Abstract
Aerosolized drug delivery has recently attracted much attention as a possible new tool for the delivery of complex nanoparticles. This study aims to investigate whether catheter-based aerosolization of islets via endobronchial systems is a feasible option in islet transplantation. Besides investigating the feasibility of islet aerosolization, we also examined cluster cell vitality and structural integrity of the islets following aerosolization. Using an ex vivo postmortem swine model, porcine pancreatic islets were isolated and aerosolized with an endoscopic spray catheter. Following aerosolization, islet cell vitality and function were assessed via Calcein AM and propidium iodide as well as insulin production after glucose exposure. In the final step, the overall feasibility of the procedure and structural integrity of cells were analyzed and evaluated with respect to clinical applicability. No significant difference was detected in the viability of control islets (90.67 ± 2.19) vs aerosolized islets (90.68 ± 1.20). Similarly, there was no significant difference in control islets (1.62 ± 0.086) vs aerosolized islets (1.42 ± 0.11) regarding insulin release after stimulation. Indocyanine green marked islets were transplanted into the lung without major difficulty. Histological analysis confirmed retained structural integrity and predominant location in the alveolar cavity. Our ex vivo data suggest that catheter-based aerosolized islet cell delivery is a promising tool for the application of cell clusters. According to our data, islet cell clusters delivery is feasible from a mechanical and physical perspective. Moreover, cell vitality and structural integrity remain largely unaffected following aerosolization. These preliminary results are encouraging and represent a first step toward endoscopically assisted islet cell implantation in the lung.
Introduction
Type I diabetes mellitus is an autoimmune disorder, which is characterized by the destruction of β cells of the pancreatic Langerhans islets. Disruptions in insulin secretion and glycemic control homeostasis result in several secondary metabolic diseases. For affected patients, human pancreatic islet allografts are a potential treatment 1,2 . Due to the limited supply of quality human islets, attempts have been made to perform islet xenotransplantation 3,4 , which requires the use of nonhuman xenogeneic pancreatic islets donors. While this is a challenging process, some promising results were obtained in the past three decades in both animal studies and clinical trials 5 –8 . However, multiple challenges with xenograft transplantations have been described, including relevant complication rates and risk of collateral damage during the transplantation procedure, e.g., portal vein damage 9 . Other risks include immunological incompatibility, oxygen deprivation, and early loss of 50%–70% of transplanted islets 10 –12 .
While a variety of solutions are currently under investigation, one of the more promising options is microencapsulation. Microencapsulation is designed to improve protection against the initial innate immune response following transplantation 13,14 . This is based on the concept of placing a mechanic and chemical barrier to prevent innate immune cell (e.g., macrophage) adhesion. Since islet cells are bedded in surrounding tissues, they have a relatively large surface that facilitates cells of the immune system to check for human leukocyte antigen (HLA) compatibility. While introduction of a barrier can decrease the extent of HLA compatibility check, it may also compromise oxygen flow and sustainability of cells, further enhancing the importance of establishing a balance between these two aspects.
Currently, the use of aerosol generation is increasingly attracting attention for the delivery of drug particles in cancer treatments. By means of aerosolization, complex particles, e.g., liposomal doxorubicin, protein macroparticles, and even entire cells can be both delivered into body cavities as well as dispersed to cover entire biological surfac 15 -19 . By means of aerosolization we can combine a new, minimally invasive method to deliver islet cells with a novel application site. This study is the first to consider islet cell transplantation in the pulmonary airways via bronchoscopy. Theoretically, the bronchial system and alveolar structure are an ideal location for islet cell transplantations: Within the alveoli, porcine islet cells have direct vascular access to the highest oxygen concentration anywhere in the body. At the same time, these cells do not have full contact with biological surfaces as they are not embedded into surface tissues due to their round-shaped morphology and small size (<200 µm). In this study, we examine the delivery of aerosolized islet cells via endoscopy catheter device with respect to technical feasibility, cell implantation in bronchial tissue, cluster cell vitality, and structural integrity after following the aerosol generation process.
Materials and Methods
Porcine Pancreatic Islet Isolation and Culture
All animal procedures were approved by the Institutional animal care and use committee at the University of California, Irvine. Porcine pancreata were procured from 8- to 15-day-old, preweaned Yorkshire pigs and isolated as previously described 20 . In brief, pancreata were procured under 10 min and stored in Hank’s balanced salt solution (HBSS Thermo-Fisher Scientific, Austin, Texas, USA) on ice. The cold ischemic time of all procured pancreata was limited to less than 1 h. Each unperfused pancreas was individually minced into 1 mm3 pieces and digested in a 37°C, 100 rpm shaking water bath for 15 min using Sigma Type V Collagenase (2.5 mg/ml, dissolved in HBSS; cat# C8051, Sigma-Aldrich, St. Louis, Missouri, USA). HBSS supplemented with 1% porcine serum (cat# 26250084, Thermo Fisher Scientific) was used to quench the digestion, and digested pancreatic tissues were filtered through a 500-µm metal mesh. After the isolation, preweaned porcine islets (PPIs) were cultured in an islet maturation media supplemented with antibiotic/antimycotic solution (Corning Inc., Midland, North Carolina, USA, cat#30-004-CI), and 10% porcine serum in a 37°C, 5% CO2 humidified incubator (cat#3110, Thermo Forma Series II 3120 Water Jacketed CO2 Incubators) for 7 days in T-150 untreated suspension flasks (cat # CLS430825, Corning Inc.) 21 . A full media change was performed on day 1, and a half media change was done on days 3 and 5. PPIs were collected on day 7 of culture for assessment.
Microcatheter
The microcatheter (MC, PW-205L, Olympus, Aomori, Japan) consists of a connecting device with a pressure line connecting the shaft to the nozzle. The nozzle head has a small central opening. Ten milliliters of each sample is delivered within a constant flow using a 10 ml syringe and high manual pressure (1 ml/s). The MC generates a polydisperse aerosol.
Ex Vivo Model
The experiments were performed using a standard ex vivo model for aerosolization experiments. This ex vivo model is well established and has been previously described in multiple studies 16,22 . A commercially available hermetic plastic box with a total volume of 3.5 l, was used. In the center of the top cover of the plastic box, a 5 mm trocar (Kii® Balloon Blunt Tip System, Applied Medical, Rancho Santa Margarita, CA, USA) was placed. The nozzle of the MC was introduced into the trocar. The plastic box was kept at a constant room temperature of 27°C for the entire procedure. A large sterile Petri dish was placed in the corner of the box to collect aerosolized material for further analysis. All experiments were repeated and independently performed three times.
Islet Morphology
An 100-µL aliquot of islets were collected for staining with 1 ml dithizone (DTZ, MP Biomedicals, Irvine, Califonria, USA, cat#150999) for 5 min [20]. DTZ-stained islets were imaged using a digitally inverted microscope (EVOS XL Digital Imaging System; Life Technologies, Austin, Texas, USA).
Islet Viability
PPIs (100 IEQ) were washed with phosphate buffered saline and incubated for 30 min in Calcein AM (CalAM, Invitrogen, Carlsbad, Califonria, USA, cat#C1430) and propidium iodide (PI, Invitrogen, cat#P3566, Carlsbad, Califonria, USA) for staining of living and dead cells, respectively 23 . After washing, the viability of CalAM/PI-stained islets was evaluated using a microplate reader (Tecan Infinite F200; Tecan, Chapel Hill, North Carolina, USA) and calculated using the following equation: CalAM-positive cells/(CalAM-positive cells + PI-positive cells) × 100.
Islet Function
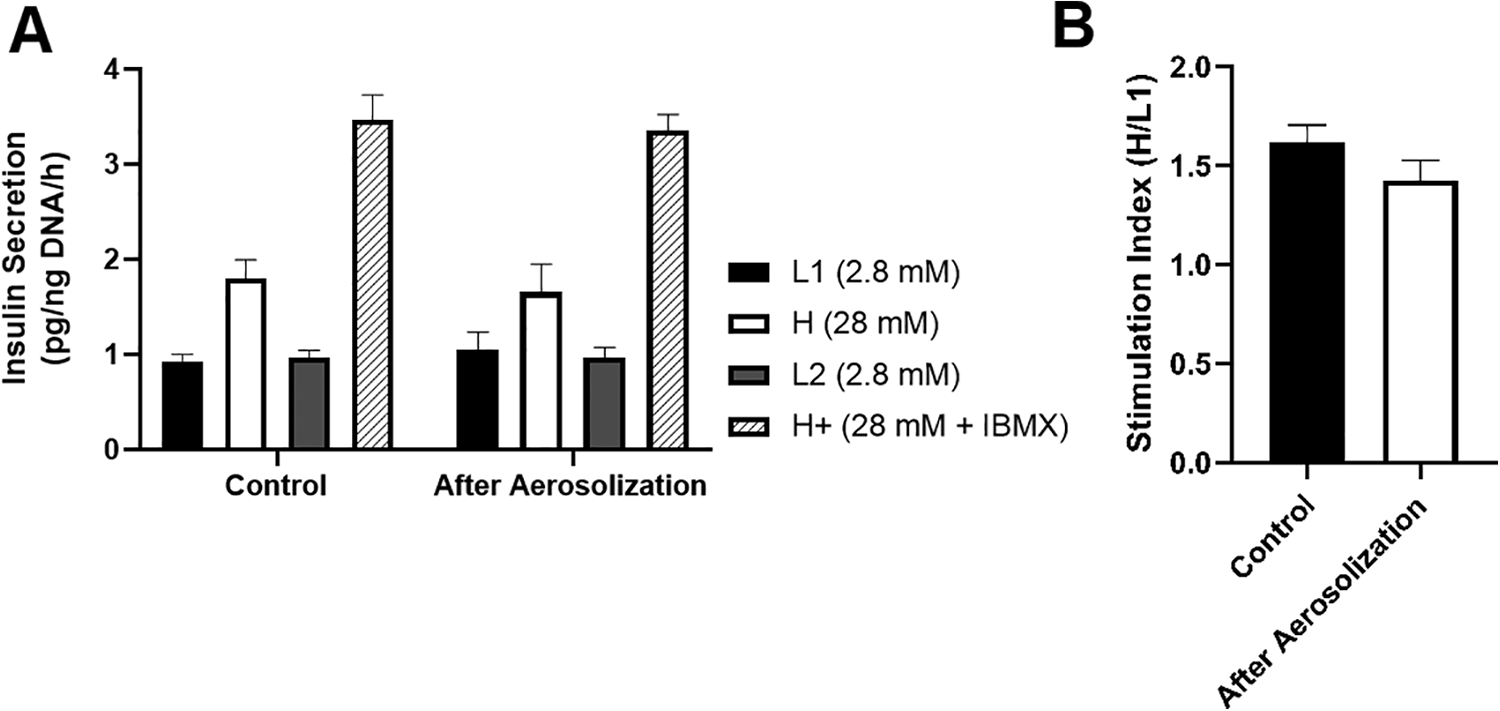
Insulin secretion of PPIs was determined by glucose-stimulated insulin release (GSIR) assay 19 . PPIs (100 IEQ) were incubated at 37°C and 5% CO2 for 1 h in the following order of glucose media: low glucose (2.8 mM; L1), high glucose (28 mM; H), second low glucose (2.8 mM; L2), and high glucose plus 3-isobutyl-1-methylxanthine (28 mM + 0.1 mM IBMX; H+). The insulin concentration from the supernatant of each media was determined using a standard porcine insulin enzyme-linked immunosorbent assay (Porcine Insulin ELISA; cat#10-1200-01, Mercodia, Winston-Salem, North Carolina, USA) and analyzed on a microplate reader (Tecan Infinite F200; Tecan, Chapel Hill, North Carolina, USA). The amount of secreted insulin in each media was normalized to the DNA content of each sample and expressed as pg of insulin/ng of DNA/h. The stimulation index was calculated by dividing the amount of insulin in the high glucose media over the first low glucose media.
Islet DNA Content
Islets were collected after GSIR assay and lysed with cell lysis buffer (10 mM Tris–HCl, 1 mM ethylenediaminetetraacetic acid, 1% Triton X-100, pH 8). Islets were then sonicated on ice for 30 s using an ultrasonic sonicator (Sonics VibraCell Ultrasonic Processor Model VC70 T, Sonics & Materials, Inc. Newtown, Connecticut, USA) to ensure total cell lysis. The islet cell lysate was centrifuged at 1,400×g for 15 min at 4°C and the supernatant was collected. DNA content from each supernatant sample was determined using a fluorescent dsDNA stain (Quant-iT PicoGreen dsDNA kit; cat #Q32850, Molecular Probes, Eugene, Oregon, USA) and quantified using a microplate reader (Tecan Infinite F200; Tecan, Chapel Hill, North Carolina, USA) 24 .
Islet Transplantation in the Lungs
Experiments were performed in two swine at 30 min postmortem (German landrace pigs). Cadavers were placed in supine position. An endoscope (GIF-HQ 190°, Olympus, Shinjuku, Tokyo, Japan) was introduced into the trachea and then into the right bronchus. A microspray catheter (PW-205 V Olympus Surgical Technologies Europe, Hamburg, Germany) was introduced into the endoscope and launched into the bronchial system (Fig. 1). A suspension of 10,000 IEQs of islet cells was dissolved into 10 ml NaCl 0.9% with 0.5 ml indocyanine green (ICG, IC-GREEN-™, Akorn Inc., Buffalo Grove, IL, USA) and filled into a sterile syringe. The suspension was then aerosolized into the bronchial system. After aerosolization thoracotomy was performed with removal of ICG marked tissue and subsequent histological analysis. Lung samples were subject to hematoxylin and eosin (H&E) staining as well as insulin staining.[Note to typesetter: Figures 1, 2, and 3 have been renumbered as Figures 2, 3, and 1 to follow sequential order. Please set the artworks accordingly.]

Intrabronchial endoscopy-assisted aerosolization of islets in swine using a microcatheter.
Islet viability after aerosolization; 100 IEQ were stained with CalAM and PI for 30 min. Stained islets were analyzed using a microplate reader. Islet viability was calculated by the following equation: CalAM-positive cells/(CalAM-positive cells + PI-positive cells) × 100. n = 3 for each group. Data expressed as mean ± SEM. CalAM: Calcein AM; PI: propidium iodide; SEM: standard error of the mean.

Function of PPIs in response to glucose challenge after aerosolization. Islet function after aerosolization was determined using glucose-stimulated insulin release assay. A triplicate of 100 IEQ was incubated for 1 h in the following order of glucose media: 2.8 mM glucose (L1), 28 mM glucose (H), 2.8 mM glucose (L2), and 28 mM + 0.1 mM IBMX glucose (H+). The amount of insulin secreted by islets from each media was quantified by ELISA and normalized to the total DNA. (A) Insulin secretion per ng DNA after 1-h incubation in varying concentrations of glucose media. (B) Stimulation index calculated as the insulin secretion in H media over L1 media. n = 3 for each group. Data expressed as mean ± SEM. ELISA: enzyme-linked immunosorbent assay; IBMX: 3-isobutyl-1-methylxanthine; PPI: preweaned porcine islet.
Histological Analysis
Lung samples transplanted with PPIs were fixed for 48 h in 10% neutral buffered formalin (cat# 22-026-354, Fisher Scientific). Formalin-fixed samples were prepared for paraffin processing by serial dehydration in increasing concentrations of ethanol solutions using a tissue processor (Leica TP1020, Leica Microsystems, Buffalo Grove, Ilinois, USA). After preparation, tissues were embedded in paraffin wax using a tissue embedder (Leica EG 1150C, Leica Microsystems, Buffalo Grove, Ilinois, USA). Paraffin-embedded tissue blocks were sectioned into 5-µm sections on a microtome (Leica RM 2255, Leica Microsystems, Buffalo Grove, Ilinois, USA). Five-micrometer sections were stained with H&E. For the detection of insulin-positive cells, 5-µm sections were prepared using an IHC kit according to the manufacturer’s protocol [Mouse and Rabbit Specific HRP/DAB (ABC) Detection IHC kit; cat#ab64264, Abcam] and incubated in insulin primary antibody (1:400; cat#ab6995, Abcam). Insulin-stained slides were counterstained with hematoxylin. All slides were imaged using an inverted microscope (Nikon Ti-E Widefield microscope, Nikon Instruments Inc. Melville, NY, USA)
Statistical Analysis
All data are expressed as mean ± standard error of the mean. An unpaired two-sample Student’s t-test was used to determine statistical significance. P-values <0.05 were considered statistically significant. All data were analyzed on GraphPad Prism (GraphPad Software 8.0.1, Inc. La Jolla, California, USA).
Results
Viability of PPIs After Aerosolization
Viability of PPIs after aerosolization via endoscopic catheter-based device was evaluated by CalAM and PI staining. There was no significant difference in the viability of control islets (90.67 ± 2.19) vs aerosolized islets (90.68 ± 1.20) (P = NS, Fig. 2).
Function of PPIs in Response to Glucose Stimulation Following Aerosolization
Insulin secretory function of PPIs after aerosolization was determined by GSIR assay. The amount of insulin secreted per hour in L1, H, L2, and H+ glucose media of control islets (L1 = 0.93 ± 0.069 pg/ng DNA/h, H = 1.80 ± 0.20 pg/ng DNA/h, L2 = 0.97 ± 0.077 pg/ng DNA/h, H+ = 3.47 ± 0.26 pg/ng DNA/h) was similar to aerosolized islets (L1 = 1.05 ± 0.18 pg/ng DNA/h, H = 1.66 ± 0.29 pg/ng DNA/h, L2 = 0.97 ± 0.11 pg/ng DNA/h, H+ = 3.36 ± 0.17 pg/ng DNA/h) (P = NS, Fig. 3A). Similarly, there was no significant difference in the stimulation indices in control islets (1.62 ± 0.086) vs aerosolized islets (1.42 ± 0.11) (P = NS, Fig. 3B).
Bronchoscopy and Histological Analysis of PPIs After Transplantation in the Lung
Placement of the MC into the bronchial system was feasible and islet cells were delivered. The injection of the suspension had to be swift as not to cause too much cell sedimentation within the syringe. No congestion of the MC was observed. H&E histology showed detachable, round-shaped cell clusters (Fig. 4A, B). Besides their characteristic shape and location of the structures, insulin staining confirmed their identity (Fig. 4C). In the alveolar space, islets were mainly detached. In contrast, in small-sized bronchioles, only very few islet cells were detached. The alveolar space, usually in the shape of a sacculus, was usually significantly larger than the islet cells. The contact surface between the islets and the alveolar wall was mostly minimal. Most of the islet surface was surrounded by air.

Histological staining of lung tissue after alveolar islet transplantation. Islets trapped in the alveolar sacculus (A) and in single alveoli (B), both depicted using hematoxylin and eosin staining. (C) Islets marked with red arrow using insulin staining.
Discussion
Delivery of pharmacological substances into the lung via aerosolization is an established concept in pulmonology. Besides topical applications, aerosolization represents one of the least invasive options of drug delivery. Thus, multiple attempts have been made to deliver aerosolized insulin into the airways 25,26 . However, to the authors’ knowledge, no attempts have been made to investigate actual islet cell transplantation by means of aerosolization in the lung. Endoscopically assisted alveolar islet cell transplantation may reduce complication rates and risks related to the transplantation procedure. While bronchoscopy is not completely risk free, the occurrence of severe complications is rare 27,28 with bleeding, respiratory depression, and pneumothorax occurring in <1% of cases. Mortality is extremely rare with a reported death rate of 0%–0.04% 17 .
Despite the overall safety in using bronchoscopy, aerosolization of larger particles, especially cells, is a challenging task with risks of losing structural integrity and cellular stability 26 . However, as our data indicate, the tear force during the spraying procedure apparently does not mechanically disrupt islet cells. This is an important finding, since we know that islet cells’ structural integrity is essential to their functionality 29 –31 . Besides the structural integrity, other biological functions of islet cells must also be kept intact.
From a technical perspective, bronchoscopy-assisted islet cell transplantation in the lung via aerosol generation is possible. Uncertainties on previously experienced challenges, e.g., immunological incompatibility and oxygen depravity, must be addressed in future in vivo models. Our preliminary data are encouraging and indicate that these challenges can be overcome by better choice of transplantation sites, e.g., alveoli and other bronchial sites.
Based on basic physiologic knowledge, we know that the alveoli contain the highest oxygen levels in the body. Also, surface contact with adjacent alveoli seem limited as most of the islet cell surface faces the air lumen and not the surrounding tissues. Thus, the presented surface antigen is minimally exposed to passing cells of the immune system. Besides, this in-lumen location serves as a partial barrier to the immune system itself since passing cells would have to enter the alveolar cavity to exert their effect.
Conclusion
This study is the first to demonstrate a feasible concept for islet cell transplantation in the lung. Implantation of islet cells into the alveolar cavity has the potential to overcome currently faced challenges in xenograft islet transplantations. While our preliminary data are promising, further studies are required to assess the biological efficiency of this method with regard to in vivo transplantations.
Footnotes
Ethical Approval
This study was approved by our institutional review board (IACUC-Approval case number: AUP-17-241).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
