Abstract
Neural progenitor cells (NPCs) overexpressing fibroblast growth factor 2 (FGF-2) have the distinct tendency to associate with the vasculature and establish multiple proliferative clusters in the perivascular environment after transplantation into the cerebral cortex. Strikingly, the vascular clusters of progenitor cells give rise to immature neurons after ischemic injury, raising prospects for the formation of ectopic neurogenic niches for repair. We investigated the spatial relationship of perivascular clusters with the host vascular structures. FGF-2-GFP-transduced NPCs were transplanted into the intact somatosensory rat cortex. Confocal microscopic analysis revealed that grafted cells preferentially contacted venules at sites with aquaporin-4-positive astrocytic endfeet and avoided contacts with desmin-positive pericytes. Electron microscopic analysis confirmed that grafted cells preferentially made contact with astroglial endfeet, and only a minority of them reached the endothelial basal lamina. These results provide new insights into the fine structural and anatomical relationship between grafted FGF-2-transduced NPCs and the host vasculature.
Keywords
Introduction
Neural progenitor cells (NPCs) have two essential properties: self-renewal and multipotency. In the postnatal mammalian brain, active neurogenesis occurs only in two restricted areas: the subventricular zone of the lateral ventricle (SVZ) and the subgranular zone of the dentate gyrus (SGZ) in the hippocampus1–3. In these regions, adult stem cells and their progeny are localized in specialized vascular niches that regulate their self-renewal and differentiation4–6.
The stem cell niche is an anatomical and functional unit that provides a specific microenvironment where stem cells replenish themselves through self-renewal and give rise to different progenitors 7 . The cellular and molecular composition of the niche in the two germinal regions has been extensively studied and characterized. In both structures, multipotent stem cells have been identified as slow-dividing glial fibrillary acidic protein (GFAP)-positive astrocytes that give rise first to transit-amplifying cells that, in turn, differentiate into neuroblasts 5 . Interestingly, SVZ astrocytes come into contact with the cerebrospinal fluid (CSF) via a process that extends between ependymal cells 5 . These astrocytes (type B cells) also come into contact with blood vessels 8 .
Neural stem cell activation, self-renewal, proliferation, and differentiation in the niche are regulated by a multitude of signals. These include bone morphogenetic proteins (BMPs) and their antagonists: Noggin, fibroblast growth factor 2 (FGF-2), epidermal growth factor (EGF), the Wnt/β-catenin pathway, Notch, and sonic hedgehog (Shh) signaling7,9. Extracellular matrix proteins such as tenascin C and laminin α2 chain along with β1 integrins have also been implicated in niche functions10,11.
An interesting feature of the niche is the intimate association of NPCs with vasculature in both the SVZ and the SGZ12,13. Blood vessels in the brain are formed by endothelial cells and perivascular support cells, including astrocytic endfeet, pericytes, and smooth muscle cells. Endogenous NPCs are frequently in contact with the vasculature at sites that lack astrocytic endfeet and pericyte coverage and receive spatial cues and regulatory signals from diverse elements of the vascular system4,14,15. Endothelial cells seem to provide attachment for NPCs, generate signals for self-renewal, and prime stem cells for the production of neurons. Such signals include Notch signaling and diffusible signals, such as vascular endothelial growth factor (VEGF), FGF-2, insulin-like growth factor 1 (IGF-1), pigment epithelium-derived factor (PEDF), brain-derived neurotrophic factor (BDNF), and other unidentified factors that affect neural precursors6,16,17. In vivo, these factors may bind to the vascular basal lamina and to fractones that extend from the vasculature in the SVZ 18 , as has been shown for FGF-2 19 . Synergistic interactions between FGF-2 and VEGF signaling may underlie the coordination of angiogenesis and neurogenesis in the SVZ and dentate gyrus20,21.
It has been shown that the postnatal cortex has a very limited ability to regenerate neural tissue after brain insults. Compensatory cortical neurogenesis has been reported after induced apoptotic degeneration 22 or after stroke23–25, but the number of newly generated neurons, if present at all 26 , remains insufficient to restore normal cortical function 27 . This may be related, in part, to the lack of specific niches that could be activated by injury-derived signals to provide new neurons for repair. To overcome this limitation, the manipulation and transplantation of NPCs have developed into a key strategy for brain repair28,29. A fundamental challenge is to find ways to construct ectopic neurogenic niches using grafted progenitor cells that would provide new neurons to regenerate the injured brain tissue.
In a previous study 28 , we developed a system to over-express FGF-2 in transplanted NPCs (FGF-2-NPCs) and showed that this manipulation significantly enhanced the capacity of these cells to generate a pool of progenitors and immature neurons in the ischemic cortex. A closer look revealed that FGF-2-NPCs had the distinct tendency to associate with the vasculature and establish multiple proliferative clusters (more than three cells associated with blood vessels) in the perivascular environment. Strikingly, the vascular clusters of progenitor cells can generate immature neurons in an ischemic environment, raising the prospect for the formation of ectopic neurogenic niches for repair. Here we propose to investigate the cellular architecture and relationship of grafted cells with the vasculature in these specialized anatomical entities.
Materials and Methods
All animal experiments were conducted in compliance with the bylaws of the Geneva Cantonal Veterinary Authority and were approved by the ethical committee of the Geneva University School of Medicine. Newborn (P0) male and female Sprague–Dawley (SD) rat pups (n = 54) and female and male Wistar pups at postnatal day 3 (P3) (n = 12) were obtained from the Geneva University School of Medicine animal facility.
Isolation and Culture of NPCs
NPCs from newborn SD rats were dissected from the SVZ. Cell suspensions were purified using a 22% Percoll (GE Healthcare Life Sciences, Uppsala, Sweden) gradient centrifugation at 250 × g as previously described 28 , and seeded onto Matrigel (BD Biosciences, San Diego, CA, USA)-coated dishes (4 × 10 5 cells per 35-mm culture dish). NPCs were allowed to expand in Neurobasal medium (Invitrogen, Carlsbad, CA, USA) supplemented with 20 ng/ml human recombinant FGF-2 (R&D Systems, Minneapolis, MN, USA), 2% B27 supplement (Invitrogen), 2 mM L-glutamine (Invitrogen), 1 mM sodium pyruvate (Invitrogen), 2 mM N-acetylcysteine (Sigma-Aldrich, St. Louis, MO, USA), and 1% penicillin–streptomycin (Invitrogen). At day 3 (DIV3), half of the culture medium was replaced with fresh medium containing 20 ng/ml of FGF-2 with the FGF-2 lentiviral vector. At DIV5, cells were then trypsinized, centrifuged, and resuspended in Neurobasal medium for transplantation.
Design and Production of Lentiviral Vectors
For the FGF-2 construct, a cDNA coding for the 18-kDa form of human FGF-2 (basic FGF) was fused to an immunoglobulin signal peptide to facilitate secretion 30 and cloned into a pWPI bicistronic lentiviral vector as previously described 31 . FGF-2 lentiviral vector titers ranging from 10 8 to 10 9 transducing units (TU)/ml were used. For transduction, we used doses ranging from 5 × 10 4 to 5 × 10 5 TU for FGF-2 lentiviral vectors on NPC dishes.
NPC Transplantation
For NPC transplantation, Wistar pups at P3 were anesthetized with a mixture of isoflurane (Forene®; Abbott, Cham, Switzerland), O2 30%, and air 70%. Approximately 30,000–50,000 (in 1 μl volume) FGF-2-NPCs were stereotactically transplanted into the somatosensory cortex with a nanoinjector (coordinates from bregma: 1.5 mm posterior, 2.0 mm lateral, 0.7 mm depth).
BrdU Injection
Bromodeoxyuridine (BrdU) (Sigma-Aldrich) was intraperitoneally (IP) injected (50 mg/kg) twice daily 2 days before sacrifice.
Immunohistochemistry and Image Processing
One or 2 weeks after NPC transplantation, rats were anesthetized by sodium pentobarbital (Abbott Laboratories, North Chicago, IL, USA). After intracardial perfusion of 0.9% saline followed by cold 4% paraformaldehyde (PFA) (Sigma-Aldrich) brains were immersed in the same fixative and postfixed overnight at 4°C. Brain sections were cut on a vibratome 1500 (40-μm-thick sections) (The Vibratome Co., St. Louis, MO, USA). Sections were incubated overnight at 4°C with a primary antibody diluted in phosphate-buffered saline (PBS; Sigma-Aldrich)/0.5% bovine serum albumin (BSA; Sigma-Aldrich)/0.3% Triton X-100 (Sigma-Aldrich).
The following primary antibodies were used: monoclonal mouse anti-nestin clone rat-401 (1:1,000; Chemicon, Temecula, CA, USA), monoclonal mouse anti-RECA-1 (rat endothelial cell antigen), clone HIS52 (1:250; AbD Serotec, Oxford, UK), polyclonal rabbit anti-glial fibrillary acidic protein (GFAP) (1:500; DakoCytomation, Glostrup, Denmark), polyclonal goat anti-green fluorescent protein (GFP) (1:5,000; Novus Biologicals, Littleton, CO, USA), monoclonal rat anti-BrdU, clone BU1/75 (ICR1) (1:100; Oxford Biotechnology Ltd, Oxfordshire, UK), and polyclonal rabbit anti-EGFR (1:50; Abcam, Cambridge, UK). The following day sections were incubated with the secondary antibody against the appropriate species: anti-rabbit Alexa 568, 647, and 488; anti-mouse Alexa 568, 488, and 647; anti-goat Alexa 555 and 647; and anti-rat Alexa 555 (Invitrogen). Nuclei were counterstained with Hoechst 33342 (Invitrogen).
Epifluorescent images were acquired with a digital camera (Retiga EX; Qimaging, Surrey, BC, Canada) linked to a fluorescent microscope (Eclipse TE2000-U; Nikon Instruments Inc., Melville, NY, USA) equipped with Nikon objectives.
Confocal images of grafted FGF-2 + GFP-positive cells were acquired with LSM 510 Meta (Zeiss, Jena, Germany) and FV1000-BX61WI (Olympus, Tokyo, Japan) laser scanning microscopes.
Electron Microscopic Analyses of Perivascular Cell Clusters
To assess the spatial organization of the perivascular cell clusters, an electron microscopy analysis of GFP-positive cells was carried out. A week after FGF-2 (GFP)-transduced NPC transplantation, the animals were deeply anesthetized with Nembutal (Abbott, North Chicago, IL, USA) and transcardially perfused with 0.9% saline followed by 2% paraformaldehyde (Sigma-Aldrich) and 0.2% glutaraldehyde (Sigma-Aldrich). Brains were then washed in 0.1 M of PBS. Horizontal 150-μm sections were cut on a vibratome.
The sections were incubated in goat anti-GFP antibody (1:500; Novus Biologicals, LLC). A biotinylated secondary anti-goat antibody (1:200; DakoCytomation) was used for visualization of the primary antibody. Subsequently, the avidin–biotin complex (ABC) method was carried out, and the peroxidase reaction was developed in 3,3′-diaminobenzidine (DAB; Sigma-Aldrich).
Immunostained sections were postfixed in 1% OsO4 (Sigma-Aldrich), dehydrated in a graded series of ethanol, and embedded in Epon 812 (Fluka, Buchs, Switzerland). Semithin sections were cut with a glass knife and stained lightly with 1% toluidine blue (Sigma-Aldrich). For the identification of individual GFP-labeled cells, ultrathin sections were cut with a diamond knife, stained with uranyl acetate (Sigma-Aldrich) and lead citrate (Sigma-Aldrich), and examined with an electron microscope JEM-100CX (JEOL, Tokyo, Japan).
Statistical Analysis
Student t-tests were carried out using the software Minitab 13.32 (Minitab Inc., State College, PA, USA). Data are given as mean ± standard error of the mean (SEM).
Results
NPC Primary Culture From Newborn Rat SVZ
In order to prepare cell suspensions for transplantation, SVZ cells were isolated from P0 rats and cultured in the presence of FGF-2 as previously described 31 . At day 3 in vitro (DIV3), NPCs had round or bipolar morphology (Fig. 1A), and the vast majority (97%) of cells expressed immature markers including nestin and epidermal growth factor receptor (EGFR) (Fig. 1B), which is characteristic in transit-amplifying type C cells in the SVZ 32 . At DIV3, cultures were transduced with the FGF-2 lentiviral vector. At DIV5, NPCs formed a monolayer, and they were trypsinized and resuspended in Neurobasal medium for transplantation.

Characterization of NPCs in culture and after transplantation into the somatosensory cortex of P3 rats. (A) Phase-contrast images of neural progenitors at day 3 in culture. (B) The majority of cells were immunopositive for nestin (green) and EGFR (red). Cell nuclei were counterstained with Hoechst 33342 (blue). (C) Epifluorescent images of coronal cortex sections without immunohistochemical staining. The majority of transplanted FGF-2-transduced GFP-positive NPCs (FGF-2 + GFP-NPCs) (green) formed clusters around the blood vessels (red dashed lines). Scale bars: 20 μm.
The Spatial Organization of Grafted Cells in Relation to the Blood Vessels
To study the relationship of grafted FGF-2-transduced GFP-positive cells (FGF-2 + GFP-NPCs) with the vasculature, we stereotaxically injected FGF-2 + GFP-NPCs into the somatosensory cortex of P3 rats. One and 2 weeks after transplantation, rats were intracardially perfused, and 40-μm-thick sections were processed for immunohistochemistry and analyzed by confocal microscopy.
Before immunohistochemical staining, coronal sections were checked for the presence of GFP-positive cells in the cortex using epifluorescent microscopy. This analysis revealed that the majority of grafted FGF-2 + GFP-NPCs were found clustered adjacent to blood vessels in the somatosensory cortex (Fig. 1C). This confirms the results of our previous publication 28 .
To investigate the proliferative activity of FGF-2 overexpressing cells, the S phase marker bromodeoxyuridine (BrdU) was intraperitoneally injected twice daily 2 days before sacrifice (Fig. 2A). Quantitative analysis showed that 2 weeks after transplantation 68.5% ± 3.2% of FGF-2-NPCs associated with blood vessels were BrdU positive compared with only 36.4% ± 8.7% for transplanted NPCs that were not associated with the vasculature (Fig. 2B). Therefore, FGF-2-transduced cells associated with vessels remained proliferative for at least 2 weeks after transplantation.

GFP-positive transplanted NPCs associate with the vasculature. (A) The clusters composed of dividing BrdU-positive (white) NPCs (green) associated with blood vessels (red). (B) Graph showing the proportion of BrdU-positive cells in cell clusters associated with vessels (vs) compared with cells that did not associate (no vs) at 2 weeks after transplantation. Error bars indicate SEM. ∗p < 0.05, t-test. (C) Desmin-positive pericytes (white) wrapping around RECA-positive vessels (red). (D) Confocal image shows the gap (indicated by red arrows) between FGF-2 + GFP-NPCs (green) and RECA-1-positive endothelial cells (red). Cell nuclei were counterstained with Hoechst 33342 (blue). (E–G) Confocal images of GFP-positive grafted cells contacted different types of blood vessels: arterioles (E), capillaries (F), and venules (G). Red, RECA-1; green, FGF-2-NPCs; white, desmin. Scale bars: 20 μm.
To further characterize perivascular clusters of grafted cells, we carried out immunofluorescence staining for desmin, a marker for pericytes. It is known that arterioles have a high density of circumferentially oriented pericytes to withstand blood pressure, whereas venules, like capillaries, exhibit irregularly arranged pericytes with multiple processes to prevent backflow of blood 33 . We used these criteria to identify vessel types. Immunohistochemical staining for desmin and RECA-1 revealed that venules were partly covered by desmin-positive pericytes whereas arterioles were entirely covered by these cells (Fig. 2A).
The average diameter of the vessels contacted by GFP-positive clusters of grafted cells (more than 3 GFP/BrdU-positive cells) was 26.89 ± 4.01 μm (Fig. 2B). All of the GFP-positive clusters (n = 65) were in contact with venules, but not with arterioles and capillaries (Fig. 2C–E).
The immunofluorescence analysis revealed that grafted FGF-2-NPCs were not in direct contact with endothelial cells, and we systematically observed a gap between GFP-positive cells and RECA-1-positive endothelial cells lining the cerebral vasculature (Fig. 2B).
To identify potential structures between grafted NPCs and endothelial cells, immunofluorescence staining for GFAP, aquaporin-4 (marker for astrocytic endfeet), and desmin (marker for pericytes) was carried out. As expected, the immunofluorescence analysis revealed that the blood vessels in the somatosensory cortex were entirely covered by GFAP-positive astrocytic endfeet (Fig. 3A).
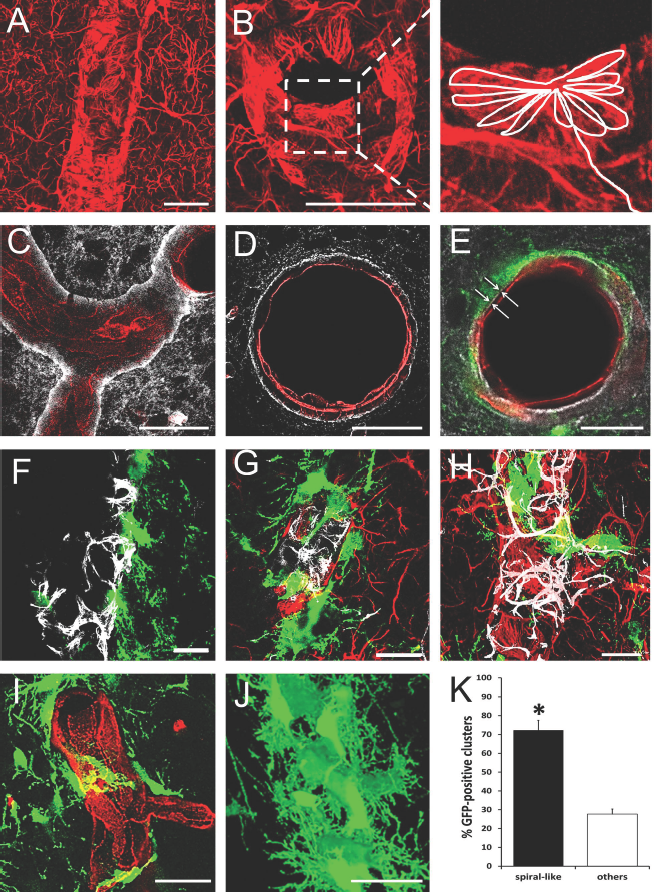
FGF-2-NPCs made contact with blood vessels at sites with astrocyte coverage, but no pericytes. (A, B) Confocal image of blood vessel covered with GFAP-positive astrocytic endfeet: longitudinal (A) and transverse (B) sections. (B) Right image: magnified view of the dashed square on (B); left image, showing schematic drawing of rosette-like astrocytic endfeet. (C, D) AQP-4-positive astrocytic endfeet (white) wrapping around RECA-1-positive blood vessel (red): longitudinal (C) and transverse (D) sections. (E) GFP-positive cells (green) in contact with RECA-1-positive blood vessel (red) via AQP-4-positive astrocytic endfeet (white) (indicated by white arrows). (F–H) FGF-2-NPCs (green) in contact with the blood vessel at sites with astrocyte coverage (red) (G, H), but no pericytes (white) (F). (I) Spiral-like FGF-2 + GFP-positive clusters (green) wrap around RECA-1-positive vessel (red). (J) 3D reconstruction of spiral-like GFP-positive perivascular clusters. Scale bars: 20 μm. (K) Graph showing the percentage of spiral-like and nonspiral (others) GFP-positive clusters (error bars indicate SEM, ∗p < 0.01, t-test).
We found that cerebral perivascular GFAP-positive endfeet display characteristic anatomical specializations displaying well-defined rosette-like structures abutting on the endothelial cells (Fig. 3B). Several astrocytes appeared to contact one endothelial cell, and in this case, rosette-like astrocytic endfeet were intermingled with each other (Fig. 3B) 34 . We also performed immunostaining for aquaporin-4 (AQP-4), a water channel highly enriched in astrocytic endfeet at the brain–blood barrier (BBB) 35 .
Confocal analysis of AQP-4-immunostained brain slices confirmed that blood vessels in the rat postnatal somatosensory cortex were completely ensheathed by AQP-4-positive astrocytic endfeet (Fig. 3C and D). Furthermore, FGF-2-transduced GFP-positive cells were associated with blood vessels via AQP-4-positive astrocytic endfeet (Fig. 3E).
Immunostaining for desmin revealed that FGF-2 + GFP-NPCs mainly contacted blood vessels at sites with astrocyte coverage but no pericytes (Fig. 3F–H).
It is known that pericytes wrap around vessels in spiral-like fashion 36 . To show that FGF-2-transduced GFP-positive NPCs formed spiral-like clusters around vessels between the pericytes, we took serial confocal images of a z-stack with a step size of 0.25 μm in depth (Fig. 3J). The criterion for spiral-like cluster was the corkscrewlike spiraling of the GFP-positive cells around the vessel (Fig. 3I and J). Quantitative analysis (65 GFP-positive clusters, n = 7 animals) revealed that 72.3% ± 5.2% of analyzed FGF-2 + GFP-positive clusters contacted the vessels in a spiral-like manner (p < 0.01, t-test) (Fig. 3K).
Ultrastructural Analysis of Perivascular Cell Clusters
Next we examined the perivascular clusters at the ultrastructural level. For this purpose, 150-μm sections were processed for GFP immunostaining prior to being embedded in Epon resin. Followed by biotinylated secondary antibody, the ABC method was carried out, and the peroxidase reaction was developed in DAB. It was easy to visualize GFP/DAB-positive cells because they had dark cytoplasm (Fig. 4). DAB-positive cells were elongated, and their cytoplasm contained mitochondria and no bundles of intermediate filaments. The nuclei had invaginations and mostly lax chromatin. The ultrastructural analysis confirmed that grafted GFP/DAB-positive cells mainly come in close contact with venules at sites with astrocytic endfeet (Fig. 4A).

Electron microscopy images of the GFP/DAB-positive cells in contact with blood vessels. (A) DAB-positive cell (green) in contact with astrocytic endfeet (blue). Endothelial cells are red. (B) DAB-positive cell in juxtaposition with the basal lamina. (C) GFP/DAB-positive cells in contact with branches of thick, extravascular basal lamina. Green, GFP/DAB-positive cells; red, endothelial cells; blue, astrocytic endfeet. L, lumen; arrows, basal lamina. Scale bars: 1 μm. (D) Graph showing the percentage of DAB-positive cells contact with the endothelial cells via astrocytic endfeet (as) and in juxtaposition with the basal lamina (bl). A total (100%) of 140 DAB-positive cells were counted (error bars indicate SEM, ∗p < 0.001, t-test).
Quantification was done by first counting the number of DAB-positive cells associated with blood vessels and then by determining the percentage of DAB-positive cells in contact with the endothelial cells via astrocytic endfeet and in juxtaposition with the basal lamina. Quantitative analysis (140 cells, n = 5 animals) showed that the large majority (88.5% ± 7.4%) of cells contacted astrocytic endfeet and only 12.5% ± 3.2% of GFP/DAB-positive cells (p < 0.001, t-test) (Fig. 4D) were in juxtaposition with the basal lamina (BL) of endothelial cells of venules (Fig. 4B).
The extracellular matrix (ECM)-rich basal lamina displayed a characteristic electron-dense pattern easily distinguished by electron microscopy 37 . Interestingly, 27.85% of vessels were in contact with transplanted cells (n = 140) and featured increased basal lamina-forming fractone-like structures (Fig. 4C). We also found that these labyrinthine-like structures enclosed astrocyte endfeet.
Discussion
The anatomical phenomenon (perivascular cell cluster formation) described here shows for the first time, to our knowledge, that grafted FGF-2-NPCs preferentially make contact with venules at sites with astrocytic endfeet and avoided contact with pericytes. The cell composition and organization of the perivascular ectopic clusters of grafted cells were similar to native neurovascular niches and contained stem cells, an extensive basal lamina, ECM, and vasculature3,7,38,39. The cerebral vasculature is a key, integral component of stem cell niche4,20. Stem cells receive spatial cues and regulatory signals from different elements of the vasculature that stimulate their self-renewal and inhibit differentiation 17 . Endothelial cells produce different mitogens and differentiation and survival factors including bFGF, IGF-1, VEGF, platelet-derived growth factor (PDGF), interleukin-8 (IL-8), and BDNF20,40,41.
There could be at least two possible reasons that grafted FGF-2-NPCs preferentially made contact with venules. First, the transplanted progenitor cells use these blood vessels as scaffolds for their migration. Kojima and colleagues showed that SVZ-derived NPCs migrate along blood vessels from the SVZ toward the ischemic area, and they suggested that the vasculature plays a significant role as a scaffold for NPC migration 42 . The second reason is that NPCs are known to prefer hypoxic conditions 43 . In various studies, it has been shown that NPCs in culture under mild hypoxia have strongly enhanced self-renewal and proliferation by activation of molecular pathways that regulate Wnt/β-catenin, octamer-binding transcription factor 4 (Oct-4), and Notch signaling44–47. Interestingly, severe hypoxia (<1% O2) arrests the proliferation of stem cells and drives them into quiescent and even apoptotic states. Conversely, atmospheric culture conditions (20% O2) lead stem cells to differentiate43,48,49.
It has also been demonstrated that stem cells in the SVZ neurovascular niche often make contact with the vasculature at regions of blood vessels lacking AQP-4-positive astrocytic endfeet and pericyte coverage, giving them direct access to vascular and blood-derived signals4,35. Such interrupted AQP-4 staining is unique for the SVZ, as blood vessels from the cortex or striatum are completely ensheathed by astrocytic endfeet 4 . Our confocal analysis of AQP-4 immunostaining of the rat postnatal somatosensory cortex confirmed that blood vessels were completely ensheathed by AQP-4-positive astrocytic endfeet (Fig. 3C and D). Thus, GFP-positive transplanted NPCs were associated with blood vessels via AQP-4-positive astrocytic endfeet where there were no desmin-positive pericytes. Since NPCs are in direct contact with astrocytes, it is likely that local astrocytes play an essential role in determining NPC fate. It is known that astrocytes produce soluble and membrane-bound factors, which could counteract stem cell differentiation and support proliferation or maintenance of pluripotency 1 .
An integral part of the stem cell niche is the basal lamina (BL) and extracellular matrix components that anchor the cells and provide essential signals for NPCs 18 . The adult SVZ stem cell niche contains collagen-1, tenascin-C, heparan sulfate proteoglycans, chondroitin sulfate proteoglycans, and integrins, which are involved in both proliferation and migration18,50–53. For example, heparan sulfate proteoglycans bind growth factors (EGFs, FGFs, IGF-II, PDGF-AA, and VEGF), morphogens and mitogens (BMP-2, -4, Shh, and Wnts), chemokines, and cytokines18,50,52,54,55. It has been shown that in the neurovascular niche a BL forms branched labyrinthine structures known as fractones7,18. We have also shown in these experiments that transplanted FGF-2-NPCs stimulated formation of fractone-like structures around the vasculature (Fig. 4C) that could bind factors 18 and might provide spatial cues within the ectopic stem cell niche.
Here we investigated the architecture and relationship of FGF-2-overexpressing transplanted cells with the vasculature in the cerebral cortex. This is a key issue if we want to find ways to construct ectopic neurogenic niches that will be similar to the native ones and be able to transiently restore the neurogenic potential of injured brain tissue. Our findings could open new possibilities to induce ectopic neurogenesis and to generate a robust source of immature neurons for brain repair.
Conclusions
In conclusion, our results provide a new insight into the relationship of grafted FGF-2-secreting NPCs and the vascular environment. Creating an artificial niche can be a useful strategy to overcome a low regenerative potential of the cerebral cortex by maintaining NPCs in a migratory and proliferative state and by increasing the pool of immature neurons available for brain repair.
Footnotes
Acknowledgements
This work was supported by the Swiss National Foundation No. IZ73Z0_128015 to Jozsef Z. Kiss. The authors declare no conflicts of interest.
