Abstract
Ischemic heart disease is the leading cause of mortality in industrialized countries. Cell transplantation could restore function of the ischemic heart likely through the mechanism of cell-induced angiogenesis. We have previously shown that cells isolated from uteri increase angiogenesis and alleviate cardiac dysfunction when transplanted after MI. However, which uterine cell type contributes to angiogenesis is unknown. Here we report that uterine-derived CD11b cells significantly increase vasculogenesis and promote myocardial healing in ischemic cardiomyopathy. We have established a novel and simple methodology for uterine CD11b cell isolation and enrichment and demonstrate that this technique can be used for purifying and establishing viable CD11b cell cultures in rats. The isolated fresh CD11b cells were transplanted into ischemic rat hearts 5 days after injury. Following transplantation, vasculogenesis significantly increased in ischemic cardiac tissue, which reduced infarct size and restored myocardial function and perfusion compared with controls. Thus, uterine CD11b cells have the potential to promote functional healing when implanted after ischemic cardiomyopathy. Importantly, we have demonstrated a novel means by which CD11b cells can be easily purified and cultured for cell transplantation.
Introduction
Ischemic heart disease is the leading cause of mortality in the world1,2. The results of clinical trials evaluating angiogenic protein and gene therapies as possible approaches to establish a “biological bypass” indicate that a more profound vasculogenesis is required3–5. Clinical and scientific research data accumulated over the last decade suggest that cell transplantation can prevent ventricular dilatation after a myocardial infarction (MI)6–9. Implanted cells are proposed to improve cardiac function by multiple mechanisms, including the induction of myogenesis 10 , angiogenesis 11 , stem cell recruitment12,13, and matrix alteration 14 . Recent research has identified cell-derived angiogenesis as a critical factor in the restoration of cardiac function reduced by ischemic cardiomyopathy15,16. This suggests that the transplantation of new cells with great angiogenic capacity could be a novel therapy to restore the cardiac function of hearts with ischemic cardiomyopathy.
The uterus is a rare site of physiological angiogenesis in the adult body. Previous studies have shown that transplantation of a heterogeneous mixture of uterine cells into rat myocardium after an MI increased angiogenesis and tissue repair and prevented cardiac dysfunction 17 . However, which uterine cell type contributed to these significant effects is unknown. Increased angiogenesis is vital to tissue repair postischemia, and cell–cell signaling initiated by CD11b cells can promote vascularization 18 . For example, in a mouse model of retinal angiogenesis, CD11b cell recruitment and cell–cell signaling were required to trigger functional angiogenesis in endothelial cells 19 . CD11b cells may also play a role in regeneration beyond angiogenesis, as they have been shown to be required for limb regeneration in salamanders 20 . In the mammalian cardiovascular system, CD11b cell depletion abrogates the normal ability of neonatal hearts to recover completely after MI 21 . These studies suggest that CD11b cells may be a potent cell source for transplantation to promote angiogenesis and regeneration in damaged tissues. We hypothesized that transplantation of uterine-derived CD11b cells would induce angiogenesis and promote myocardial healing in ischemic cardiomyopathy.
In the current study, we isolated CD11b-positive and CD11b-negative cells from uterine tissue using their specific ability to grow out from heterogeneous primary cultures, thus allowing for their purification without the use of antibodies. The isolated cells were implanted into rodent hearts with ischemic cardiomyopathy to investigate their angiogenic and tissue regenerative capacities. We found that CD11b-positive cells enhanced vasculogenesis and better improved cardiac function and perfusion in a rat cardiac ischemia/reperfusion (I/R) injury model compared to CD11b-negative cells and a control. This suggests that uterine CD11b cells may have therapeutic potential in regenerative medicine.
Materials and Methods
Uterine Cell Collection
All animal protocols were approved by the Animal Care Committee of the Toronto General Research Institute, University Health Network (Toronto, ON, Canada). Female C57BL/6 mice aged 8–20 weeks and Sprague–Dawley rats aged 3 months were purchased from Charles River Laboratories (Saint-Constant, QC, Canada) and used in both in vitro and in vivo studies.
For the in vitro studies, the animals were sacrificed and the uteri were dissected and rinsed in a 10-cm dish with warm Iscove's modified Dulbecco's medium (IMDM; Sigma-Aldrich, Oakville, ON, Canada). The uterine horns were cut longitudinally and rinsed to wash away uterine fluid. Tissue was then placed in a 1.5-ml microfuge, minced into pieces (~3 mm3 in length) using curved scissors, transferred to a 50-ml tube with 10 ml of digestion buffer [IMDM with 0.2% collagenase II (Worthington Biochemical, Lakewood, NJ, USA), 0.25% trypsin (Sigma-Aldrich), and 0.5 U/ml DNase (Life Technologies, Burlington, ON, Canada)], mixed, and incubated at 37°C. After 45 min, the cells were “shaken off” of the tissue by repeatedly pipetting the mixture using a 10-ml pipette. Increased turbidity indicated the presence of cells and debris. The pieces of tissue were allowed to settle to the bottom of the tube, and the supernatant was collected in a new tube, passed through a 40-μm nylon mesh, topped-up with IMDM with 10% fetal bovine serum (FBS; Life Technologies), centrifuged (234 × g) to pellet cells, and washed. Additional cells were collected from the remaining undigested tissue pieces by adding 10 ml of fresh digestion buffer and repeating the procedure using 15-min incubation periods. Approximately 2 h was required for maximum yield from mouse and 3 h from rat uteri. Cells collected from each enzyme incubation step were pooled, and hemolysis was performed using Gey's Solution (Sigma-Aldrich). A yield of approximately 2 × 106 uterine cells from one female mouse, or 2–2.5 × 107 uterine cells/rat with ~90% viability assayed by trypan blue (Sigma-Aldrich) exclusion was obtained.
Uterine Cell Culture
Uterine cells were cultured in IMDM with 10% FBS. Where specified, cells were supplemented with 50 ng/ml of species-matched recombinant macrophage colony-stimulating factor (M-CSF; PeproTech, Rocky Hill, NJ, USA) or cultured using banked supernatants from uterine cell or STO cell cultures (see below). Uterine cells were seeded at a density of 1 × 106 in 10-cm dishes.
STO Cell Culture
STO cells [mouse embryonic fibroblast cell line; American Type Culture Collection (ATCC), Manassas, VA, USA] were cultured using routine procedures in IMDM with 10% FBS for supernatant collection. Growth-arrested STO cells for use as feeder layer cultures were prepared by incubation for 3 h with mitomycin C (10 μg/ml; Sigma-Aldrich). For feeder layer cultures, STO cells were plated at 6 × 105/well in six-well plates (Sarstedt, Newton, NC, USA) a day before introducing uterine cells for coculture.
Rounded Cell Isolation Technique
After cultured uterine cells reached confluence, cells exclusively adopted a round, loosely adherent structure. Rounded cells were collected by gently rinsing the culture dish and harvesting the supernatant.
Immunohistochemistry for Cell Identification In Vivo and In Vitro
For localization of CD11b cells in the uterine tissue, uteri were fixed using 4% paraformaldehyde (PFA; Sigma-Aldrich), frozen, sectioned, and used for identification using antibodies and for hematoxylin and eosin (H&E; Sigma-Aldrich) staining. For identification of CD11b cells in culture, phycoerythrin (PE)-conjugated anti-mouse CD11b antibody (1:200; STEMCELL Technologies, Vancouver, BC, Canada) was used for staining, and the positive cells were visualized by a Nikon Eclipse Ti fluorescent microscope (Tokyo, Japan) in five randomly selected fields per section. For immunofluorescence staining, the sections were dried at room temperature briefly, followed by fixation in 2% PFA for 10 min at 4°C. The slides were then incubated with the primary antibody at room temperature for 2 h. The nuclei were identified with 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich).
Flow Cytometry
For quantification of CD11b-positive cells, flow cytometry (FACSCanto II; BD Biosciences, Mississauga, ON, Canada) was used to analyze uterine cells labeled with PE-conjugated anti-mouse CD11b antibody (1:200; STEMCELL Technologies) and F4/80 antibody (1:200; BD Pharmingen, San Jose, CA, USA). Alexa 488-conjugated donkey anti-rat (1:400; Life Technologies) secondary antibody was used for the detection of F4/80.
Filopodia Extension Assay
Culture dishes were coated with FBS by incubation with FBS overnight. The following morning, the FBS was aspirated and the plates were rinsed with phosphate-buffered saline (PBS; Sigma-Aldrich). Rounded CD11b cells were cultured in the FBS-coated dishes and photographed.
Magnetic Cell Isolation
For mice, uterine CD11b cells were also purified using a commercial CD11b isolation kit composed of a targeting antibody and magnetic nanoparticles with an EasySep magnetic column according to the manufacturer's instructions (STEMCELL Technologies,). The purity of cell fractions was determined by flow cytometry. CD11b-positive cell fractions were ~99% pure, while CD11b-negative cells were ~95% pure.
Rat I/R Model
For the in vivo studies, I/R injury was induced as previously described 22 . Briefly, animals were anesthetized with 2% isoflurane (Pharmaceutical Partners of Canada Inc., Richmond Hill, ON, Canada), intubated, and a thoracotomy was performed. The left coronary artery was ligated with a 7-0 proline suture to induce tissue ischemia, confirmed by paling of the left ventricle. After 1 h, the ligature was released, and the tissue underwent reperfusion and returned to its normal red color. The chest was closed, animals were monitored, and analgesics were administered. Five days later, under general anesthesia, 50 μl of serum-free medium containing 9 × 105 CD11b-positive or CD11b-negative cells from uterine tissue or medium only (control) was delivered intramyocardially (n = 6/group).
Myocardial Function and Perfusion Measurement
Myocardial function was assessed by echocardiography (Echo). Using Echo, left ventricular internal end-diastolic (LVIDD) and end-systolic dimensions (LVIDS) were measured on the day of I/R, 5 days prior to cell transplantation, the day of cell transplantation, and then 7, 14, 21, and 28 days after cell transplantation. The percentage of fractional shortening (%FS) was calculated as follows: %FS = (LVIDD – LVIDS)/LVIDD × 100).
Myocardial perfusion was evaluated using myocardial contrast echocardiography on day 28 after cell transplantation. Rats were sedated, and 200 μl of DEFINITY microbubble (Bristol-Myers Squibb Canada, Montreal, QC, Canada) solution was infused into the tail vein at a rate of 1.2 ml/h. Digital images were used to measure signal intensity offline at a pulsing interval of 1,800–2,000 ms (plateau intensity). The anterior wall represented the infarct region, and the posterior wall represented the noninfarct (normal) region. The intensity ratio was calculated (signal intensity in the anterior wall divided by that in the posterior wall) to estimate myocardial perfusion as we previously described 23 .
Infarct Area and Blood Vessel Density Measurement
Infarct area and blood vessel density were measured on day 28 after cell transplantation. After the Echo functional analysis was complete, rats were sacrificed and their hearts were rapidly excised and fixed in 10% formaldehyde. Computerized planimetry (measuring infarct area) and immunohistochemical staining (measuring blood vessel density) were performed as we previously described24,25. Briefly, the infarct border region sections obtained on day 28 after cell transplantation were immunolabeled for α smooth muscle actin (α-SMA) to stain arterioles and factor VIII to stain capillaries. To estimate blood vessel density areas, total α-SMA- and factor VIII-positive areas were measured and expressed as percentages of total areas. Infarct areas were measured by computerized planimetry and presented as a percentage of the left ventricular free wall area, calculated as follows: LV infarct area/LV area × 100.
Statistics
Data are expressed as mean ± SD. Student's t-test was used for comparison of means between two groups. Comparisons of means among three groups were done by analysis of variance (ANOVA) using GraphPad Prism 5 software (La Jolla, CA, USA). If the F test was significant, Tukey's (one-way for perfusion, infarct area, and blood vessel density) and Bonferroni's (two-way repeated-measures for Echo function studies) post hoc tests were used to identify any statistically significant differences between groups. Differences were considered statistically significant at p < 0.05.
Results
CD11b Cells Are Present Throughout Uterine Tissue
Murine uteri contain a population of detectable CD11b-positive cells, which were identified by staining with CD11b antibody. These CD11b cells were present throughout the uterus, in the myometrium, stroma, and endometrium (Fig. 1A–D). CD11b-positive cells were also identified by flow cytometry (Fig. 1E).
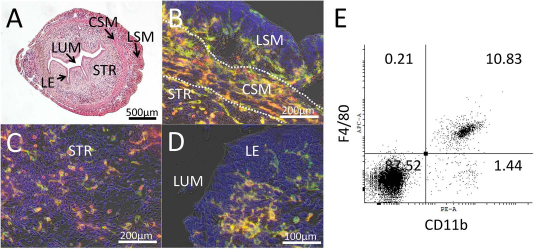
Uterine tissue contains CD11b-positive macrophage-like cells. Hematoxylin and eosin staining (A) and immunohistochemistry localizing CD11b cells that coexpress F4/80 (green) and CD11b (red) expression in the myometrium (B), stroma (C), and endometrium (D) of mouse uterine sections. CSM, circular smooth muscle; LE, luminal epithelium; LSM, longitudinal smooth muscle; LUM, lumen; STR, stroma. (E) The uterine macrophage population can be identified by flow cytometry double staining for CD11b and F4/80.
Uterine CD11b Cells Become Round in Cell Culture
Murine uteri were enzymatically digested to collect cells for culture. Uterine cell cultures contained a population of round, phase-bright cells that increased in number over time (Fig. 2A–C). Time-lapse image capture demonstrated adherent cells that transitioned to a round phenotype (Fig. 2D–F). Antibody labeling showed that adherent cells undergoing rounding (Fig. 2G) and rounded cells (Fig. 2H) were exclusively CD11b positive. These results suggest that rounded cells derived from monolayer adherent uterine cell culture are CD11b-positive cells.

Uterine CD11b-positive cells transition from an adherent morphology to a rounded shape in heterogeneous cell culture. Plates (10 cm) were seeded with 1 × 106 heterogeneous cells from uterine tissue and cultured. Media changes were minimized, and the culture was observed at 1 day (A), 7 days (B), and 28 days (C). Round, phase-bright cells were observed as the culture progressed. (D–F) Time-lapse video showed that these cells transitioned from an adherent to a rounded phenotype. Immunohistochemistry demonstrated that all rounding and rounded cells in early (G) and late-stage (H) cultures were CD11b positive. All images are mouse uterine cell cultures.
To confirm this result, CD11b-positive and -negative uterine cells were magnetically fractioned, isolated, and compared for rounding ability. Uterine cultures lacking CD11b cells did not round after 21 days in culture, while unfractionated cultures did show significant numbers of rounding cells (p < 0.01) (Fig. 3A–C). We then investigated whether the rounding of some cells was a result of extensive time in culture or if it was a result of confluence. When uterine cells were cultured on an STO feeder layer, CD11b-positive cultures showed significant numbers of rounding cells after only 7 days in culture, while CD11b-negative cells showed no evidence of rounding cells (p < 0.01) (Fig. 3D–F).

Uterine cell rounding is unique to CD11b-positive cells. Unfractioned mouse uterine cells (A) and mouse uterine cells depleted of CD11b cells by magnetic cell sorting (B) were cultured using 1 × 106 cells/well in a six-well plate for 21 days, and cell rounding was quantified. (C) Rounded cells were observed in unfractioned cultures, but their presence in CD11b-depleted cultures was minimal (**p < 0.01). Similarly, CD11b-positive (D) and -negative (E) mouse uterine cells were cultured using 1 × 106 cells/well in six-well plates on a STO cell feeder layer (prepared from 6 × 105 mitomycin-treated STO cells/well), and the number of rounding cells was counted after 7 days in culture. (F) Rounding cells were detected in CD11b-positive cultures but not in CD11b-negative ones (**p < 0.01).
When rounded CD11b-positive cells were observed at high magnification, it was noted that these cells were gently tethered by filopodia at their base (Fig. 4A and B). These cells could be collected by gently rinsing the dish (Fig. 4C), and they could be cultured (Fig. 4D). Purified cells were viable and had functional filopodia capable of extension (Fig. 4E).

The cell rounding phenotype can be used to isolate CD11b-positive cells, and isolated cells retain functional filopodia. (A) Rounded mouse uterine CD11b cells are phase-bright cells. (B) High magnification shows that these cells are gently anchored by filopodia (arrows) at their base. (C) Rinsing the plate detaches rounded CD11b-postitive cells, allowing for their collection. (D) These collected cells can be cultured. (E) Filopodia extension was observed when rounded mouse CD11b-positive cells were isolated and plated on fetal bovine serum (FBS)-coated dishes.
Increased Myocardial Vascular Density and Perfusion Following CD11b Cell Transplantation
Twenty-eight days after cell transplantation, α-SMA immunostaining of the infarct border region identified large vascular structures (arterioles) with smooth muscle cells within their walls, and factor VIII immunostaining identified small vascular structures, mainly capillaries (Fig. 5A–F). The staining areas of both types of vessels within the border region of the infarct were significantly increased following transplantation of CD11b-negative cells compared with the implantation of control medium (p < 0.01 for both arterioles and capillaries) (Fig. 5G and H). However, the greatest increase in vascular areas (both arterioles and capillaries) was observed when CD11b-positive cells were transplanted (p < 0.01 vs. CD11b-negative cells and control medium) (Fig. 5G and H).
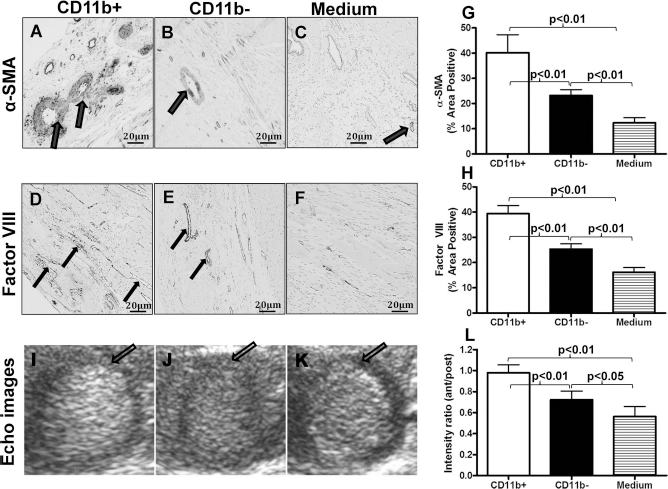
Vascular density and myocardial perfusion following cell transplantation. (A–F) Photomicrographs of representative myocardial sections 28 days after cell or medium injection. The infarct border region was immunolabeled with α-SMA for arterioles or factor VIII for capillaries (large arrows for arterioles and small arrows for capillaries). (G) The percentage of arteriolar-positive areas was higher in the CD11b-negative group compared to the control medium group (p < 0.01), but was highest in the CD11b-positive group (p < 0.01 vs. control medium and CD11b-negative groups). (H) The percentage of capillary-positive areas was also higher in the CD11b-negative group compared to the control medium group (p < 0.01), but was highest in the CD11b-positive group (p < 0.01 vs. control medium and CD11b-negative groups). (I–K) Representative images obtained by myocardial contrast echocardiography performed 28 days after cell or medium transplantation (arrows indicate anterior wall). (L) Myocardial perfusion (ratio of signal intensity in anterior wall (infarct region) to posterior wall (noninfarct region) 28 days after transplantation was measured by myocardial contrast echocardiography. By day 28, transplantation of CD11b-negative cells improved cardiac perfusion compared to implantation of the control medium (p < 0.05), but the greatest improvement occurred following CD11b-positive cell transplantation (p < 0.01 vs. CD11b-negative group and control medium group; n = 6/group).
Contrast echocardiography was employed to assess myocardial perfusion by comparing the ratio of the signal intensity in the anterior wall (infarct region) to the posterior wall (noninfarct region) 28 days after cell transplantation (Fig. 5I–K). Consistent with the increased blood vessel density areas, the signal intensity ratio was significantly higher 28 days following transplantation of CD11b-negative cells compared with control medium (p < 0.05) (Fig. 5L). A further increase in perfusion occurred when CD11b-positive cells were transplanted (p < 0.01 vs. CD11b-negative cells and control medium) (Fig. 5L). These data demonstrate that macrophage transplantation increased blood vessel density and enhanced myocardial perfusion.
Improved Cardiac Function and Reduced Infarct Area Following CD11b Cell Transplantation
Cardiac function was determined by echocardiographic examination. Fractional shortening (FS) was similar among all three groups before I/R (day –5) and decreased to a similar degree 5 days following I/R (day 0, prior to cell transplantation) (Fig. 6A). However, during the 21 to 28 days after cell transplantation, FS was significantly greater in the rats that received CD11b-negative cells compared with those that received the control medium (p < 0.05) (Fig. 6A). The greatest improvement in FS was seen following CD11b-positive cell transplantation (p < 0.01 vs. control medium and p < 0.05 vs. CD11b-negative cells). Left ventricular dimensions were also assessed at various time points before and after cell transplantation (Fig. 6B and C). The control medium group exhibited the largest systolic and diastolic dimensions, while the CD11b-positive cell transplantation group exhibited the smallest dimensions (p < 0.05, CD11b-positive vs. CD11b-negative groups; p < 0.05, CD11b-negative vs. control medium group; p < 0.01, CD11b-postive vs. control medium group).

Cardiac function and infarct area following macrophage transplantation. Cells or medium were injected on day 5 after ischemia/reperfusion (I/R) injury. Echocardiography was performed before I/R (day –5) and 0, 7, 14, 21, and 28 days after transplantation. By days 21 to 28, implantation of CD11b-negative cells improved fractional shortening (A) compared to the control medium (*p < 0.05), but the greatest improvement was found following CD11b-positive cell transplantation (**p < 0.01 vs. control medium group, #p < 0.05 vs. CD11b-negative group). Left ventricular internal systolic dimensions (LVIDS) (B) and diastolic dimensions (LVIDD) (C) were also measured before I/R (day –5) and 0, 7, 14, 21, and 28 days after transplantation. At day 28, the control medium group exhibited the largest systolic and diastolic dimensions, while the CD11b-positive cell group exhibited the smallest dimensions (#p < 0.05, CD11b-positive vs. CD11b-negative groups; *p < 0.05, CD11b-negative vs. control medium group; **p < 0.01, CD11b-postive vs. control medium group; n = 6/group). (D) Representative heart slices obtained 28 days after cell or medium transplantation. Arrows indicate the location of the infarct in individual slices. (E) Infarct areas were smaller in the CD11b-negative group compared to the control medium group (*p < 0.05), but were smallest in the CD11b-positive group (**p < 0.01 vs. control medium group, #p < 0.05 vs. CD11b-negative group; n = 6/group).
Figure 6D shows representative heart slices obtained 28 days after cell or medium transplantation. Arrows indicate the location of the infarct in individual slices. Computerized planimetry (performed 28 days after cell transplantation on explanted hearts fixed at physiologic pressures) showed that the infarct area was larger in animals that received control medium than in those that received CD11b-negative cell transplantation (p < 0.05) (Fig. 6E). Importantly, the infarct area was the smallest in animals that received CD11b-positive cell transplantation (p < 0.01 vs. control medium and p < 0.05 vs. CD11b-negative cell transplantation) (Fig. 6E). These data demonstrate that CD11b cell transplantation produced the greatest improvement in ventricular function and reduced adverse LV remodeling.
Discussion
In the current study, we demonstrated that CD11b-positive cells are able to grow out from the monolayer of cultured uterine cells. These cells were pure in population and were able to be implanted into ischemic myocardium, where they induced significant vasculogenesis and restored cardiac function.
Cell transplantation for ischemic cardiomyopathy has been under extensive investigation because of its potential to restore heart function after a MI6–9. Although several mechanisms have been proposed to explain the functional improvements associated with cell transplantation, it is likely that angiogenesis derived from the implanted cells plays a particularly important role. Vasculogenesis particularly arteriogenesis, is a necessary process that, concomitant with the growth and regression of the human endometrium, provides an extensive blood supply to this rapidly changing tissue 26 . The angiogenic mechanisms in the uterus are well established and of a significant magnitude, and this effect could be exploited to promote the compensatory growth of blood vessels in ischemic cardiomyopathy. We have previously demonstrated that a heterogeneous mixture of uterine cells could increase angiogenesis and tissue repair and prevent cardiac dysfunction when transplanted after an MI 17 . However, it is unknown which cell type contributed to the angiogenesis. Importantly, the extent and type of angiogenesis induced could be determined by the type of uterine cells implanted.
Inflammatory cells such as monocytes and CD11b cells and their cytokine products have been shown to enhance vascularization in ischemic tissue27,28 and promote infarct healing16,29, myocyte protection30,31, and, possibly, regeneration16,30,32. In a mouse sciatic nerve microcrush injury model, CD11b cells were required for axon regeneration 33 . We previously reported that improvement in cardiac function after bone marrow cell therapy was associated with an increase in myocardial inflammation, including the rapid and robust influx of macrophages into the infarcted region 34 . These unique tissue repair and regenerative properties of CD11b cells could be applied to myocardial tissue repair and improve cardiac function and perfusion. In the present study, we demonstrate that CD11b cells are significantly vasculogenic. Of particular note, CD11b-positive cells induced more functional arteriolar structures (larger vessels with smooth muscle contributions to the media) than CD11b-negative cells. These findings suggest that uterine CD11b-positive cells have a significant angiogenic potential that might be exploited to induce angiogenesis in the ischemic heart. Transplantation of these cells into ischemic rat hearts was indeed associated with enhanced vascularization and myocardial perfusion. This was accompanied by reduced scar area, which, in turn, contributed to better infarct healing and improvement of cardiac function compared with the transplantation of CD11b-negative uterine cells.
The overlapping and diverse functions of CD11b cells in tissue repair and regeneration are complex because they produce a wide range of biologically active molecules that contribute to both the beneficial and detrimental outcomes of inflammation. While the role of CD11b cells in tissue repair and regeneration is less well characterized than their role in innate immunity, it is becoming clear that these cells do play an important regulatory role in the tissue repair process 35 . Thus, control of CD11b cell activity is essential for effective healing and repair after MI.
CD11b cell isolation from solid tissues, such as the uterus, is most commonly done using antibody-targeted approaches such as magnetic isolation or fluorescence-activated sorting36,37. However, the unique cellular properties of CD11b cells allow for pure isolation without using antibodies from some tissues. Primary cultures of brain, liver, and testicular cells produce rounded CD11b cells that can be selectively purified from culture by rinsing the plate or tapping36–40. Here we demonstrate that uterine CD11b cells also have the ability to round in heterogeneous primary cultures, thus allowing for their purification without the use of antibodies.
The rounding phenomenon of CD11b cells described here was discovered serendipitously when a dish of uterine cells was deprived of fresh media for 3 weeks. When this culture was examined under a microscope, we observed rounded, phase-bright CD11b cells. We then undertook further experimentation to understand the mechanisms of this phenomenon. We discovered that a feeder layer-like relationship with sufficient area coverage of the dish surface is necessary to produce loosely adherent and rounded CD11b cells. By seeding a high density of uterine cells in media supplemented with recombinant M-CSF, a confluent stromal/fibroblastic environment with optimal CD11b cell rounding was achieved without waiting for cell growth to cover the dish surface or paracrine conditioning.
In conclusion, we describe a new method for the isolation of uterine CD11b cells. Implanting these cells into ischemic rat hearts reduced infarct size, increased vasculogenesis, and improved myocardial function and perfusion. Importantly, CD11b cells may have immune-privileged properties, decreasing the likelihood of rejection following transplantation 41 . Thus, uterine CD11b cells may represent an important reservoir of angiogenic cells with significant therapeutic potential for the treatment of ischemic heart disease.
Footnotes
Acknowledgment
We thank Dr. Leigh Botly for help with manuscript editing and preparation. This work was supported by a grant from the Heart and Stroke Foundation of Ontario (No. G15009023) as well as a grant from the Canadian Institutes of Health Research (No. MOP86661). R.-K.L. holds a Tier 1 Canada Research Chair in Cardiac Regeneration. The authors declare no conflicts of interest.
