Abstract
Traumatic brain injury (TBI) is a common cause of mortality and long-term morbidity in children and adolescents. Posttraumatic stress disorder (PTSD) frequently develops in these patients, leading to a variety of neuropsychiatric syndromes. Currently, few therapeutic strategies are available to treat juveniles with PTSD and other developmental neuropsychiatric disorders. In the present investigation, postnatal day 14 (P14) Wistar rats were subjected to TBI induced by a controlled cortical impact (CCI) (velocity = 3 m/s, depth = 2.0 mm, contact time = 150 ms). This TBI injury resulted in not only cortical damages, but also posttrauma social behavior deficits. Three days after TBI, rats were treated with intracranial transplantation of either mouse iPSC-derived neural progenitor cells under normal culture conditions (N-iPSC-NPCs) or mouse iPSC-derived neural progenitor cells pretreated with hypoxic preconditioning (HP-iPSC-NPCs). Compared to TBI animals that received N-iPSC-NPCs or vehicle treatment, HP-iPSC-NPC-transplanted animals showed a unique benefit of improved performance in social interaction, social novelty, and social transmission of food preference tests. Western blotting showed that HP-iPSC-NPCs expressed significantly higher levels of the social behavior-related genes oxytocin and the oxytocin receptor. Overall, HP-iPSC-NPC transplantation exhibits a great potential as a regenerative therapy to improve neuropsychiatric outcomes after juvenile TBI.
Keywords
Introduction
Traumatic brain injury (TBI) is a leading cause of human death and disability in the US and worldwide (9,41). TBI among the juvenile population occurs at a rate of over 1 per 1,000 and is associated with an elevated risk of long-term morbidity (30). Although the exact etiology of the neuropsychiatric deficits observed post-TBI remains unclear, recent investigations have demonstrated that early childhood TBI can result in cognitive and social developmental deficits, potentially resulting in the development of depression (3,44). A number of pathogenic triggers, such as pain, gestational inflammation, encephalopathy, perinatal asphyxia, and neurodegenerative insults, may cause developmental cognitive dysfunction, social behavior problems, and neuropsychiatric deficits in children and young adults (1,5,20,23,46,53). The cognitive and social developmental problems associated with brain trauma can persist for many years following injury, from early life to young adulthood (4,17). Unfortunately, despite the fact that many investigations have explored pathological signaling pathways and cell death mechanisms associated with TBI, current TBI therapeutic strategies are limited (22,31).
Cell-based regenerative therapy, which utilizes stem cells/progenitors including neural stem cells (NSCs) and induced pluripotent/embryonic stem cell-derived neural progenitor cells (iPSC-NPCs and ESC-NPCs), promises multiple benefits for animals with brain injury such as stroke and TBI (24,34). Cell transplantation provides neurotrophic support and the potential for cell replacement in injury and peri-injury regions. It may also promote endogenous repair following brain trauma (6,37,49). There are primarily two possible goals for cell-based regenerative therapy: 1) repair of damaged neurovascular networks with functionally differentiated cells via transplantation and/or stimulated regenerative activities and 2) restoration of impaired brain function. Whether transplanted cells can attenuate post-TBI neuropsychiatric alterations has rarely been investigated.
iPSC-NPCs are considered an appropriate cell source for transplantation and have shown encouraging effects on central nervous system (CNS) diseases in animal models. Our previous reports have shown that iPSC-NPCs exhibit regenerative activity following transplantation into the ischemic brain of adult and neonatal animals (7). To improve the survival and regenerative properties of transplanted cells, we have previously developed an effective strategy of preconditioning cells with sublethal hypoxia before transplantation (14). Additionally, hypoxic preconditioning has been shown to induce differentiation of neuron-like cells from iPSC-NPCs. In the present investigation, the ability of iPSC-NPCs pretreated with regular or hypoxic culture conditions to attenuate neuropsychiatric deficits associated with TBI in a juvenile model of postnatal day 14 (P14) rat pups was examined.
Materials and Methods
TBI Model of Juvenile Rats
Male P14 Wistar rats (Charles River Laboratories, Wilmington, MA, USA) were anesthetized using 1.5% isoflurane (Piramal Healthcare, Andhra Pradesh, India). Pups were positioned in a stereotaxic frame (Smiths Medical, Arden Hills, MN, USA), and after cutting a midline skin incision, a 3.5-mm circular craniotomy was performed midway between lambda and bregma, 2.0 mm to the right of the central suture using an electric drill. Controlled cortical impact (CCI) was induced with an electric impact device (PCI3000 precision impactor; Hatteras Instruments, Cary, NC, USA) as previously described (19,26). The impact (velocity = 3 m/s, depth = 2.0 mm, contact time = 150 ms) led to evident damage in the cortical regions, specifically the sensorimotor cortex (19,26). Animal procedures were completed in accordance with protocols approved by Emory University's Institutional Animal Care and Use Committee (IACUC). All surgeries were performed by the same animal surgeon. For immunostaining and Western blot analysis, 6–8 animals were utilized per group; for behavior tests, 12–16 animals per group were utilized in each test.
Cell Preparation
Mouse iPSC-NPCs were differentiated from WP5 iPSCs (derived from mouse embryonic fibroblasts; Stemgent Inc., Cambridge, MA, USA) as previously described (35). The cells used in this study were harvested for transplantation during passages 18 to 25. WP5 iPSCs were cultured in Dulbecco's modified Eagle's medium (DMEM; Cellgro, Manassas, VA, USA) supplemented with 10% fetal bovine serum (FBS; Sigma-Aldrich, St. Louis, MO, USA), 2 mM glutamine (Life Technologies, Carlsbad, CA, USA), 0.1 mM nonessential amino acids (Life Technologies), 1 mM sodium pyruvate, 55 μM 2-mercaptoethanol, 2,000 U/ml leukemia inhibitory factor (LIF; Millipore, Billerica, MA, USA), and 100 U/ml penicillin/streptomycin (pen/strep; Sigma-Aldrich). For the cell tracking study, mCherry-expressing iPSCs were produced by transfection using a CAG–mCherry-woodchuck hepatitis virus posttranscriptional regulatory element-internal ribosome entry sequencepuromycin (CAG-mCherry-WPRE-IRES-PURO) plasmid (Addgene, Cambridge, MA, USA) and subjected to cell population purification procedures by puromycin (Sigma-Aldrich). These mCherry iPSCs were cultured in the same medium as WP5 iPSCs. Cultures were maintained in a 37°C incubator containing 5% carbon dioxide. All cells used in this study were harvested for transplantation after a designated “4–/4+” retinoic acid (RA, 1 μM; Sigma-Aldrich) protocol (35).
Hypoxic Preconditioning and Administration of iPSC-NPCs
Hypoxic preconditioning (HP) of stem cells prior to transplantation was performed to enhance their survival and regenerative properties (21,42,45). For hypoxic exposure, the oxygen in the hypoxia chamber (MedRep, Newnan, GA, USA) was maintained at 0.1–0.3% with a residual gas mixture composed of 5% carbon dioxide balanced with nitrogen for 8 h prior to harvesting (right after 4–/4+ retinoic acid induction protocol). There was approximately a 1-h reoxygenation period during harvest before transplantation. Control iPSC-NPCs were cultured in normoxic conditions (21% oxygen). The iPSC-NPCs were rinsed with phosphate-buffered saline (PBS; Sigma-Aldrich) and harvested by trypsinization using 0.25% trypsin/0.1% ethylenediaminetetraacetic acid (EDTA; Life Technologies).
Animals were transplanted with iPSC-NPCs under anesthesia. Three days after TBI induction, all animals were anesthetized under 2% isoflurane. The skull area over the pericontusion regions was cleaned and removed. Traumatic tissue was visually identified through the skull. Intracranial delivery of the cells was performed as previously described with modifications for the trauma model (7). HP-iPSC-NPCs, control N-iPSC-NPCs (4 × 105 cells in 4 μl for each treatment), or an equal amount of vehicle was delivered into the contusion and pericontusion regions of each animal. Cells were delivered through injections at four sites in the pericontusion region using a Hamilton syringe (Reno, NV, USA). Control animals received injections of DMEM-Sato Base Growth Media (Sigma-Aldrich) at four sites as a vehicle control. The incision was closed with surgical adhesive (Vetbond, Maplewood, MN, USA).
Astrocyte and iPSC-NPC Cocultures
A mouse astrocyte layer with high confluence (>90%) was used to support maturation of iPSC-NPC-derived neurons for in vitro characterization. Primary cultures of cerebral cortical astrocytes from newborn C57BL/6 mice (within 24 h of birth; Charles River Laboratories, Wilmington, MA, USA) were prepared according to previous publications with minor modifications to culture medium (16). Briefly, meninges-free cerebral cortices were cut into small cubes and suspended in DMEM (Cellgro) supplemented with 15% FBS (Sigma-Aldrich) and 3.5 mM glucose (Sigma-Aldrich). After dissociation using surgical steel scalpels (Swann-Morton, Sheffield, UK), the cell suspension was passed through both 70-μm (Corning Inc., Corning, NY, USA) and 10-μm nylon filters (Sterlitech, Kent, WA, USA). Cells were then seeded 3 × 105 cells/ml in 35-mm tissue culture dishes (Corning) for characterization and 24- or 48-well plates (Corning) for cell survival and viability tests. Cultures were incubated at 37°C with 5% CO2. Culture medium was changed twice per week with DMEM containing 15% FBS for the first 2 weeks and 8% FBS afterward. The astrocyte layer was ready for iPSC-NPC coculture after 3 weeks. After seeding iPSC-NPCs, the medium was changed from the astrocyte culture medium to the Sato medium for NPCs (52). Immunostaining was performed on the mCherry-labeled cells as previously described (7). We previously characterized the neural differentiation of these iPSCs using electrophysiological recording and immunostaining, showing the expression of the neural precursor/neuronal marker β-tubulin III, neurofilament, neuronal nuclei, synapsin-1, synaptosomal-associated protein 25 (SNAP25), and some trophic factors including fibroblast growth factor (FGF), glial cell line-derived neurotrophic factor (GDNF), brain-derived neurotrophic factor (BDNF), and others (7). Along the neuronal differentiation lineage in vitro, differentiated cells fired evoked repetitive action potentials (7).
Immunohistochemical Staining and Cresyl Violet Staining
For cell tracking study and histopathological examination by immunostaining, animals were sacrificed, and coronal sections were prepared using the rodent brain matrix (Harvard Bioscience, Holliston, MA, USA). Sections were then frozen on dry ice and preserved at −80°C. Frozen brain sections were sliced into 10-μm coronal sections using a cryostat (Leica CM 1950; Leica Microsystems, Buffalo Grove, IL, USA). The sections were air dried and fixed with 10% buffered formalin (Thermo Fisher Scientific, Waltham, MA, USA). Brain sections were then submerged in an ethanol/acetic acid solution (2:1) (Sigma-Aldrich) for 10 min, washed three times with PBS, and incubated with 0.2% Triton X-100 (G-Biosciences, St. Louis, MO, USA) for 10 min. Slides were then blocked with 1% fish gelatin (Sigma-Aldrich) in PBS for 1 h at room temperature and incubated with PBS-diluted mouse anti-neuronal nuclei (NeuN; 1:400; Millipore) and rabbit anti-mCherry (1:500; Abcam, Cambridge, UK) primary antibodies at 4°C overnight. Primary antibodies were removed by rinsing with PBS and replaced with secondary antibodies conjugated to Alexa Fluor 488 (1:200; Life Technologies) or cyanine 3 (Cy3; 1:1,000; Life Technologies) for 1 h at room temperature before rinsing again with PBS. Hoechst 33342 (1:20,000; Life Technologies) was used to stain nuclei. After a final rinse, slides were mounted with ProLong Antifade mounting medium (Life Technologies).
Terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining kit (Promega, Madison, WI, USA) was used to detect fragmented DNA and cell death by costaining with Hoechst 33342 or NeuN in pericontusion regions. To confirm the contusion location and study the histopathological changes, cresyl violet (Nissl; Sigma-Aldrich) staining was performed. Sections were then fixed with a 1:1 mixture of 10% formalin (Thermo Fisher Scientific) and acetic acid (VWR International, Suwanee, GA, USA) for 10 min. After washing with distilled water for 5 min, slices were stained with a working solution containing buffer solution [0.1 M acetic acid and 0.1 M sodium acetate (Sigma-Aldrich); volume ratio = 94:6] and cresyl violet acetate at a ratio of 5:1. The sections were then dehydrated in 100% ethyl alcohol and mounted. The cortical damage was then examined after Nissl and TUNEL staining on the sections.
Western Blotting Analysis of Social Behavior-Related Genes
After sacrifice, animals were subjected to transcardial perfusion using PBS (pH 7.4). Pericontusion brain tissue was lysed in a buffer containing 0.02 M Na4P2O7, 10 mM Tris-HCl (pH 7.4), 100 mM NaCl, 1 mM EDTA (pH 8.0), 1% Triton, 1 mM ethylene glycol tetraacetic acid (EGTA), 2 mM Na3VO4, and a protease inhibitor cocktail (all from Sigma-Aldrich). The supernatant was collected after centrifugation at 15,000 × g for 15 min at 4°C. Protein concentration was determined with a bicinchoninic acid assay (Pierce Biotechnology, Rockford, IL, USA). Equivalent amounts of protein were separated by molecular weight on a sodium dodecyl sulfate (SDS)-polyacrylamide gradient gel (Sigma-Aldrich) and then transferred to a polyvinylidene difluoride (PVDF) membrane (Thermo Fisher Scientific). The blot was incubated in 5% bovine serum albumin (BSA; Sigma-Aldrich) for 1 h and then treated with primary antibodies overnight at 4°C. Primary antibodies used and their associated dilutions are as follows: rabbit anti-oxytocin (Abcam) at 1:500, goat anti-oxytocin receptor (Santa Cruz Biotechnology, Dallas, TX, USA) at 1:500, and mouse anti-actin (Sigma-Aldrich) at 1:5,000. After washing with Tris-buffered saline/Tween 20 (TBST; Sigma-Aldrich), membranes were incubated with AP-conjugated secondary antibodies (GE Healthcare, Piscataway, NJ, USA) for 2 h at room temperature. After completion of a final wash with TBST, signal bands were detected with bromochloroidolylphosphate/nitroblue tetrazolium (BCIP/NBP) solution (Sigma-Aldrich). The band intensity was measured using ImageJ (NIH, Bethesda, MD, USA) and normalized to actin.
Sensorimotor Functional Tests
Adhesive removal test, corner test, and cylinder test were performed to measure deficits in the sensorimotor function or locomotor asymmetry function for every set of TBI animals.
Adhesive Removal Test
The adhesive removal test measures sensorimotor function as previously described (27) with modifications for rat pups. Briefly, a small adhesive dot was placed on each forepaw, and the amount of time (seconds) needed to contact and remove the tape from each forepaw was recorded. Different from our previous mice protocol for the testing of stroke animals, rat pups were not trained before TBI induction in this study. P31 animals were tested at 17 days after TBI by an investigator who was blinded to the experimental groups. The test was performed four times per rat, and the average time was used in the analysis. In this behavioral study, 12–16 animals were randomly assigned to each group before surgery and transplantation.
Corner Test
The corner test for the whisker sensorimotor function analysis was performed as described previously (8) with slight modifications for rat pups. Two cardboard plates (30 cm × 20 cm × 0.3 cm) were attached at a 30° angle from each other and placed in a home cage. Each testing rat was placed between the two plates and allowed to freely move to the corner where the two plates meet. The numbers of right and left turns were counted. Ten trials were performed for each rat, and the total turns were summarized for comparisons.
Cylinder Test
A unilateral injury to the motor cortex results in an asymmetry in the forelimb used to support the body during rearing, which can be measured using the cylinder test. The rats were placed in a glass cylinder (9.5 cm diameter and 11 cm height), and the number of times each forelimb or both forelimbs was used to support the body on the wall of the cylinder was counted for 5 min. The animals were evaluated in P31 rats at 17 days after TBI or sham procedure. Two mirrors were placed behind the cylinder to view all directions. The numbers of impaired and nonimpaired forelimb contacts were counted and calculated as a percentage of total contacts.
Social Behavior Tests
Social Interaction Test
Animals were placed into a test cage, and social interaction was recorded on day 17 post-TBI. Social sniffing was defined as the “experimental” rat (one male Wistar rat) sniffing any body part of its test partner (another male nonlittermate Wistar rat). Social grooming was defined as the experimental rat licking and/or chewing the fur of its test partner while placing its forepaws on its partner's back or neck. Following/chasing behaviors were also recorded and defined as any time when the experimental rat walked or ran in the direction of its test partner, who either stayed in place or moved away in response. The duration of active social interaction was accessed during a 10-min testing period.
Sociability and Social Novelty Test
To test general sociability and social novelty, a P31 rat was placed into a plastic three-chambered box (Clever Sys Inc., Reston, VA, USA) and allowed to explore all of the chambers for 10 min, after which the rat completed two 10-min tests. During the first 10-min testing period, a nonlittermate P31 rat (former stranger) locked in a wire cage was placed into the left chamber, and the “experimental” rat was allowed to explore all three chambers. Exploration during this period was used to quantify sociability. During the second testing period, a different locked nonlittermate P31 rat (new stranger) was placed into the right chamber. Using a top-view behavior monitoring system and an automated behavior recognition software (Clever Sys Inc.), the time spent with the former stranger rat and the new stranger rat was recorded, and the results were used to access the animal's social novelty.
Social Transmission of Food Preference (STFP) Test
The STFP test is a test of olfactory memory and social transmission in mice and rats (12,15,54). Two “demonstrator” rats were removed from each test cage and individually housed overnight with water but without food (18 h). The demonstrator rats were then placed into cages containing cocoa-flavored food in small glass jars (3.9 cm diameter, 3.4 cm high). The glass jars were set in shallow Petri dishes so that food scattered by digging was retained. Demonstrator rats were left to eat the cued food (cocoa) for 1 h. Dishes were weighed pre- and posttesting to access the amount of food consumed. The demonstrator rat was then placed in an experimental cage. Individually, an “observer” rat from the same home cage as the demonstrator rats was placed in the cage containing the demonstrator rat and left there for 1 h. The observer rat was then removed. All observer rats were then returned to their home cage, and demonstrator rats were individually housed. Six hours after the social interaction session, the observer rat was food deprived for 18 h (overnight). The following morning, each rat was placed individually in a clean cage (45 cm × 28 cm × 12 cm) containing two dishes in either back corner of the cage. The dishes contained either cocoa-flavored food (cued) or a normal diet (noncued). Rats were allowed to eat for 1 h. Dishes were weighed pre- and posttesting to determine the amount of food consumed. Food preference was calculated as the amount of cued, noncued, and total food (noncued food plus cued food) consumed.
Statistical Analysis
For comparison between two groups, we used the Student two-tailed t-test. Graph Prism version 5.0 was used to make graphs and to perform statistical analysis. One-way analysis of variance (ANOVA) was used for data analysis, and multiple comparisons (four groups) were corrected with Bonferroni's test for pairwise comparisons. Significance was assumed at a value of p < 0.05 in all statistical analyses. Randomization was performed, and the sample size was further determined using power analysis (Power and Precision 4; Biostat, Inc., Englewood, NJ, USA).
Results
Tracking Transplanted iPSC-NPCs in the Post-TBI Brain
The juvenile TBI model was induced using a CCI targeting the right sensorimotor cortex in P14 rats. The impact resulted in extensive cortical damage as revealed by Nissl and TUNEL staining, leading to sensorimotor functional deficits as shown in our previous reports (19). For the treatment of TBI, we used genetically modified iPSCs that constitutively express mCherry with red fluorescence, thereby facilitating the tracking of these cells after transplantation (Fig. 1A, B). After the induction of neural differentiation using a 4-/4+ retinoid acid protocol (35), the mouse iPSC-NPCs differentiated into neuron-like cells after 8 days in vitro (Fig. 1C–E). iPSC-NPCs were either primed with sublethal hypoxia (0.1–0.3% oxygen for 8 h) or remained in normoxic conditions during the last day of the 4–/4+ protocol. Cells were intracranially transplanted into the pericontusion and contusion region 3 days post-TBI. Transplanted cells survived in the traumatic core and the pericontusion region and could be detected as mCherry positive 14 days following transplantation (Fig. 2).

iPSC-NPCs in vitro expansion and differentiation. Phase contrast (A) and fluorescent (B) images of mouse induced pluripotent stem cells (iPSCs) cultured and expanded without feeder cells. (B) iPSCs were labeled with fluorescent mCherry (red). Scale bar: 20 μm. (C) Diagram for the procedure timeline of iPSC neural induction in vitro. Phase contrast (D) and fluorescent (E) images of iPSC-neural progenitor cell (NPC)-derived neuron-like cells on a mouse astrocyte layer. Scale bar: 10 μm. RA, retinoic acid; HP, hypoxic preconditioning.
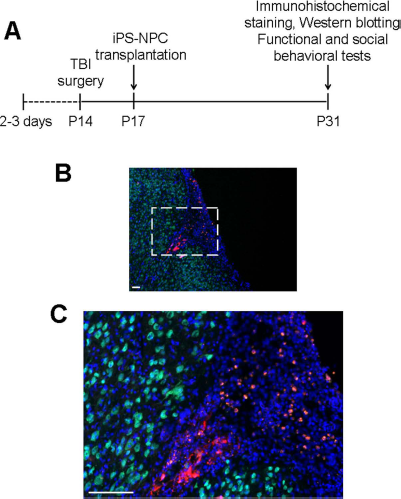
Tracking iPSC-NPCs after transplantation into the post-TBI brain. (A) The experimental timeline for traumatic brain injury (TBI) and post-TBI procedures/examinations of juvenile rats. (B) Representative image showing transplanted cells with mCherry labeling in the contusion and pericontusion regions. Red: mCherry; green: neuronal nuclei (NeuN) of mature neurons; blue: Hoechst 33342. Scale bar: 100 μm. (C) Enlarged image from the framed area in (B). Red: mCherry; green: NeuN; blue: Hoechst 33342. Scale bar: 100 μm.
Transplantation of iPSC-NPCs Improved Social Activities After TBI
In the TBI model, the traumatic insult led to damage in the ipsilateral sensorimotor cortex, and consequently, animals were found to have significant sensorimotor deficits at 7 days post-TBI (age of P21) (19). In the present investigation at 17 days post-TBI (P31), most of the functional deficits were no longer detectable, presumably due to spontaneous recovery via endogenous neuroregeneration and neuroplasticity. There was no significant difference among TBI control and cell transplantation groups in these tests (Fig. 3). On the other hand, when social activities were examined in these animals, significant social behavior dysfunctions were detected 17 days after TBI, and iPSC-NPC transplantation showed significant benefits of correcting social behaviors (Fig. 4). Interestingly, transplantation of HP-iPSC-NPCs provided even greater improvements in most of the social activities tested, including social following, grooming, and the total social time (Fig. 4).

Sensorimotor functional measurements after TBI and iPSC-NPC transplantation. Sensorimotor and locomotor asymmetry functional evaluations were performed in postnatal day 31 (P31) animals. In the adhesive removal test (A), TBI alone and TBI with transplantation of normal (N)-iPSC-NPCs or hypoxic preconditioned (HP)-iPSC-NPCs all showed significant and similar sensorimotor deficits on the lesioned side (left paw) compared to sham controls. In both the corner test and cylinder test (B, C), no significant difference was detected among all groups of P31 animals. Mean ± SEM; N = 12–16 per group; *p < 0.05 versus sham.
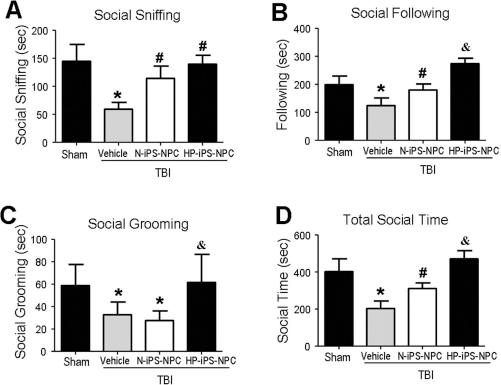
Social behavior changes after TBI and iPSC-NPC transplantation. According to the experimental timeline shown in Figure 2A, social interaction tests were performed at postnatal day 31 (P31). Transplantation of N-iPSC-NPCs or HP-iPSC-NPCs improved social behaviors including social sniffing (A) and following (B), and increased total social time (D). HP-iPSC-NPCs, however, showed superior effects on social following (B), social grooming (C), as well as total social time (D). Mean ± SEM; N = 12–16 per group; *p < 0.05 versus sham; #p < 0.05 versus TBI vehicle controls; &p < 0.05 versus N-iPSC-NPC group.
In additional sociability and social novelty tests, we assessed the rat's general sociability and interest in social novelty after TBI (Fig. 5A). Seventeen days post-TBI, vehicle-treated control animals showed a preference for staying with a nonstranger, resulting in significantly less time to stay with a stranger (Fig. 5B). HP-iPSC-NPC-treated TBI animals, on the other hand, spent longer time with stranger animals and remained in the stranger's chamber longer than TBI controls and N-iPSC-NPC-treated TBI animals (Fig. 5B, C).

Sociability and social novelty behavioral changes after TBI. The three-chamber test, which measures sociability and social novelty, was performed 17 days post-TBI. (A) During testing, a camera monitoring system was used to track the contact time of the animal in the chamber with the first and second “stranger” animals. (B) Interaction time between the testing animal with the first stranger was interpreted as sociability. This behavior was significantly reduced after TBI but increased in rats that received iPSC-NPC treatments. (C) The interaction time between the testing animal with the second stranger was summarized as social novelty. There was a significant improvement after iPSC-NPC transplantation. In both tests, transplantation of HP-iPSC-NPCs resulted in significant improvement compared to N-iPSC-NPC treatment. Mean ± SEM; N = 12–16; *p < 0.05 versus sham; #p < 0.05 versus TBI vehicle controls; &p < 0.05 versus N-iPSC-NPC group.
The social transmission of food preference (STFP) is a test that examines the combined olfactory memory and social transmission behavior of rodents (12,15,54). The behavior test requires the observer rat to detect olfactory cues on the breath of the demonstrator rat during their interaction. Subsequent food preference serves as a measure of memory for these olfactory cues that transmit social information (Fig. 6A). TBI control rats tested as observers were not able to distinguish between the food eaten by the demonstrator and the novel food (Fig. 6B), while iPSC-NPC-treated TBI rats showed an increased ability to recognize and consume the cued food. Furthermore, treatment with HP-iPSC-NPCs showed an even stronger transmitted effect on food preference than N-iPSC-NPC treatment, such that HP-iPSC-NPC-treated animals more readily recognized and consumed cued food (Fig. 6C).

Alterations of the social transmission pattern after TBI. (A) The social transmission of food preference (STFP) test was performed to test social olfactory memory after TBI in P31 rats. (B) iPSC-NPC treatment increased cued and decreased noncued food consumptions. (C) In the analysis of the ratio of consumed cued cocoa-flavored food against noncued food, both iPSC-NPC treatments increased the ratio, while the HP-iPSC-NPC treatment resulted in an even greater increase than the N-iPSC-NPC group. Mean ± SEM; N = 12–16; *p < 0.05 versus vehicle controls; #p < 0.05 versus N-iPSC-NPC group.
HP-iPSC-NPC Transplantation Increased Oxytocin/Oxytocin Receptor Following Juvenile TBI
To understand the mechanism underlying the beneficial social behavioral effects of iPSC-NPC transplantation after TBI, we analyzed the expression of social behavior genes in iPSC-NPCs. Western blot analysis showed that both oxytocin and oxytocin receptor levels were significantly increased in HP-iPSC-NPCs compared to N-iPSC-NPCs (Fig. 7A–C). More importantly, the levels of oxytocin and the oxytocin receptor were significantly higher in the pericontusion regions of the cortex where HP-iPSC-NPCs were transplanted. This upregulation of the oxytocin/oxytocin receptor system may provide a molecular mechanism for the observed social improvements (Fig. 7D, E).

Expression of oxytocin/oxytocin receptor in iPSC-NPCs and the pericontusion region. (A) Western blot analysis was applied to measure protein levels of the social behavior-associated genes oxytocin and the oxytocin receptor with and without hypoxic preconditioning (HP). Oxytocin (B) and oxytocin receptor (C) protein levels were significantly higher in iPSC-NPCs with hypoxic pretreatment and showed further increases 24 h later. *p < 0.05 versus N-iPSC-NPCs; #p < 0.05 versus 0 h HP-iPSC-NPCs. In the pericontusion region 14 days after transplantation, oxytocin (D) and oxytocin receptor (E) levels were significantly enhanced with a greater effect of the HP-iPSC-NPCs. *p < 0.05 versus vehicle controls; #p < 0.05 versus N-iPSC-NPC group. Mean ± SEM; N = 6–8.
Discussion
The present investigation examined the development and treatment of neuropsychiatric symptoms after juvenile TBI. We have shown that TBI induction in juvenile P14 rats causes several social behavior deficits. Our data suggest a therapeutic potential of cell-based therapy to treat neuropsychiatric deficits after juvenile TBI. Stem cells have been used in clinical trials to treat TBI patients (48). Although iPSCs hold great promise as a cell source for autologous replacement to treat central nervous system (CNS) disorders including ischemia (11), myocardial infarction (28), TBI (10), intracerebral hemorrhage (39), Huntington's disease (36), and spinal cord injury (2,40), information on the therapeutic benefits of cell-based therapy on brain injury at a juvenile age is limited. Few previous studies have addressed the social behavior changes associated with juvenile TBI, and unfortunately, the available treatments are very limited.
Past investigations demonstrated that transplanted iPSC-NPCs were able to differentiate into neuronal and nonneuronal cells that may integrate into the neural circuits and upregulate growth/regenerative factors in the host brain environment (14,50,55). Our previous data have also shown the social behavior benefits of transplantation treatment using bone marrow mesenchymal stem cells (BM-MSCs) in a neonatal stroke model (51). Building upon these past findings, we here show that cell transplantation causes noticeable improvements in social novelty, sociability/interactions, and social transmission in post-TBI rats.
Impaired social behavior is associated with decreased quality of life and declines in pathological state (13,29). Even weeks and months after TBI, disinhibition, impulsiveness, reduced anger control, aggressiveness, and poor social judgment can result (32). Furthermore, TBI can make associating specific emotions with event memories difficult, leading to impaired social behavior (18). Human and animal studies have suggested that the oxytocin/oxytocin receptor system may play a key role in regulating social behaviors, including social interaction, social stress/anxiety, social memory/recognition, sexual, and aggressive behaviors (33,47). Once these genes are translated, the secretion of oxytocin and the membrane localization of the oxytocin receptor become important for their essential biological functions. Oxytocin and its receptors are distributed in various brain regions (25) associated with the control of stress, anxiety, and with social behaviors, including social memory and aggression. In addition, increased levels of oxytocin induce cellular resistance to oxidative stress (43) and differentiation (38). These results suggest that transplanted cells with high oxytocin expression may hold a supportive role by optimizing the host vasculature milieu and promoting healing by altering the cellular response to ischemia. In our present study, we show that transplantation of iPSC-NPCs effectively attenuates TBI-associated deficits during development. Furthermore, we found that oxytocin and oxytocin receptor levels were both significantly increased in HP-iPSC-NPCs in vitro and in vivo. Taken together, these findings suggest that the upregulation of oxytocin/oxytocin receptors by a preconditioning procedure may have dual beneficial roles.
Investigations from our and many other groups have shown that hypoxia-preconditioned cells show increased therapeutic potentials. HP-iPSC-NPCs show: 1) increased HIF-1α; 2) upregulated downstream regenerative factors such as BDNF, GDNF, vascular endothelial growth factor (VEGF), and/or erythropoietin (EPO); and 3) increased expression of social behavior genes such as oxytocin and the oxytocin receptor (14,50,55). Hypoxic preconditioning also suppresses inflammatory cytokine/chemokine expressions in stem cells and neural progenitor cells in host tissues (50). Our investigation shows that hypoxic preconditioning of iPSC-NPCs further improves the efficacy of iPSC-NPC transplantation therapy against post-TBI neuropsychiatric disorders. Furthermore, this study provides new evidence to support the idea that hypoxic preconditioning can increase the expression of social behavioral genes that may mediate attenuation of sociability deficits observed in TBI animals. Together, these findings support the combining of cell-based therapy with hypoxic preconditioning to treat juvenile TBI.
Footnotes
Acknowledgments
This work was supported by NIH grants NS091585 (L.W.), NS062097 (L.W.), and NS085568 (L.W.); AHA Grant-in-Aid 12GRNT12060222 (S.P.Y.); VA National Merit grant RX000666 (S.P.Y.); AHA Predoctoral/Postdoctoral Fellowships 15POST25710112 (Z.Z.W.), 15POST 25680013 (J.H.L.), and 14PRE18830026 (T.C.D.); National Natural Science Foundation of China 81371355/81500989 (Y.Z.); and Beijing Natural Science Foundation 7142045 (Y.Z.). The authors declare no conflicts of interest.
