Abstract
The aim of this study was to investigate a cell delivery system for repair of severe chronic osteochondral defects using magnetically labeled mesenchymal stem cells (m-MSCs), with the aid of an external magnetic device, through the accumulation of a small number of m-MSCs into a desired area and to detect the suitable number of autologous m-MSCs needed for repair of the defect. Twenty-six male Japanese white rabbits aged 6 months were used. An osteochondral defect was created bilaterally at the weight-bearing surface of the medial femoral condyle of the rabbits' knees (3 mm diameter; 4 mm depth). At 4 weeks after creation of the defect, autogenic transplantation of the m-MSCs into the defect area was performed, followed by 10-min exposure to an external magnetic device, where animals were divided into four groups: high (1 × 106 m-MSCs), medium (2 × 105 m-MSCs), low (4 × 104 m-MSCs), and control (PBS injection). At 4 and 12 weeks posttransplantation of m-MSCs, repaired tissue was assessed histologically using the Fortier score with toluidine blue staining. Transplantation of a low number of m-MSCs was not enough to improve osteogenesis and chondrogenesis, but the medium and high groups improved repair of the chronic defect with chondrogenic tissues and showed histologically significantly better results than the control and low groups. The use of a magnetic targeting system for delivering m-MSCs has the potential to overcome the clinical hurdles for repair of the severe chronic osteochondral defect. Furthermore, this system is predicted to produce good clinical outcomes for humans, not only to repair osteochondral defects but also to repair a variety of damaged tissues.
Keywords
Introduction
An osteochondral defect comprises damage to the articular cartilage as well as the underlying subchondral bone. Most osteochondral lesions are located in patients' medial femoral condyles (58%), patella (11%), and trochlea (6%) (12). In order to repair an osteochondral defect, the needs of the bone, cartilage, and the bone–cartilage interface must be taken into account (30).
Recently, most studies on cartilage regeneration have depended on cell-based therapy, with either natural or synthetic scaffolds or growth factors (31,32), as well as the use of cell-based gene therapy for tissue engineering and regeneration (10). Among the different cell populations used for cartilage regeneration, mesenchymal stem cells (MSCs) have attracted a great deal of interest because of their potential use in regenerative medicine and tissue engineering (5) and their multilineage differentiation potential, especially their chondrogenic differentiation properties (15). Autologous chondrocyte implantation is not suitable for treating defects including subchondral bone (10). MSCs can be obtained easily from bone marrow aspirate and other mesenchymal tissues (4). Mobilization of injected MSCs into the injured tissues has been reported (1). Intra-articular injection of MSCs is the most convenient method, depending upon its secretions of bioactive factors, that allows for integration into the tissues (29), particularly since it is less invasive. However, the efficacy of this procedure is potentially limited.
A novel cell delivery system for musculoskeletal regeneration was developed in our department. Kobayashi et al. succeeded in delivering magnetically labeled mesenchymal stem cells (m-MSCs) into the desired site with the aid of an external magnetic force (20). Furthermore, Kobayashi et al. found that the cell layer containing an extracellular matrix was formed on degenerated human cartilage in vitro using magnetically labeled MSCs with the aid of an external magnetic device (21). Also, Kamei et al. found that chondrogenic differentiation of m-MSCs was not affected negatively by the use of magnetic labeling with a ferucarbotran–protamine complex (16). No report has been published previously presenting a cell magnetic targeting system for repair of a severe chronic defect at the weight-bearing portion of the medial femoral condyle, despite the fact that it is the most common location for the occurrence of osteochondral lesions. Consequently, the objective of this study was to ascertain the appropriate number of m-MSCs needed to repair a chronic osteochondral defect in the weight-bearing portion of the medial femoral condyle with the aid of an external magnetic device in a rabbit model, through accumulation of cells into the desired site.
Materials and Methods
This study followed the National Institute of Health Guide for animal experimentation and was approved by the ethical committee of research facilities for laboratory animal science (Graduate school of Biomedical and Health Sciences, Hiroshima University, Japan).
Isolation of MSCs
Isolation and in vitro expansion of bone marrow-derived mesenchymal stem cells (BM-MSCs) has been previously described (16). In brief, 5 ml of a blood sample of bone marrow iliac crest of 21 male Japanese white rabbits, aged 6 months, was aspirated with 1 ml of heparin (1,000 units/1 ml; Mochida Pharmaceutical Co. Ltd., Tokyo, Japan) using an 18-gauge needle. The sample was centrifuged for 5 min at 420 × g, and the resultant supernatant, including heparin, was discarded. The extract was resuspended in 2 ml of culture medium composed of Dulbecco's modified Eagle medium (DMEM; Gibco®, Life Technologies™, Grand Island, NY, USA) with 10% fetal bovine serum (FBS; Sigma-Aldrich, St. Louis, MO, USA) and 1% antibiotics (penicillin, streptomycin, and amphotericin B; Nacalai Tesque, Inc., Kyoto, Japan). Then 2 ml of the blood sample was suspended in 8 ml of culture medium on 10-cm2 tissue culture dishes (BD Falcon, Franklin Lakes, NJ, USA). The dishes were incubated for 4 weeks under a humidified atmosphere and 5% CO2 at 37°C. The medium was changed after 7 days to remove detached cells and was thereafter changed every 3 days. The cells proliferated in isolated colonies through approximately 2 weeks. The cells were detached by treatment with 0.25% trypsin (Gibco®, Life Technologies™), and seeded onto new culture dishes. After a confluent cell layer was formed again, the cells were reseeded onto 10-cm2 culture dishes under the same conditions. Cells at passage 3 were used in the current study.
Magnetic Labeling of MSCs
Ferucarbotran (Resovist® Inj.; Fujifilm RI Pharma Co. Ltd., Kanagawa prefecture, Japan) is an intracellular magnetic labeling contrast agent composed of superparamagnetic iron oxide nanoparticles (SPIONs) [0.5 mmol (28 mg) Fe/ml]. Ferucarbotran was added to serum-free Roswell Park Memorial Institute (RPMI)-1640 medium (BioSource, Camarillo, CA, USA), consisting of 25 mmol/L of 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), L-glutamine, minimum essential medium (MEM) nonessential amino acids, and sodium pyruvate. Then an equal volume of ferucarbotran was added to the culture medium. MSCs were labeled overnight with ferucarbotran only without transfection with any of the incorporation facilitators such as protamine sulfate or poly-L-lysine (PLL). After that, cells were washed twice with phosphate-buffered saline (PBS; Wako Pure Chemical Industries Ltd., Osaka, Japan). After magnetic labeling, MSCs were referred to as m-MSCs (Fig. 1).

Morphology of mesenchymal stem cells after labeling with ferucarbotran overnight (original magnification: 40×). Scale bar: 500 μm.
Cell Proliferation and Viability
Cell proliferation was assessed by using a cell counting method, whereby 5 × 104 MSCs and m-MSCs were seeded separately into six-well plates at day 0, medium was changed every 2 days, collected at day 7, and then cells were counted by using a hemocytometer to ascertain whether magnetic labeling affected cell proliferation or not. Cell proliferation of MSCs versus m-MSCs was compared by a Student's t-test. Data were represented as mean ± standard deviation. Also, viability of m-MSCs was assessed using Guava ViaCount reagent by guava easy-Cyte™ Single Sample Flow Cytometer (EMD Millipore Co., Hayward, CA, USA).
External Magnetic Device
A magnetic drug delivery system (MDDS) has been recently developed to navigate magnetically seeded drugs around diseased parts of the human body. This type of medical technique enables magnetically seeded drugs to be correctly delivered to a target in a very short time. This dramatically reduces the total dosage, and the side effects are alleviated as well. Since the magnetically seeded drugs that are moving deep within a blood vessel need to be controlled from outside of the human body in MDDS, a magnet that generates a very powerful magnetic field in a limited area is necessary. To improve the magnetic drug delivery performance, a portable high-temperature superconducting (HTS) bulk magnet system with high magnetic fields has been developed. An external magnet system (Hitachi Ltd., Ibaraki, Japan) mainly consists of small bulk high-temperature superconductors and a compact cryocooler. In our study, the bulk magnet was activated successfully using field-cooling magnetization under a superconducting solenoid magnet (33) (Fig. 2A).

(A) External magnetic device comprised of a compact cryocooler and small bulk high-temperature superconductors (3 cm in diameter) to generate a very powerful magnetic field in a limited area. (B) Transplantation of m-MSCs into osteochondral defect of the medial femoral condyle with exposure to magnetic device.
Creation of Severe Osteochondral Defect in Animal Model
Twenty-six male Japanese white rabbits (Kitayama Labes Co. Ltd., Nagano prefecture, Japan) aged 6 months and ranging from 2.5 to 2.99 kg were used in this study and were maintained freely in stainless steel cages. Anesthesia was induced by intravenous injection of pentobarbital (30 mg/kg body weight; Somnopentyl, Kyoritsu Seiyaku Ltd., Tokyo, Japan). In order to expose the knee joint, the medial parapatellar approach was used, and lateral dislocation of patella was carried out. An osteochondral defect (diameter 3 mm and depth 4 mm) was created in the medial femoral condyle at the weight-bearing surface in both knees by a stainless steel drill. At 4 weeks after creation of the defect, autogenic transplantation of m-MSCs was performed with exposure to an external magnetic device (1.5 T) for 10 min, with the rabbits divided into four groups: the high group, wherein 1 × 106 m-MSCs were transplanted into the defect site by the external magnetic device (Fig. 2B); the medium group, with 2 × 105 m-MSCs; the low group, with 4 × 104 m-MSCs; and the control group, wherein animals were only injected with PBS. m-MSCs were suspended in 25 μl of PBS. Closure of the joint capsule and skin was performed using nonabsorbable suture material. The rabbits were then returned to their cages.
Histological Evaluation
The rabbits were sacrificed at 4 and 12 weeks after transplantation. Femoral condyles were fixed in 4% paraformaldehyde phosphate-buffered solution (Wako Pure Chemical Industries Ltd.) for 48 h. The samples were decalcified with ethylenediaminetetraacetic acid (EDTA, 10%; Nacalai Tesque, Inc.) solution for 8 weeks and were embedded in paraffin blocks. The samples were cut into 5-μm sections sagittally. For histological evaluation, sections were stained with toluidine blue and Safranin O/fast green stains (Muto Pure Chemicals Co. Ltd., Tokyo, Japan). The repaired tissue was assessed histologically, using toluidine blue staining to produce histological scoring on the Fortier scale (7) (Table 1).
Histological Scoring of Repaired Tissue Using the Fortier Scale

Immunohistochemical Evaluation
To confirm the nature of the repaired tissue as to whether it consisted of hyaline cartilage or fibrocartilage, sections were assessed immunohistochemically using a collagen type II antibody. In brief, the sections were covered with 3% H2O2 (Wako Pure Chemical Industries Ltd.) at room temperature for 10 min to block nonspecific reactions. Subsequently, the sections were incubated in anti-human collagen type II antibody diluted with PBS (1:250; Daiichi Fine Chemical, Toyama, Japan) at room temperature (RT) for 1 h. After being washed with PBS three times, the samples were incubated in a working dilution of a few drops of anti-mouse secondary antibody diluted with PBS (1:200) [VECTASTAIN® Elite ABC Mouse IgG kit (pk-6102); Vector Laboratories, Burlingame, CA, USA] at RT for 30 min. Finally, the samples were incubated in 3,3′-diaminobenzidine tetrahydrochloride (DAB) dilution (Vector® DAB substrate; 0.5 mg/ml, with 0.01% H2O2 and buffer stock solution as solvent, pH 7.6; Vector Laboratories, Inc.) for 1–5 min depending upon the degree of color change (brown stain), indicating expression of collagen type II.
Statistical Analysis
Statistical analysis was performed for overall histological scoring (median) using the Kruskal–Wallis test and between groups of histological scoring using the Steel–Dwass test. Cell proliferation and viability were analyzed using the Student's t-test with a 95% confidence interval. For all statistical analyses, SPSS version 20 was used (IBM, Armonk, NY, USA). Values of p < 0.05 were considered significant in all tests.
Results
Cell Proliferation and Viability
We found that proliferation of m-MSCs was significantly higher than that of MSCs (p = 0.0065). Therefore, proliferation of MSCs was affected positively by using ferucarbotran for magnetic labeling. Data represent the increase in cell number from original plating as MSCs versus m-MSCs (1.31 ± 0.02 vs. 1.66 ± 0.1 × 104 cells) (Fig. 3). In addition, cell viability of MSCs was not affected when ferucarbotran was added, compared with unlabeled cells (data not shown).
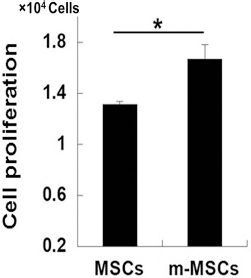
Cell proliferation was assessed between 0 and 7 days, and m-MSCs had significantly greater proliferation than MSCs. Data represented the increase in cell number from original plating as MSCs versus m-MSCs (1.31 ± 0.02 vs. 1.66 ± 0.1 × 104 cells). *p < 0.05.
Macroscopic Findings
At 4 weeks, there was partial repair of tissue (more than 50%) in the medium and high groups, whereas there was no tissue repair in the control and low groups. At 12 weeks, there was complete repair of the defect at the level of the adjacent cartilage, with smooth white tissue in the medium and high groups. However, a little fibrous tissue was formed in the defect of the control and low groups. Furthermore, asymmetry of the osteochondral defect surfaces was minimized at 12 weeks compared to those at 4 weeks. Therefore, identification of the defect demarcation was more difficult at 12 weeks than at 4 weeks (Fig. 4).

Macroscopic findings at 4 (upper row) and 12 (lower row) weeks after transplantation of m-MSCs into defect site. (A, B) Control group; (C, D) low group: 4 × 104 m-MSCs; (E, F) medium group: 2 × 105 m-MSCs; (G, H) high group: 1 × 106 m-MSCs. Control and low groups still have significant gross defect in comparison with the medium and high groups (original magnification: 24×). Scale bars: 500 μm.
Histological Assessment
At 4 weeks, partial repair of the defect with hyaline cartilage at the periphery of the defect with subchondral repair occurred in the medium and high groups, while minimal fibrous tissue was formed in the control and low groups. At 12 weeks, in the control and low groups, the defect site was just replaced by little fibrous tissue without integration with the surrounding tissue, while in the medium and high groups there was complete repair of the subchondral bone, which was covered with a layer of hyaline cartilage, well stained with toluidine blue stain and Safranin O/fast green stain, and there was a complete osteochondral junction at the same level as a normal one (Figs. 5 and 6). When assessment was performed histologically at 4 weeks using the Fortier score, the repaired tissue produced the following results: control group: 18; low group: 18; medium group: 12; high group: 15 (p = 0.0007). At 12 weeks, the results were control group: 16; low group: 17; medium group: 9; high group: 9 (p = 0.0001). At 4 and 12 weeks, the medium and high groups were histologically significantly better than the control and low groups, while there was no significant difference between the medium and high groups nor between the control and low groups (Fig. 7).

Microscopic findings at 4 (upper row) and 12 (lower row) weeks after transplantation of m-MSCs using toluidine blue stain. (A, B) Control group; (C, D) low group: 4 × 104 m-MSCs; (E, F) medium group: 2 × 105 m-MSCs; (G, H) high group: 1 × 106 m-MSCs. Black arrows indicate demarcation of the defect (original magnification: 40×). Scale bars: 500 μm.

Microscopic findings at 4 (upper row) and 12 (lower row) weeks after transplantation of m-MSCs using Safranin O/fast green stain. (A, B) Control group; (C, D) low group: 4 × 104 m-MSCs; (E, F) medium group: 2 × 105 m-MSCs; (G, H) high group: 1 × 106 m-MSCs. Black arrows indicate demarcation of the defect (original magnification: 40×). Scale bars: 500 μm.

Fortier scale for histological assessment at 4 (upper row) and 12 (lower row) weeks. Medium and high groups were significantly better than the control and low groups at 4 and 12 weeks. *p < 0.05.
Immunohistochemical Assessment
At both 4 and 12 weeks in the control and low groups, no evidence of the presence of collagen type II was reported. However, there was evidence of collagen type II at the periphery of the defect in the medium and high groups at 4 weeks, while at 12 weeks in the medium and high groups, chondrocytes embedded in collagen type II completely covered the subchondral bone (Figs. 8–10).

Immunohistochemical staining using collagen type II at 4 (upper row) and 12 (lower row) weeks after transplantation of m-MSCs. (A, B) Control group; (C, D) low group: 4 × 104 m-MSCs; (E, F) medium group: 2 × 105 m-MSCs; (G, H) high group: 1 × 106 m-MSCs (original magnification: 40×). Scale bars: 500 μm.

Higher magnification (100×) for the medium and high groups using toluidine blue, Safranin O/fast green stains and collagen type II at 4 (upper two rows) and 12 (lower two rows) weeks. At 4 weeks, (A–C) medium group and (D–F) high group. At 12 weeks, (G–I) medium group and (J–L) high group revealed complete repair of the defect and good integration with the surrounding tissues. Black arrows indicate demarcation between host and repaired tissues. Scale bars: 200 μm.
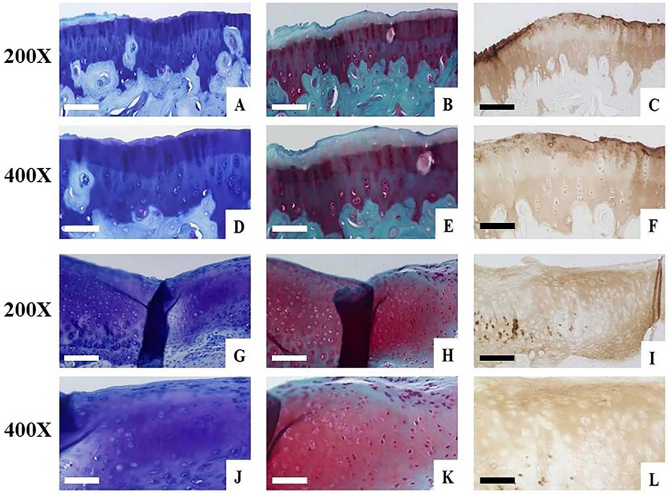
Magnification with high power (200× and 400×) for the medium (upper two rows) and high (lower two rows) groups, respectively. Histological assessment was performed using toluidine blue, Safranin O/fast green stains, and collagen type II at 12 weeks and showed distribution of chondrocytes in the repaired tissues and positivity of collagen type II. (A–C) and (G–I) show repaired tissue of the medium and high groups, respectively (original magnification: 200×). (D–F) and (J–L) show higher power images of the medium and high cell dose groups, respectively (original magnification: 400×). Scale bars: 100 μm (A–C, G–I) and 50 μm (D–F, J–L).
Discussion
Our study aimed to investigate the delivery of m-MSCs into a severe chronic osteochondral defect at the weight-bearing portion of the medial femoral condyle using a magnetic targeting system and to achieve the appropriate number of m-MSCs required for repair of the defect (3 mm in diameter; 4 mm in depth). We found that autogenic transplantation of 2 × 105 m-MSCs (medium group) was potentially suitable to attain complete repair of the subchondral bone covered by a layer of hyaline cartilage, which markedly stained with toluidine blue stain to the same level as a normal bone and cartilage with the aid of an external magnetic device. In addition to the complete osteochondral junction, complete integration of the repaired tissue with the surrounding tissue and bone–cartilage interface was achieved, more so than in the case of fibrous tissue formation in the control and low groups. While transplantation of 4 × 104 m-MSCs (low group) is not sufficient to repair the defect and stimulate osteogenesis and chondrogenesis, we cannot find any significant difference between the low and control groups. Also, the outcome for collagen type II stain was positive; chondrocytes were embedded in collagen type II in the medium and high groups.
Many recent studies have focused on cartilage repair using MSCs, but such attention is vital. In clinical application, Wakitani et al. reported that transplantation of 1 × 107 MSCs embedded in collagen gel with high tibial osteotomy improved the repair outcome of cartilage defects (14 mm × 35 mm) in the medial femoral condyle with white tissue, although the cartilage was softer than normal, and the surgery was more invasive (34). Nishimori et al. mentioned that repair of a chronic osteochondral defect (2 mm × 2.5 mm × 1 mm depth) in the patellar groove of a rat model demands combination therapy of an intra-articular injection of MSCs (1 × 106) with subchondral drilling, while traditional surgical techniques caused potential changes to the subchondral bone (27). In equines, McIlwraith et al. found that clinical use of BM-MSCs in conjunction with microfracture of cartilage defects (1 cm2) in the medial femoral condyle may be potentially beneficial for repair with tissue firmness, although there was no significant difference clinically or histologically between the two groups of microfracture with or without BM-MSCs (20 × 106) (26). For human clinical application, Wong et al. tried to repair chronic cartilage defects (5 cm2) in the medial femoral condyle by combining microfracture, high tibial osteotomy, and intra-articular injection of MSCs (1.47 × 107) suspended in hyaluronan, but only 36% of patients have 50% of cartilage cover, due to some MSCs not being attached to the desired site (35).
There are significant advantages of using a scaffold and growth factors with MSCs according to the concept of successful tissue engineering, but we cannot ignore the potential disadvantages of scaffolds, such as possible toxicity and harmful biodegradation by-products that cause inflammation and cell death (9,19). Indeed, several studies have revealed that there is no difference between using a scaffold with or without MSCs in a rabbit model (17,24). From previous studies, we can conclude that most surgical treatments, scaffolds, and growth factors do not improve MSCs for cartilage repair.
However, the exact mechanisms that direct MSCs for tissue homing are not known. Intra-articular injection of MSCs into the knee joint is the least invasive suitable treatment and to avoid potential complications of systemic administration of MSCs like loss of cells due to liver and lung entrapment. The function of MSCs, according to a previous study, is executed via secretion of bioactive factors, which have been shown to inhibit tissue scarring, suppress apoptosis, stimulate angiogenesis, and enhance mitosis (28). This procedure is a good option, but is not satisfactory due to the wide dispersion of MSCs inside the whole joint. In addition, the adverse effect of an intra-articular injection of too many MSCs inside a rat joint revealed free scar tissues (1). While this phenomenon is not clearly understood, we surmise that it depends upon the relationship between the size of the joint, defect size, and number of injected MSCs to produce free scar bodies, so we suggest that transplantation of 14.3 × 107 m-MSCs with the aid of an external magnetic device will be enough to repair an osteochondral defect (5 cm2) in human clinical application.
Hori et al. found that transplantation of 5 × 105 magnetically labeled synovium derived cells (M-SDCs) in the patellar groove (2 mm diameter; 1 mm depth) using an intra-articular magnet has great chondrogenic potential (13). However, we do not know if isolated SDCs from osteoarthritic joints have the same proliferation and differentiation as healthy tissue. Furthermore, it is more invasive to implant and remove an intra-articular magnet, and doing so causes discomfort to the patient. Therefore, implantation of magnets is best avoided in human patients.
According to previous reports, repair of bone, cartilage, and calcified cartilage are the major challenges for osteochondral defects. Chen et al. performed the first fabrication of scaffolds using PLGA–collagen hybrid mesh for osteochondral tissue engineering. However, the success of such materials has been limited due to lack of porosity (6). In our study, complete formation of the osteochondral junction and integration of the repaired tissue with the surrounding tissue were achieved in the medium and high groups at 12 weeks. This means that complete bone–cartilage interface occurred.
The fundamental aim of the magnetic targeting system is to deliver m-MSCs and to try to reduce the number of cells required. Few previous reports have referred to the use of an external magnetic device to direct m-MSCs into a cartilage defect in the patella and patellar groove. Kobayashi et al. proved that accumulation and direction of m-MSCs to the wall of a flask containing an aqueous suspension was accomplished by applying an external magnetic force (20).
Also, Kobayashi et al. found that most of the cells accumulated in the osteochondral defect (3 mm diameter, 2 mm depth) in an ex vivo study in which cells were stained with Prussian blue, indicating that cells contained iron; however, using a weak magnetic device (0.6 T) made it difficult to handle a large number of m-MSCs (3 × 106) relative to the size of the defect (20).
In the previous studies, ferumoxide has been used as a SPION coated with dextran, in combination with PLL as a transfection agent for humans because ferumoxide does not show sufficient cellular uptake to enable tracking of nonphagocytic cells (3). Conflicting studies have been published, with one revealing that ferumoxide has an inhibitory effect on the chondrogenesis of MSCs (23), but another reported that magnetic labeling with ferumoxide has no inhibitory effect on chondrogenesis of MSCs (2).
Kamei et al. prefer to use a ferucarbotran–protamine complex for magnetic labeling of MSCs (5 × 106) to repair chronic cartilage defects (6 mm diameter). The group applied this method in the patella of a porcine model following an ex vivo study, which revealed that a higher adhesion rate of m-MSCs (95%) was exposed to an external magnetic force (16).
Ferucarbotran is a SPION coated with carboxydextran, while ferumoxide (Feridex) is coated with dextran. It has been suggested that the additional carboxyl groups associated with ferucarbotran might lead to a higher affinity to the cell membrane so that cells could be labeled with or without the need of an incorporation facilitator (25). In our study, we used ferucarbotran for magnetic labeling without using any transfection agent, which was already approved for human clinical application in Japan. Our data showed that using ferucarbotran for magnetic labeling improved proliferation of MSCs; this finding was in agreement with those of other investigators who mentioned that ferucarbotran promotes cell growth due to its ability to diminish intracellular H2O2 through intrinsic peroxidase-like activity (8,14).
In our department, a cell magnetic targeting system was used not only to repair cartilage but also to repair a fracture model. Kodama et al. found that union of femur fracture model was detected when m-MSCs were transplanted, while no union was observed when MSCs or PBS were applied because m-MSCs were recruited to the fracture site, while cells were barely detectable in the MSC group after 3 days. For checking the distribution of transplanted m-MSCs using luciferase-based in vivo bioluminescence imaging, Kodama et al. found that MSC-derived photons accumulated at the fracture site in the m-MSC group in comparison with diffused photons around the femur in the MSC group (22). Also, other investigators engrafted ferromagnetic-labeled matrix-associated stem cell implants (MASI) into osteochondral defects with in vivo visualization using MRI (11,18). MRI will be an established method for clinical application for in vivo monitoring of transplanted cells.
Limitations of this study include that we did not perform in vitro monitoring of m-MSCs to confirm magnetic labeling by using Berlin blue stain and that we did not perform MSC or m-MSC injection without the external magnet.
In future investigations, we will try to evaluate the effect of magnetic labeling on anti-inflammatory activities of MSCs through assessment of interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), IL-6, IL-4, and IL-10 either in synovial tissue or repaired tissue. Also, we would like to study the difference between autologous and allogeneic transplantation of m-MSCs to assess the immune response. Further research will be required for application of a magnetic delivery system to deep organs to decide if there are any limitations for using magnet tracking with these organs.
Conclusion
Complete repair of the severe chronic defect including cartilage and subchondral bone was confirmed with transplantation of 2 × 105 m-MSCs. Ultimately, using an external magnetic device to deliver m-MSCs into diseased parts will be the gold standard treatment option, not only to repair a severe chronic osteochondral defect but also to repair different organs of the body.
Footnotes
Acknowledgments
Special thanks to Prof. Mitsuo Ochi for his encouragement. This research was partly supported by the Highway program for the realization of regenerative medicine from the Japan Science and Technology Agency. It was also supported by the Egyptian Cultural Affairs and Missions Sector through the Egyptian Joint Supervision Scholarship. No benefits in any form have been received from commercial part related directly or indirectly to this article. The authors declare no conflicts of interest.
