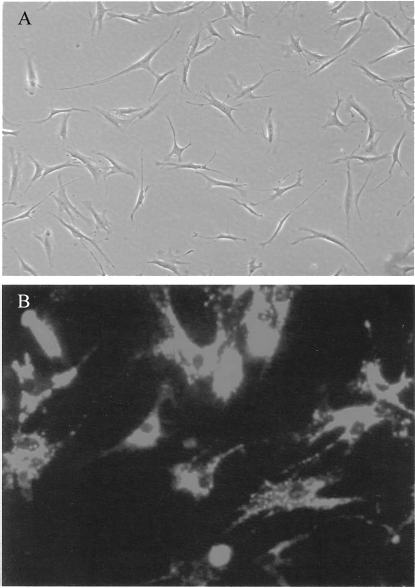
Appearance of APCs in culture. APCs display fibroblast-like morphology in culture (A). Prior to transplantation, APCs were labeled with a PHK26 fluorescence membrane dye (B). Original magnification 400×.
Preparation of Nerve Guides
Nerve guides were prepared using a solvent evaporation technique as previously described (5). Briefly, polycaprolactone (PCL) was dissolved in tetrahydrofuran (THF) at a concentration of 0.22 g of PCL/ml of THF. Particles of sodium chloride (NaCl) (diameter ranged from 38 to 50 μm) were added to the polymer solution in a 1:1 PCL/NaCl weight ratio and the polymer/salt solution was mixed. To prepare the guides, a 16-gauge stainless steel wire (1 cm in length) was immersed three times into the polymer solution and the solvent was allowed to evaporate overnight at room temperature. The wire containing the conduit was immersed in deionized distilled water for 48 h with the water being replaced three times daily. The nerve guides were then carefully removed from the wire and were trimmed for a final length of 8 mm. Before further use, the guides were allowed to dry for 2 days at room temperature.
To promote the interaction of host-derived axons, Schwann cells, and APCs with the internal surface of the guide, the peptide sequence Arg-Gly-Asp (RGD) was incorporated into the lumen of the conduit employing a protocol as previously described (28). Briefly, the internal lumen of the guide was first treated for 3 h with a 10% w/v solution of 1,6-hexanediamine prepared in isopropanol. Then, the lumen of the conduit was thoroughly rinsed with deionized distilled water and the guide was allowed to dry overnight at room temperature. Peptide and 1-ethyl-3-(dimethylaminopropyl) carbodi-imide (EDC) were combined in a 1:1 molar ratio and N-hydroxysuccinimide, (NHS) was added to that solution in a molar ratio of EDC/NHS of 4:1. RGD was added to the solution for a final density of 708 ng RGD/mm2 of internal surface area. To avoid the leakage of solution while the peptide solution was incubated in the lumen of the guide, both ends of the conduit were sealed with removable silicone glue. The final solution was incubated inside the lumen of the guide for 3 h at room temperature, with 90° rotations every 15 min. The solution was removed and the lumen of the guide was thoroughly rinsed with deionized distilled water. The guides were sterilized with ethanol prior to implantation.
Surgeries on Sciatic Nerve and Cell Transplantation
Twenty-two male athymic rats were purchased from Taconics (Hudson, NY). At the time of implantation, the rats were 8 weeks old with an average weight of 223.5 ± 49.2 g. Animals were randomly assigned into four experimental groups: empty defect (n = 3), autograft (n = 4), nerve guide (n = 6), and nerve guide with 1 × 106 APCs (n = 9). The rats were anesthetized by inhalation of isofluorane and the right sciatic nerve was exposed; 6 mm of sciatic nerve was surgically removed and each rat received one of the above treatments. For the autograft, the 6-mm piece of sciatic nerve was excised, flipped 180°, and implanted. For the cell transplantation group, the nerve conduit was sutured into both nerve stumps prior to cell transplantation. PBS (5 μl) containing 1 million PKH26-labeled APCs were delivered into the middle of the conduit (treatment group referred as guide with APCs) using a 10-μl micropipette.
Immunohistochemistry
Twelve weeks postinjury a section of sciatic nerve containing the injury site and a piece of nerve from the noninjured leg (control leg) were excised from each rat. The samples of tissue were either immediately frozen or placed in 10% buffered formalin for paraffin embedding. Both frozen and paraffin embedded samples were sectioned at a thickness of 10 μm.
To determine the fate of the transplanted cells, sections of tissue containing the regenerated nerve were blocked with 5% horse serum for 1 h at room temperature. Mouse anti-human lamin A/C (1:100; Vector Laboratories) prepared in 2.5% horse serum was added to the slides and the slides were incubated overnight at 4°C. Next day, the tissue sections were treated with biotinylated anti-mouse IgG (1:300; 90 min; Vector Laboratories). Streptavidin-FITC (1:300; Invitrogen) was added and incubated for 20 min at room temperature. The sections of tissue were individually stained with antibodies against neurofilament (NF; 1:300; Chemicon), and glial fibrillary protein (GFAP; 1:1000; Chemicon) to determine the final phenotype of the surviving cells. For NF and GFAP, the secondary antibody used was Cy5-conjugated anti-rabbit IgG (1:500; 1 h; Molecular Probes). All sections were counterstained using DAPI nuclear stain.
Oil-Red-O Staining
To detect the presence of adipocytes in the culture of APCs, the cells were seeded at a density of 4000 cells per well in a 24-well plate. After 12 h, APCs were fixed for 10 min with 2% paraformaldehyde. The fixed APCs were exposed for 20 min to a solution containing 2% w/v Oil-red-O prepared in isopropanol. The percent of APCs positive for Oil-red-O was quantified by visual examination under the microscope.
For immunohistochemistry, frozen tissue sections were fixed with 2% paraformaldehyde for 10 min, immersed for 20 min in the 2% w/v Oil-red-O solution, rinsed with PBS, and attached to a coverslip.
Muscle Atrophy
Gastrocnemius Muscle Weight Ratio
Athymic rats were euthanized 12 weeks postsurgery with an overdose of sodium pentobarbital. Left (control) and right (treatment) gastrocnemius muscles were excised and weighed. The tissue was either immediately frozen in liquid nitrogen or immersed in formalin for paraffin embedding. Per each rat, a gastrocnemius muscle weight ratio (E/C) was calculated based on the weight of the gastrocnemius muscle on the experimental leg (right leg) versus the control leg (left leg) according to the following formula: E/C = weight experimental/weight control muscle.
Histological Analysis of Gastrocnemius Muscles
Experimental and control gastrocnemius muscles were sectioned at a thickness of 10 μm and were stained using hematoxylin and eosin (H&E). For both control and experimental muscle, the midportion of the muscle was analyzed by taking 10 images per tissue section with the 40× objective. Each image was manually analyzed by counting the total number of whole myofibers. A ratio of myofibers was calculated based on the average number of fibers in the experimental leg versus the average number of fibers in the control leg according to the formula: ratio number of myofibers = number of fibers experimental muscle/number of fibers control muscle.
Cross-Sectional Area of the Newly Formed Nerve at the Center of the Lesion
The cross-sectional area of the regenerated nerve at the center of the lesion was determined for those rats that received the guide, the guide with APCs, and the autograft. Sections of nerve were stained with H&E and for these treatment groups; images at a magnification 4× were captured at the center of the newly formed nerve. The images were processed using computer software (MetaMorph, Molecular Devices Corporation, Sunnyvale, CA) and, for each image taken, the cross-sectional area of the nerve was calculated using an internal calibration curve.
Sciatic Functional Index
The recovery of function at the toe level was addressed using the sciatic functional index (SFI) formula, which provides information on the ability of the rat to spread its toes. Three parameters in the formula permit the assessment of the level of impairment: print length (PL), toe spread (TS), and intermediary toe spread (ITS). The day prior to surgery and every 3 weeks postsurgery, waterproof paint was placed on the hindlimb toes of each rat. Hindlimb prints were recorded on a piece of paper while the rat walked through a corridor. For each foot print, measurements of PL, TS, and ITS were manually determined. Five prints were analyzed per each leg for a total of 10 prints per rat at each time point. A value of SFI was calculated per each print according to the formula:
where EPL is experimental print length, NPL is normal print length, ETS is experimental toe spread, NTS is normal toe spread, EIT is experimental intermediary toe spread, and NIT is normal intermediary toe spread.
Statistical Analysis
Unless otherwise specified, the results are reported as mean ± SD. Minitab version 13 (Minitab Inc., State College, PA) was used in the statistical analysis of the data. Analysis of variance (ANOVA) was performed to assess changes in the SFI, cross-sectional area of regenerated nerve, gastrocnemius muscle weight ratio, and ratio of myofibers among treatment groups. Tukey's post-hoc analysis was conducted to assess differences among treatment groups. Statistical significance was set at a level of p ≤ 0.05.
Results
Characterization of APCs Prior to Transplantation
APCs were isolated from samples of human adipose tissue obtained from cosmetic surgical procedures. The tissue was processed through dissociation with collagenase and centrifugation until the stromal fraction, which contains the APCs, was obtained. Figure 1 shows the morphology of APCs when cultured in a tissue culture flask. Cells at early passage (passage <5) were used in the study. The absence of adipocytes in the injected samples was confirmed using the Oil-red-O stain (data not shown).
Nerve Guides
Nerve guides were manufactured using a procedure previously described (5). Figure 2 shows a cross-sectional view of the nerve guides obtained using a scanning electron micrograph. The guides utilized in the study were 50% porous and contained an internal diameter of 1.5 mm, an outside diameter of 1.8 mm, and were 8 mm long. A surface modification technique was utilized to incorporate RGD into the internal lumen of the guide (28). The binding of RGD to PCL was confirmed using X-ray Photoelectron Spectroscopy (XPS) and these results have been published (28). Human APCs have been shown to bind to RGD-treated surfaces (28).

Nerve guides were prepared from polycaprolactone. Scanning electron micrographs showing a cross-sectional view (A, C) of the nerve guides prepared for this study. The implanted guides contained an internal diameter of 1.5 mm and were 8 mm long. The high porosity observed in the internal surface of the guide (B) is attributed to the addition of a porogen into the polymer solution.
Survival of APCs in the Injured Sciatic Nerve
Regeneration within the gap was assessed based on physical observation and histological results. The results of the histology demonstrated the full regeneration of the sciatic nerve across the gap in those rats that received the autograft, the nerve guide, and the nerve guide with APCs. No regeneration was observed in those rats in which the defect was left empty (Fig. 3A).

Images of regenerated sciatic nerve. Images of the area of peripheral nerve surgery as observed 12 weeks postsurgery. No regeneration was visible in those rats in which the gap was left untreated (A). Full regeneration of the sciatic nerve was obtained in those rats that received the autograft (B), the nerve guide (C), and the nerve guide containing APCs (D), as confirmed by immunohistochemistry.
The survival of APCs in the injured peripheral nerve was histologically assessed using antibodies against human lamin A/C, as well as confirmation of the PKH26-labeled cells. As shown in Figure 4A, APCs survived transplantation for up to 12 weeks in the injured peripheral nerve. In frozen sections, a high background was observed using the red excitation filter, which made it challenging to identify the PKH26-labeled cells on those samples. However, in paraffin embedded samples, PHK26+ cells were observed and colocalized with the DAPI nuclear stain and lamin A/C (Fig. 4A). Autograft sections did not show any signals indicative of human cells (e.g., lamin A/C immunostaining or PKH26 excitation at 560 nm, data not shown).

Survival and differentiation of APCs after transplantation. Twelve weeks postinjury APCs were identified in the newly formed peripheral nerve using the PKH26 membrane stain (red) and identifying colocalization with antibodies against human lamin A/C (green) (A, original magnification 200×). (B) APCs were observed around neurofilament (NF)-positive axons (green) in sections of regenerated nerve, but no colocalization with PKH26 was evident (original magnification 200×). (C) Human lamin A/C (green) and GFAP (red) were found in this section, with the top arrow indicating overlap, the middle arrow indicating GFAP+ only, and the bottom arrow indicating human lamin A/C+ only (original magnification 600×). (D) The presence of adipocytes stained with Oil-red-O was observed in the newly formed nerve as indicated by the arrow (D). Blue: DAPI stain.
Differentiation of APCs in the Injured Sciatic Nerve
Immunohistochemistry was employed to determine the final phenotype of the transplanted cells. For this purpose, we utilized antibodies against human lamin A/C to detect the presence of the transplanted APCs in the sections of regenerated nerve. We counterstained the slides with antibodies against GFAP, which identifies premyelinated and nonmyelinating Schwann cells and with antibodies against NF, which identifies growing axons. No colocalization was observed between the PKH26 and NF stainings (Fig. 4B). Results from the stainings indicate that no significant colocalization was observed between the lamin A/C and GFAP staining, which indicates that the surviving cells did not differentiate into Schwann cells (Fig. 4C). However, it must be noted that there was an occasional colocalization as evidenced by the top arrow in Figure 4C. However, the presence of cells containing lipid vacuoles was observed in the newly formed nerve (Fig. 4D). Therefore, it is possible that a portion of the transplanted cells differentiated into adipocytes, but further studies need to be conducted.
Cross-Sectional Area of the Regenerated Nerve
For the rats that received the autograft, the guide, and the guide with cells, the cross-sectional area of the regenerated nerve measured at the center of the injury was found to be 1.683 ± 0.400, 0.406 ± 0.323, and 0.913 ± 0.234 mm2, respectively. The largest cross-sectional area was observed for the rats that received the autograft. As illustrated in Figure 5, the rats that received the guide with APCs (Fig. 5A) had a larger cross-sectional area in comparison with the rats that received the guide without cells (Fig. 5B) with values of p = 0.049.
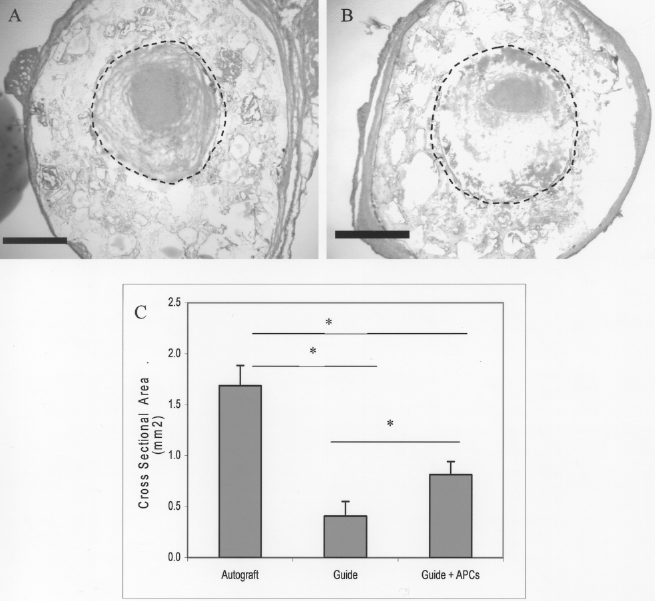
Cross-sectional area of regenerated nerve. Cross-sectional view of regenerated nerve stained using hematoxylin and eosin. The location of the lumen of the guide is indicated by the circle. At the center of the lesion, the regenerated nerve was thicker on the rodents that received guides with APCs (A, scale bar: 680 μm), in comparison with rodents that received empty guides (B, scale bar: 720 μm). The transplantation of APCs into the injured sciatic nerve produced an increased in the cross-sectional area of the regenerated nerve in comparison with the guide without cells, with the highest cross-sectional area of regenerated nerve observed in those rats that received the autograft (C). *p ≤ 0.05. Original magnification 4×.
Analysis of the Gastrocnemius Muscle
The transection of the sciatic nerve produced the loss of neural innervation of the gastrocnemius muscle, which led to a decrease in gastrocnemius muscle mass. This decrease in muscle mass was quantified in this study by weighing the excised gastrocnemius muscles and calculating a ratio of muscle mass (referred as E/C) based on the mass of the experimental muscle versus the mass of the muscle in the control leg. As the E/C value approaches 1, less muscle atrophy is present. The smallest E/C ratio was obtained by the empty treatment group in which nerve regeneration was never observed (0.192 ± 0.024) (Fig. 6A). The highest E/C ratio and therefore less muscle atrophy was present in the rats that received the autograft (0.666 ± 0.070). For the rats that received nerve guides and guides with APCs, the E/C ratio was 0.487 ± 0.151 and 0.522 ± 0.108, respectively (p = 0.632).

Analysis of gastrocnemius muscle atrophy. Muscle atrophy was assessed based on the analysis of experimental and control gastrocnemius muscle weight and their histology. H&E stains of cross sections of the gastrocnemius muscle are shown in (A) autograft, (B) empty guide, and (C) treatment with ASCs plus guide (original magnification 40×). Also, the gastrocnemius muscle weight ratio, referred as E/C, was calculated as the ratio of muscle weight on the experimental leg versus the weight on the control leg (D). According to the E/C ratio, the highest muscle atrophy was observed on those rats in which the defect was left empty, whereas less atrophy was observed on rodents that received the autograft (D). The ratio of number of muscle fibers per area in the experimental leg versus the control leg showed a high fiber ratio, indicative of more muscle atrophy, in those rats in which the defect was left empty. The smallest fiber ratio, and therefore less atrophy, was observed among those rats that received the autograft and the guide with cells (E). An inverse correlation was found between the individual rodent values of E/C and the muscle fiber ratio, with a value of p < 0.001 (F). *p ≤ 0.05.
The resected gastrocnemius muscles were further analyzed by sectioning and staining with hematoxylin and eosin (H&E). Images of stained sections containing the middle portion of the muscle were taken and for each image the number of myofibers per unit area was quantified. The ratio of myofibers in the experimental leg versus number of fibers in the control leg was calculated per rat. As this ratio approaches 1, less muscle atrophy is expected. The ratio of fibers per area was the highest in those rats in which the defect was left empty, with a mean ratio ± SD of 4.29 ± 1.45 (Fig. 6B). For the group of rats that received the empty guides, the fiber ratio was 3.03 ± 1.46, which is not statistically different from the empty defect (p = 0.774). However, values approaching 1 were obtained for the autograft and guide with cells, with a fiber ratio of 1.41 ± 0.71 and 1.46 ± 0.27, respectively.
Performance From Sciatic Functional Index (SFI)
The results of the sciatic functional index are shown in Figure 7C; error bars were omitted in the figure to make clear the general trend of the data. As shown in the figure, for all four treatment groups in the study, the SFI decreased from an initial mean ± SD of −8.31 ± 2.68 to −64.51 ± 11.20 at 3 weeks after the transection of the sciatic nerve. Significant improvements in the SFI were observed 3 weeks after surgery between the treatment group that received the guide with cells and the empty defect (p = 0.010); however, these improvements were not evident at week 6 or week 12. Indeed, 12 weeks postsurgery the SFI values were similar among all treatment groups (p = 0.864).
Discussion
The autograft is the gold standard for nerve repair and its success has been attributed to the presence of Schwann cells that secrete factors that promote axonal growth through the gap and to the structural support provided by the extracellular matrix (ECM) proteins present in the autograft, which provides binding sites and directionality to the growing axons (9). Various studies have shown that nerve guidance tubes alone cannot efficiently promote full regeneration of a peripheral nerve over a gap longer than 3 cm in length (in humans). However, the incorporation of Schwann cells into the tube has significantly improved the conduit's performance in animal studies (16). Although autologous Schwann cell transplantation offers great promise for the repair of peripheral nerves, the isolation of Schwann cells involves a secondary surgery and renders a secondary nerve nonfunctional (14, 15). Furthermore, it is clinically challenging to obtain high numbers of pure Schwann cells needed for repair.
Stem cells and progenitor cells are seen as an attractive alternative to the use of autologous Schwann cells. Bone marrow-derived stem cells, skin precursor cells, and hair follicle stem cells have been transplanted into an injured peripheral nerve model (11). However, unlike other precursor cells, APCs can be isolated in sufficient amounts making these adult progenitor cells clinically feasible for cell transplantation therapies. Not only complications related to cell expansion prior to transplantation are eliminated by the use of these progenitor cells, but the use of autologous cell transplantation eliminates the need for immunosuppressant.
To the best of our knowledge, this is the first study in which human APCs are transplanted into a rat model of nerve injury. Our study demonstrates that human-derived APCs survive transplantation for up to 12 weeks in the injured peripheral nerve as observed by the immunohistochemical results obtained using lamin A/C antibodies.
In our study, we selected a 6-mm defect with the purpose of determining if factors released at the site of injury could potentially induce APC transdifferentiation. The potential for APCs to differentiate into Schwann cells was assessed by colocalizing the anti-human lamin A/C staining with the staining against GFAP and neurofilament. We did not identify any colocalization of PKH26 and NF (Fig. 4B). Furthermore, very minimal colocalization was observed between lamin A/C-positive and GFAP-positive cells, indicative that the transplanted cells did not differentiate into Schwann cells at the site of injury. However, there were rare events indicating colocalization of the human lamin A/C-positive cells with GFAP regions (Fig. 4C). It is impossible to stain every cell in every section, so there is the potential that some APCs may have differentiated into Schwann cells; further studies must be conducted. To identify a possible differentiation of the transplanted cells into adipocytes, tissue sections were stained using Oil-red-O. In this study, we were able to observe the presence of adipocytes dispersed in the regenerated nerve (Fig. 4D). No adipose tissue formation was observed in the newly formed nerve or in the surrounding areas.
Functional recovery was assessed using SFI, which has been widely employed to document changes in the ability of the rodent to spread its toe as a result of the reinnervation of deinnervated areas in the leg and toes. SFI values close to zero represent full recovery of function and values in the proximity to −100 represents full impairment. A significance difference in SFI values was only observed 3 weeks postsurgery between the rats that received the guide with cells and those that received the empty defect (p = 0.009) (refer to Fig. 7C). However, this difference was not present at later time points. For all treatment groups, the average SFI value at the end of the study was −64.86 ± 2.70. The proximity of this value to −100 indicates that proper nerve reinnervation of leg and toes did not occur, or that pain sensation impeded toe spread. At 12 weeks postsurgery, we observed that rodents that received the guide with cells were able to spread the toe in the injured leg at stance (Fig. 7A). However, we also observed that the same rodents closed the paw during locomotion (Fig. 7B). We cannot rule out the possibility that pain sensation may have contributed to the inability of the rodent to spread its toes during walking, and therefore to the low SFI values observed at the end of the study. Nevertheless, we recommend the use of time points longer than 3 months to follow up the SFI recovery. Spontaneous regeneration of the sciatic nerve has been documented to occur at rate of 2–3 mm per day in rats (18). We also recommend the analysis of thermal and mechanical sensation using paw withdrawal latency after heat treatment and paw withdrawal response to von Frey filaments.
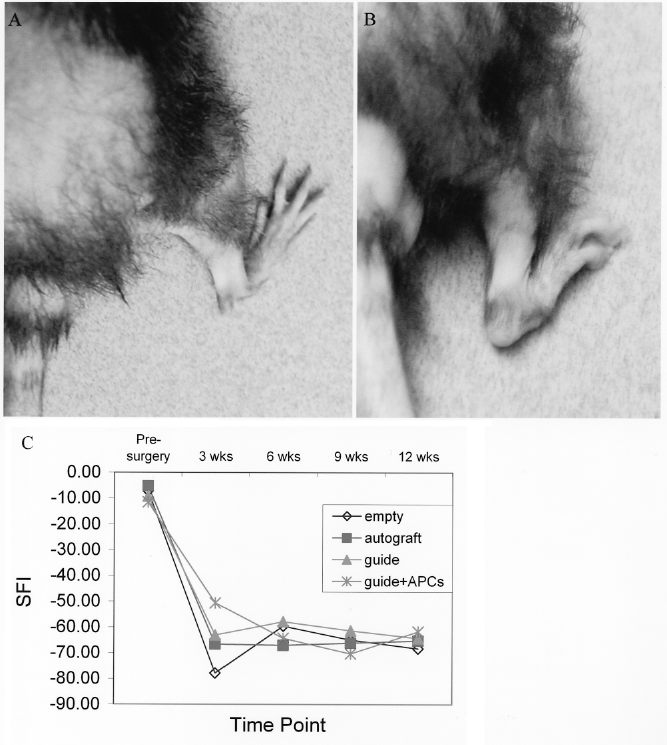
Walking track analysis. Twelve weeks postinjury, rodents that received guides with APCs were able to spread the toe during resting conditions (A). However, these rodents maintained the paw closed during walking (B). (C) Pre- and postsurgical SFI values represented as a function of time. SFI values significantly different (p ≤ 0.05) between the guide with APCs and the empty defect were observed 3 weeks postsurgery. No differences in SFI among treatment groups were observed 12 weeks postsurgery (p > 0.05). *p ≤ 0.05.
Muscle atrophy was assessed using the gastrocnemius muscle weight ratio and by analyzing the number of myofibers per area for all treatment groups. The myofiber ratio was more a more sensitive measurement of atrophy in comparison with the bulk muscle weight. An inverse relationship was observed between the muscle weight ratio and the myofiber ratio, as shown in Figure 6. The greatest loss of muscle mass was observed in those rats in which the defect was left empty and the least amount of muscle atrophy was observed on those rodents that received the autograft. For the rats that received the nerve guide with APCs, the muscle weight ratio was not statistically different from those that received the empty guide (p = 0.632). However, the myofiber ratio indicates that less muscle atrophy in rodents that received the autograft and the guide with cells, with the fiber ratio closer to a value of one.
The formation of a thicker nerve can be attributed to APC transplantation. Indeed, the regenerated nerve after cell transplantation was more than double the size of the nerve formed with the conduit alone. Although some disperse lipid vacuoles were observed in the newly formed nerve, no adipose tissue formation was identified in the nerve or surrounding areas after APC transplantation.
In summary, we determined that APCs survive transplantation for up to 12 weeks in the injured peripheral nerve. This study showed that at 12 weeks postinjury, the environment at the injury site cannot promote the differentiation of the cells into the ectodermal lineage. However, cell differentiation is not required for the stem and progenitor cells to promote repair, as it has been documented that stem cells release factors and supporting extracellular matrix components that induce tissue repair (6). Furthermore, adipocytes secrete angiopoietin-1, which supports neurite outgrowth; although not measured in this study, it may be possible that APCs also induced neurite outgrowth (21). Although no cell differentiation was promoted, a modest decrease in muscle atrophy and the formation of a thicker nerve was observed due to the transplanted cells.
Conclusions
Research on cell-based therapies for neural tissue repair has shown promising results. The results of this study indicate that APCs survive for up to 12 weeks in the injured peripheral nerve. However, the environment around the lesion site did not appear to be sufficient to promote significant in situ differentiation of APCs toward the ectodermal lineage. A modest decrease in muscle atrophy and the formation of a more robust nerve can be attributed transplantation of APCs into the injured peripheral nerve. Future experiments will be conducted to assess the possibility that APCs can improve nerve repair by releasing factors that promote axonal growth as it has been documented that adipocytes secrete factors that promote neurite outgrowth.
Footnotes
Acknowledgments
This work was supported by an NIH institutional postdoctoral fellowship (L.Y.S.) through the Pittsburgh Tissue Engineering Initiative (PTEI), the American Association for Hand Surgery (K.G.M.), and the National Science Foundation (K.G.M). The authors would like to thank Nicole Martik for the assistance provided in the calculations of SFI, and the Center of Biologic Imaging (CBI) at the University of Pittsburgh.

