Abstract
With the use of cell replacement therapies as a realistic prospect for conditions such as Parkinson's and Huntington's diseases, the logistics of the delivery of cell suspensions to deep brain targets is a topic for consideration. Because of the large cannulae required for such procedures, we need to consider the behavior of cell suspensions within the cannulae if we are to ensure that the injected cells are distributed as intended within the target tissue. We have investigated the behavior of primary embryonic cell suspensions of neural tissue, in cannulae of different diameters, using a protocol designed to mimic the handling and injection of cells during clinical application. Internal cannula diameter had a large effect on the distribution of cells during their dispensation from the syringe. In vertical or near vertical cannulae, cells settled toward the tip of the needle, and were dispensed unevenly, with the majority of cells emerging in the first 10-20% of the injectate. In horizontal or near-horizontal cannulae, we observed the opposite effect, such that few cells were dispensed in the first 80% of the injectate, and the majority emerged in the final 10-20%. Use of a glass cannula showed that the results obtained using the horizontal cannula were caused by settling and adherence of the cells on the side of the cannulae, such that during dispensation, the overlying, cell-free solution was dispensed first, prior to the emergence of the cells. We show that the behavior of cells in such cannulae is affected by the cannula diameter, and by the material of the cannula itself. In horizontal cannulae, uneven expulsion of cells from the needle can be ameliorated by regular rotation of the cannula during the procedure. We discuss the potential impact of these observations on the translation of cell therapies to the clinic.
Keywords
Introduction
The prospect of replacement cell therapies for diseases of the central nervous system (CNS) is both real and exciting. Neurodegenerative diseases, such as Parkinson's disease (PD), Huntington's disease (HD), multisystem atrophy, amyotrophic lateral sclerosis, multiple sclerosis, or retinal degeneration, and brain injuries, such as ischemia or traumatic brain injury, might soon be treated by the implantation of cells into affected brain areas. The aim of these therapies is to replace lost cells (whether neurons or glia) or to use cells as neurochemical or trophic delivery systems in the depths of the brain, and thereby alleviate the clinical symptoms of the conditions concerned.
Currently, such therapies are mainly experimental, not in widespread use, and depend on primary sources of cells such as human embryos. However, the development of safe, reproducible stem cells capable of providing a steady source of implantable cells is becoming a reality and, with it, the prospect of moving CNS cell therapies into the mainstream (2, 4, 6, 12, 23, 36, 40, 41).
In animal models, behavioral deficits associated with parkinsonian or striatal lesions have been shown to be ameliorated by grafting with embryonic ventral mesencephalon or ganglionic eminence, respectively, at the appropriate early developmental stages (10, 13, 38). In rats and mice, this involves the injection of a few microliters of a cell suspension containing several hundreds of thousands of cells, into brain areas up to 5–6 mm from the brain surface. The cannulae used are small, typically 0.15–0.25 mm internal diameter (ID), and the loading and positioning of the cannula and subsequent injection of the cells into the brain is relatively quick, such that the entire procedure may be completed in 5–10 min.
Translation of such cell therapies from animal models to the clinic poses a number of challenges (34), not least the scaling of cell injection protocols for use in the human brain. As in animal models, injection of cells and substances into the brain in the clinic is performed using stereotaxic surgery. Cells are preloaded into a syringe, which is then attached to a stereotaxic frame mounted on the patient's head. Subcortical, target structures such as the caudate nucleus or corpus striatum are deep in the brain and require a long, thin cannula of sufficient rigidity to penetrate accurately to the target site without injury to the overlying structure. In addition, the number of cells to be injected will be in the millions or tens of millions, and a cannula or syringe of considerable volume is required (25). The behavior of cells in suspension within such large-volume cannulae is the subject of the present work.
We have investigated the behavior of cell suspensions prepared from embryonic rat CNS using metal and glass cannulae of different IDs. Cannulae were loaded with cell suspension before being attached to a cannula holder and positioned either vertically (±30°) or horizontally (±30°) and then allowed to sit for up to 20 min. This protocol was intended to mimic the positioning and handling of cannulae during stereotaxic surgery for patients being treated either sitting upright or prone (which can vary in different deep brain stimulation and implantation surgeries, both under general and local anesthesia). Following the settling period, cells were dispensed from the cannula in 8 × 2.5-μl aliquots over a period of 2 min in order to determine their distribution within the cannula after settling and how this might affect the distribution of cells along the injection track. The use of glass cannulae allowed direct visualization of cell settling and for photographs to be taken prior to analysis of cell distribution within the cannulae at different time points during both settling and collection.
Materials and Methods
Cell Suspensions
Whole embryonic rat brains of either 12 days (E12) or 14 days (E14) gestation were collected from time-mated dams (adult rats supplied by Charles River, Margate, UK), dissected in Hank's balanced salt solution (Life Technologies, Paisley, UK). The brains were then cut into smaller pieces and transferred to a 1.5-ml centrifuge tube containing 1 ml of Dulbecco's minimum Eagle's medium (DMEM; Life Technologies). After a further two washes in DMEM, tissue was incubated for 20 min in 1 ml of prewarmed TrypLE Select enzyme (Life Technologies) containing 20 U/ml of the endonuclease dornase alfa (DA; Roche, Welwyn Garden City, UK).
Tissue digestion was stopped by washing 2 × 1 min in prewarmed dissociation medium (DMEM containing 20 U/ ml DA). Following the final wash, tissue was placed in 200 μl of dissociation medium and dissociated by repeated trituration of 150 μl of the suspension each time using standard, plastic pipette tips (first P1000 then P200) to a “near single ” cell suspension. Cell numbers and viability were determined using 2×2-μl samples in trypan blue (Life Technologies) exclusion assays. Finally, cells were gently spun down using centrifugation and resuspended in dissociation medium to a final cell concentration of 100,000 cells/ μl. In order to assess the suitability of rat embryonic suspensions for this work in a pilot stage, cell suspensions were also prepared from human embryonic brain tissue using the same protocol, and the cell diameters from human and rat preparations were compared. Human fetal tissue was collected with local research ethics committee approval (Bro Taf, Pontyclun, Mid Glamorgan, UK), with full consent of the maternal donor, as part of the MRC-sponsored, South Wales initiative for transplantation (SWIFT) program, operating under license from the UK Human Tissue Authority. All animal experiments were conducted in accordance with local ethical review and Home Office licenses according to the UK Animals Scientific Procedures Act 1986.
Latex Beads
Previous work used microfluidic techniques to measure the density of single cells and employed polystyrene beads with an equivalent density showing that they performed like live cells (erythrocytes) (16). As human erythrocytes are similar in size to the cells used here (6-8 μm), the same nonionic, polystyrene (white) beads (Life Technologies) were used to better visualize settling using the current protocol, although cell adhesion properties may differ.
Cannulae
Metal cannulae were prepared from stainless steel tubing (Cooper's Needle Works, Birmingham, UK) cut to length and filed to a smooth, beveled end. The gauges used were 18 (ID 0.83 mm), 21 (ID 0.51 mm), and 25 (ID 0.26 mm), which for convenience are denoted as 0.8-, 0.5-, and 0.25-mm cannulae. For a given volume of cell suspension, as the ID of the cannula decreases, there is a dramatic increase in the length of cannula required. In the present experiment, the approximate volume per unit length for each cannula was as follows: 0.8-mm ID: 5 μl/ cm, 0.5-mm ID: 2 μl/cm, and 0.25-mm ID: 0.5 μl/cm.
Glass cannulae consisted of capillary electrode tubes with a nominal ID of 0.8 mm (Clark Electromedical Instruments, Edenbridge, Kent, UK). Between experiments, cannulae were cleaned by flushing with sterile saline solution followed by 70% ethanol, before air drying.
Loading and Dispensing
Cannulae were attached via a short length of silicone tubing to a 100-μl Hamilton syringe mounted on an electronic syringe driver (model 22; Harvard Apparatus, Holliston, MA, USA). To avoid capillary and mixing effects, the syringe, tubing, and cannula were not filled with priming fluid. To avoid the effects of temperature changes during the experiment, all equipment, media, and cell suspensions were allowed to equilibrate to room temperature before use. For loading of cells, the cannula was clamped in a vertical position and, following gentle mixing of the cell suspension, loaded with 20 μl (0.8- and 0.5-mm ID cannulae) or 5 μl (0.25-mm ID) of cell suspension. The cannula was then immediately reoriented to one of five angles designed to mimic the range of orientations encountered during intracranial surgery. The orientations used were vertical; 30° from vertical; horizontal; 30° from horizontal, tip up; and 30° from horizontal, tip down. Cells were then allowed to settle for 20 min, the period of time estimated for the transfer and attachment of the syringe to the stereotaxic frame and the lowering of the needle into the brain prior to injection. After the settling period, cells were extruded from the cannula in order to determine their distribution within the cannula and how this might affect their distribution following injection. The syringe driver was set to dispense at 10 μl/min, and 8 × 2.5-μl aliquots were collected into eight sequentially numbered PCR tubes. The number of cells collected in each aliquot was determined by counting on a hemocytometer. Each 2.5-pl aliquot was diluted using physiological phosphate-buffered saline to a final volume of 20 μl, and 10-μl samples were then loaded on opposite sides of a hemocytometer, and the numbers of cells counted using 10× objective on an inverted microscope (Olympus CK40). The mean cell count was then used to estimate the total number of cells collected in each aliquot. The collected data are presented as the proportion of cells collected in each aliquot expressed as a percentage of the total collected.
Rotation
In order to ameliorate settling in horizontal orientations, 0.8-mm metal or glass cannulae were loaded with cell suspension as previously described, then rotated through 180° along their long axes once every minute during the 20-min settling period. After 20 min, cells were collected in eight aliquots and counted as previously.
Photography
Photographs of glass cannulae were taken during settling and extrusion of the cell suspension in both vertical and horizontal orientations, using a Canon 95 IS compact camera.
Statistical Analysis
To determine the effects of orientation and cannula size on the distribution of cells in the collected aliquots, data were analyzed by multifactorial analysis of variance (GenStat 16th edition; VSN International, Oxford, UK) with separate analyses undertaken for each experiment with cell source, cannula orientation, and internal diameter as between-sample factors and aliquot as the within-sample factor. Post hoc comparisons were undertaken using Newman-Keuls and Sidak's tests as appropriate.
Results
Cell Diameters
To determine whether or not rat embryonic cells were a suitable proxy for human cells, a comparison was made of cell diameters in cell suspensions prepared from either rat or human embryonic neural tissues. Rat embryos were aged E12-14, and human embryos were of an equivalent developmental age ranging from 7 to 9 weeks postconception.
As shown in Table 1, rat cells were on average 12% larger than human cells in suspension [t(1121) = 6.210, p < 0.001], and rat cell suspensions displayed a wider range of cells sizes [F(752, 371) = 1.418, p < 0.001]. Although significant, not least due to the large sample sizes, the differences between human and rat cell diameters were small, and the range of human cell diameters was encompassed by that of the rat. Rat cells were considered to be a suitable surrogate for the current work.
Comparison of Rat and Human Cells in Suspension

Significant differences by
Student's t-test and
F ratio test, respectively, both p < 0.001.
Cannula Experiments
Settling experiments were all conducted using a cell suspension containing 100,000 cells/μl and using a 20-min wait period between cannula filling and collection of the cells in eight aliquots. The following data report the proportion of cells collected in each aliquot in each experiment. The data from each experiment consist of a minimum of three replicates, and error bars represent the standard errors of the means.
The 0.8-mm Metal Cannula
The distribution of cells collected from each aliquot dispensed from 0.8-mm cannulae at each orientation are shown in Figure 1. There was a marked difference between the proportions of each suspension extruded in each aliquot depending upon the cannula angle [Angle × Aliquot, F(28, 91) = 147.33, p < 0.001].
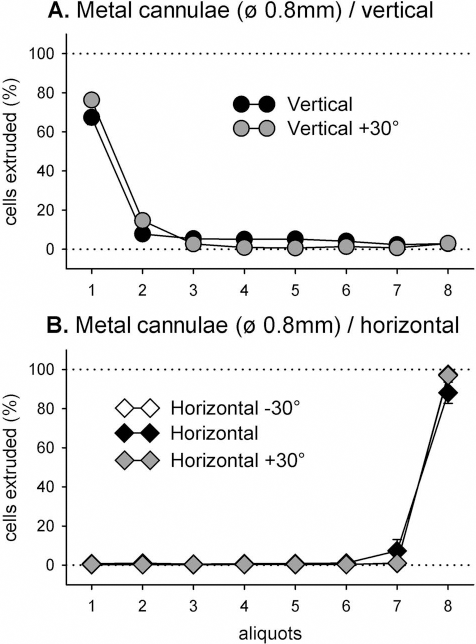
The 0.8-mm cannula quantitation. Proportions of total cells (%) collected in each aliquot, following a 20-min settling period in vertical (A) and horizontal (B) orientations of the cannulae. Vertical bars are SEMs.
Vertical Orientations
Inspection of Figure 1A indicates that cells dispensed from the cannulae oriented either vertically or at 30° from vertical exhibited a marked skew in the numbers of cells toward the first two collected aliquots. On average 65–75% of cells were counted in the first extruded aliquot, with fewer than 5% counted in the final three aliquots combined.
Horizontal Orientations
Conversely, the distributions of cells extruded from cannulae oriented either horizontally or 30° above or below the horizontal were also greatly skewed, but in the opposite direction toward the later aliquots. On average <1% of cells were collected in each the first six aliquots, <10% in the seventh aliquot, and around 90% in the final aliquot.
The 0.8-mm Glass Cannula
In order to visualize and illustrate the behavior of cell suspensions in the 0.8-mm cannula, the initial experiment was repeated using a 0.8-mm glass cannula. Photographs were taken during the 20-min settling period and after each aliquot was dispensed during the expulsion phase.
Vertical Cannula
Figure 2 shows the 0.8-mm glass cannula in a vertical orientation, photographed at 2-min intervals during settling. Cells were initially well mixed (T0). Settling of cells in the cannula was evident after 4 min and was pronounced 6 min after loading. After 20 min, the majority of cells were located near the end of the cannula, in the bottom 3–4 mm (of the total 40 mm) of the height of the liquid in the cannula. As a result, most cells were extruded within the first (2.5 ml) aliquot.

Photographs of the settling of cell suspensions in the 0.8-mm vertical glass cannula. The settling of cell suspensions in the 0.8-mm vertical glass cannula is shown, starting immediately after the filling of the cannula with cell suspension (T0) then at 2-min intervals over the 20-min settling period. Settling was evident after 4 min and prominent after 8 min. Cell clumping was also evident after 8 min followed by continued settling toward the cannula tip.
Horizontal Cannula (Settling)
Figure 3 shows settling of the cell suspension in a horizontally oriented glass cannula during the 20-min settling period. Cells were initially well mixed, but settled quickly so that after 20 min there was a dense layer of cells at the bottom of the cannula, above which was clear, mainly cell-free solution. The use of white-colored latex beads of similar size and density to the cells (16) allowed higher power images of the settling process to be obtained. A clear layer of fluid was visible at the top of the cannula after just 2 min, and by 6 min, beads were only visible in the bottom half of the cannula. The settled layer at the bottom of the cannula was clearly visible after just 4 min, suggesting that even after a short period of settling, some cells might be adherent to the cannula wall.

Photographs of settling in the 0.8-mm horizontal glass cannula. The settling of cell suspensions in the 0.8-mm horizontal glass cannula is shown, starting immediately after the filling of the cannula with cell suspension (T0) then at 2-min intervals over the 20-min settling period. Top: A layer of clear fluid at the top edge of the cannula was apparent after 4 min, which became progressively larger as the cells settled in to a compact layer on the bottom surface. Bottom: Settling at high magnification visualized using white latex beads of similar size and density to embryonic neural cells. Settling is apparent after 2 min. At 4 min, a layer of settled cells is visible. At 8 min, the top half of the cannula is clear of cells.
Vertical Cannulae (Expulsion)
Figure 4 shows the behavior of the settled cell suspension during extrusion of the cells after 20 min of settling. The layer of settled cells appears to be adherent to the lower surface of the cannula. As the needle is evacuated, the overlying, largely cell-free medium is expelled first, and the layer of settled cells accumulates in front of the advancing plunger, only emerging once all of the overlying fluid has been dispensed.

Photographs of the expulsion of the settled cell suspension from a 0.8-mm horizontal glass cannula. The expulsion of the settled cell suspension from a 0.8-mm horizontal glass cannula is shown, starting immediately after the 20-min settling period (T0), then at 15-s intervals during the collection of each of the eight 2.5-μl aliquots. The settled cells collect together as the plunger advances to emerge only after all of the overlying fluid has been expelled.
Quantitation
Results were similar to those seen with the metal cannula. In the glass cannula, oriented either vertically or at 30° from vertical, on average, 50–75% of cells were collected in the first two extruded aliquots, with most of the remaining cells emerging in aliquots 2–7, and less than 5% of cells collected in the final aliquot (Fig. 5A). In 0.8-mm glass cannulae oriented either horizontally, at 30° from horizontal with the needle tip pointing up, or at 30° from horizontal with the needle tip pointing down, on average, <1% of cells were collected in the first six aliquots, <10% in the seventh aliquot, and >80% of cells emerging in the final aliquot (Fig. 5B). The effect of cannula angle on the proportions of cells extruded in each aliquot was again very highly significant [Angle × Aliquot, F(28, 119) = 35.98, p < 0.001].
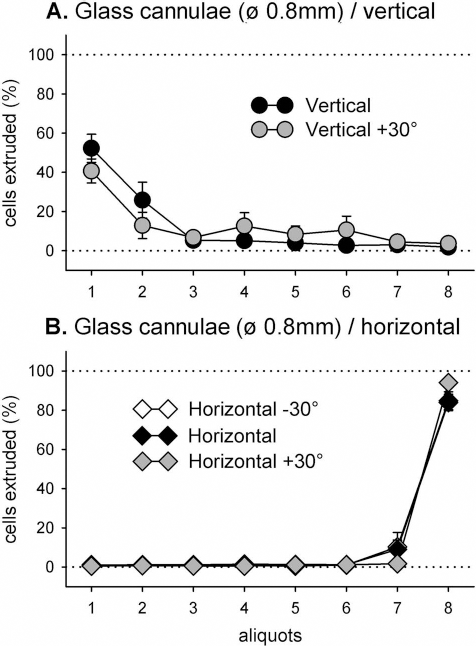
The 0.8-mm glass cannula quantitation. Proportions (%) of total cells collected in each aliquot, following a 20-min settling period in vertical (A) and horizontal (B) orientations of the cannulae. Vertical bars are SEMs.
In summary, settling of cells in 0.8-mm cannulae, whether made of metal or glass, resulted in a skewed distribution of the extruded cells in all cannula orientations. In vertical orientations, settling of the cells toward the cannula tip resulted in extrusion of most of the cells in the first one to two aliquots. In horizontally oriented aliquots, the settled cells were adherent to the lower wall of the cannula, remaining stuck to the wall until physically dislodged by the advancing plunger, and resulting in a collection of the cells in the final one to two aliquots. In a surgical situation, such behavior of cells within the cannula would be likely to lead to uneven distribution of cells in the host brain during implantation.
The 0.5-mm Metal Cannula
Using the 0.5-mm cannulae oriented either vertically or at 30° from vertical, there was again a marked settling of the cells in the cannula, resulting in a skewed distribution of cell numbers in the collected aliquots (Fig. 6A). On average, 30–45% of cells were collected in the first extruded aliquot. Most of the remaining cells emerged in aliquots 2–7, with less than 5% of cells being collected in the final aliquot. Conversely, when the cannulae were oriented either horizontally or 30° above or below the horizontal, there was again a skewed distribution of cell numbers in the collected aliquots opposite to that seen in the vertical orientations (Fig. 6B). On average <1% of cells were collected in the first six aliquots, <20% in the seventh aliquot, and >70% of cells emerging in the final aliquot. The differences between the distributions of cell extrusions at different cannula orientations was again highly significant [F(28, 91) = 53.91, p < 0.001] and was similar in pattern to that seen using the 0.8-mm metal cannula, though not quite as marked (compare Fig. 1 vs. Fig. 6).
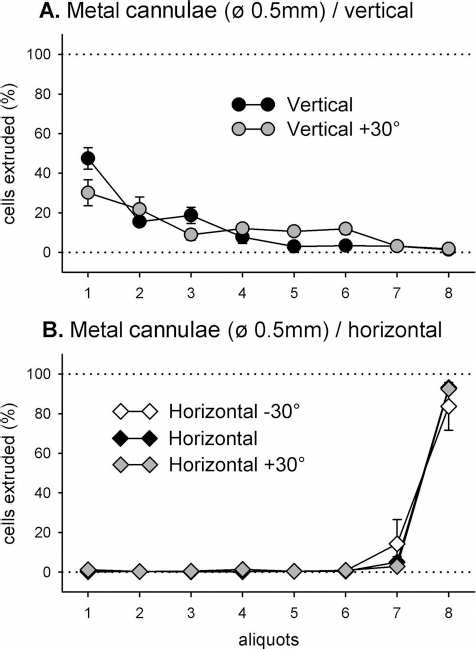
The 0.5-mm metal cannula quantitation. Proportions of total cells (%) collected in each aliquot, following a 20-min settling period in (near) vertical (A) and horizontal (B) orientations of the cannulae. Vertical bars are SEMs.
The 0.25-mm Metal Cannula
Using the thinnest metal cannulae of 0.25 mm ID, of the same order of magnitude as those used in rodent studies, when oriented either vertically or at 30° from vertical there was still an observable settling of the cells in the cannula resulting in a skewed distribution of cell numbers in the collected aliquots (Fig. 7A). Conversely, when the 0.25-mm metal cannulae were oriented either horizontally or ±30° from horizontal, there was again a skewed distribution of cell numbers, but in the opposite direction toward the later collected aliquots (Fig. 7B). Although the effect of angle on the profiles of cell extrusion was again highly significant [Angle × Aliquots, F(28, 133) = 8.20, p < 0.001], the skews in either direction were not nearly as pronounced as that seen with larger cannulae (compare Fig. 7 vs. Figs. 1 and 6).

The 0.25-mm metal cannula quantitation. Proportions of total cells (%) collected in each aliquot, following a 20-min settling period in vertical (A) and horizontal (B) orientations of the cannulae. Vertical bars are SEMs.
In summary, a reduction of internal cannula diameter from 0.8 to 0.5 mm had little effect in horizontally oriented cannulae, but in vertically oriented cannulae, more cells were collected in earlier aliquots. In cannulae of 0.25 mm ID, the effect was more dramatic, and the skewed distribution of collected cells was largely alleviated [Diameter × Angle × Aliquot, F(84, 434) = 15.43, p < 0.001]. Thus, in vertically oriented cannulae, cells were widely distributed across the first six aliquots, while in horizontally oriented cannulae, cells were widely distributed across aliquots 2–8. Note that in standard rat stereotaxic surgery, the predominant orientation is vertical, although there have been specific surgical targets where a relatively oblique orientation has been used (11).
Cannula Rotation
It was hypothesized that, in order to limit the effects of settling in the horizontally oriented cannula, periodic rotation of the cannula along its axis might serve to maintain cells in suspension and to prevent the adherence of settled cells to the wall of the cannula. Using the 0.8-mm glass or 0.8-mm metal cannulae loaded with cell suspension as previously, the horizontally oriented cannula was rotated through 180° at 1-min intervals throughout the 20-min waiting period and the contents collected in eight aliquots as previously described. Figure 8 shows the results of rotations from both types of cannula (A and B, respectively). In the metal cannula, rotation appeared to ameliorate cell settling. Unlike from the nonrotated cannula, cells were collected in all aliquots, with a small peak in aliquot 1. It fell to a minimum in aliquots 6 and 7, and a large peak as previously in aliquot 8 [Rotation × Aliquots, F(7, 84) = 9.87, p < 0.001]. This is consistent with some settling as previously observed, but with a proportion of cells remaining suspended in solution and emerging early in the injection with the overlying fluid.

The effect of rotation in the 0.8-mm horizontal cannula. Proportions of total cells (%) collected in each aliquot, following a 20-min waiting period during which time the cannula was rotated around its long axis at intervals of 1 min. (A) Metal cannula. (B) Glass cannula. Data are plotted against nonrotated data, collected previously, for comparison. Error bars are SEMs.
In the glass cannula, cell distribution was even more evenly distributed across the eight aliquots in the rotating than the stationary cannulae [Rotation × Aliquots, F(7, 63) = 130.45, p < 0.001] such that settling was almost completely eliminated. This suggests that the adhesion (and/or cohesion) of the settled cell layer is less in the glass than in the metal cannula [Cannulae × Rotation × Aliquots, F(7, 147) = 130.45, p < 0.001].
Discussion
Cell therapies targeting deep anatomical structures such as subcortical nuclei in the brain will require the use of long cannulae that have either large IDs or are attached to large-diameter syringes at their distal ends. When such cannulae are loaded with cell suspensions, the possibility of cell sedimentation during the course of the surgical procedure, and the potential for uneven deposition of cells along the injection tract during implantation needs to be considered. Unequal deposition of cells may result in poorer survival or poorer integration of cells in the host brain or in a failure to properly access the target region for which the therapy is intended. In recent cell therapy trials for PD, posttherapy side effects in the form of graft-induced dyskinesias have been observed, which may, in part, have been due to the formation of dopamine “hot spots ” in the host brain resulting in abnormal activation of the neural circuits involved (26). Additionally, when targeting deep structures, it is desirable to avoid repeated penetrations with the injection cannula. In order to minimize damage to both the target tissue and overlying structures and in order to achieve widespread distribution of cells in the target tissue, injection protocols usually involve injection of cells at several different depths along the same needle track. As a result, both the volume of cells loaded into the syringe and the size of cannula required will be large, and the time between initial loading of the cannula and expulsion of the last cell deposit may be lengthy.
Note that the potential for tissue trauma when injecting with large needles must be considered. In animal models, fine glass cannulae (<100 μm) have been shown to cause less damage than larger metal cannulae to the host brain (30, 31). However, glass cannot be used in the clinic because of lack of rigidity and the possibility of fragmentation within the host. Additionally, the use of a very fine cannula would require more extensive dissociation of the donor tissue with the potential for trauma to the donor cells and a potential decrease of both viability and survival. Experiments in rats using different diameter metal cannulae showed a detrimental effect on graft survival using a 2.5-mm cannula (7) as a result of which subsequent clinical trials adopted a 1.0-mm diameter cannula.
Thus, metal cannulae with IDs in the 0.25–1.0-mm range provide a balance between tissue trauma and optimal cell delivery, and there have been a number of clinical trials for the treatment of PD, ALS, and HD, which illustrate this point. Trials using nondissociated cells have used relatively large injection cannulae with IDs of 0.33 mm (5, 14), 0.4 mm (36), 0.5 mm (9, 28), 0.6 mm (15, 32), 0.69 mm (34), 0.8 mm (27), and 1.0 mm (19). However, for reasons detailed previously, trials using dissociated cell suspensions have also employed large cannulae in the range 0.5 mm to 0.8 mm ID (8, 24, 28, 33, 35, 37).
The behavior of cell suspensions within such cannulae will be influenced by a number of factors. In the current article, we have considered the material of the cannula itself, cannula diameter, cannula angle during surgery, and cannula rotation. The amount of time between filling of the cannula and injection of the cells into the target area will also be important, though this is not reported in the literature. The protocol used in the present work was designed, following discussions with neurosurgeons, to represent a maximal interval between filling of the cannula and cell injection, allowing time for the syringe to be transported to the patient, affixed to the stereotaxic frame, and lowered into the target structure. In practice, the surgical apparatus and protocols used by others may be longer or shorter than those used here. Although the specific combinations of cell preparation, density, timing, instruments, and angles of delivery will not match exactly those used in any particular clinical trial, the large and consistent results observed indicate that these factors must be considered and empirically validated for each variant prior to use in surgical practice.
The present results show clearly that there may be considerable settling of embryonic neural cell suspensions in large-diameter cannulae (0.8 and 0.5 mm ID), regardless of the angle of orientation, but that settling had opposing effects, depending on the gross orientation of the cannula. In a vertical cannula, settling caused an accumulation of cells near the tip of the needle, and during ejection of the cells from the cannula, the majority of cells emerged in the first part of the injection. In a surgical setting, where cells are intended to be injected as multiple deposits, from deepest to shallowest, along the same injection track, this would result in a bolus of cells dispensed at the bottom of the needle track and few cells implanted in the overlying tissue (Fig. 9A). By contrast, in a horizontal cannula, the reverse situation was observed, with few cells emerging in the early part of the injection and the great majority of cells emerging in the last one to two aliquots of the injection. In a prone patient, this would result in few or no cells dispensed in the deepest parts of the target tissue and the majority of the transplanted cells deposited at the top of the needle track (Fig. 9B). Deviation of the cannula by ±30° from either the vertical or horizontal did not significantly alter the observed skewed distribution of the collected cells.
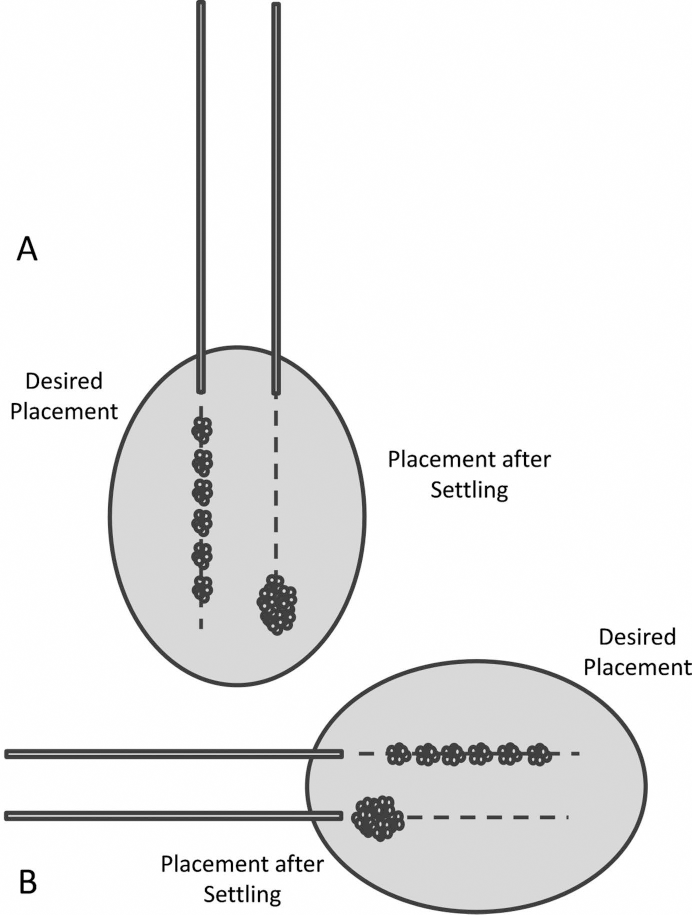
The effects of settling on tissue distribution in the target tissue. Where the desired placement is to distribute cells in multiple deposits throughout the target structure (gray shaded area in diagram), settling may disrupt the even distribution of cells. (A) In a vertical orientation, settling will cause the majority of cells to be deposited at the bottom of the injection tract (dashed line). (B) In a horizontal orientation, settling will cause the majority of cells to be deposited at the top of the injection tract.
Observation of settling and expulsion of cell suspensions in a 0.8-mm ID glass cannula provided an explanation for the skewed distribution of collected cells when using the 0.8-mm ID metal cannula. In the vertical glass cannula, cells could clearly be seen collecting near the tip of the needle, which then quickly emerged within the first few microliters on expulsion. In the horizontal orientation, the skewed distribution of collected cells was seen to be caused by adherence of the settled cells in a layer on the wall of the cannula, where they remained until they were forced to detach by the advancing plunger. As a result, during expulsion, the overlying, cell-poor fluid was dispensed first, while the settled cells collected in front of the moving plunger, emerging en masse, in the last one or two aliquots.
Use of a 0.5-mm ID cannula partially alleviated settling effects in the vertical, but not horizontal orientations, whereas use of a smaller cannula of 0.25 mm ID significantly alleviated settling effects in all orientations. This resulted in a much more even distribution of cells in the collected aliquots. Based on what was observed previously in the glass cannula, this was likely due to the final packed volume of the settled cells, which was large relative to the internal volume of the cannula, leading to a correspondingly small volume of cell-free fluid within the needle.
Routine use of small-diameter cannulae would seem an obvious solution to the cell-settling phenomenon described here. Provided that the settled volume of cells is close to the internal volume of the cannula, and there are no significant areas of cell-free medium within the syringe, the skewed distribution of ejected cells could be largely avoided. However, it should be noted that, although this small size is similar or larger than the cannulae used in most rodent experiments on which clinical trials are initially developed (including preclinical validation of human cells following rodent xenotransplantation), 0.25 mm ID is significantly smaller than any clinical instrument in current use. A cannula of small ID becomes impractical for injections larger than a few tens of microliters in volume because as the ID of the cannula decreases, the length of cannula required increases as the square of the cannula's radius. Thus, for a modest injection volume of 20 ml, a cannula of 0.25 mm ID needs to be more than 41 cm long. A cannula with an ID of 0.1 mm would require a cannula 200 cm long. While it is possible to use a small-diameter cannula connected to a syringe, this merely has the effect of transferring the bulk of the cell suspension from the cannula to the syringe itself and the potential for the settling effects within the syringe still remains. A similar effect might be achieved by increasing the cell concentration used, likewise to increase the settled cell volume within the needle. There are limitations to this approach also, as there will be physical limits to the concentration of cells that can be loaded and ejected from the cannula without blocking. The much reduced buffering and nutrient capacity of smaller volumes of the injection medium may also be detrimental to the health and survival of the cells.
We have shown here that the skewed distribution of cells in horizontally oriented cannulae can be ameliorated by rotation of the cannula at regular intervals during the procedure. This effect was more pronounced in glass cannulae than in metal, suggesting that the interaction between the cells and the cannula material is a factor worthy of further investigation. Whether the adherence of the cell layer to the wall of the cannula (and/or the adherence of cells to each other) is due to chemical, physical, or charge effects has not been studied here, but all of these might conceivably be manipulated to decrease adhesion even if settling effects in solution cannot be avoided. The present data suggest that, in prone patients, the use of cannula rotation during the injection is a viable strategy. Indeed, there are examples of the use of this strategy in the literature. In trials carried out in Sweden using the 0.8-mm ID Rehncrona injection device (8, 39), the needle was rotated at regular intervals during the procedure (personal communication with Dr. S. Rehncrona). Similarly, in a clinical trial for PD carried out by Mendez et al., the injection protocol incorporates rotations of the cannula between deposits (29). Postmortem examination of the brains of patients from both of these studies were able to show surviving grafts well distributed within the target tissue (3, 20). Similarly, in a clinical trial for stroke using a cannula with an ID of only 0.25 mm, attached to either a 250-μl or 100-μl syringe, the syringe and cannula were rotated during the procedure in order to “maintain a mixture of cells within the solute ” (18). In order to counter settling, it is clearly important to keep the main volume of cell suspension agitated in some way. For a syringe with a fixed needle, rotation of the syringe at regular intervals during the procedure is likely to be sufficient. As an alternative, where a large volume syringe is connected via fine caliber tubing to a cannula, rocking or rotation of the syringe at intervals might also be possible.
The present study is essentially empirical, and a detailed consideration of the physics of cell sedimentation is beyond the scope of the current work. However, we can infer how changes in cell size or medium viscosity might affect the rate of settling within the cannula from Stokes' law on the settling speed of small spheres in a fluid medium. According to this, the settling velocity (V) of a cell is given by the formula

where g is gravitational acceleration, D is the cell diameter, ρ c is cell density, ρ is fluid density, and μ is fluid viscosity (21). Based on this relationship, for a given cell density, settling velocity increases with the square of the cell diameter, and larger cells settle more quickly than small ones. Similarly, we would expect the settling velocity of clumps of cells to be high relative to single cells of the same type. This is an important consideration. In the clinical setting, cell suspensions might well be only partially dissociated in order to minimize cell trauma and may contain clumps of cells likely to settle more quickly than the relatively well dissociated cell suspensions studied here.
In contrast, the viscosity of the medium has an inverse, linear effect on settling such that a doubling of medium viscosity will effectively halve the settling velocity of the cells. Therefore, slowing down the sedimentation rate of cells within the cannula by increasing the viscosity of the medium is another strategy worth consideration. Alginates (naturally occurring complex sugars extracted from brown seaweeds) can be used to increase the viscosity of physiological solutions, without toxicity to the cells concerned. These have been used in a variety of in vitro applications (e.g., microfluidics) and for the injection of cells into the vascular system in animal models (22) or as hydrogels to construct tissue scaffolds (1, 17).
There are a number of other factors, not investigated in the present work, which might have an effect on cell behavior in large cannulae. In the translation of the current protocols to the clinic, different factors might further ameliorate or exacerbate cell settling. The time between filling of the cannula and injection is clearly critical and, based on the current data, should be kept as short as possible. Higher cell concentrations will result in larger packing volumes of the settled cells and, by default, better distribution of the cells from the needle. However, if these are too high, then cell health and even flow of the cell suspension within the needle may be compromised. The effects of temperature may also need to be considered. All of the experiments reported here were carried out at room temperature, but in a clinical setting, cell suspensions may be kept chilled or on ice before loading into the injection cannula (at room temperature) and then be lowered into the tissue to be injected, which is at body temperature. The increase in temperature may cause expansion of the cell preparation, and convection currents may be set up within the cannula, and the effects of this on the settling process is unclear. Cannulae made from different materials might also have an effect. In the current work, the ameliorating effect of rotation was greater in glass cannulae than in metal. Whether this was due to the surface properties of the material itself or the effects of other factors such as electrical charge or temperature stability is not known. In a horizontal cannula, adhesion of the cell layer to the cannula wall prevented the movement of cells with the overlying fluid during injection, so that low friction coatings of the internal wall of the cannula, such as silicone or Teflon®, might be considered to overcome this.
The present results do not preclude the use of large cannulae for cell therapies in the clinic; indeed, such cannulae have already been used in a number of trials with successful outcomes. Rather, the data point to the limitations of large cannulae, particularly for large volumes of single cell suspensions and where lengthy, multideposit procedures are proposed. When using such cannulae, procedures should be validated using the actual instruments and delivery protocols proposed, so that potential problems of uneven cell delivery may be identified and addressed before commencement of the clinical trial.
Footnotes
Acknowledgments
This work was funded by grants from the UK Medical Research Council, the European Union TransEUro 7th Framework program, and by Aide à la Mobilité des Etudiants AMIE par la Région Ile de France. The authors declare no conflict of interest.
