Abstract
We previously demonstrated that short-term administration of a combination of anti-LFA-1 and anti-CD154 monoclonal antibodies (mAbs) induces tolerance to neonatal porcine islet (NPI) xenografts that is mediated by regulatory T cells (Tregs) in B6 mice. In this study, we examined whether the coinhibitory molecule PD-1 is required for the induction and maintenance of tolerance to NPI xenografts. We also determined whether tolerance to NPI xenografts could be extended to allogeneic mouse or xenogeneic rat islet grafts since we previously demonstrated that tolerance to NPI xenografts could be extended to second-party NPI xenografts. Finally, we determined whether tolerance to NPI xenografts could be extended to allogeneic mouse or second-party porcine skin grafts. Diabetic B6 mice were transplanted with 2,000 NPIs under the kidney capsule and treated with short-term administration of a combination of anti-LFA-1 and anti-CD154 mAbs. Some of these mice were also treated simultaneously with anti-PD-1 mAb at >150 days posttransplantation. Spleen cells from some of the tolerant B6 mice were used for proliferation assays or were injected into B6 rag-/- mice with established islet grafts from allogeneic or xenogeneic donors. All B6 mice treated with anti-LFA-1 and anti-CD154 mAbs achieved and maintained normoglycemia until the end of the study; however, some mice that were treated with anti-PD-1 mAb became diabetic. All B6 rag-/- mouse recipients of first- and second-party NPIs maintained normoglycemia after reconstitution with spleen cells from tolerant B6 mice, while all B6 rag-/- mouse recipients of allogeneic mouse or xenogeneic rat islets rejected their grafts after cell reconstitution. Tolerant B6 mice rejected their allogeneic mouse or xenogeneic second-party porcine skin grafts while remaining normoglycemic until the end of the study. These results show that porcine islet-specific tolerance is dependent on PD-1, which could not be extended to skin grafts.
Keywords
Introduction
The widespread application of islet transplantation as a treatment for type 1 diabetes is currently limited by the shortage of human donors and the chronic use of harmful immunosuppressive drugs to prevent rejection (31,34). Neonatal porcine islets (NPIs) are being considered as an alternative source of islets for clinical transplantation because an abundant number of islets can be easily isolated and maintained in culture (21). In addition, NPIs are capable of reversing diabetes in both small (20,21,27) and large animals (5,19,37-39), including the preclinical nonhuman primate model (5,37-39). Previously, we demonstrated that a combination of anti-lymphocyte function-associated antigen-1 (anti-LFA-1) and anti-CD154 monoclonal antibody (mAb) therapy could promote long-term survival of both encapsulated and nonencapsulated NPI transplants in immune-competent B6 mice (20,27). We also reported that the protection observed in B6 mice was mediated by CD4+ T cells expressing CD25, forkhead box P3 (FOXP3), and programmed cell death-1 (PD-1) regulatory markers (2), indicating that regulatory T cells (Tregs) may be mediating the tolerance to NPI xenografts. These Tregs may depend on PD-1 to exert their tolerogenic effect on NPI xenografts. Thus, in this study we determined whether PD-1 may be one target for the beneficial effect of anti-LFA-1 and anti-CD154 mAb therapy on NPI xenografts.
PD-1 is a coinhibitory molecule initially found to be upregulated in T-cell hybridoma undergoing apoptosis (14). Several studies showed that PD-1 is important in the induction of tolerance to mouse skin (32), heart (25,32,36), and islet (9) allogeneic transplants. PD-1 was also a key component in the protection observed in a rat to mouse islet xenograft model (24); however, the role of PD-1 has not been investigated in porcine islet xenotransplantation. In this study, we examined the importance of PD-1 in the induction and maintenance of tolerance to NPI xenografts in B6 mice treated with anti-LFA-1 and anti-CD154 mAbs. We also determined whether tolerance to NPI xenografts could be extended to allogeneic mouse or xenogeneic rat islet grafts since we previously demonstrated that tolerance to NPI xenografts could be extended to second-party NPI xenografts. Finally, we determined whether tolerance to NPI xenografts could be extended to second-party porcine and allogeneic mouse skin grafts. Our results show that PD-1 is important in both the induction and maintenance of tolerance to NPI xenografts in B6 mice treated with anti-LFA-1 and anti-CD154 mAbs. We also found that tolerance to NPI xenografts is specific to NPIs, but not to mouse or rat islets and second-party porcine or mouse skin grafts.
Materials and Methods
Animals
Six- to 8-week-old male immune-competent B6 (C57BL/6J, H-2b) and immune-deficient B6 rag-/-(B6.129S7-Rag1tm1Mom/J, H-2b) mice (Jackson Laboratory, Bar Harbor, ME, USA) were used as recipients. These mice were rendered diabetic by a single intraperitoneal injection of 180 or 175 mg/kg body weight of streptozotocin (Sigma-Aldrich, St. Louis, MO, USA) for B6 or B6 rag-/- mice, respectively. Blood glucose levels of the mice were monitored two times a week, and mice were considered diabetic when their blood glucose levels reached ≥20 mmol/L for 2 consecutive days following streptozotocin injection. All mice were fed with standard laboratory food and were cared for according to the guidelines established by the University of Alberta Animal Care and Use Committee and the Canadian Council on Animal Care Committee. Islet or skin donors were 3-day-old Duroc cross neonatal pigs (>1.5 kg body weight) obtained from different sows, 8- to 10-week-old male BALB/c (BALB/cCr//AltBM, H-2d) mice, both purchased from the University of Alberta (Edmonton, Alberta, Canada), and male Wistar–Furth rats (WF, RT1U, 200-300 g body weight; Harlan Laboratories, Indianapolis, IN, USA).
Islet Isolation and Transplantation
Islets were isolated as previously described (3,12,21, 30,33). A total of 500 rodent islets or 2,000 NPIs were transplanted under the left kidney capsule of diabetic B6 or B6 rag-/- mice as previously described (2,21,30,45). The blood glucose levels of recipient mice were monitored two times a week for >100 days posttransplantation. Islet transplantation was considered successful when the blood glucose levels of recipient mice fell below or equal to 8.5 mmol/L. At > 100 days posttransplantation, the kidney that contained the islet graft was removed from mice that maintained normal blood glucose levels to determine whether the islet transplant was responsible for the maintenance of normoglycemia. The islet transplant was considered rejected when the blood glucose levels of recipient mice increased to more than 12 mmol/L for 2 consecutive days after transplantation. Rejection of the islet transplant was confirmed by histological analysis of the graft.
Monoclonal Antibody Therapies
Anti-LFA-1 mAb (KBA; rat IgG2a from ascites; 200 μg on days 0, 1, 7, 14 posttransplantation) and anti-CD154 mAb (MR-1; hamster IgG1; 250 μg on days −1, +1 and two times a week for an additional 4 weeks posttransplantation; BioXcell, West Lebanon, NH, USA) were injected into the peritoneal cavity of B6 mouse recipients of NPIs. Anti-PD-1 mAb was administered in combination with anti-LFA-1 and anti-CD154 mAbs as regimen ***1-500 μg on day 0 and 250 μg on days 2, 4, 6, 8, and 10 posttransplantation (J43, hamster IgG2; BioXcell) or regimen ***2-500 μg per injection every other day starting on days 0 to 14 posttransplantation for a total of eight injections (J43). The same dose and number of injections were used to determine the role of PD-1 in the maintenance of tolerance except that injection began at >150 days posttransplantation. In addition, another clone of anti-PD-1 mAb was used in combination with anti-LFA-1 and anti-CD154 mAbs as regimen ***3-500 μg per injection every other day starting on day 0 to 14 posttransplantation for a total of eight injections (4F10, rat IgG2a, κ chain).
In Vitro Proliferation Assays
The proliferation of lymphocytes was determined by combining 5 × 105 responder cells with 3 × 105 irradiated (2,500 rad) stimulator cells in 96-well flat-bottom plates and were incubated under physiological conditions on days 3, 4, and 5 following our published protocol (42). Spleen cells from tolerant or naive B6 mice were used as responder cells, and spleen cells from naive BALB/c mice, WF rats, the same neonatal pigs as the islet donors (first party) or different neonatal pigs (second party) were used as stimulator cells. Spleen cells from first-party NPI donors were also used as responder cells, and spleen cells from second-party NPI donors were used as stimulator cells and vice versa to ensure the mismatch between these donors. Positive control for these experiments included stimulation of spleen cells from tolerant and naive B6 mice with concanavalin A (Con A; 10 μg/ml; Sigma-Aldrich). In addition, other positive controls included stimulation of spleen cells from primed B6 mice with Con A or with the same porcine spleen cells used to prime the immune response. Spleen cells that were not stimulated and cultured in media alone served as negative controls.
Adoptive Transfer Studies
Diabetic B6 rag-/- mice were transplanted with 500 islets from BALB/c mice or WF rats, or 2,000 islets from first-party or second-party neonatal pigs. At 150 days posttransplantation, all mice with normal blood glucose levels (≤8.5 mmol/L) received intraperitoneal injection of 50 × 106 spleen cells collected from tolerant or naive B6 mice. After cell injection, the blood glucose levels of B6 rag-/- mice were monitored three times a week for 60 days or until the mice became diabetic, indicating that the islet transplant had been rejected, then the kidney bearing the islet transplant was removed for histological analysis. For those mice that remained normoglycemic for 60 days after cell injection, the kidney that contained the islet graft was removed, and their blood glucose levels were monitored until they became diabetic, which indicated that normoglycemia was maintained by the islet graft. The presence of adoptively transferred immune cells was confirmed using flow cytometry.
Skin Transplantation
Tolerant B6 mice were transplanted with full-thickness skin from BALB/c mice or second-party neonatal pigs at 100 days post-first-party NPI transplantation. Skin grafts were inspected daily until they were rejected. In a parallel experiment, skin transplantation was also performed on naive B6 mice and B6 rag-/- mice as positive and negative control groups of rejection, respectively.
Immunohistological Analysis
The presence of insulin-producing cells and FOXP3+ immune cells in the grafts was examined following our published protocol (2,3,27). Briefly, the kidney bearing the islet graft was fixed in 10% buffered formalin (Fisher Scientific, Fair Lawn, NJ, USA) solution and embedded in paraffin (Fisher Scientific), and the other half was cryopreserved in frozen tissue-embedding media (Fisher Scientific). Then 5-μm sections were stained with guinea pig anti-insulin primary antibody (1:1,000; Dako Laboratories, Burlington, ON, Canada) or rat anti-mouse FOXP3 antibody (1:25 dilutions; eBioscience, San Diego, CA, USA) for 30 min, followed by the addition of biotinylated goat antiguinea pig IgG (1:200; Vector Laboratories, Burlingame, CA, USA) or biotinylated goat anti-rat IgG secondary antibody (1:200; Southern Biotechnology Associates, Inc., Birmingham, AL, USA). Avidin–biotin complex/horseradish peroxidase (ABC/HP; Vector Laboratories) and 3,3-diaminobenzidine tetrahydrochloride (DAB; BioGenex, San Ramon, CA, USA) were used to detect positive-stained cells (brown color). Paraffin-embedded tissue sections were counterstained with Harris' hematoxylin (Cedarlane, Burlington, ON, Canada) and eosin (Fisher Scientific), and cryopreserved tissue sections were counterstained with Harris' hematoxylin.
Detection of Anti-Donor Antibodies by Flow Cytometry
The levels of mouse anti-donor IgG antibody in naive and transplant recipient mice were determined using flow cytometry as previously described (2,3,27). Briefly, spleen cells (1 × 106) from islet or skin donors were incubated with naive or transplant recipient's serum (1:128 dilution for xenogeneic donor and recipient combination and 1:64 dilution for allogeneic combination) for 1 h at 37°C, 5% CO2, and 95% air. After incubation, the cells were then washed with phosphate-buffered saline (PBS; Sigma-Aldrich) and incubated with fluorescein isothiocyanate (FITC)-conjugated anti-mouse IgG antibody (1:200; Southern Biotechnology Associates, Inc. for xenogeneic or 1:100; Jackson Immuno Research Laboratories, Inc., West Grove, PA, USA, for allogeneic combination) for 1 h at 4°C. The levels of IgG antibody were detected from single-parameter fluorescence histograms on a BD FACS Calibur flow cytometry machine (BD Biosciences Pharmingen, Mississauga, ON, Canada) after gating on viable lymphocytes. Control groups for this study included donor spleen cells that were not incubated with mouse serum and those incubated with a FITC-conjugated secondary antibody only. The mean fluorescence intensity (MFI) for each sample was determined, and the fold changes in the MFI when compared to the MFI obtained in cells incubated with secondary antibody only were calculated since the levels of mouse IgG antibody detected in this negative control group were almost similar to those obtained in cells not incubated with mouse serum.
Characterization of Lymphocytes by Flow Cytometry
Spleen cells from B6 mouse recipients of NPI and B6 rag-/- mice reconstituted with spleen cells from naive or transplanted B6 mice were isolated at the end of the study as described previously (2,29). One million spleen cells were incubated with antibodies specific to immune cell markers, and the percentages of the cells that expressed the markers were determined using flow cytometry by gating on live cells (2). Control groups for this experiment included spleen cells from naive and transplanted B6 mice as well as reconstituted B6 rag-/- mice that were not incubated with the antibodies.
Statistical Analysis
Statistical differences between groups in in vitro proliferation assays, expression of immune cell markers, and levels of anti-donor IgG antibody were determined using nonparametric Kruskal–Wallis test. Statistical differences in graft survival among the groups were analyzed using the Kaplan–Meier log rank test. All statistical tests were performed using GraphPad Prism software (La Jolla, CA, USA). A value of p < 0.05 was considered statistically significant.
Results
PD-1 Is Important in the Induction and Maintenance of Tolerance to NPI Xenografts
We first examined whether induction of tolerance to NPI xenografts is dependent on PD-1 by interfering with the PD-1 interaction to its ligands, programmed death ligand 1 (PD-L1) and PD-L2, using anti-PD-1 mAb. First, we administered anti-PD-1 mAb (J43) for a short time in combination with anti-LFA-1 and anti-CD154 mAbs. Similar to what we found in B6 mice treated with anti-LFA-1 and anti-CD154 mAbs (n = 8), B6 mouse recipients of NPI simultaneously treated with short-term administration of anti-LFA-1, anti-CD154, and anti-PD-1 mAbs (J43, regimen 1, n = 8) achieved and maintained normal blood glucose levels for more than 100 days posttransplantation (Table 1). Histological analysis of the NPI xenografts from these mice showed intact islets with insulin-producing cells and no immune cell infiltration (Fig. 1A). Similar results were observed in the NPI xenografts from tolerant B6 mice treated with anti-LFA-1 and anti-CD154 mAbs and immune-deficient B6 rag-/- mouse recipients of the same NPIs (Fig. 1B, C, respectively). In addition, the amount of anti-porcine IgG antibodies detected from these mice (MFI: 25.1 ± 0.8, fold change in MFI: 1.39 ± 0.05) (Fig. 1G, n = 8) was comparable to the amount found in tolerant B6 mice treated with anti-LFA-1 and anti-CD154 mAbs (MFI: 26.8 ± 0.6, fold change in MFI: 1.49 ± 0.03) (Fig. 1G, n = 8) and naive B6 mice (MFI: 22.4 ± 0.8, fold change in MFI: 1.24 ± 0.04) (Fig. 1G, n = 5). We next assessed whether the amount of anti-PD-1 mAb administered to the mice was not enough to abrogate the protective effect of the combined mAb therapy. We then modified the regimen for anti-PD-1 mAb following the protocol published by Mai et al. (24). Increasing the dose and time of administration of anti-PD-1 mAb abolished the tolerance to NPI xenografts induced by the combination of anti-LFA-1 and anti-CD154 mAbs. We found that four out of eight B6 mouse recipients of NPIs simultaneously treated with anti-LFA-1, anti-CD154, and anti-PD-1 mAbs (clone J43, regimen 2) did not achieve normoglycemia, while the remaining four mice achieved normoglycemia within 5 to 8 weeks posttransplantation (Table 1). When the kidney bearing the NPI xenograft was removed from mice that maintained normoglycemia for >100 days posttransplantation, all four mice became diabetic, demonstrating that normoglycemia in these mice was maintained by the NPI xenograft. Examination of the NPI xenografts from mice that maintained normoglycemia for >100 days posttransplantation revealed some insulin-producing β-cells and infiltration of mononuclear cells (Fig. 1D). We also found that none of the mice simultaneously treated with anti-LFA-1, anti-CD154, and the other clone of anti-PD-1 mAbs (clone 4F10, regimen 3) achieved normoglycemia (Table 1). Histological analysis of the NPI xenografts harvested from these mice showed absence of insulin-positive stained cells in the graft site and abundant immune cell infiltration (Fig. 1E) contrary to what was observed in the NPI xenografts of tolerant B6 mice treated with anti-LFA-1 and anti-CD154 mAbs (Fig. 1B). The levels of mouse anti-pig IgG antibody detected from all mice treated simultaneously with anti-LFA-1, anti-CD154, and anti-PD-1 mAbs (regimens 2 and 3) were significantly (p < 0.001) higher, (MFI: 51.6 ± 0.8, fold change in MFI: 2.87 ± 0.05, and MFI: 79.3 ± 1.4, fold change in MFI: 4.40 ± 0.08, respectively) (Fig. 1G, n = 8) than those observed in tolerant B6 mice treated with anti-LFA-1 and anti-CD154 mAbs (MFI: 26.8 ± 0.6, fold change in MFI: 1.49 ± 0.03) (Fig. 1G, n = 8). Collectively, these results indicate that PD-1 may play a role in the induction of tolerance to NPI xenografts rendered by anti-LFA-1 and anti-CD154 mAb therapy.
Effect of anti-PD-1 treatment during the induction and maintenance of tolerance to NPI xenografts. NPI xenografts from B6 mice treated with a combination of anti-LFA-1 and anti-CD154 mAbs with (A, J43 regimen 1) or without anti-PD-1 mAb (B). (C) NPI xenograft from immune-deficient B6 rag-/- mouse. Intact islets positive for insulin-stained cells are shown. (D, E) NPI xenograft from tolerant B6 mouse simultaneously treated with anti-LFA-1, anti-CD154, and anti-PD-1 mAbs (J43, regimen 2 and 4F10, regimen 3, respectively). (F) NPI xenograft from tolerant B6 mouse treated with anti-PD-1 mAb (J43, regimen 2) at >150 days posttransplantation. Representative NPI xenografts from eight mice for each group are shown, and all images were taken at the same magnification. Scale bar: 100 μm. (G) Mouse anti-porcine IgG antibodies in naive nontransplanted B6 mice (G, black bar, n = 5) and tolerant B6 mice treated with anti-LFA-1 and anti-CD154 mAbs alone (clear bar, n = 8) or with anti-PD-1 mAb (regimen 1, perpendicular line bar, n = 8). Mouse anti-porcine IgG antibodies in tolerant B6 mice simultaneously treated with anti-PD-1 mAb (J43, regimen 2, light gray bar, n = 8 and 4F10, regimen 3, dark gray bar, n = 8) are also shown. (H) Mouse anti-porcine IgG antibodies detected in tolerant B6 mice treated with anti-PD-1 mAb (J43, regimen 2, light gray bar, n = 8) at 150 days posttransplantation compared with mouse anti-porcine IgG antibodies in untreated tolerant B6 mice (clear bar, n = 8). (I) Mean percentage of FOXP3+PD-1+ cells in CD4+CD25+ spleen cells from naive (black bar, 31.2% ± 2.5%, n = 5) and tolerant B6 mice (clear bar, 90.4% ± 0.9%, p < 0.001, n = 5). (J) Mean percentage of CD25+FOXP3+ cells in CD4+PD-1+ spleen cells from naive B6 mice (black bar, 17.4% ± 1.4%, n = 5) compared to the same spleen cell population from tolerant B6 mice (clear bar, 87.8% ± 1.3%, p < 0.001, n = 5). Survival of NPI Xenografts in B6 Mice Simultaneously Treated With a Combination of Anti-LFA-1, Anti-CD154, and Anti-PD-1 mAbs All mAbs were administered intraperitoneally. p < 0.05 versus group of mice treated with anti-LFA-1 and anti-CD154 mAbs with or without anti-PD-1 mAb (regimen 1); p < 0.05 versus group of mice treated with anti-LFA-1 and anti-CD154 mAbs plus anti-PD-1 mAb (regimen 2).

NPI Xenograft Survival in Tolerant B6 Mice Treated With Anti-PD-1 mAb

B6 mice were transplanted with 2,000 NPIs and treated with anti-LFA-1 mAb (KBA; rat IgG2a from ascites; 200 μg on days 0, 1, 7, 14 posttransplantation) and anti-CD154 mAb (MR-1; hamster IgG1; 250 μg on days −1, +1 and 2 times a week for an additional 4 weeks posttransplantation). At >150 days, some mice were treated with anti-PD-1 mAb.
p < 0.01 versus untreated mice.
Combination of Anti-LFA-1 and Anti-CD154 mAbs Induces Porcine-Specific Unresponsiveness
Previously we demonstrated that tolerance induced by anti-LFA-1 and anti-CD154 mAbs can be extended to second-party NPI donors. We next determined whether the specificity of tolerance to NPI xenografts could also be extended to mouse islet allografts and rat islet xenografts using in vitro and in vivo experimental models. We found that on days 3, 4, and 5 of culture, lymphocytes from spleen of tolerant B6 mice responded strongly when stimulated with BALB/c mouse or WF rat spleen cells (Fig. 2A, n = 3). These responses were comparable to the proliferation of lymphocytes from naive B6 mice stimulated with the same BALB/c mouse and WF rat spleen cells on days 3, 4, and 5 of culture (Fig. 2B, n = 3). In contrast, weak proliferation of lymphocytes from tolerant B6 mouse recipients of NPI was observed after stimulation with first-party or second-party porcine donors (Fig. 2C, n = 3). The responses observed in this group were comparable to those seen in lymphocytes of naive B6 mice stimulated with the same cell populations on the same days of culture (Fig. 2D, n = 3). However, a strong response was observed from lymphocytes of pig-primed B6 mice stimulated with spleen cells from first-party porcine donors (Fig. 2D, n = 3). As expected, we observed robust proliferation of lymphocytes from first-party (Fig. 2E, n = 3) and second-party porcine islet donors (Fig. 2F, n = 3) after stimulation with lymphocytes from second- and first-party porcine islet donors, respectively on days 3, 4, and 5 of culture. These results confirmed the mismatch between the first- and second-party porcine donors.
Proliferation of lymphocytes from naive and tolerant B6 mice in vitro. (A) Proliferation of lymphocytes expressed as counts per minute (CPM) from tolerant B6 mice after stimulation with ConA (white diamonds), allogeneic BALB/c mouse (white squares), or xenogeneic WF rat spleen cells (white triangles). (B) Proliferation of lymphocytes from naive B6 mice after stimulation with ConA (black diamonds), BALB/c mouse (black squares), or WF rat spleen cells (black triangles). (C) Proliferation observed when lymphocytes from tolerant B6 mice were stimulated with ConA (white diamonds) or spleen cells from first- or second-party porcine donors (white circles and triangles, respectively). (D) Proliferation of lymphocytes from naive B6 mice stimulated with ConA (black diamonds) or spleen cells from first- or second-party porcine donors (black circles and triangles, respectively). Proliferation of pig-primed B6 mouse lymphocytes after stimulation with pig spleen cells used to prime the immune response is also shown (D, black reverse triangles). Proliferation of lymphocytes from first-party (E, white circles) and second-party (F, black circles) porcine islet donors after stimulation with spleen cells from second- and first-party porcine donors, respectively. Proliferation of lymphocytes from tolerant (white hexagons) and naive (black hexagons) B6 mice cultured in media alone are also shown. Results are shown as the mean CPM ± SEM of three samples per group.
We also found that all of the B6 rag-/- mice transplanted with islets achieved and maintained normal blood glucose levels for >150 days posttransplantation. Mice that received BALB/c mouse or WF rat islets achieved normoglycemia within 1 day posttransplantation, while mice transplanted with first- or second-party NPI achieved normoglycemia at 78 days posttransplantation due to the immature nature of the porcine islets. B6 rag-/- mice with established allogeneic BALB/c mouse islet grafts were reconstituted with spleen cells from tolerant B6 mice with first-party NPI xenograft. These mice became diabetic within 14 and 25 days post-cell injection with a mean survival time (MST) of 20.3 ± 1.9 days (Table 3, Fig. 3A, n = 7). B6 rag-/- mice with the same allogeneic BALB/c mouse islet grafts and were injected with spleen cells from naive B6 mice also became diabetic between 9 and 14 days post-cell injection (MST is 11.6 ± 0.8 days) (Table 3, Fig. 3A, n = 5). The difference in the rate of rejection between these two groups was found to be statistically significant (p < 0.002). Similarly, B6 rag-/- mice transplanted with rat islets and reconstituted with spleen cells from tolerant B6 mice with first-party NPI xenograft rejected their grafts by 9.6 ± 0.6 days post-cell injection (Table 3, Fig. 3A, n = 7). B6 rag-/- mice with rat islet xenografts and received spleen cells from naive B6 mice also rejected their grafts between 10 and 15 days post-cell injection (MST is 12.0 ± 0.8 days) (Table 3, Fig. 3A, n = 5). As expected based on our previous findings (2), B6 rag-/- mice with first- or second-party NPI xenografts that were injected with spleen cells from tolerant B6 mice transplanted with first-party NPI xenografts maintained normoglycemia until the end of the study (>60 days post-cell injection) (Table 3, Fig. 3A, n = 9 and n = 10, respectively). While all B6 rag-/- mouse recipients of first- or second-party NPI xenografts that received spleen cells from naive B6 mice rejected their grafts by 12.3 ± 1.1 or 13.0 ± 1.2 days, respectively (Table 3, Fig. 3A, n = 4 and n = 3, respectively). Mononuclear cell infiltrate and absence of islets were observed in B6 rag-/- mice transplanted with allogeneic BALB/c mouse (Fig. 3B) or xenogeneic WF rat islets (Fig. 3C) and reconstituted with spleen cells from tolerant B6 mice. In contrast, the islet grafts from B6 rag-/- mice with established first-party (Fig. 3D) and second-party (Fig. 3E) NPI xenografts that were reconstituted with spleen cells from tolerant B6 mice with first-party NPI xenografts had intact islets staining strongly positive for insulin. While the grafts from B6 rag-/- mice with established first- or second-party NPI xenografts that received spleen cells from naive B6 mice had mononuclear cell infiltrate, no insulin-positive cells remained in the grafts (Fig. 3F).
Survival of allogeneic BALB/c mouse, xenogeneic WF rat, and xenogeneic porcine islet grafts in B6 rag-/- mice reconstituted with spleen cells from naive and tolerant B6 mice. (A) Survival of allogeneic mouse, xenogeneic rat, or porcine islet grafts in B6 rag-/-mice reconstituted with spleen cells from naive or tolerant B6 mice. (B) Representative islet allograft, (C) rat islet xenograft, (D) xenogeneic first-party NPI graft, and (E) xenogeneic second-party NPI graft from B6 rag-/- mice reconstituted with spleen cells from tolerant B6 mice. (F) Representative NPI xenograft from B6 rag-/- mice reconstituted with spleen cells from naive B6 mice. Scale bar: 100 μm, and dark structures represent insulin-positive-stained cells. (G) IgG antibodies detected in B6 rag-/- mice with BALB/c mouse islet grafts and reconstituted with spleen cells from tolerant and naive B6 mice (light gray and black bars, n = 7, respectively). (H) Mouse anti-rat IgG antibodies in B6 rag-/- mice transplanted with WF rat islets and reconstituted with spleen cells from tolerant (light gray bar, n = 7) or naive (black bars, n = 7) B6 mice. (I) IgG antibodies detected in B6 rag-/- mice transplanted with first-party (light gray bar, n = 9) or second-party (dark gray bar, n = 10) NPIs and reconstituted with spleen cells from tolerant B6 mice. The average level of mouse anti-porcine IgG antibody in B6 rag-/- mice with NPI xenografts and reconstituted with spleen cells from naive B6 mice is also shown (black bar, n = 7). Survival of Mouse Islet, Rat Islet, and NPI Grafts in B6 rag-/- Mice Reconstituted With Spleen Cells From Tolerant or Naive B6 Mice Diabetic B6 rag-/- mice were transplanted with 500 islets from BALB/c mice or WF rats, or 2,000 islets from first-party or second-party neonatal pigs. At 150 days posttransplantation, all mice with normal blood glucose levels (≤8.5 mmol/L) received intraperitoneal injection of 50 × 106 spleen cells from tolerant or naive B6 mice. After cell injection, the blood glucose levels of B6 rag-/- mice were monitored for 60 days or until the mice became diabetic. For those mice that remained normoglycemic for 60 days after cell injection, the kidney that contained the islet graft was removed, and their blood glucose levels were monitored until they became diabetic. p < 0.002 between the groups of B6 rag-/- mice transplanted with allogeneic BALB/c mouse islets injected with spleen cells from tolerant or naive nontransplanted B6 mice.
The levels of anti-mouse (MFI: 101.0 ± 4.3, fold change in MFI: 5.61 ± 0.24) (Fig. 3G, n = 7) and anti-rat (MFI: 69.0 ± 5.5, fold change in MFI: 3.83 ± 0.30) (Fig. 3H, n = 7) IgG antibody in B6 rag-/- mice with allogeneic BALB/c mouse or xenogeneic WF rat islet grafts that received spleen cells from tolerant B6 mice with first-party NPI xenograft were comparable in recipients of the same islets and reconstituted with spleen cells from naive B6 mice (MFI: 102 ± 5.0, fold change in MFI: 5.71 ± 0.28 and MFI: 72.6 ± 4.8, fold change in MFI: 4.03 ± 0.27) (Fig. 3G, n = 5 and Fig. 3H, n = 5, respectively). In contrast, the levels of anti-porcine IgG antibody in reconstituted B6 rag-/- mice with first-party (MFI: 22.9 ± 0.8, fold change in MFI: 1.27 ± 0.04) (Fig. 3I, n = 9) or second-party (MFI: 22.8 ± 0.8, fold change in MFI: 1.27 ± 0.05) (Fig. 3I, n = 10) NPI xenografts were significantly (p < 0.001) lower than those observed in B6 rag-/- mice with NPI xenografts that received spleen cells from naive B6 mice (MFI: 145.1 ± 6.1, fold change in MFI: 8.06 ± 0.34) (Fig. 3I, n = 7).
Tolerance Induced by a Combination of Anti-LFA-1 and Anti-CD154 mAbs Is Specific to Porcine Islet but not to Porcine Skin Xenografts
Tolerant B6 mice with first-party NPI xenograft received full-thickness skin graft from either a second-party neonatal pig or allogeneic BALB/c mouse on day 100 post-first-party NPI transplantation (Fig. 4A, B). All second-party porcine skin xenografts were rejected by 15.8 ± 1.7 days post-skin transplantation (Table 4, Fig. 4C, n = 5). While all naive B6 mice transplanted with the same porcine skin rejected their grafts significantly (p < 0.002) faster (6.8 ± 0.7 days posttransplantation) (Table 4, Fig. 4C, n = 5) compared to the rate of rejection observed in tolerant B6 mice. All B6 rag-/- mice transplanted with the second-party porcine skin xenografts maintained their skin grafts until the end of the study (>60 days posttransplantation) (Table 4, Fig. 4C, n = 3). We also found that all tolerant B6 mice transplanted with full-thickness BALB/c mouse skin rejected their grafts by 10.8 ± 0.7 days post-skin transplantation (Table 4, Fig. 4C, n = 5) similar to what was observed in naive B6 mice (10.6 ± 0.8 days posttransplantation) (Table 4, Fig. 4C, n = 5). Despite the rejection of second-party porcine and BALB/c mouse skin grafts, tolerant B6 mice remained normoglycemic until the end of the study (>200 days post-NPI transplantation) (Fig. 4A, B, respectively), and they only became diabetic when the first-party NPI xenograft was removed. The NPI xenografts from these mice had intact islets (Fig. 4D, E) with immune cells expressing FOXP3 surrounding, but not infiltrating, the NPI xenografts (Fig. 4F, G). Despite the rejection of second-party porcine skin grafts in tolerant B6 mice, the levels of IgG antibody against second-party porcine skin donors were significantly (p < 0.01) lower (MFI: 40.2 ± 1.6, fold change in MFI: 2.23 ± 0.09) (Fig. 4H, n = 5) compared to the levels of IgG antibody against the same porcine skin xenografts in naive B6 mice (MFI: 94.8 ± 5.3, fold change in MFI: 5.27 ± 0.30) (Fig. 4H, n = 5). The levels of anti-mouse IgG antibody (MFI: 98.1 ± 4.0, fold change in MFI: 5.45 ± 0.22) (Fig. 4I, n = 5) in tolerant B6 mice that rejected the allogeneic BALB/c mouse skin grafts were comparable to those detected in naive B6 mice that rejected the same mouse skin grafts (MFI: 104.4 ± 4.1, fold change in MFI: 5.80 ± 0.23) (Fig. 4I, n = 5).
Survival of porcine and skin grafts in naive and tolerant B6 mice. (A) Blood glucose levels of tolerant B6 mice transplanted with second-party porcine skin (first arrow). (B) Blood glucose levels of tolerant B6 mice transplanted with BALB/c mouse skin (first arrow). Second arrow in (A) and (B) represents the time when the left kidney bearing the first-party NPI xenograft was surgically removed. (C) Survival of porcine and mouse skin grafts in naive and tolerant B6 mice as well as in B6 rag-/- mice. (D) Representative first-party NPI xenograft from tolerant B6 mice transplanted with second-party porcine skin. (E) Representative NPI xenograft from tolerant B6 mice transplanted with BALB/c mouse skin. Insulin-positive cells are shown as dark-stained cells. (F) FOXP3+ cells (dark-stained cells) in NPI xenograft of tolerant B6 mouse with second-party porcine skin graft. (G) FOXP3+ cells in NPI xenograft of tolerant B6 mouse with BALB/c mouse skin graft. (F and G) Representative images are shown. Scale bars: 100 μm. (H) Mouse anti-porcine IgG antibodies in tolerant (clear bar, n = 5) and naive (black bar, n = 5) B6 mice with second-party porcine skin graft. (I) Mouse anti-mouse IgG antibodies in tolerant (clear bar, n = 5) and naive (black bar, n = 5) B6 mice transplanted with BALB/c mouse skin. Survival of Allogeneic BALB/c Mouse or Second-Party Porcine Skin Grafts in Tolerant B6 Mice With First-Party NPI Xenograft, Naive Nontransplanted B6 or Naive Nontransplanted B6 rag-/- Mice Tolerant B6 mice were transplanted with full-thickness skin from BALB/c mouse or second-party neonatal porcine donors at 100 days post-first-party NPI transplantation. Skin grafts were inspected daily until they were rejected. Skin transplantation was also performed on naive B6 mice and B6 rag-/- mice as positive and negative control groups of rejection, respectively. p < 0.002 between tolerant and naive B6 mice transplanted with full-thickness skin graft from the same second-party neonatal porcine donors.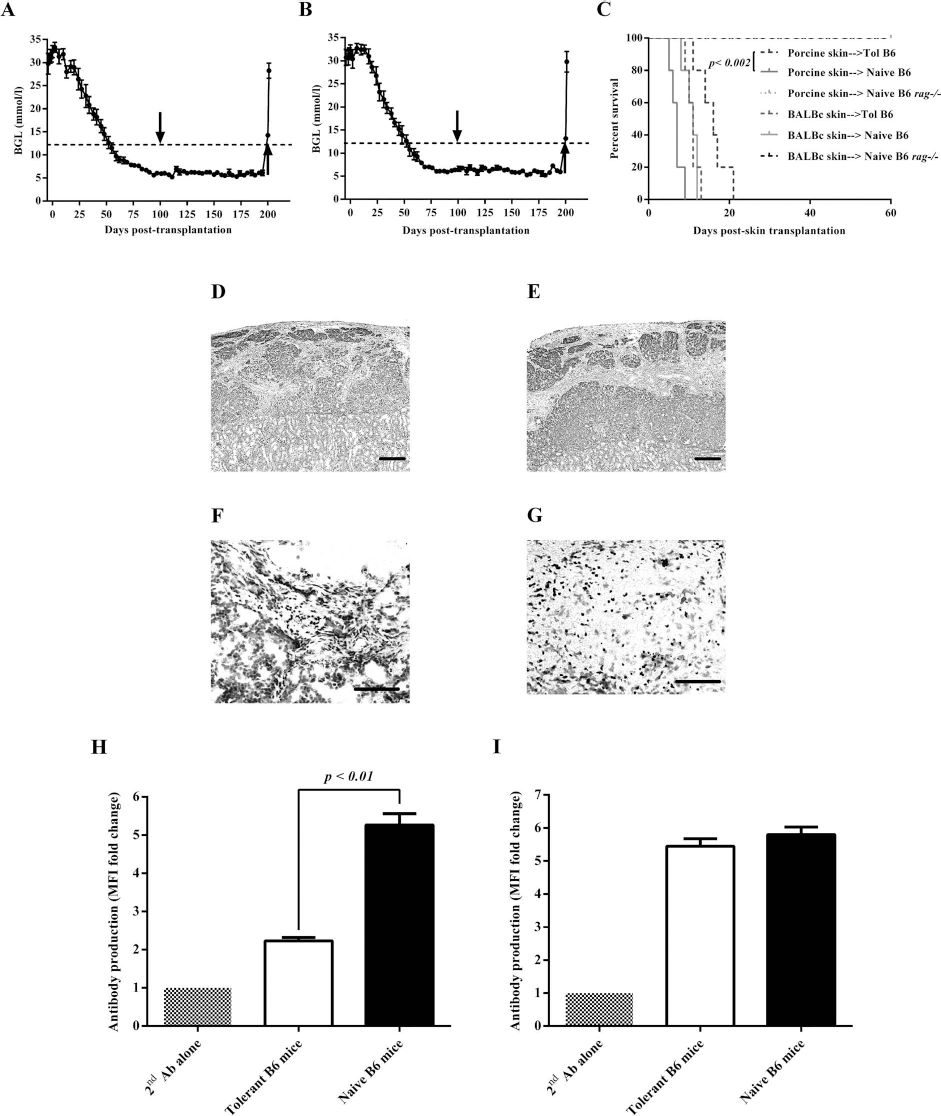

Discussion
Our previous studies demonstrated that transient therapies directed against T-lymphocyte activation and function using mAbs resulted in long-term NPI xenograft survival, especially regarding highly phylogenetically disparate xenograft donors (2,3,20,27). In particular, we recently showed that short-term administration of a combination of anti-LFA-1 and anti-CD154 mAb therapy resulted in tolerance to NPI xenografts that is mediated by Tregs (2). In the current study, we demonstrated that tolerance induced by the combined mAb therapy is dependent on PD-1 and is specific to porcine islets from either first- or second-party donors.
The importance of PD-1 has previously been reported in the induction and maintenance of peripheral tolerance (1,2,9,13,15,16,24,25,32,36,41,46,48). Previously we showed that spleen cells isolated from B6 mice treated with anti-LFA-1 and anti-CD154 mAbs that eventually rejected their NPI xenografts had significantly lower expression of PD-1 compared to what we observed in tolerant B6 mice that had significantly higher expression of PD-1 particularly on CD4+ T cells (2). These results lead us to examine the importance of PD-1 in the induction and maintenance of tolerance to NPI xenografts. Our data showed that PD-1 is important in both the induction and maintenance of tolerance to NPI xenografts. Fifty and 100% of tolerant mice treated with anti-PD-1 mAb (J43, regimen 2 and clone 4F10, regimen 3, respectively) rejected their islet xenografts and failed to achieve normoglycemia. The difference in the effect of the two clones (J43 and 4F10) of anti-PD-1 mAb, which are blocking and not depleting antibodies, could be due to the difference in the strength of negative signals these antibodies deliver to the target cells, considering that these two mAbs target the same epitope (24). We also showed that 100% of tolerant B6 mice rejected their NPI xenografts and eventually became diabetic within 16 to 27 days (with a mean survival time of 21.5 ± 1.2 days postinjection) when given anti-PD-1 mAb beginning at >150 days posttransplantation. We previously observed comparable time in the abrogation of tolerance when tolerant B6 mice were treated with depleting anti-CD25 mAb, which resulted in a mean survival time of 25.3 ± 2.5 days postinjection (2). In this study, we further characterized the spleen cells from tolerant B6 mice and found that the majority (88-90%) of CD4+CD25+ or CD4+PD-1+ cells also coexpressed foxP3+PD-1+ or CD25+FOXP3+ cell markers, respectively, suggesting that PD-1 may be a target of the beneficial effect of a combination of anti-LFA-1 and anti-CD154 mAbs on NPI xenografts.
Similar to what we found, species-specific tolerance to islet xenografts was also reported earlier by other groups (10,11,23). For example, Goss et al. (11) showed that 50% of recipients that were treated with antilymphocyte serum (ALS) did not reject their third-party Lewis rat islet xenografts, while Gordon et al. (10) demonstrated that the combination of donor-specific transfusion (DST) and anti-CD154 mAb induces species-specific prolongation of rat islet xenografts. Lehnert et al. (23) also demonstrated that a combination of anti-CD154 mAb with cytotoxic T-lymphocyte antigen 4 fusion protein (CTLA4Fc) produced indefinite survival of rat islet xenografts in 100% of mouse recipients. It is possible that rat or porcine islets of different strains share a wide range of species-specific antigens that are recognized through indirect antigen presentation in the mouse host. Thus, tolerance induced to the repertoire of indirectly presented xenograft-associated antigens may demonstrate a high degree of cross-reactivity with unrelated donors (28). It is interesting to note that while islet allograft rejection has both major histocompatibility complex (MHC) class II-dependent and -independent mechanisms of rejection, the response to porcine xenografts appears to be greatly reliant on the indirect MHC class II-restricted pathway (29). Despite being finite in nature, the indirect antixenograft response appears to be extremely virulent, but once tolerance to the islet xenograft is achieved, the state of unresponsiveness appears to be universal to the species.
Our results indicate that maintenance of tolerance induced by combined anti-LFA-1 and anti-CD154 mAbs requires the presence of porcine antigens responsible for induction of tolerance and suggest for a perpetual role for host antigen-presenting cells (APCs) in maintaining antigen exposure to Tregs (17,44). Similar to what was previously demonstrated, we showed that antigen-specific suppression can be adoptively transferred (4,6) as was found in our adoptive transfer experiments where suppression activity of Tregs was observed in B6 rag-/-mouse recipients of either first- or second-party NPI xenografts. It is possible that due to the lack of porcine-specific antigens in BALB/c mouse or WF rat islet grafts, the Tregs were not activated, and suppression did not take place. Therefore, the antigen specificity observed both in vitro and in vivo could be explained by the hypothesis that Tregs are activated only in hosts that have the antigens for which they are specific.
Unlike the previous study by Gordon et al. (10), which showed that combined DST with anti-CD154 mAb therapy could be effective in prolonging the survival of rat skin xenografts, we showed that tolerance to porcine islets induced by the combined anti-LFA-1 and anti-CD154 mAbs could not be extended to porcine skin xenografts. The rejection of skin may require different costimulation pathways or additional pathways for T-cell activation. Gordon et al. (10) suggested that such pathways might be mediated by APCs found in skin but not in islets, an example would be the epidermal Langerhans cells. Our results suggest that tolerance to NPI xenografts induced by short-term administration of combined anti-LFA-1 and anti-CD154 mAbs is specific to antigens that are common to porcine islets but not to porcine skin. In addition, all of the tolerant B6 mice retransplanted with second-party allogeneic BALB/c mouse skin or xenogeneic porcine skin maintained normoglycemia, showing that tolerance to NPI xenografts could not be broken during the rejection process of mouse or porcine skin xenografts. We sought evidence of Tregs in the islet graft sites of these mice and showed the presence of FOXP3+ cells surrounding the protected NPI xenografts. This finding suggests that cell-mediated regulation, at least in part, might be operating within the tolerated islet grafts. As was suggested, FOXP3+ T cells in the graft play a key role in dominant tolerance induced by combined DST and anti-CD154 mAb (22). It is possible that Tregs in NPI transplant site has created an immunological privilege site that protects the islet xenograft from potentially damaging immune reactions during the rejection process of BALB/c mouse or porcine skin grafts. Such an idea has been advocated for how tumors may avoid immunological control (7). However, a significant delay in the rejection of porcine skin xenografts in tolerant B6 mice compared to the rejection of the same porcine skin grafts in naive B6 mouse recipients was observed, which could be partially due to the expression of some porcine antigens that may be common between the porcine islets and porcine skin. We also observed significantly lower levels of IgG antibody against the second-party porcine skin in tolerant B6 mice compared to the levels of IgG antibody against the same porcine skin xenografts in naive B6 mice. This could be due to a cell-mediated immune regulation conducted by a subpopulation of Tregs (CD4+ CD25+CD90+FOXP3+), as was shown by Vevis et al. (43). The role of this Treg subpopulation remains to be determined in our model. In addition, it remains to be determined whether tolerance to NPI xenografts induced by combined anti-LFA-1 and anti-CD154 mAbs could be extended to vascular solid organs such as heart or kidney. This is not possible in our model, but perhaps it could be tested in porcine to nonhuman primate models when a tolerizing regimen in nonhuman primates is developed.
In conclusion, the data presented here indicate that tolerance induced by short-term administration of a combination of anti-LFA-1 and anti-CD154 mAbs is species and tissue specific. In addition, our data showed the critical role of PD-1 in both the induction and maintenance of tolerance to NPI xenografts induced by a combination of anti-LFA-1 and anti-CD154 mAbs through CD4+CD25+FOXP3+PD-1+ Tregs. We think that these Tregs regulate the immune response to NPI by suppressing the effector T cells that are reactive to porcine islet antigens through the expression of PD-1 and secretion of interleukin-10 (IL-10) (2). In the presence of porcine antigens and costimulatory signals provided by the interactions of CD154 with CD40 and LFA-1 and intercellular adhesion molecule-1 (ICAM-1), CD4+ T cells are activated resulting in the rejection of porcine islet grafts. In the absence of these costimulatory signals using respective mAb to CD154 and LFA-1, at the time of transplantation, Tregs are generated possibly through the conversion of T effector cells to a regulatory phenotype, a process that is known as infectious tolerance (8,18,26,35,40). It is possible that any new T cells reactive to NPIs that enter the repertoire after transplantation, when immunoregulation is the dominant mechanism of tolerance as long as the porcine islet antigen is present, will be converted to Tregs, thereby propagating and reenforcing the tolerant state throughout the posttransplant period (47). This was evident in our previous (2) and current studies that show significantly higher percentage of CD4+ T cells with regulatory cell markers (i.e., CD25, FOXP3, PD1) detected from tolerant B6 mice compared to naive mice and untreated mice that were not tolerant to NPI. Blocking the interaction between PD-1 and its ligands using monoclonal antibodies to PD-1 resulted in the rejection of NPI xenografts. In our previous studies, we also observed higher expression of glucocorticoid-induced TNFR family-related gene (GITR) and cytotoxic T-lymphocyte-associated protein 4 (CTLA4) coinhibitory molecules in CD4+CD25+ Tregs from tolerant B6 mice. It remains to be determined whether these Tregs also play an important role in the induction and maintenance of tolerance to NPI xenografts. Further, the role of IL-10 in the induction and maintenance of tolerance to NPI xenografts remains to be confirmed. Taken together, our results suggest the possibility of development of clinical protocols that simultaneously target both LFA-1 and CD154 pathways for preventing rejection and induction of robust tolerance to NPI xenografts mediated by Tregs.
Footnotes
Acknowledgments
We acknowledge the technical assistance of Deb Dixon and Dawne Colwell and thoughtful discussions with Dr. Tsunehiro Kobayashi. We are grateful to the Canadian Diabetes Association, which provided major funding for this work as well as the Edmonton Civic Employees' Charitable Assistance Fund, Stollery Children's Hospital Foundation, the MacLachlan Fund University Hospital Foundation, Canadian Institutes of Health Research, Colliers International, Ken and Denise Cantor as well as Ewa and John Burton, who provided additional support. The Muttart Diabetes Research Training Centre provided scholarship for H.A. The authors declare no conflicts of interest.
