Abstract
Stem cell transplantation has enormous potential for the treatment of neurodegenerative disorders like Parkinson's disease (PD). Mesenchymal stem cells (MSCs) have attracted much attention because they can secrete a wide variety of cellular factors that promote cell growth. In this study, we prepared a conditioned medium (CM) using lyophilized MSC culture medium that contained the secretome of MSCs and applied this CM to the culture of neural stem cells (CM-NSCs) for the transplantation of PD model rats. Quantitative realtime PCR, Western blot, and immunocytochemistry were used to identify cell differentiation and expression of dopaminergic neuron-specific genes in vitro. Behavioral tests including rotational behavior and MWM training tests were also performed to assess the recovery. Our results indicated that combined treatment of CM and neural stem cell transplantation can significantly reduce apomorphine-induced rotational asymmetry and improve spatial learning ability. The CM-NSCs were able to differentiate into dopaminergic neurons in the ventral tegmental area (VTA) and medial forebrain bundle (MFB), and migrated around the lesion site. They showed a higher activity than untreated NSCs in cell survival, migration, and behavior improvement in the dopa-deficit rat model. These findings suggest that the neural stem cells treated with conditioned medium possess a great potential as a graft candidate for the treatment of Parkinson's disease.
Keywords
Introduction
Parkinson's disease (PD) is the second most common chronic progressive neurodegenerative disorder and is characterized primarily by major loss of nigrostriatal dopaminergic neurons (20). It is estimated that 200 out of 100,000 people worldwide have PD (29). In typical cases, the onset of symptoms is asymmetrical, with tremor and rigidity affecting limbs on one side of the body first. Although the limbs on the other side of the body will eventually be affected, it can often be several years before the symptoms occur. Although pharmacological treatment with L-DOPA as well as surgical treatments can alleviate parkinsonian symptoms, these methods still have their own limitations in repairing the dopaminergic pathway and preventing its degeneration.
The brain has limited ability to generate new functional neurons in response to injury, although endogenous neural progenitor cells are present in multiple locations in the adult brain (25). Therefore, the use of neural stem cells (NSCs) for therapeutic transplantation is a promising strategy for the treatment of CNS injuries and neurodegenerative disorders because of their potential to replace lost or dysfunctional cells (2,17). To understand the basic mechanisms of cell transplantation therapy clearly, some researchers speculate that the microenvironment of the damaged part of the brain will influence the phenotype of the cells or that the transplanted cells will influence the microenvironment of the diseased or injured brain (7,12). Others postulate that NSCs may be important cellular factories for providing a neuroprotective microenvironment, but may not necessarily replace lost neurons (10,14,15,38,40).
Mesenchymal stem cells (MSCs) are another kind of cell for transplantation because of their easy accessibility, multipotency, and immunomodulatory properties (9,39). They can rapidly proliferate and differentiate intocells of the neuroectodermal lineage (6). However, recent reports have revealed that MSCs promote tissue repair by secretion of factors that enhance regeneration of injured cells, stimulate proliferation and differentiation of endogenous stem-like progenitor cells found in most tissues, and decrease inflammatory and immune reactions (37). Therefore, the changes in the tissue microenvironment may be more important than transdifferentiation in effecting tissue repair (28,32,36,41).
In this study, we grafted rat NSCs cocultured with conditioned medium (CM) produced by MSCs into the ventral tegmental area (VTA) and medial forebrain bundle (MFB) in PD model rats generated by injection of 6-hydroxydopamine (6-OHDA) (19). As this is a combined therapy involving transplantation of pretreated NSCs into animals, it may imply a possible relationship between cell transplantation and the environment it creates for recovery from neural injury or disease.
Materials and Methods
All experiments were performed in accordance with the guidelines and regulations of Shanghai University Ethics Committee, who also approved the study protocol.
Materials
Hank's balanced salt solution (HBSS) and phosphate-buffered saline (PBS) were prepared according to Sambrook and Russell (35). NH4Cl, EDTA, methanol, hydrogen peroxide, HCl, and sodium borate were purchased from Sinopharm Chemical Reagent Co. Ltd. (Shanghai, China). L-Glutamine, Tween-20, ascorbate, and 4′,6-diamidino-2-phenylindole (DAPI) were purchased from Sigma-Aldrich (Shanghai, China). Plastic flasks were purchased from Corning (Corning, NY, USA). Glass microsyringes are products from Hewlett-Packard (Palo Alto, CA, USA).
Animals
Male Sprague–Dawley (SD) rats weighing 60–80 g and 180–200 g (Fudan Laboratory Center, Shanghai, China) were used for MSC isolation and generation of the PD model rats, respectively. The animals were cared for according to the guidelines set forth by the Committee on Laboratory Resources, National Institutes of Health (NIH; Bethesda, MD, USA). All animals used as PD models had free access to food and water both before and after the operation. The neonatal rats used for NSC isolation were also SD rats from Fudan Laboratory Center.
MSC Culture
Rat MSCs were isolated according to a method previously described with some modifications (13). Briefly, tibias and femurs were dissected from adult Sprague–Dawley rats (60–80 g). The ends of the bones were cut, and the marrow was squeezed with 5 ml D-Hank's solution using a needle and syringe. The marrow was resuspended in 0.84% NH4Cl solution. After 5 min, about 2 × 106 marrow cells were seeded into 25-cm2 plastic flasks containing Dulbecco's modified Eagle medium (DMEM; Hyclone, Logan, UT, USA) supplemented with 10% fetal bovine serum (FBS; Gibco, Grand Island, NY, USA), 2 mM L-glutamine, 100 U/ml penicillin, and 100 mg/ml streptomycin (Gibco). Cells were incubated at 37°C with 5% humidified CO2 for 24 h, and the nonadherent cells were removed by replacing the medium. The medium was replaced every 3 or 4 days for about 2 weeks. When cells grew to confluency, they were harvested with 0.25% trypsin (Hyclone) and 1 mM ethylenediaminetetraacetic acid (EDTA) for 5 min at 37°C and reseeded into 25-cm2 plastic flasks at the split ratio of 1:2. All of the experiments described below were performed using cells from the third to the fifth passage.
Preparation of MSC-Derived Conditioned Medium
MSCs were grown to 80–90% confluency in 10-cm dishes and cultured in complete culture medium for 2 days. The attached cells were washed with PBS three times, and the medium was replaced with basal culture medium DMEM plus 1% penicillin/streptomycin (Gibco). After 48 h, the medium containing MSC-secreted factors was collected and centrifuged at 2,000 × g for 10 min followed by filtering the supernatant through a 0.22-μm filtration unit (Millipore, Bedford, MA, USA). The collected medium was concentrated by freeze-drying to preserve the activity of secreted factors. The concentrated medium was stored at −80°C for future analysis (16).
NSC Culture
Rat NSCs were obtained from hippocampi of neonatal rats and cultured in DMEM/F12 basic medium (Gibco), supplemented with 1% penicillin/streptomycin (Gibco), 20 ng/ml epidermal growth factor (EGF; Invitrogen, Carlsbad, CA, USA), 10 ng/ml basic fibroblast growth factor (bFGF; Invitrogen), and 1% B27 (Invitrogen) for 5–7 days. The medium was replaced at half volume with fresh medium every 3 days.
Quantitative Real-Time RT-PCR
Total RNA of NSCs treated with conditioned medium (CM-NSCs), NSCs (1 × 106 cells), and brain tissue from the transplantation sites was extracted with TRIzol (Invitrogen) and first-strand complementary DNA was obtained using a ReverseTra Ace kit (ToYoBo, Co. Ltd., Osaka, Japan). qPCR was performed in a 20-μl reaction mixture solution containing 10 μl of SYBR® Premix Ex Taq™ (TaKaRa Bio Inc., Dalian, China), 1 μl cDNA, 2 μl primers, and 7 μl ddH2O. PCR was initiated at 94°C for 5 min and followed by 40 cycles of amplification (30 s at 94°C, 25 s at 57°C, 20 s at 72°C). Real-time quantitative PCR was performed using Rotor-Gene 3000 (Corbett Research, Cambridge, UK) detection system and SYBR® Premix Ex Taq™ (TaKaRa Bio Inc.). Each sample was tested in triplicate. Relative gene expression was analyzed using the 2–ΔΔCt method and β-actin served as an internal reference. Primer sequences and the sizes of the products are as follows: TH (248 bp): 5′-CCCAGGACATTGGACTTG-3′ and 5′-CACAGGCTGGTAGGTTTG-3′; β-actin (203 bp): 5′-ACGTTGACATCCGTAAAGAC-3′ and 5′-CTGGAA GGTGGACAGTGAG-3′; and GFAP (387 bp): 5′-GTGG GCAGGTGGGAGCTTGATTCT-3′ and 5′-GTGGGCAG GTGGGAGCTTGATTCT-3′.
Western Blot Analysis
Protein was isolated from the cells and brain tissues from the transplantation sites with TRIzol (Invitrogen) according to the manufacturer's manual. Protein concentrations were tested by a bicinchoninic acid assay (BCA) protein assay (Pierce, New York, NY, USA). Samples (30 μg protein/lane) were concentrated and separated by SDS-PAGE on 5% and 15% acrylamide gels (Sangon, Shanghai, China) and then transferred to Immun-Blot polyvinylidene difluoride membranes (PVDF; Bio-Rad, Hercules, CA, USA). Membranes were blocked in 5% nonfat dried milk (Sangon) in PBS with 0.1% Tween-20 (PBST) for 1 h at room temperature, followed by incubation with primary antibodies at 4°C overnight. The primary antibodies included polyclonal rabbit β-actin antibody (1:1,000; Abcam, Cambridge, UK), mouse tyrosine hydroxylase (TH) antibody (1:1,000; Sigma-Aldrich), and rabbit glial fibrillary acidic protein (GFAP) antibody (1:800; Sigma-Aldrich). After being washed three times with PBST, the membranes were incubated with goat anti-rabbit IRDye® 800CW antibody or goat anti-mouse IRDye® 700CW antibody (Li-Cor Biosciences, Lincoln, NE, USA) diluted 1:10,000 in blocking buffer (1 h) and washed three times. Protein bands were scanned and analyzed through Odyssey Infrared Imaging System (Li-Cor Biosciences).
Rat Model of Parkinson's Disease
PD rats were produced by the unilateral injection of 6-OHDA (Sigma-Aldrich) into the right MFB (AP: 4.4 mm; L: 1.2 mm; DV: 7.8 mm) and VTA (AP: 4.8 mm; L: 1.0 mm; DV: 7.8 mm). The sham-operated rats received vehicle only (0.1% ascorbate in 0.9% saline) at the same coordinates. 6-OHDA administration was performed through a glass microsyringe (5 μl) with a dose of 10 μg/5 μl at each site at a rate of 0.5 μl/min. The needle was kept in place for 5 min before slowly being retracted to allow for toxin infusion and to prevent reflux. Four weeks after injection of 6-OHDA, the rotarod behavior of the rats was tested. Only those rotating at rates of over six turns per min in response to 0.5 mg/kg of apomorphine (Sigma-Aldrich, St. Louis, MO, USA) were selected for transplantation of NSCs and CM-NSCs (19,20).
Cell Transplantation
On day 28 postlesioning, surgery was performed after chloral hydrate (Sigma-Aldrich) anesthesia. A suspension of cells (5 μl per site, 1 × 103 cells/μl) was injected at a rate of 0.5 μl/min at two transplantation sites (right MFB and VTA) with a 5-μl glass microsyringe. The needle was kept in situ for 5 min to allow the cells to diffuse. For the vehicle group, the suspension was 0.9% normal saline without cells. For the NSC group, the suspension included normally cultured NSCs at a concentration of 1 × 103 cells/μl. For the CM-NSC group, the suspension included NSCs cultured with conditioned medium derived from MSCs with the same concentration of 1 × 103 cells/μl.
BrdU Administration
After implantation of CM-NSCs for 24 h, rats (n = 8) received injections of 5-bromo-2′-deoxyuridine (BrdU; Sigma-Aldrich) solution in PBS at a dose of 50 mg/kg with 0.007 M NaOH twice daily, consecutively, for 6 days before they were sacrificed on the seventh day.
Immunocytochemistry
Cells used for in vitro studies were fixed with 4% paraformaldehyde (Sigma-Aldrich), permeabilized with 0.1% Triton X-100 (Sigma-Aldrich) in PBS, and blocked with 5% normal goat serum (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA). Then cells were subjected to immunofluorescent staining using mouse anti-TH (1:1,000, Sigma-Aldrich) monoclonal antibodies. Tetramethylrhodamine isothiocyanate (TRITC)-conjugated goat anti-mouse secondary antibody (Santa Cruz Biotechnology Inc.) was used at 1:200. Cell nuclei were stained with DAPI to count the cell number. Images were taken under a fluorescent microscope (ECLIPSE TE2000-U; Nikon Co., Tokyo, Japan) and confocal microscope (FV1000; Olympus Co., Tokyo, Japan).
Brain Immunohistochemistry and BrdU Labeling
Six and 8 weeks after transplantation, brain samples were promptly fixed in 4% paraformaldehyde (Sigma-Aldrich), dehydrated in gradient sucrose solution, and then embedded in optimum cutting temperature (OCT) compound (Sakura Finetek, Torrance, CA, USA). Frozen sections (20 μm) were obtained using a cryostat microtome (Thermo Scientific, Rochester, NY, USA). After being incubated in methanol and 3% hydrogen peroxide solution and blocked with normal goat serum, sections were incubated subsequently with mouse anti-TH (1:400; Sigma-Aldrich) monoclonal antibody. For BrdU detection, pretreatment of sections was followed to denature the DNA with 2 N HCl for 45 min at 37°C and then with 0.1 M sodium borate buffer (pH 8.5) for 10 min. Afterward, sections were incubated with a rabbit monoclonal antibody against BrdU (1:300; Biogenex, Inc. San Francisco, CA, USA). Secondary antibodies used to visualize the antigen were either goat anti-mouse FITC-conjugated (Santa Cruz Biotechnology Inc.) or goat anti-rabbit Cy5-conjugated (Beyotime, Shanghai, China), both with a dilution of 1:200. Images were taken under a fluorescent microscope (ECLIPSE TE2000-U; Nikon Co.) or confocal microscope (FV1000; Olympus Co.). Image Pro Plus 6.0 (IPP; Media Cybernetics, Inc., Bethesda MD, USA) was used to analyze and quantify the expression of target genes. Positively stained regions of the full image were obtained and then integrated optical density (IOD) was measured.
DiI Staining
DiI (Sigma-Aldrich) was dissolved in ethanol to reach a final concentration of 1 mM. The NSCs were incubated with DiI solution (5 μl/ml) for 40 min at 37°C with 5% CO2 before they were washed with PBS three times. Images were taken under a fluorescent microscope or confocal microscope as mentioned above.
Behavioral Testing: Rotarod Test and Morris Water Maze (MWM)
Rats were tested for rotational behavior induced by intraperitoneal injection of apomorphine (0.5 mg/kg; Sigma-Aldrich) in a session lasting over 30 min at 2, 4, 6, and 8 weeks after transplantation. Rats with a rotational speed of 6/min or even higher were considered as the successful PD models. Eight weeks after transplantation, rats were trained for the MWM test. Each day, a trial was started by placing each rat in the water facing the pool wall in one of the four quadrants (designated NE, NW, SE, SW), which were defined in the computer software (EthoVision Noldus, Wageningen, The Netherlands) such that each quadrant was equal in dimension. The daily order of entry into each quadrant was random, and all four quadrants were used every day in three trials. For each trial, the rat was allowed to swim for 90 s to find the hidden platform. If the rat found the platform successfully, it would be allowed to have a 30-s rest on the platform. If unsuccessful within time permitted, the rat was given a score of 90 s and then physically placed on the platform and also allowed to have the 30-s rest period. In either case the rat was immediately arranged for the next trial after the rest period.
Statistical Analysis
All data were presented as mean ± SD. Data were analyzed by one-way ANOVA with Scheffe's post hoc test. Datasets consisting of two groups only were compared through unpaired Student's t-tests. A value of p < 0.05 was considered statistically significant. SigmaPlot (Systat Software, San Jose, CA, USA) was used for the statistical analyses.
Results
CM-NSCs Differentiate Into Dopaminergic Neurons In Vitro
To produce the conditioned culture medium, rat MSCs were isolated as described previously (11). When the isolated MSCs had been passaged three to five times, the culture medium was collected and lyophilized. To determine the optimal concentration of lyophilized MSC culture medium in the CM that may affect the differentiation of NSCs, we performed a concentration gradient assay with the freeze-dried medium from 50 ng/ml to 500 ng/ml, and found that the optimal concentration should be 250 ng/ml, which was thereafter used in the following experiments. To examine the effects of CM on differentiation of NSCs in vitro, immunostaining was performed to detect the expression level of neuron-specific markers. Results indicated that the number of dopaminergic neurons (TH-positive staining) obviously increased after treatment for 10 days in conditioned medium (Fig. 1B,C) compared with those treated with normal culture medium (Fig. 1E,F). The proportion of TH-positive cells was significantly larger after CM treatment (15.74 ± 1.17%) than that of the control group (10.98 ± 1.38%) (p < 0.05) (Fig. 1G). Furthermore, the expressions of TH and GFAP genes were quantitatively analyzed by real-time RT-PCR, and the expression of TH was obviously upregulated in CM-NSCs, while GFAP expression was remarkably downregulated compared with control in NSCs (Fig. 2A,B). Also, Western blot results indicated a marked increase in TH and a decrease in GFAP (Fig. 2C,D), which corresponds with the realtime PCR results. Together, these data suggest that CM containing rat MSC-secreted factors could induce NSC differentiation into dopaminergic neurons.
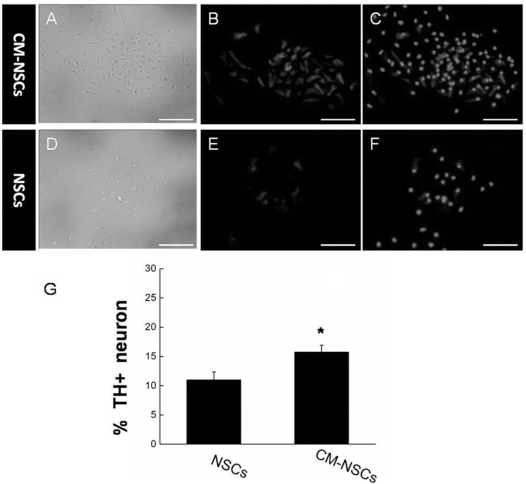
NSCs induced by CM can differentiate into dopaminergic neurons. (A–C) Expression of TH in the CM-NSC group. (D–F) Expression of TH in the NSC group. (G) Quantification of TH-positive cells. The percentages were 15.74 ± 1.17% in CM-NSCs and 10.98 ± 1.38% in NSCs, respectively. Scale bars: 60 μm. Mean ± SD, n = 5. *p < 0.05 by unpaired two-tailed Student's t-test.

TH and GFAP expression in NSCs and CM-NSCs. The expression of TH was evaluated through real-time PCR (A) and Western blot (C). Quantification at RNA level (B) and protein level (D) indicates an increase in TH and a decrease in GFAP expression. All experiments were performed in triplicate. *p < 0.05; **p < 0.01.
Increased Neurogenesis Is Observed in the CM-NSC-Transplanted Area of VTA and MFB
In view of the fact that conditioned medium is conducive to NSC differentiation into dopaminergic neurons, we assumed that it might be useful for the treatment of PD. We therefore generated a PD model rat for CM-NSC transplantation by unilateral injection of 6-OHDA as previously described (19). CM-NSCs were labeled with DiI as previously described (19) to track living cells and their proliferation and differentiation after transplantation. One week after DiI-labeled CM-NSCs and NSCs were transplanted into PD model rats in the VTA and MFB, BrdU immunostaining was used to check the difference in neurogenesis. Results indicated that there was obvious neuronal proliferation (Fig. 3I) in the CM-NSC group (Fig. 3C and G) in contrast to the NSC group (Fig. 3D and H).

Neurogenesis was increased in the CM-NSC- and NSC-transplanted VTA and MFB. (A–C) CM-NSCs in the transplanted regions labeled with DiI, TH antibody, and BrdU, respectively. (E–G) NSCs in the transplanted regions labeled with DiI, TH antibody, and BrdU, respectively. (D) Merged images of (A–C) and (H) merged images of (E–G). Arrows indicate cells labeled with all three markers. (I) Analysis of neurogenesis increases treated with CM-NSCs and NSCs. Immunostaining of coronal sections was performed in both CM-NSC and NSC groups 7 days posttransplantation. Scale bars: 50 μm.
The Migration and Differentiation of NSCs Transplanted Into the MFB and VTA
We wondered if CM-NSCs transplanted into the brain can survive longer than untreated NSCs. To this end, both DiI-labeled NSCs and CM-NSCs were transplanted into the VTA and MFB of PD model rats. After 6 and 8 weeks, transplanted CM-NSCs and NSCs were identified via labeled DiI fluorescence and immunohistochemistry (Fig. 4). Results showed a remarkably higher ratio of TH-positive neurons in the CM-NSC group (Fig. 4A, B, E, and F) after both 6 and 8 weeks of treatment than in the control group (Fig. 4C, D, G, and H). Furthermore, migration of transplanted cells in the VTA and MFB was observed. Compared with the NSC group, the CM-NSC group covered a larger area of expansion in the eighth week (Fig. 4F and H). Statistical analysis showed a significant increase in the CM-NSC group compared with the control (p < 0.01) (Fig. 4N).

Differentiation and migration of NSCs transplanted into the VTA and MFB in PD model rats. (A–D) DiI labeling of CM-NSCs (A, B) and NSCs (C, D) 6 and 8 weeks after the operation. (E–H) Immnuostaining with TH antibody of the corresponding samples above. (I–L) Merge of DiI and TH antibody labeling. (M) Percentage of TH-positive cells of NSC and CM-NSC groups in the sixth and eighth week, respectively. (N) Cell diffusion area of the NSC and CM-NSC groups in the sixth week and the eighth week. Scale bars: 50 μm. Mean ± SD, n = 5. **p < 0.01 by unpaired two-tailed Student's t-test.
CM-NSCs Are Beneficial to the Functional Recovery of PD Model Rats
To assess the functional recovery of PD model rats with transplanted CM-NSCs and NSCs, the rotational behavior of rats induced by intraperitoneal (IP) injection of apomorphine was tested as previously described (19) (Fig. 5). Results show that apomorphine-induced rotation numbers decreased significantly in both NSC- and CM-NSC-transplanted animals compared with the vehicle group (p < 0.01, n = 6). Additionally, the number of rotations of the CM-NSC group was significantly less than that of the NSC group 2, 4, 6, and 8 weeks after treatment (p < 0.05, n = 6). This result indicates that CM-NSCs play a more effective role in the functional recovery from PD in PD model rats. Next, the learning and memory of PD rats were also tested via the MWM test 8 weeks after the operation. During the MWM training period, rats with both transplanted NSCs and CM-NSCs gradually learned to spend less time reaching the platform than the vehicle group (Fig. 6A). Furthermore, differences in latency between the NSC group and the CM-NSC group began to appear on the fourth day (p < 0.05), indicating that the learning and memory ability of PD rats had been improved after CM-NSC transplantation (Fig. 6A). However, analysis of swimming paths showed no significant difference between the experimental group and control group on the first day (Fig. 6B). After training, differences among the CM-NSC group, the NSC group, and the vehicle group began to appear on the fourth day (Fig. 6B). These data suggest that CM-NSCs are beneficial in the recovery of motor function in a rat model of PD.

Rotarod test of PD rats after transplantation of CM-NSCs and NSCs. Apomorphine-induced rotation test shows a significant difference both between vehicle and NSC groups (**p < 0.01) and between NSC and CM-NSC groups (*p < 0.05) during the entire time course of the experiment (2 weeks, 4 weeks, 6 weeks, 8 weeks). Mean ± SD, n = 6. The p values by unpaired two-tailed Student's t-test.

Morris water maze test of vehicle, NSC-, and CM-NSC-treated PD rats. (A) Effects of CM-NSC transplantation on MWM training. Difference in latency to find platform between groups does not appear until day 4. Significance was observed both between vehicle and NSC groups (**p < 0.01) and between NSC and CM-NSC groups (*p < 0.05). (B) Representative swimming distances illustrate the impairment and recovery of learning and memory. Differences between the vehicle, NSC, and CM-NSC groups do not appear until day 4, corresponding to the results in (A). Mean ± SD, n = 5. The p values by unpaired two-tailed Student's t-test.
CM-NSCs Differentiated Into Dopaminergic Neurons in the Lesion Area
To explore the fate of transplanted NSCs, we investigated the expression of the dopaminergic neuron marker, TH, in the CM-NSC-transplanted regions at the RNA and protein levels, respectively. Results show that the TH expression in the CM-NSC-treated group was significantly upregulated in contrast to the control group (Fig. 7A) using quantitative real-time PCR analysis. Similarly, the Western blot assay also displayed marked upregulation in protein level expression for the CM-NSC-treated group (Fig. 7B).

CM-NSCs are beneficial to dopaminergic neuron differentiation in the transplanted area. (A) The expression of TH mRNA was tested by RT-PCR, and (C) the quantitative analysis was performed using real-time PCR assay from transplanted tissue in PD rats at 6 weeks after the operation. The expression of TH protein was identified using Western blot analysis (B), and the quantitative assay (D) at 6 weeks after the transplantation. Upregulation of TH expression at both RNA and protein levels was observed in the CM-NSC group compared with the NSC group and vehicle group.
Discussion
In the past decades, it has been shown that adult stem cells possess greater plasticity in the field of stem cell transplantation than their own natural evolving paradigms in embryonic development (42). However, adult stem cells have their own pros and cons over embryonic stem cells in potential applications and development of cellular therapeutics. A distinct advantage of adult stem cells is that the patient's own cells can be proliferated and then reintroduced back into his/her own body. Using the patient's own adult stem cells will cause no rejection by the immune system. The main disadvantage of such therapy, however, is that adult stem cells are highly committed and are few in number in mature tissues where they have generally differentiated into different types of cells (1). Recently, people have been paying increasing attention to MSCs from bone marrow and other related tissues (4,5,34). Due to their easy isolation, proliferation potential, and favorable immunological properties, MSCs have proven highly promising in repairing tissue damage in response to injuries and diseases. The ability of MSCs to repair skeletal defects was originally evaluated in animal models. Subsequently, MSCs were demonstrated to be efficient as therapeutic cells in animal models of various neurological disorders and other diseases (3). Until now, two major mechanisms have been hypothesized to be responsible for tissue repair via MSC transplantation: transdifferentation and paracrine effects. Recently, more evidence has emerged in support of the latter view, while the transdifferentation potential of MSCs was proven to be invalid (27). For example, a number of reports demonstrated a low or transient level of MSC engraftment in vivo. Although the mechanisms of neural damage repair by MSC transplantation are still unclear (26), we could speculate in this study that the effects of MSC transplantation are mediated by the supply of cell protective, angiogenic, and mitogenic factors in a paracrine manner just like the maintenance of hematopoietic stem cells (31).
In the present study, we combined the idea of differentiation with paracrine mechanisms for the treatment of a PD rat by transplantation of NSCs treated with MSC-secreted factors. Our results showed that we successfully constructed MSC-derived conditioned medium in which NSCs could effectively be induced to differentiate into dopaminergic neurons, providing an important reference for regulation and directed differentiation of NSCs, and thus possibly increasing the success rate of transplantation. Croft and Przyborski (11) reported that MSCs can provide instructive signals to direct the differentiation of NSCs and promote axonal development when MSCs and NSCs are both in an in vitro coculture system. This is in agreement with our observation that the culture of NSCs in MSC-derived conditioned medium leads to a significant increase in neurogenesis (Fig. 1G). In addition, we also found an obvious neuronal proliferation in the CM-NSC group compared with the control group using BrdU immunostaining (Fig. 3E,F), suggesting a positive contribution to the improvement of the microenvironment for transplanted cells. Such effects may well explain recent reports that MSCs and related cells from bone marrow could improve recovery from injury in nervous system. (8,18,22,23,30,33); MSC-derived CM, which contained multiple secreted factors, has a protective effect in promoting cell survival and migration (21). Our results showed that after treatment with CM, the number of viable cells was significantly elevated (Fig. 4M), and the area of cell migration also significantly increased 8 weeks posttransplantation (Fig. 4N). The key to successful transplantation is cell survival in migration. In this study, we detected the status of transplanted cells by DiI labeling and a neuron-specific immunohistochemical marker and confirmed cell migration and incorporation, which was consistent with our previous observations (19). MSC-derived CM also has a positive effect on the functional recovery in PD rats. We tested the behavior of CM-NSC-transplanted mice in motor, learning, and memory tasks, and the results showed that CM-NSCs are propitious to functional recovery of PD rats (Figs. 5 and 6). Subsequently, we confirmed the presence of dopaminergic neurons at the molecular level (Fig. 7). Strikingly, we found in our study that CM-NSCs play an important role in promoting cellular differentiation and integration into the damaged area. Similar research can be found in the treatment of Alzheimer's disease (AD) mice. Lee et al. reported that MSC transplantation into AD mice significantly improved spatial learning and memory, suggesting that MSCs may produce a neuroprotective effect (24). It is for this reason that we can explain why the functional recovery mediated by CM-NSCs is better than that of NSCs alone.
In summary, we have constructed an MSC-derived CM that is conducive to the generation of dopaminergic neurons in vitro and in vivo. Cell transplantation experiments showed that MSC-derived CM has a protective effect in promoting cell survival, migration, and integration into damaged areas in the brains of PD rats. Behavioral analysis further indicated that CM-NSCs play a critical role in functional recovery from PD. These results may provide an important basis for further exploring the mechanisms of neural damage repair mediated by CM-NSCs and present CM-NSCs as a promising graft candidate for future clinical implementation.
Footnotes
Acknowledgments
This work was funded by the National Science Foundation of China (81471162, 81070737, 81271253), the Science and Technology Commission of Shanghai (14JC1 402400, 09PJ1407200), and the Key Innovation Project of Shanghai Municipal Education Commission (Grant No. 14ZZ090). The authors declare no conflicts of interest.
