Abstract
Intracerebral cell transplantation is increasingly finding a clinical translation. However, the number of cells surviving after implantation is low (5–10%) compared to the number of cells injected. Although significant efforts have been made with regard to the investigation of apoptosis of cells after implantation, very little optimization of cell preparation and administration has been undertaken. Moreover, there is a general neglect of the biophysical aspects of cell injection. Cell transplantation can only be an efficient therapeutic approach if an optimal transfer of cells from the dish to the brain can be ensured. We therefore focused on the in vitro aspects of cell preparation of a clinical-grade human neural stem cell (NSC) line for intracerebral cell implantation. NSCs were suspended in five different vehicles: phosphate-buffered saline (PBS), Dulbecco's modified Eagle medium (DMEM), artificial cerebral spinal fluid (aCSF), HypoThermosol, and Pluronic. Suspension accuracy, consistency, and cell settling were determined for different cell volume fractions in addition to cell viability, cell membrane damage, and clumping. Maintenance of cells in suspension was evaluated while being stored for 8 h on ice, at room temperature, or physiological normothermia. Significant differences between suspension vehicles and cellular volume fractions were evident. HypoThermosol and Pluronic performed best, with PBS, aCSF, and DMEM exhibiting less consistency, especially in maintaining a suspension and preserving viability under different storage conditions. These results provide the basis to further investigate these preparation parameters during the intracerebral delivery of NSCs to provide an optimized delivery process that can ensure an efficient clinical translation.
Keywords
Introduction
Clinical implementation of cell therapy is on the rise. The isolation of cell lines has produced homogeneous populations of cells that comply with good manufacturing practices (GMPs) and are suitable for clinical translation (25). Extensive in vitro characterizations define the biological properties of the cells and highlight their potential mechanism(s) of repair (55). However, transplanted cells can only unfold their therapeutic potential if these are transferred successfully from the dish to the host. Overall, the efficiency of cell transplantation is low, with typically only 5–10% of injected cells surviving (53). Apart from a few cases (3,21,28,4), translational efforts to date have mostly demonstrated the safety of intracerebral implantation (31), although achieving therapeutic efficacy that would warrant the treatment of large numbers of patients remains a challenge (5).
Delivery of cells to the brain is hence a technical challenge that will be pivotal to successful clinical translation (30,55). Most of the techniques used today were developed for fetal tissue transplants. From fetal tissue grafts, it is known that the preparation of material for implantation will influence viability (6). Suspension medium (16,59), cell concentration (41,42), time between preparation and implantation (22), clumping of cells (9), as well as needle size (1) can influence the viability of cells in the suspension and their survival upon implantation. However, it is important to contrast fetal tissue transplantation, where small fragments from primary tissue are injected without significant processing and need for a homogeneous suspension (i.e., equal amount of cells spread through the vehicle), and cell implantation in which cells are isolated from the tissue through processing to yield individual cells and a full suspension is intended (5,38).
Essentially, three technical stages can be distinguished to transfer cells from the dish to the brain (Fig. 1A): 1) in vitro preparation and maintenance of a cell suspension (cell harvesting, vehicle suspension, maintenance of cells in vehicle prior to implantation, concentration of suspension, and sedimentation of cells in vehicle), 2) uptake and ejection of a cell suspension from a syringe through a needle (speed of uptake, bore size, time delay in syringe, sedimentation, adhesion of cells to syringe/needle, ejection efficiency and shear stress on cells, viability of cells upon ejection), 3) intracerebral injection and retention of cells (volume of injection, speed of injection, diffusion from site of injection, retention of injectate, survival and differentiation of cells, inflammatory response to injection, blood–brain barrier integrity, and potential for an immune response). An optimal delivery of cells accounts for all these aspects. Merely focusing on one stage can yield “optimal” conditions that are not conducive to the entire process. For instance, in vitro, a wide-bore needle will afford a better viability (1), but wide-bore needles are problematic in vivo due to more extensive tissue damage caused during injection (29). It is hence essential that a systematic investigation of intracerebral cell implantation considers all three stages to define optimal parameters (Fig. 1B).
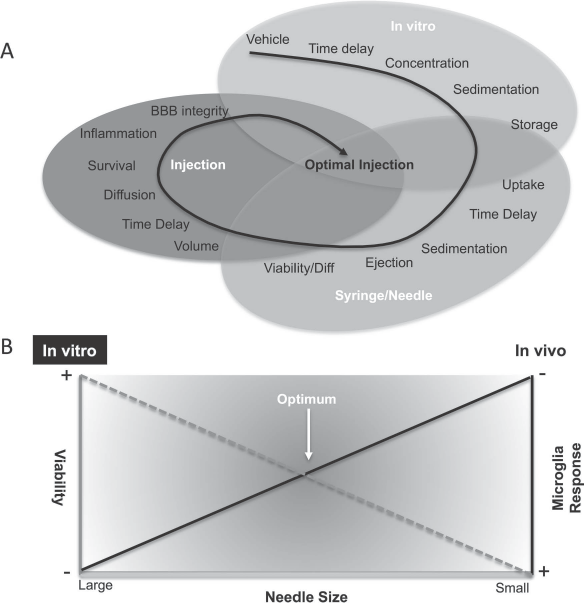
Technical considerations for intracerebral cell transplantation. (A) Individual steps that can affect the preparation of a cell suspension, its transfer to the brain, and intracerebral injection. (B) Certain favorable in vitro conditions, such as wide-bore needle to maintain high cell viability, might be in opposition to in vivo needs, where a wide-bore needle might induce significant parenchymal damage and provoke a microglial response that is detrimental to cell survival. A balance between these factors is hence required to define optimal conditions for the whole procedure.
Still, the first step in these three stages, the preparation and maintenance of a cell suspension, provides the necessary conditions to pursue further steps. We here investigate the effect of six different suspension vehicles that serve as carriers for the single-cell suspension of human neural stem cells on viability, cell membrane damage, as well as the quality of the suspension. The density and viscosity of the vehicles were also determined, as they affect the shear stress that cells are exposed to during uptake and ejection from a syringe/needle (2,4). The results presented form the basis for further testing of cell suspensions during uptake and ejection from syringe/needles used for intracerebral cell implantation.
Materials and Methods
Neural Stem Cells
The human striatal neural stem cell (NSC) line STROC05 (derived and kindly provided by ReNeuron, Guildford, UK) were cultured and passaged, as previously described in detail (19,26). In brief, the cell line was derived under GMP conditions from the whole ganglionic eminence of a human fetus 12 weeks old, and was conditionally immortalized using cMyc-ERTAM under the control of 4-hydroxytamoxifen (4-OHT, 100 nM; Sigma-Aldrich, St. Louis, MO, USA) (44,45). In the absence of 4-OHT, NSCs will cease proliferation and undergo differentiation. NSCs were expanded on laminin (10 μg/ml; Sigma-Aldrich)-coated flasks until 80–85% confluency was reached. Recombinant human basic fibroblast growth factor (bFGF, 10 ng/ml; PeproTech, Rocky Hill, NJ, USA) and epidermal growth factor (EGF, 20 ng/ml; PeproTech) were used as mitogens (see Table 1 for complete description of media components). All culturing was performed without the addition of antibiotics at 37°C in 5% CO2. Cells were tested monthly for mycoplasma using a PCR kit (Sigma-Aldrich) or colorimetric analysis (Plasmotest; InvivoGen, San Diego, CA, USA) and found to be negative.

For differentiation medium, bFGF, EGF, and 4-hydroxy-tamoxifen were omitted.
Suspension Vehicles
Cell suspensions for intracerebral transplantation consist of cells that biophysically are “solid spheres” dispersed (i.e., dispersed phase) in a liquid (i.e., dispersion medium serving as vehicle). Solid particles <1 μm dispersed in a liquid phase will form a sol, whereas cells larger than >1 μm in diameter are prone to settling (i.e., are unstable) and hence do not produce a colloid (i.e., a suspension that does not sediment), but form a suspension. Cell suspensions for intracerebral injections share biophysical characteristics of natural cell suspensions, such as blood (i.e., blood cells as a solid in plasma as a liquid) or semen (sperm in seminal fluid).
In their simplest preparations, vehicles used for intracerebral injections are merely buffer solutions (i.e., aqueous solutions maintaining pH). Phosphate-buffered saline (PBS) is a simple buffer solution frequently used for cell transplantation. A buffer solution commonly used for sampling from the human brain in microdialysis studies consists of artificial cerebral spinal fluid (aCSF) and might hence provide a buffer solution specifically formulated for the brain. In contrast, cells for suspension are grown in culture using a medium specifically formulated for their expansion and maintenance. Indeed, Dulbecco's modified Eagle medium (DMEM) with Ham's nutrient mixture F12 is extensively used as a basal medium supplemented with specific factors to control in vitro cell proliferation. For cell transplantation, DMEM is typically supplemented with N-acetyl-cysteine, which promotes survival without affecting proliferation (39). HypoThermosol, in contrast, represents a specific proprietary formulation of a suspension vehicle to preserve cell vitality during cryopreservation (7,37). To specifically control shear forces in suspension, Pluronic F68 is a nonionic surfactant commonly used to prepare cell suspensions (33). Although many other vehicles are in common use, the following vehicles were evaluated due their clinical relevance, distinct characteristics, and previous use for intracerebral cell transplantation: 1) 0.01 M PBS (P4417; Sigma-Aldrich), 2) aCSF (59-7319; Harvard Apparatus, Holliston, MA, USA), 3) DMEM/F12 (21331020; Invitrogen, Carlsbad, CA, USA) with N-acetyl-cysteine (NAC, A9165; Sigma-Aldrich), 4) HypoThermosol (HTS, H4416; Sigma-Aldrich), 5) 0.5% Pluronic F68 (P1300; Sigma-Aldrich) in PBS. All solutions were sterilized using a 0.2-μm filter.
Vehicle Density
To maintain a homogeneous dispersion of cells in the liquid phase, density is a crucial measure that influences buoyancy, as well as sedimentation. Ideally, density matching reduces buoyancy and settling (i.e., sedimentation) of cells. Conversely, in vivo density of the liquid phase will influence permeation of cells into tissue. The density of water is ~1 g/ml, whereas blood is ~1.05 g/ml (20). The weight of vehicles was measured by weighing 1 ml of each medium on a precision scale (Ohaus, Parsipanny, NJ, USA). A total of three samples were measured. To measure the density of STROC05, 104, 105, and 106 cells were suspended in 10 μl of PBS. The density of PBS was subtracted from the PBS + cells measure, and the density for a single cell was calculated. The average of all three measurements was taken and multiplied by the number of cells (i.e., 2.55 ° 108 cells) required to fill 1 ml. Density was calculated using the following formula:
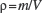
where V = volume; m = mass (g); ρ = density.
Vehicle Viscosity
Viscosity measures the internal friction of a solution that produces its resistance to gradual transformation through shear stress. In the context of cell suspensions, the viscosity of a vehicle is hence likely to affect cellular stress due to passing through a thin needle but also affects the quality and consistency of the suspension. Water has a reported viscosity of 1 centipoise (cP), whereas blood ranges between 3 and 4 cP at 37°C (20), and semen is as high as 9.35 cP (32). Viscosity measurements were performed using the Falling Ball Viscometer (GV-2100; Gilmont Instruments, Barrington, IL, USA) (18). The viscometer consists of a glass tube filled with 7 ml of solution through which a glass (0.2–2 cP) or a stainless steel (1–10 cP) ball is allowed to sink to the bottom at 37°C. The time for the ball to sink to the bottom was recorded with a stopwatch. All measurements were conducted in triplicate. The solution's viscosity in cP is estimated based on the time required to reach the bottom using the following formula:

where μ = viscosity in cP; ρt = density of ball (2.53 g/ml); ρ = density of liquid; t = time descendant; K = viscometer constant (0.3).
Volume Fraction Measurement
Often in cell transplantation studies, a concentration of cells within the cell suspension is reported. However, concentration typically refers to solutes or solvents in a solution rather than solids (i.e., cells) in a solution. As cell size varies widely either due to their site (e.g., bone marrow vs. neural) or species of origin (e.g., mouse vs. human), the maximum concentration suspended within 1 μl of vehicle will hence be quite different. It is therefore more appropriate to express the cellular component within a suspension as a volume fraction, akin to packed cell volume or erythrocyte volume fraction used to define the hematocrit in blood. The cellular volume fraction in blood is approximately 45% for men and 40% for women (54). To calculate the volume occupied by the cell fraction in 1 μl of 0.1 mM PBS, the STROC05 cell diameter was measured in a representative fraction (all live cells). Measurements were undertaken in iso-osmotic conditions. The diameter was used to calculate the radius and the volume of a sphere using the following formula:

where V = volume; ∏ = 3.14; r = radius.
An additional validation of volume fraction was achieved by suspending 107 cells in 200 μl of total volume and spinning these down at 5,000 rpm. The pellet volume was subsequently compared to the overall volume.
Preparation of Cell Suspension and Viability Measurement
At 80–85% of cellular confluence, cells were harvested from their flask after adding 5 ml of Accutase (Sigma-Aldrich) in a T175 flask (BD Biosciences, San Jose, CA, USA). Cells were spun down at 405 ° g (Centrifuge 5804; Eppendorf, Hauppauge, NY, USA), and supernatant was aspirated prior to resuspension in 10 ml of culture media. A 5-μl sample of cell suspension was acquired and mixed with 5 μl of Trypan blue (Sigma-Aldrich). Using a hemocytometer, cells in the sample were counted to estimate the total cell number of live (unlabeled) and dead cells (labeled). The viability was expressed as percentage of live cells in the total number counted in the sample.
Cells were spun down again with the supernatant aspirated to afford suspension in a given vehicle at a concentration corresponding to the appropriate volume fraction (Fig. 2A). The following formula was used to calculate the appropriate volume of suspension to be added to the cell pellet to yield a given concentration/volume fraction:
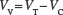
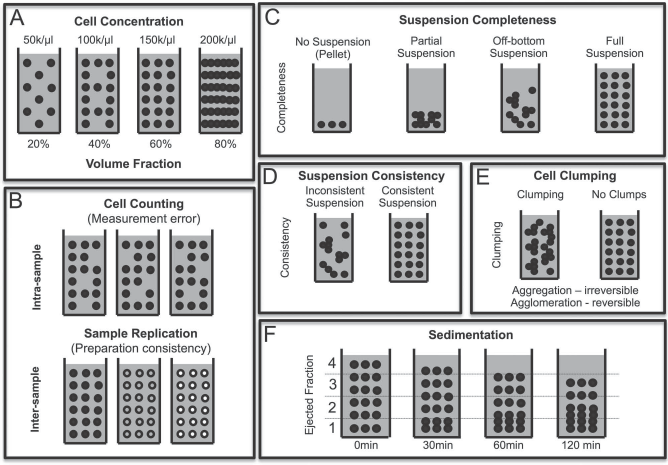
Characterization of cell suspensions. (A) Cell density provides an indication as to the number of cells being delivered and is essential to establish a relationship between cell number and efficacy. Conversely, volume fraction determines the space occupied by cells and contrasts this with space available for dispersion medium, but also potentially is important to determine what additional space is available for a second cell type or scaffold particles to be included in the same suspension. Cell density can range from very dilute (<20% of occupied volume) to highly concentrated (>80%). (B) Cell counting is used to estimate the number of cells present within a given suspension volume. To gain accurate counts, several technical replicates might be needed for any given preparation. However, to determine how reliable and accurate a suspension with a given dispersion medium (i.e., suspension vehicle) is, several biological replicates are required. (C) Suspension completeness refers to the degree that cells are in suspension rather than a pellet (i.e., no suspension). Ideally cells are in full suspension to ensure that each volume fraction delivered by injection contains therapeutic entities (i.e., cells). (D) The quality of the suspension is defined by its consistency. An inconsistent suspension is characterized by different fractions containing different amounts of cells, whereas a consistent and full suspension will contain an equal amount of cells in all fractions of volume. (E) As cells interact, these can agglomerate into clumps (i.e., clusters of cells). Agglomeration refers to reversible cell attachment, whereas aggregation indicates an irreversible attachment that would lead to cell death if these cells were separated. (F) Sedimentation of cells occurs through settling, where due to a mismatch in material density and/or lack of traction (i.e., viscosity) cells settle through gravity. In this case, over time, cells from upper volume fractions will gradually move to lower ones until they form a pellet (i.e., no suspension).
where VV = volume of vehicle to be added; VT = total volume of preparation; VC = total cell volume. Total cell volume is defined by the number of cells in a pellet ° volume of one cell. Note VC/VV = cell volume fraction, whereas the number of cells in a given volume gives the cell concentration. Importantly, adding VT to a cell pellet would not yield an accurate cell concentration, as it neglects to account for the volume of cells.
After preparation of a cell suspension for implantation, the suspension is typically stored until needed, either at hypothermia on ice (2°–4°C), at room temperature (20°–21°C), or normothermia at a physiological temperature (37°C). Cell viability was measured for a 20% cell volume fraction (50,000 cells/μl) suspension every 2 h for 8 h.
Suspension Accuracy
The accuracy of suspension refers to the error between the target cell density (e.g., 50,000 cells/μl) and the measured cell density (Fig. 2B). To define this variability in preparation (i.e., intersample/biological variability), four independent preparations (10 μl) were assessed using a hemocytometer with each being sampled using three technical replicates (1 μl volume each) to account for intrasample variability. For subsequent experiments, a variation of 10% was considered acceptable, and greater variability led to a resuspension of the sample.
Suspension Completeness
The completeness of the suspension was defined by cells being present throughout the sample (Fig. 2C). If cells were only present at the bottom of the sample preparation (i.e., a pellet), this was defined as no suspension. Cells present only in the bottom 50% of the sample was referred to as partial suspension, and some cells being present in the top half (in the absence of a pellet) was considered an off-bottom suspension.
Suspension Consistency
Upon preparation, the consistency of the suspension is defined by an equivalent local cell concentration throughout the sample (Fig. 2D). Each equivalent part of the sample should hence contain an equal amount of cells.
Cell Clumping
In suspension, individual cells can interact with other cells through Brownian motion and adhere to each other (Fig. 2E). Adherence of cells to each other can either be through agglomeration, which is a reversible process, or aggregation, which is an irreversible process (uncommon for cells even in tissue) (52). Clumping was quantified by counting cells in clusters of 2, 3–5, and >5 cells on a hemocytometer.
Sedimentation
As cell suspensions are heterogeneous mixtures of cells and liquids, with time the solid component tends to settle (i.e., sediment at the bottom of the solution) (Fig. 2F). In the context of cell transplants, a rapid sedimentation potentially compromises the delivery of a homogeneous sample, with the last ejection fraction merely consisting of vehicle. Ensuring that sedimentation is minimal during the injection procedure, and also when preparing for injection, are important characteristics that influence the suitability of vehicle for suspension. Apart from vehicle characteristics, such as density and viscosity, cell volume fraction is also expected to affect sedimentation, with higher volume fractions exhibiting a reduced propensity for sedimentation. Sedimentation was measured here by preparing cell suspensions for different vehicles at 20% (50,000 cells/μl) and 60% (150,000 cells/μl) volume fraction. Cell content was measured by counting cells in four fractions (0–2, 2–4, 4–6, 6–8 μl) of a 10-μl suspension ejected at 0, 30, 60, and 120 min after uptake into a 10-μl Hamilton syringe with a 26-gauge needle (701 N; Hamilton, Reno, NV, USA) attached vertically to a stand with a three-prong holder. In addition to sedimentation, cell viability (see below) and cluster formation were measured.
Resuspension
Cells in suspension will settle to some degree between preparation and use. It is hence necessary to resuspend cells to ensure a full and consistent suspension. A simple and controllable approach for resuspension is to use a pipette to take up and expel a certain volume of the suspension. However, pipetting can expose cells to shear stress that can reduce viability and lead to cell membrane damage. The effect of 1, 3, 5, and 10 times pipetting a sample (100 μl) using a standard laboratory P200 pipette (Eppendorf) with an appropriate tip (Fisherbrand SureOne; Thermo Fisher Scientific, Waltham, MA, USA) for resuspension was hence investigated in 20% cell volume fraction suspensions (50,000 cells/μl). Samples from resuspensions were either taken hourly for 8 h from the same preparation or from a preparation that was only resuspended every 4 h, as frequency of resuspension could also affect viability, membrane damage, and cell clumping.
Membrane Damage
The effect of mechanical stress on cell damage was investigated using lactate dehydrogenase (LDH) release (TOX7; Sigma-Aldrich), as previously described (58), in combination with measurement of cell viability in the same sample. For this, every hour and for a total of 8 h the stock solution was resuspended by (five times) pipetting. Samples were collected immediately after resuspension to measure viability and LDH. For the LDH assay, 100,000 cells were seeded in a 48-well plate. After 24 h, the LDH assay was conducted on the supernatant using a microplate reader (Bio Tek Instruments, Winooski, VT, USA).
Statistical Analyses
All data were graphed and analyzed in Prism v6 for Mac (GraphPad, La Jolla, CA, USA). One-way analyse of variance (ANOVAs) compared multiple conditions for a single independent and dependent variable (e.g., vehicle density, viscosity, suspension viability, and LDH) with Bonferroni post hoc testing. Two-way ANOVAs were used to determine statistical differences between more than one independent variable (e.g., sedimentation, clumping) using Tukey's post hoc test. Repeated-measures ANOVAs compared measurements that occurred over time (e.g., pipetting viability, temperature effects) with Tukey's post hoc test. A value of p < 0.05 was considered significant.
Results
Suspension Vehicle Viscosity and Cell Volume Fraction
The biophysical and chemical properties of a suspension vehicle influence their suitability to achieve a full homogeneous suspension that is resistant to settling of cells and protects them from shear stress during uptake and ejection. The cardinal biophysical property of a suspension vehicle is its density (Fig. 3A), which determines if the disperse phase (i.e., cells) will be buoyant or settle. There was no significant difference between PBS, aCSF, DMEM, and HTS, but Pluronic (r = 0.97) was significantly less dense (p < 0.05). NSCs had a measured density of 2.04 and hence undergo sedimentation in these vehicles. Vehicle density also influences viscosity, which can reduce sedimentation, as well as protecting from shear stress (Fig. 3B). PBS, aCSF, and DMEM exhibited similar viscosities. However, the viscosity of Pluronic was significantly higher (p < 0.05) than PBS, aCSF, and DMEM, but HTS revealed a viscosity >3 times higher (p < 0.001), akin to that of blood.
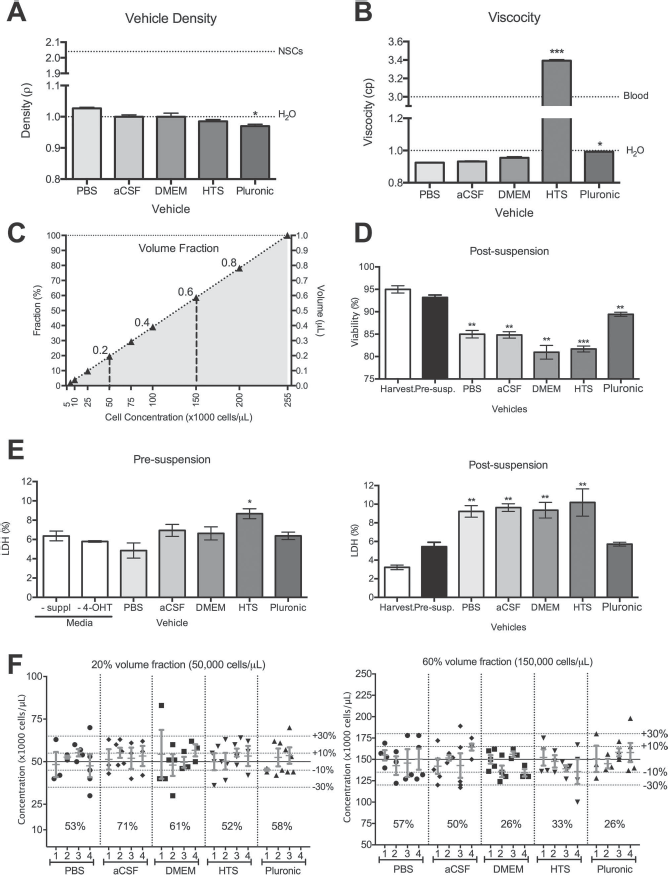
Accuracy of preparing cell suspensions. (A) Comparison of vehicle density against density of water (H2O) and neural stem cells (NSCs). HTS and Pluronic exhibited densities below that of water. The density of NSCs was more than twice that of the vehicles used for suspension. (B) Comparison of vehicle viscosity against H2O and blood. Only HTS had a viscosity within the range of blood; all other vehicles yielded low viscosities. (C) Determination of cell volume fraction (%) for different cell concentrations in addition to its corresponding volume in 1 μl of suspension. (D) Effect of vehicles on cell viability during the suspension process. Upon harvesting cells from the flask, viability is ~95%; merely resuspending this in media only slightly impacts viability. However, suspension in any of the tested vehicles significantly affects viability, with Pluronic exerting the least impact. (E) This reduction in viability is not a direct result of the vehicles themselves on cells, as indicated by a very minor increase in cell membrane damage (measured using LDH) when these are added to cells in culture (presuspension). However, suspension of these (i.e., use of a pipette to equally distribute cells within the dispersion medium) leads to a significant increase in membrane damage, unless cells are suspended in Pluronic. (F) The accuracy of estimating cell concentrations for a 20% and 60% volume fraction was measured for individual suspensions using three technical replicates (i.e., intrasample variability), as well as across different preparations (i.e., intersample variability). These measurements were compared to the target concentrations, as well as ±10% and 30% confidence intervals. The percentage accuracy of a suspension vehicle falling within a 10% confidence interval upon first suspension was calculated from these measurements. *p < 0.05; **p < 0.01; ***p < 0.001.
As cells provide the dispersed phase for the suspension, a crucial question that arises is how many cells can actually be suspended within a given volume. To this effect, it is essential to measure the volume occupied by a given number of cells. Based on an average cell diameter of 19.29 ± 0.75 μm, an individual cell volume of 3,912 μm3 (0.000003912 μl) is calculated. A theoretical maximum of 255,623 cells could hence fit within 1 μl. Accordingly, cell volume fractions can be calculated for different cell concentrations (Fig. 3C). A cell concentration of 50,000 cells corresponds to ~20% cell volume fraction (i.e., fairly dilute), whereas 150,000 cells occupy 60% of available space with suspension vehicle only using up to 40% of the volume. It is important to note that even in a cell pellet some fluid will be present in between approximately spherical cells; hence, a certain estimation error is inherent in these theoretical calculations.
Suspending Reduces Viability
The process of cell suspension induces cellular stress that can lead to a reduction in viability of cells. Indeed, at the point of cell harvesting from the cell culture dish, viability was ~95% (providing the standard control value for all other measurements), with viability being only marginally (2%) reduced due to resuspension (Fig. 3D). Suspension of cells (20% cell volume fraction) in vehicle, however, led to a significant (p < 0.01) decrease in viability by 5–14%. The most significant reduction in viability (as measured by Trypan blue) was observed for DMEM and HTS. Exposure of cells in culture to suspension vehicle indicated a slight increase in cell membrane damage, as indicated by LDH release (Fig. 3E). Only HTS significantly (p < 0.05) increased LDH release by ~4% compared to PBS. Upon suspension, apart from Pluronic, suspension of cells in all vehicles resulted in an equivalent cell membrane damage, as indicated by the 4% increase in LDH release (p < 0.01). Pluronic hence provides some protection against suspension-induced cell membrane damage that can lead to a loss of viability.
Quality of Cell Suspensions
The suspension vehicle can also affect the quality and accuracy of the suspension. The aim is to achieve a full and homogeneous suspension in which all parts of the suspension have equal amounts of cells. In contrast, the accuracy of the suspension is defined by the number of cells within a given volume being within a 10% tolerance limit from the anticipated cell concentration. In a first suspension of a 20% cell volume fraction, over half of all samples taken were not within the 10% accuracy limit, and there was substantial variation between technical replicates for each suspension for all four preparations tested (Fig. 3F). Nevertheless, the mean values estimated for each preparation were within the 10% accuracy limit and indicated that suspension accuracy for each vehicle is equivalent. The number of estimates outside the 10% accuracy limit was reduced by half for DMEM, HTS, and Pluronic for a 60% cell volume fraction. However, no improvement in suspension accuracy was observed for PBS and aCSF.
To further probe the influence of cell volume fraction on the accuracy of cell delivery, suspensions with four different volume fractions (20%, 40%, 60%, 80%) were prepared and assessed immediately after suspension (0 h) or upon resuspension after 8 h of settling (8 h) (Fig. 4). Freshly prepared suspensions were close approximations (<10% limit) to the target cell concentration for a given condition. However, resuspension after 8 h indicated differences between suspension vehicles, as well as cell fractions. At a dilute 20% volume fraction, there was no significant difference in the estimation of cell concentration between 0 and 8 h. Little difference in viability was seen between time points, although HTS exhibited a lower viability compared to Pluronic (p < 0.05). At 40% volume fraction, the estimated cell concentration after resuspension (8 h) was higher for PBS (p < 0.01), aCSF (p < 0.05), and Pluronic (p < 0.05). Only PBS indicated a reduction in viability by 8 h (p < 0.05). At 60% volume fraction, PBS (p < 0.01), DMEM (p < 0.05), and HTS (p < 0.05) underestimated the cell concentration at 8 h, whereas aCSF (p < 0.001) and Pluronic (p < 0.05) overestimated the concentration. Still, viability was fairly consistent with only aCSF indicating a significant reduction in viability (p < 0.05). At a highly concentrated 80% volume fraction, apart from HTS and Pluronic, all other conditions overestimated the cell concentration at 8 h (p < 0.05). Only aCSF exhibited reduced viability at 8 versus 0 h (p < 0.05). aCSF hence provided the least consistent suspension estimate after resuspension at higher volume fractions and also was the only vehicle where viability was significantly reduced. In contrast, HTS followed by Pluronic provided the most consistent suspensions, but HTS showed some reduced viability, although this was not statistically significant.
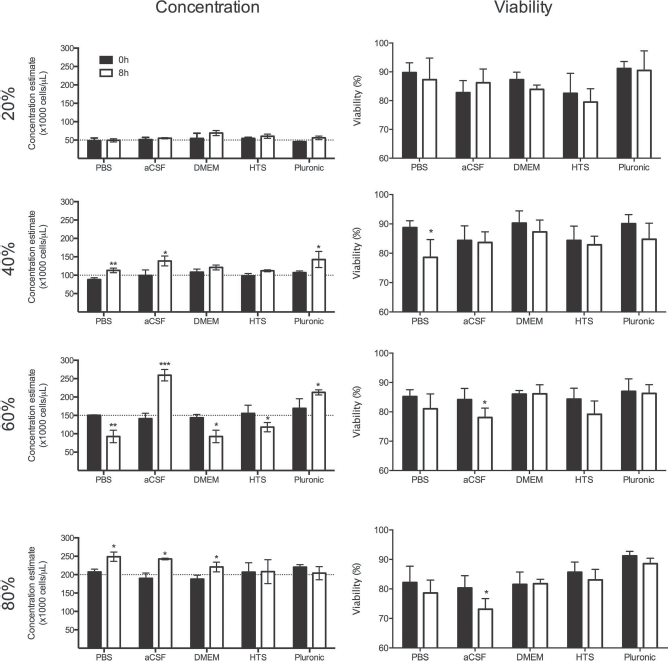
Cell volume fractions. Different cell volume fractions (20%, 40%, 60%, 80%) in suspension vehicles and their influence on achieving and maintaining target concentrations (dashed lines) at the point of suspension (0 h) and after 8 h in suspension (8 h), as well as viability. Pluronic achieved the most accurate concentration, while preserving the best viability. In contrast, aCSF was least accurate in achieving and maintaining a suspension and also reflected the greatest decrease in cell viability. *p < 0.05; **p < 0.01; ***p < 0.001.
Settling of Cells in Suspension
A drawback with cell suspensions, where the dispersed phase and dispersion medium densities are not well matched, is that the dispersed phase (i.e., cells) will eventually settle (i.e., lead to a sedimentation, resulting in a partial or no suspension of cells). Depending on the mismatch of density and the viscosity of the dispersion medium, this can occur fairly rapidly and potentially affect the accuracy of cell delivery. In all tested vehicles here, there was evidence of settlement of cells over a 120-min period (Fig. 5). This settling of cells was more evident in the 20% volume fraction conditions than in the 60% volume fraction due to the greater voids that can be filled by cells in the lower fractions. Over a 120-min period, DMEM exhibited the most settling of cells with the first fraction having tripled its concentration (p < 0.001) and the fourth fraction having only 10% of its starting concentration (p < 0.001). In contrast, HTS maintained the most consistent suspension, although a 50% increase in cell concentration was evident in the first three fractions (p < 0.01). The movement of cells within the dispersion medium also leads to an agglomeration of cells (i.e., clumping) that conversely was most evident in HTS at 20% volume fraction and least evident in Pluronic (Supplementary Figure 1, available at https://www.researchgate.net/publication/283081632_Rossetti_et_al_Cell_Transplantation_Supplementary_Material). At 60% volume fraction, significant clumping was evident in all conditions, especially PBS and aCSF, but least in Pluronic (Supplementary Figure 2, available at https://www.researchgate.net/publication/283081632_Rossetti_et_al_Cell_Transplantation_Supplementary_Material). Viability in the different ejection fractions was fairly consistent across all conditions (Fig. 6). Only DMEM saw a 15% decrease in viable cells with a 20% volume fraction (p < 0.01), although it was stable across all conditions at a 60% volume fraction. The 20% volume fraction overall saw an interaction between the last fraction of ejection and time between suspension and ejection. However with a 60% volume fraction, this effect was only evident for Pluronic (p < 0.05). Nevertheless, these viability effects are subtle with changes between 2% and 8% with >80% of cells being alive. DMEM exhibited a poor performance for maintaining a cell suspension, and settling of cells was associated with a reduction in viability. HTS in contrast provided the most consistent cell suspension while preserving a high viability, although there was evidence of cell clumping. These results indicate that cells can rapidly settle depending on the suspension vehicle and that this process can affect viability.

Settling of cells: sedimentation effects. Settling of cells in different suspension vehicles for 20% and 60% volume fractions was assessed across four fractions of cells straight after suspension (0 min delay), as well as after 30, 60, and 120 min. HTS provided the greatest consistency, whereas DMEM was the least consistent. *p < 0.05; **p < 0.01; ***p < 0.001.
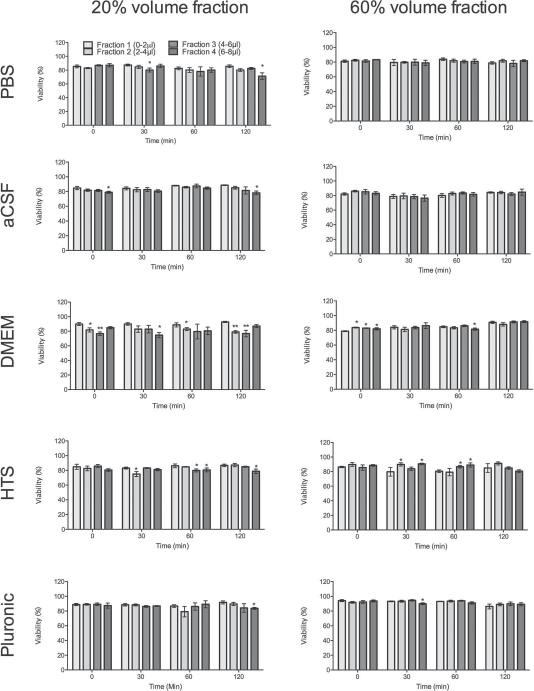
Settling of cells: viability effects. To determine if settling of cells could affect cell fate after injection, the viability of cells was assessed across four equal fractions of the cell suspension for both 20% and 60% cell volume fractions. Cell viability was consistent across almost all conditions, although fraction 4 consistently showed a decrease in cell viability after 120 min in suspension. *p < 0.05; **p < 0.01.
Resuspension of Cells After Settling
To avoid the injection of settled cells or inaccurate uptake into a syringe, a resuspension of cells might be required. This is typically achieved by pipetting the cell suspension/pellet. To determine if this pipetting affects viability, cell membrane damage, as well as clumping, pelleted cells were resuspended by pipetting the suspension 1, 3, 5, or 10 times once every hour for 7 h (Fig. 7). There was an interaction between the number of times cells were pipetted and elapsed time (p < 0.0001); viability was most reduced with 10 times pipetting and having performed this resuspension seven times (i.e., after 7 h). The most significant effect on cell viability due to repeated resuspending was observed with Pluronic that showed a 30% reduction with only a single pipetting (p < 0.001). aCSF, DMEM, and HTS were vehicles that preserved viability the best, although some reduction in viability was evident after pipetting 10 times (p < 0.05). Indeed, pipetting 10 times affected viability in all conditions, but there was little difference in viability for pipetting 1 to 5 times. For pipetting 5 times, HTS maintained the best viability followed by DMEM, whereas PBS and Pluronic performed the poorest (p < 0.05). Interestingly, PBS and Pluronic also showed the least clumping, whereas HTS showed the most (6–8%) clumping overall (Supplementary Figure 3, available at https://www.researchgate.net/publication/283081632_Rossetti_et_al_Cell_Transplantation_Supplementary_Material). However, with pipetting 10 times, a gradual and steady increase in two cell clumps was evident with Pluronic, reaching up to 14% of all cells (p < 0.001). In all conditions, very few cell clumps of >5 cells were observed, indicating that even a single pipetting will be sufficient to maintain single-cell suspensions. Importantly, the effect on cell viability through resuspension can be mitigated by avoiding too many resuspensions (Supplementary Figure 4, available at https://www.researchgate.net/publication/283081632_Rossetti_et_al_Cell_Transplantation_Supplementary_Material). HTS therefore mitigates the effects of multiple resuspensions most effectively with pipetting 5 times being the upper limit, although increased clumping is observed. Frequent resuspension resulted in increased membrane damage (as shown by LDH), but this effect was mitigated by a protective effect of Pluronic. However, resuspending cells less frequently reduced the amount of membrane damage in other vehicles.

Resuspension: viability effects. To determine if resuspension of cells using a pipette affects viability, cells in different suspension vehicles were exposed to different (1, 3, 5, 10) amounts of pipetting each hour for a duration of 8 h. In all cases, 10 pipettings affected cell viability, but 5 pipettings were pretty consistent across vehicles. Pluronic overall provided least protection from the pipetting, whereas HTS provided the most consistent viability across all pipetting conditions, with only 10 pipettings after 5 h exhibiting a reduction in viability.
Maintaining a Viable Suspension
Once prepared, cell suspensions are stored prior to their use for injections either on ice, at room temperature, or at physiological temperature (Supplementary Figure 5, available at https://www.researchgate.net/publication/283081632_Rossetti_et_al_Cell_Transplantation_Supplementary_Material). Cells stored in suspension at physiological temperature declined over an 8-h period for all suspension vehicles (Fig. 8). DMEM underwent the most dramatic loss of viability with no cells alive after 8 h (p < 0.0001), whereas Pluronic preserved viability best with only a 15% reduction (p < 0.05). Storage on ice led to a dramatic loss of cell viability, but only for aCSF (p < 0.001) and DMEM (p < 0.001), whereas the other dispersion media only had a minimal loss in viability (<5%). However, room temperature consistently maintained viability in all suspension vehicles. To maintain a viable suspension, it is therefore safest to maintain cells at room temperature irrespective of the dispersion media. However, from all suspension vehicles, Pluronic and HTS were the most tolerant to different environmental temperatures.

Effects of temperature of suspension storage on cell viability. Suspended cells can be maintained on ice (2–4°C) at room temperature (20–21°C) or at physiological temperature/normothermal conditions (37°C) while awaiting transplantation. On-ice maintenance significantly affected viability for all conditions. Normothermal conditions also affected longer maintenance conditions for aCSF and DMEM. In contrast, room temperature yielded consistently good viability for all suspension vehicles. *p < 0.05; **p < 0.01; ***p < 0.001.
Discussion
An essential step in the bench-to-bedside translation of cell therapy is to ensure that cells are appropriately prepared for implantation. For cells grown in culture, the most common practice for cell transplantation is to prepare single-cell suspensions. A systematic approach to compare different vehicles or carriers that serve as dispersion media for cells (the dispersed phase), as well as biophysical characteristics of the suspensions at different volume fractions will help to develop an optimized approach to guarantee that implanted cells retain their therapeutic properties upon implantation. We here demonstrate that suspension vehicles differ markedly in their performance in terms of suspension accuracy, cell membrane damage, and maintenance of viability, but also settling of cells over time, clumping, and maintaining a viable suspension while awaiting transplantation.
Biophysical Aspects of Suspending Cells for Transplantation
Transferring of cells from a cell culture dish to the in vivo environment necessitates that these are suspended in a fluid that affords their uptake and ejection from a small bore needle into the host tissue. The biophysical characteristics of these suspension vehicles hence becomes a key component to ensure that cells are not damaged or altered in any other way (16). Several “natural” cell suspensions, such as blood or semen, are cell delivery systems that can provide indications as to the biophysical properties (i.e., viscosity, mitigation of shear stress, volume fractions, and fluid compositions) that will be important for cell suspensions in the context of cell transplantation. Indeed, viscosity of the fluid is the seminal biophysical property that influences the overall biophysical properties of a suspension in terms of its resistance to cell settling, protection against shear stress, as well as flow properties through a syringe/needle (13,63). Blood typically has a cP value between 3 and 4, whereas semen is as high as 9. Most simple vehicles (PBS, aCSF, DMEM) tested here revealed viscosities of less than 1 cP, with Pluronic exhibiting a viscosity of 1, equivalent to that of water. Only HTS here revealed a viscosity in the range of blood, potentially providing better suspension characteristics than all others tested.
Foremost of all, the density and viscosity of the fluid define its properties to maintain a suspension of cells. Ideally, the density of the fluid and cells is equivalent to prevent buoyancy or settling of cells (13). The density of cells here was higher than all tested vehicles and will hence promote settling of cells. In the case of buoyancy or settling of cells, there is a risk of an inhomogeneous suspension that can result in the first fraction carrying almost all the cells and the last fraction only being vehicle (47,57). In this case, there is no therapeutic relevance of the last fraction being injected. In the case of buoyancy, cells might separate from the fluid, as seen in a density-gradient separation assay (27), and hence there is little benefit of having a fluid being injected. Within 30 min of suspension at 20% cell volume fraction, all vehicles tested here, apart from HTS, showed a significant settling. HTS maintained a fairly stable suspension for over 120 min. In contrast, a higher cell volume fraction (60%) limited settling of cells. During implantation, there is a risk that some material is immediately ejected from the brain (17), making the delivery of sufficient cells to the brain an essential consideration to ensure therapeutic efficacy. Apart from the delivery process, it is also important to consider that upon implantation, cells will remain within the suspension vehicle and ideally infiltrate the surrounding parenchyma. Although the infiltration can be improved by convention-enhanced delivery (62), once the needle for injection is removed, it is the biophysical properties of the suspension vehicle that will determine if cells settle to the bottom of the deposit or if these line the injection tract and can infiltrate surrounding tissue (47). Instead of simple suspension vehicles, biomaterials designed to prevent settling of cells and reflux through cross-linking into a hydrogel in situ could improve the retention of delivered cells (11,49).
Although the dispersion medium's viscosity influences the cell suspension, the proportion of cells contained within the suspension will also contribute to the overall biophysical properties of the cell suspension. A cell suspension with a negligible cell volume fraction (<20%) is hence likely to be dominated by the biophysical properties of the suspension vehicle, whereas a cell suspension with a high proportion of cells (>60%) is dominated mostly by cellular properties. It is therefore important to account for cell volume fraction in order to understand the cells’ contribution to the biophysical properties of the cell suspension (24). It is noteworthy that most cell types differ in their physical size depending on their origin and hence cell concentration, for one type of cells cannot easily be equated with cell volume fraction for another. Cell volume fraction is therefore relevant in the context of understanding the suspended cells’ biophysical aspects within the cell suspension (40), whereas cell concentration and total number of delivered cells is the key measure in the context of understanding therapeutic efficacy in relation to treatment dose.
Although a high cell volume fraction is desirable to ensure an efficient delivery and suspension consistency (40), preparation and storage of the cell suspension prior to injection will release cellular waste (e.g., reactive oxygen species) that needs to be safely dissipated in suspension fluid. The cell volume fraction (i.e., hematocrit) in blood is hence only ~40%. A cell suspension with a cell volume fraction of 80% here was difficult to handle due to its viscosity. Considering that these were mostly cells, it behaved almost akin to a cell pellet. There was less of an issue with a 60% volume fraction, which was very viscous but easier to manipulate using a pipette or syringe/needle. Higher cell concentrations (200,000 cells/μl) can lead to better cell survival (56), but this might have to be balanced against the overall volume and number of cells being injected into a specific brain location to meet the cells’ metabolic demands. The volume fraction and handling of the suspension are even more important in the context of in situ tissue engineering, where not only cells are implanted, but also potentially multiple biomaterials, such as poly-lactic-glycolic acid (PLGA) particles or hydrogels (10–12). In this case, part of the volume fraction needs to account for the dosing of this material. Lower cell volume fraction might hence be required for these approaches.
Performance Characteristics of a Promising Suspension Vehicle
The biophysical properties of suspension vehicles or carriers will greatly influence the performance characteristics of the cell suspension. A key performance characteristic is damage to cell membranes and its effect on the viability of injected cells. Certain vehicles will affect the cell membrane (16), especially of fragile cells. Indeed, we here demonstrated that HTS marginally increased cell membrane damage, as measured by the LDH assay, by 2–3% in comparison to other vehicles. Damage to cell membranes can also occur due to shear stress exerted by the flow of the cell suspension in and out of the needle/syringe used for injection and also by the pipette used to prepare the cell suspension. Extensive pipetting is typically used to fully suspend cells, as well as to resuspend these after settling. In these cases, the vehicle should protect cells from shear stress. A small number of pipettings (<5) did not significantly decrease viability, but resuspension every hour increased membrane damage in almost all vehicles, apart from Pluronic. However, 10 pipettings for resuspension resulted in a significant decrease in viability for all suspension media. Reducing the number of resuspensions mitigates these effects indicating that the shear stress accumulating from the number of pipettings, as well as incidence, exerted a compounding negative effect on cells. Although there remains an insufficient understanding of flow-induced shear stress and its effects on cell membrane damage (14,15), there are indications that higher cell volume fractions (60%) undergo more significant strain than lower (20%) cell volume fractions (34). A further factor in this equation is the bore size of the tip or needle through which cells are passing. For instance, large-bore needles reduce shear stress and improve the number of viable cells (4,57) with glass capillaries being more favorable than metal needles (42). A further optimization step based on controlling shear stress as cells flow through the different sizes of syringe/needle for injection is hence desirable (4).
Still, cell viability in a cell suspension is not only affected by effects of the solution or shear stress-related membrane damage; the environmental temperature at which the preparation is stored also dramatically influences cell viability prior to injection. All dispersion media here exhibited a significant decrease in viability over 8 h of cell suspension, indicating that on-ice storage (i.e., hypothermic conditions) is not a safe means to maintain cells prior to injection. However, HTS, which is specifically designed to be cryoprotective, preserved viability the best. Interestingly even a physiological temperature (i.e., normothermia) led to a reduction in cell viability for PBS, aCSF, and DMEM. This is likely the result of normal cell metabolism, which leads to the discharge of waste products into the dispersion medium, and a gradual build-up will lead to an initiation of apoptotic events (7). Indeed, isotonic solutions, such as PBS and DMEM, were designed to balance cells under normothermic conditions by assuming that oxygen and carbon dioxide are balanced, nutrients are present, and waste products are easily removed. In the absence of these additional conditions, the solutions will not buffer cells appropriately. In contrast, at hypothermic conditions, metabolism, enzymatic degradation, and oxygen demand are reduced, but adenosine triphosphate (ATP)-driven membrane pumps controlling the ionic balance are deregulated, hence leading to the induction of apoptosis (7,8). Cells at different temperatures will therefore have different needs for a storage solution (37). Nevertheless, viability here was well preserved in all suspension vehicles at room temperature, which hence might provide an adequate condition to maintain cells for up to 8 h. Still, a more detailed analysis and the development of a dispersion medium specifically designed for the injection of cell suspensions is likely to be a major advance for the field.
In vitro performance characteristics for preparing cell suspensions for intracerebral transplantation can be guided by the above observations. Ideally, a suspension vehicle should have a density close to 2 to limit settling of cells, with a viscosity >3 cP to reduce potential shear stress but also to facilitate maintaining cells in suspension for a long time at 20–40% cell volume fraction. The suspension vehicles should contain appropriate molecules that balance its metabolism while cells are awaiting transplantation. Suspension accuracy should be within 10% of the aimed cell concentration required to exert a therapeutic effect and require pipetting less than five times to achieve a suspension that can be maintained for at least 2 h without resuspension to minimize potential shear stress effects on cells. Cell viability is ideally maintained throughout the duration of suspension without evidence of an increase in cell membrane damage. Although these factors define certain parameters that are conducive to maintaining a cell suspension for injection, further refinement will be specified by the survival, distribution, and differentiation of cells after transplantation.
Relevance to Intracerebral Cell Transplantation
Cell transplantation is a technically demanding procedure that is highly dependent on the reliable transfer of cells from the culture dish to the living brain without affecting their therapeutic properties. Preparation of a cell suspension is the key to this process, as it allows the uptake of cells into a syringe/needle that can be used for intracerebral injection. The cell suspension is highly dependent on the vehicle used, but its quality will determine if cells sediment by settling over time and if they maintain viability while awaiting implantation. Nevertheless, preparation of a cell suspension, as described here, is only the first step in the process. Uptake of the cell suspension into a syringe/needle, duration of injection and potential for sedimentation inside the syringe, shear stress caused by flow through the small bore needle, can also compromise the quality of the suspension prior to its delivery. These aspects hence will require further investigation with the cell suspension described here. Injection parameters, such as speed of ejection, implant volume, as well as induction of an inflammatory response, can also compromise a successful transplantation procedure (23,29). The work presented here provides the foundation for these further investigations.
Conclusion
Preparing a cell suspension that maintains cells in a homogeneous distribution through the vehicle to ensure an efficient delivery while maintaining cell viability is essential to ensure an effective clinical translation of cell therapy. We here described key parameters to define the quality of cell suspensions and compared multiple vehicles in which to suspend human NSCs. From the tested vehicles, HTS provided the most favorable conditions, with Pluronic also showing promise despite significant settling of cells. Simple buffer solutions (PBS, aCSF, and DMEM) performed least favorably. Nevertheless, specifically designed dispersion media might rely on these solutions as starting material to incorporate additives that fulfill specific design characteristics. For instance, alginate, PEG, hyaluronic acid, or extracellular matrix have been used to reduce shear stress of cells during flow through needles (2,4,11,4,61), whereas other materials might aim to provide nutrients or survival factors to promote the health and survival of cells once transplanted (39,48,51).
It is important to note here that additional assays might be required to account for other factors that could influence cell viability and suspension properties, such as cryopreservation or cell isolation (46,60). Although the derivation of the cells used here was performed under GMP conditions, the assays performed here are non-GMP. Validation of the utility of these assays, as well as the implementation of GMP conditions, might be required to incorporate some of this process in a clinical translation pathway (43,50). Although the assays presented here focused on intracerebral cell injection of neural stem cells, it is likely that some of the basic principles in cell suspension will apply to other cells types (e.g., sedimentation), whereas others (e.g., cellular resistance to shear stress) might be cell type and application specific. For instance, intravascular injections can use a larger needle diameter and can accommodate a greater amount of volume than intracerebral injections, hence defining somewhat different design criteria for some of the parameters described here. Preparing and characterizing the cell suspension is a fundamental step to ensure the safe transfer of cells from the dish to the brain, but further investigations aimed at the technical aspects ensuring an efficient and safe intracerebral injection of cells will also be required to ensure a successful clinical translation.
Footnotes
Acknowledgment
The authors thank Drs. Sachin Velankar, Douglas Kondziolka, Lawrence Wechsler, and Mark Richardson for useful discussions on the biomechanics of suspension vehicles, as well as intracerebral injections in patients. This work was partially supported by the National Institute for Neurological Disorders and Stroke (R01NS08226). T.R. conducted measurements of suspension accuracy, viability, and LDH. F.N. assisted with measurements and the overall design of the study. M.M. planned the study, oversaw the experiments, provided funding, and wrote the manuscript. Supplementary material can be accessed at: https://www.researchgate.net/publication/283081632_Rossetti_et_al_Cell_Transplantation_Supplementary_Material.
