Abstract
Abnormal angiogenesis is a primary cause of many eye diseases, including diabetic retinopathy, age-related macular degeneration, and retinopathy of prematurity. Mesenchymal stem cells (MSCs) are currently being investigated as a treatment for several such retinal diseases based on their neuroprotective and angiogenic potentials. In this study, we evaluated the role of systemically injected human placental amniotic membrane-derived MSCs (AMSCs) on pathological neovascularization of proliferative retinopathy. We determined that AMSCs secrete higher levels of transforming growth factor-β (TGF-β1) than other MSCs, and the secreted TGF-β1 directly suppresses the proliferation of endothelial cells under pathological conditions in vitro. Moreover, in a mouse model of oxygen-induced retinopathy, intraperitoneally injected AMSCs migrated into the retina and suppressed excessive neovascularization of the vasculature via expression of TGF-β1, and the antineovascular effect of AMSCs was blocked by treatment with TGF-β1 siRNA. These findings are the first to demonstrate that TGF-β1 secreted from AMSCs is one of the key factors to suppress retinal neovascularization in proliferative retinopathy and further elucidate the therapeutic function of AMSCs for the treatment of retinal neovascular diseases.
Keywords
Introduction
Retinal neovascularization is the common causative pathology of age-related macular degeneration (AMD), proliferative diabetic retinopathy, retinopathy of prematurity, and ischemic retinal vein occlusion. This neovascularization often leads to vitreous hemorrhage and retinal detachment, with subsequent visual loss (22). Although trophic factors have been shown to play a central role in angiogenesis, the underlying mechanism of retinal neovascularization remains to be elucidated. Under normal conditions, angiogenic homeostasis is controlled by the balance between stimulators and inhibitors of angiogenesis. Under pathological conditions, however, the retina increases the production of angiogenic stimulators and reduces the production of angiogenic inhibitors, and this disrupted balance leads to neovascularization (12,42). Historically, attempts to treat retinal neovascularization have focused on inhibition of excessive neovascularization or the removal of abnormally formed vessels (3,18).
Transplantation of mesenchymal stem cells (MSCs) is considered to be a useful method for angiogenic therapy and tissue repair (41). It is well known that MSCs are able to differentiate into vascular cell lineages. In addition to their potential to differentiate, MSCs secrete soluble factors, such as growth factors, cytokines, and chemokines, which are primarily responsible for many of the observed therapeutic benefits through paracrine effects (21,30). Notably, several researchers have reported that MSCs can modulate angiogenesis in retinal diseases through secreting angiogenesis-related factors, such as vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), hepatocyte growth factor (HGF), and transforming growth factor-β (TGF-β1) (31,35).
TGF-β1, a multifunctional growth factor, is secreted by MSCs and has been reported to play a key role in MSC-mediated modulation of the immune response (5). Moreover, TGF-β1 has recently been shown to function as a potent inducer of angiogenesis, specifically as a differentiation-promoting/growth-inhibiting factor and stimulator of vascular maturation. TGF-β1 also regulates the differentiation of smooth muscle cells (SMCs) through Smad3 and p38/MAPK signaling and can induce the proliferation and migration of SMCs, which contributes to the formation and maturation of stable vessels (38). In proliferative retinopathy, TGF-β1 has been proposed to act as a suppressor of retinal neovascularization (40).
Amniotic MSCs (AMSCs) are derived from the amniotic membrane of the human placenta (4). The abundance of available placental tissue and their ease of collection from the tissue make AMSCs an attractive source of human stem cells that presents few ethical issues (2). Moreover, AMSCs express human leukocyte antigen (HLA)-ABC (major histocompatibility complex I, MHC I) and HLA-G, but not HLA-DR (MHC II), on their cell surfaces, and therefore may generate a less intense immune response than adult MSCs (6,34). Overall, these properties make AMSCs an ideal cell type for the treatment and repair of neovascular retinopathy.
Here we examine the possible effects of AMSCs on retinal neovascularization by characterizing TGF-β1 expression by AMSCs under pathological conditions. AMSCs showed a more potent suppressive effect on the proliferation of endothelial cells under pathological conditions than under normal conditions. Furthermore, we confirmed the suppressive effect of AMSCs on in vivo neovascularization using an oxygen-induced retinopathy (OIR) mouse model. When AMSCs were treated with TGF-β1 siRNA and transplanted into OIR mice, they failed to reduce neovascular tuft formation. In contrast, non-siRNA-silenced AMSCs were capable of carrying out this function when transplanted in OIR mice. These findings demonstrate that TGF-β1 secreted from AMSCs under pathological conditions is a crucial factor to suppress neovascularization. Furthermore, the effects of AMSC treatment on retinopathy shown in this study help to elucidate their angiogenesis-modulatory properties.
Materials and Methods
AMSC Culture and Expansion
All procedures done in this study were reviewed and approved by the Institutional Review Board (IRB) of CHA General Hospital, Seoul, Korea. Human placenta was collected immediately following delivery in CHA General Hospital, Seoul, Korea. Each informed consent form was received prior to the use of samples. Upon receiving the placenta (one sample, single donor, male), each tissue (decidua basalis) was carefully dissected and rinsed with phosphate-buffered saline (PBS; Welgene, Daegu, Republic of Korea). The harvested pieces of tissue were cut into small pieces and digested with 0.5% collagenase IV (Sigma-Aldrich, St. Louis, MO, USA) in a shaker incubator at 37°C for 30 min. The harvested AMSCs were cultured in a T 25 flask (2 × 105 cells/mm3; Nunc, Rochester, NY, USA) in α-MEM (Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (Gibco), 100 μg/ml each of penicillin and streptomycin (Gibco), 25 ng/ml FGF4 (R&D Systems, Minneapolis, MN, USA), and 1 μg/ml heparin (Sigma-Aldrich). All cultures were maintained at 37°C in an incubator with 5% CO2, and the culture medium was changed every day. In each experiment, AMSCs were grown at an approximate density of 80%, and cells of no more than passage 5 (P5) were used for experiments. To track the injected cells, AMSCs were incubated with 2 mM of CM-DiI (1,1′-dioctadecyl 3,3,3′,3′-tetramethy lindocarbocyanine pechlorate), a lipophilic membrane-bound fluorescent dye (Molecular Probes, Eugene, OR, USA) for 30 min at 37°C (Supplementary Fig. 1A; available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0).

Characterization of AMSCs. (A) FACS analysis of the immunophenotypic profiles of AMSCs. AMSCs were negative for SSEA4, TRA-1-81, and TRA-1-60 (ES markers) and CD34 (hematopoietic and endothelial cell markers). They were positive for CD9 (nontrophoblast marker) and CD13, CD90, CD105, and CD73 (low) (MSC markers). (B) Comparison of TGF-β1 expression in AMSCs, hBM-MSCs, and hAdipo-MSCs.
Immunophenotypic Analysis
Flow cytometry analysis was performed on AMSCs at P5. Cells (2 × 105) were incubated with either fluorescein isothiocyanate (FITC) or phycoerythrin (PE) conjugated with antibodies: SSEA4, TRA-1-60, TRA-1-81, CD34, CD13, CD90, CD9, CD73, and CD105 (1:1,000; BD, Becton Dickinson, San Jose, CA, USA) for 30 min. Ten thousand events were acquired per antibody with fluorescence-aided cell sorting (FACS) in a Calibur Flow Cytometer (BD, Hialeah, FL, USA). The results were then analyzed using CellQuest Pro software (BD Biosciences, San Jose, CA, USA).
In Vitro Stimulation of AMSCs
AMSCs were seeded at 2 × 105 cells/ml, cultured overnight in six-well plates (Becton Dickinson, Franklin Lakes, NJ, USA), and then stimulated with the proinflammatory cytokines TNF-α (10 ng/ml; R&D Systems) and IFN-γ (100 ng/ml; Peprotech, Rocky Hill, NJ, USA) for 6 h. After stimulation, the treated medium was aspirated and replaced with fresh medium. Concentrations and times were chosen after titration (data not shown). The supernatants were collected and assayed using enzyme-linked immunoassay (ELISA), described below.
Treatment with siRNA Targeting TGF-β1
Using the human nucleotide sequences for TGF-β1 mRNA obtained from GenBank (GI:260655621) and referring to the design strategy of siRNA, two pairs of 21-bp reverse repeat sequences targeting TGF-β1 mRNA were designed and synthesized by Bioneer (Seoul, Korea). siRNA targeted the TGF-β1 coding sequence as follows: sense: 5′-CAGAGUACACACAGCAUAU-3′; antisense, 5′-AUA UGCUGUGUGUACUCUG-3′. siRNA (at 100 pmol final concentration) was transfected with Lipofectamine 2000 (Invitrogen, Gaithersburg, MD, USA) according to the manufacturer's instructions. The efficacy of TGF-β1 gene silencing was assessed by quantitative real-time PCR (qRT-PCR) at 24 h after transfection.
Coculture of Human Umbilical Vein Endothelial Cells (HUVEC) with AMSCs
Human umbilical vein endothelial cells (HUVECs) (pooled donors) were generously provided by Dr. Hyung-Min Chung at CHA University (Seoul, Korea). Cells were grown on plates in the EGM-MV Bullet Kit (5% FBS in EBM medium with 12 μg/ml BBE, 1 μg/ml hydrocortisone, and 1 μl/ml GA-1000; Clonetics, San Diego, CA, USA). HUVECs (six passages) with ~70% confluency were used for most experiments. Transwell (BD) six-well dishes with a membrane pore size of 0.4 μm were used. AMSCs (2 × 105 cells/well) were seeded in the Transwell inserts and allowed to attach overnight. For each treatment, AMSCs were treated with/without siRNA or proinflammatory cytokines. siRNA treatment was conducted overnight, before stimulation with the proinflammatory cytokines (for 6 h). The plates were left to incubate overnight before the proliferation assay was carried out. The HUVECs (2 × 105 cells/well) were seeded on the bottom of Transwells overnight, and AMSCs and HUVECs were cocultured for the inhibition assay.
Inhibition of HUVEC Proliferation
HUVECs were seeded in a six-well plate (2 × 105 cells/well) in growth medium. After a starvation phase of 16 h (1% FCS), cells were stimulated with VEGF165 (20 ng/ml; R&D Systems) and incubated with a 5′-bromodeoxyuridine (BrdU; 1:1,000; Sigma-Aldrich) solution at a final concentration of 0.01 mM for 24 h. Cell proliferation was assessed using a BrdU-based proliferation assay. The incorporated BrdU is stained with a specific anti-BrdU (1:1,000; Abcam, Cambridge, MA, USA) fluorescent antibody. The absorbance was read with a tunable microplate reader (VersaMax; Molecular Devices, Sunnyvale, CA, USA).
The Oxygen-Induced Retinopathy (OIR) Model
All experiments were performed in accordance with the “Guidelines for the Care and Use of Laboratory Animals” at CHA General Hospital, and all experimental procedures were approved by the CHA General Hospital Research Institute Animal Care and Use Committee. OIR was induced in C57BL/6J mice according to a protocol described by Smith et al. (43). Briefly, on P7, pups (mixed gender) and their mothers were transferred from room air to an environment of 75% oxygen for 5 days and afterward returned to room air. The hyperoxic environment was created and maintained using a chamber from Jeung Do B&P (Seoul, Korea). Body weights were recorded on P12 and P17 (Supplementary Table 1; available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0).
Intraperitoneal Transplantation of AMSCs
Immediately after their return to room air on P12, pups were matched for their body weights and were randomly assigned into groups that received an intraperitoneal (IP) transplantation of either 1 × 106 cells diluted in 50 μl of PBS (hAMSC group, n = 8) or 50 μl of PBS (Control group, n = 8). Transplantation was performed in the right lower abdominal quadrant with careful handling to reduce loss of solution through backflow. The retinas of recipient mice were examined at P17.
Immunohistochemistry and Visualization of the Vasculature
Retinas were harvested from P13 to P17 for imaging of the vasculature, localization, and characterization of the injected or endogenous cells. The P17 retinas contained many AMSCs. Quantification of vascular obliteration and preretinal neovascular tufts at P17 was performed. Normal and OIR pups were placed on a 60-mm plate that was located over ice to maintain the pups motionless during the procedure. This represented an alternative to anesthesia. The animals were sacrificed using isoflurane (induction 5%, maintenance 1.5%; Hana Pharm. Co., Ltd., Seoul, South Korea) followed by cervical dislocation. The anterior segment and vitreous humor were quickly removed into ice-cold PBS and the eyecups immersed and fixed in ice-cold 4% paraformaldehyde for 1 h. Immunohistological assays to stain blood vessels were conducted as previously described (29). Briefly, retinas were fixed in 4% paraformaldehyde (PFA; Sigma-Aldrich) for 1 h, followed by blocking in 10% normal goat serum (NGS; Invitrogen) for 1 h at room temperature, followed by overnight incubation with Griffonia simplicifolia lectin (GS-lectin) (1:500; Vector Laboratories Inc., Burlingame, CA, USA) conjugated to a fluorescent label to identify vessels (Invitrogen). After washes, retinas were counterstained with 4′,6-diamidino-2-phenylindole (DAPI) (1:400; Invitrogen) to visualize the cell nuclei. Retinas were laid flat with radial-relaxing incisions to obtain whole-mount preparations. In some cases, for tracking of the injected cells, only blocking was conducted, without the vessel staining, to prevent fluorescence interference. Images were acquired using a microscope fitted with a digital camera system (Nikon, Tokyo, Japan), connected to a Windows PC for quantitative analyses using Adobe Photoshop CS5 software.
RNA Isolation and Reverse Transcriptase-PCR
RNA was isolated from the cells and retinal tissue using TRIzol reagent (Invitrogen) according to the manufacturer's instructions. The retinal RNA was extracted from the retina of one eye from each mouse in nine litters. We converted 100 ng of purified total RNA from samples into cDNA by using SuperScript™ III (Invitrogen) and stored all cDNA samples at −80°C. RNA from human bone marrow MSCs (hBM-MSCs; 38-year-old male) and human adipose MSCs (hAdipo-MSCs; 22-year-old female) at passage 5 were kindly donated by Prof. Hyung-Min Chung (CHA University, Korea). The method for hBM-MSC and hAdipo-MSC culture is described in supplementary methods (available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0). Reverse transcriptase-PCR was carried out with 20 μl of AccuPower PCR PreMix (Bioneer). The PCR was performed in a Mygenie96 thermal cycler (Bioneer). Band intensities were quantified with the ImageJ software (NIH, Bethesda, MD, USA).
Quantitative Real-Time PCR
Quantitative real-time PCR was performed using a SYBR-Green reaction kit (Roche, Lewes, UK) according to the manufacturer's instructions in a LightCycler (Roche), and the relative expression levels of a housekeeping gene and the genes for VEGF, TGF-β1, pigment epithelium-derived factor (PEDF), and angiopoietin 1 (ANG-1) were determined. Primer sequences are shown in Supplementary Table 2 (available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0). Analysis of relative gene expression data was calculated using the 2–ΔΔCt method. The values for target gene expression levels were normalized against the expression level of a housekeeping gene. We assigned the expression ratio for the control samples in the data set as the standard level and calculated relative expression levels for all other samples based on this standard level.
ELISA
The dissected, snap-frozen retinas were homogenized on ice in RIPA buffer containing a proteinase inhibitor cocktail (Sigma-Aldrich). Owing to the low amount of total protein in the pups' retinas, retinas from three mice were collected for one sample, and a total of three samples were obtained from each of nine litters in each group. The samples were centrifuged for 10 min at 8,000 × g at 4°C. The TGF-β1 present in the retina samples and culture supernatants were assayed using Human TGF-β1 Quantikine kits (R&D Systems), according to the manufacturer's instructions. The TGF-β1 concentrations were calculated by comparison with standard samples. The TGF-β1 average for each group represents three experiments.
Statistics
Statistical analyses were conducted on a CHA University mainframe computer using the Statistical Analysis System software (SAS; SAS Korea, Inc., Seoul, Korea), version Enterprise 4.0. Data are presented as the means ± SEM. All statistics were calculated using independent t-tests or analysis of variance (ANOVA) with least significant differences (LSD) tests for post hoc analysis. A value of p < 0.05 was considered significant.
Results
Characteristics of AMSCs
Flow cytometric analysis of the immunophenotypic surface profiles of AMSCs showed that they were negative for SSEA4, TRA-1-60, and TRA-1-81 (embryonic stem cell markers) and CD34 (a hematopoietic and endothelial cell marker). They were also positive for CD13 and CD90 (MSC markers), CD105 (a part of TGF-β receptor complex, a MSC marker), and CD9 (a nontrophoblast marker), but low for CD73 (a marker of lymphocyte differentiation, a MSC marker) (Fig. 1A). This expression profile confirmed that AMSCs fit into the criteria generally defined for multipotent MSCs and that they are heterogeneous (32). In order to test their differential potentials, AMSCs were subject to differentiation into three lineages: adipocytes, osteoblasts, and chondrocytes. After they were cultured in respective differentiation media, AMSCs generated all of the three different fates upon their positive staining and the expression for specific cell types (the data are not shown because they are part of results of other manuscripts under review).
The expression levels of antineovascular factors (including those that promote neovessel stabilization) in AMSCs were compared with those of hBM-MSCs and hAdipo-MSCs. All sources of MSCs expressed comparable level of PEDF (pigment epithelium-derived factor), whereas hBM-MSCs and hAdipo-MSCs expressed ANG-1 as half and as double as AMSCs did, respectively (Supplementary Fig. 1; available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0). Expression of TGF-β1, a suppressor of retinal neovascularization, was much stronger in AMSCs compared to hBM-MSCs and hAdipo-MSCs (Fig. 1B).
Response of AMSCs to Pathological Conditions In Vitro
To investigate whether AMSCs maintain their antineovascular properties under pathological conditions in which proinflammatory cytokines are present (7), cytokine expression was analyzed in retinas of mice that underwent the OIR protocol. Multiple inflammatory cytokines were upregulated, particularly tumor necrosis factor-α (TNF-α) and interferon-γ (IFN-γ) (Supplementary Fig. 2; available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0). Based on this result, the effects of IFN-γ and TNF-α on AMSCs were examined in vitro. The mRNA was extracted from AMSCs 24 h after treatment with IFN-γ and TNF-α. The real-time PCR analysis showed that expression of TGF-β1 was remarkably upregulated in the AMSCs exposed to IFN-γ and TNF-α (p < 0.001) (Fig. 2A). Consistent with this finding, increased secretion of TGF-β1 protein was confirmed by ELISA (271.82 ± 6.35 pg/ml vs. 70.45 ± 2.36 pg/ml, p < 0.001) (Fig. 2B). However, the IFN-γ- and TNF-α-stimulated AMSCSs exhibited no significant differences in PEDF mRNA levels and slightly decreased levels of ANG-1 (Fig. 2A). In addition, the expression of hypoxia and/or angiogenesis-associated factors in the pathogenic condition was examined. Among them, the angiogenesis regulators including ANG-2, hypoxia inducible factor-2α (HIF-2α), HGF, C-X-C chemokine receptor type 4 (CXCR4), and VEGF were significantly induced by TNF-α and IFN-γ stimulation (Supplementary Fig. 2; available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0). To explore the effects of increased VEGF expression on the AMSCs in the pathogenic condition, the AMSCs were treated with different concentrations of external VEGF. Although most of the angiogenic factors except for HIF-1α did not respond to a low dose of VEGF in expression, the high dose of VEGF significantly induced ANG-1, ANG-2, HIFs, HGF, CXCR4, and VEGF itself (Supplementary Fig. 3; available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0). However, TGF-β, PEDF, and bFGF were not affected in this experiment. These data suggest that AMSCs respond to environmental changes by enhancing expression and secretion of TGF-β1 and that TGF-β1-mediated VEGF induction may further induce angiogenic factors including HIFs, ANGs, and VEGF.
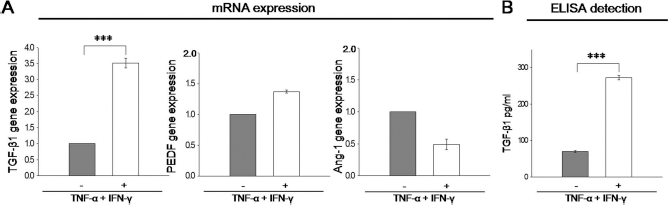
Response of AMSCs to pathological conditions in vitro. (A) In vitro mRNA expression of antineovascular factors was determined by real-time PCR after stimulation with IFN-γ and TNF-α. TGF-β1 levels were significantly increased after stimulation, while the expression levels of other factors were not significantly changed. (B) Protein levels of TGF-β1 in the culture supernatant of AMSCs were determined by ELISA. TGF-β1 levels in the culture supernatant of stimulated AMSCs were significantly increased compared to normal cells. ***p < 0.001 for each comparison. Data were normalized to the level (=1.0) of mRNA in control cells (–) and expressed as the means ± SEM.

TGF-β1 mediated suppression of AMSCs on HUVEC proliferation. (A) HUVECs cocultured with AMSCs exhibited significantly decreased proliferation compared to HUVECs cultured alone. AMSCs pretreated with IFN-γ and TNF-α (AMSC stimulation) showed greater suppressive effects on HUVEC proliferation than normal AMSCs. (B) TGF-β1-siRNA suppressed the expression of TGF-β1 in AMSCs. The RNA was extracted from TGF-β1-siRNA-transfected AMSCs or normal AMSCs and analyzed for mRNA expression of TGF-β1. The expression level of TGF-β1 was significantly decreased in TGF-β1-siRNA-transfected AMSCs, but was not affected by transfection of hAMSCs with scrambled siRNA. (C) TGF-β1-siRNA-mediated suppression was maintained up to 5 days after transfection without alteration in expression of PEDF and ANG-1. (D) Transfection of TGF-β1 siRNA blocked the suppressive effects of IFN-γ- and TNF-α-treated AMSCs, but the suppressive effects of untreated normal AMSCs were not affected by transfection of TGF-β1 siRNA. ***p < 0.001; *p < 0.05 for each comparison. Data are expressed as the means ± SEM.
Suppression Effect of AMSCs on HUVEC Proliferation
To investigate whether TGF-β1 induction by AMSCs plays an important role in their suppression of neovascularization, HUVECs were cocultured with normal or IFN-γ- and TNF-α-treated AMSCs. IFN-γ and TNF-α treatment of AMSCs (AMSC stimulation) was performed 1 day before coculture. Inhibitory effects on proliferation were measured by the BrdU-based proliferation assay, and the results demonstrated the efficiency of AMSC-mediated suppression of HUVEC proliferation. As shown in Figure 3, small but significant suppression of proliferation was observed in HUVECs cocultured with normal AMSCs compared to HUVECs cultured alone. More importantly, IFN-γ- and TNF-α-treated AMSCs suppressed the proliferation of HUVECs to a greater extent than normal AMSCs (Fig. 3A). This suggests that the AMSCs exposed to retinopathy-like conditions suppress the proliferation of HUVECs more efficiently than the AMSCs under normal conditions.
Next, the mechanism of the suppressive effect of AMSCs was examined. To evaluate the role of TGF-β1, TGF-β1 expression was silenced by siRNA transfection. The transfection efficiency of TGF-β1 siRNA in the AMSC culture was confirmed by quantitative realtime PCR, which showed that TGF-β1 siRNA decreased TGF-β1 expression by 80–90% or undetectable compared to normal and scramble controls and that the TGF-β1 silencing in AMSCs was maintained at least up to 5 days without alteration of PEDF and ANG-1 expressions (Fig. 3B,C). When HUVECs were cocultured with the IFN-γ- and TNF-α-stimulated AMSCs that were modified in TGF-β1 expression, the stimulated and TGF-β1 siRNA-treated AMSCs lost the ability to suppress HUVEC proliferation compared to the stimulated AMSCs alone (p = 0.007) (Fig. 3D). It is important to note that TGF-β1 siRNA treatment generated no significant differences in their suppressive capacity in the absence of IFN-γ and TNF-α stimulation (p = 0.77). Taken together, these results indicate that AMSCs play a suppressive role in HUVEC proliferation under retinopathic conditions, likely via the upregulation of TGF-β1 expression.
Cytokine Modulation by AMSCs Under Retinopathic Conditions In Vivo
To evaluate the possible effects of AMSCs in vivo, cytokine expression was assessed in the retinas of mice exposed to OIR after AMSC treatment. First, we confirmed that expression of proinflammatory factors, TNF-α and IFN-γ, was significantly elevated in OIR-induced retina as we expected (Supplementary Fig. 4; available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0). AMSCs were injected into the peritoneal (IP) cavity of each mouse at P12, and retinal tissue was assessed at P17. The expression of antineovascular factors in the retina was then evaluated by real-time PCR. Consistent with the in vitro data, mRNA expression of TGF-β1 was significantly increased in the retinas of AMSC-treated mice (Fig. 4A). Expression of PEDF in the AMSC-treated group was not different from the control group, and no expression of ANG-1 was detected in the retinal tissue. Quantitative analysis of TGF-β1 expression in retinal tissue lysate by ELISA showed that TGF-β1 reached levels of 65.23 ± 11.16 pg/ml in the AMSC-treated group, while the mean level of TGF-β1 in the control group was 27.73 ± 3.57 pg/ml (p < 0.01) (Fig. 4B). Thus, analysis of TGF-β1 mRNA and protein levels indicate that TGF-β1 is secreted by AMSCs in response to retinopathic conditions in vivo.

Effect of AMSCs on the expression of antineovascular factors in vivo. (A) The retinal mRNA expression levels of the indicated antineovascular factors were determined by quantitative real-time PCR. TGF-β1 was significantly increased in the retinas of AMSC-treated mice. (B) Protein levels of TGF-β1 in the mouse retina were determined by ELISA. TGF-β1 levels in the retinas of AMSC-treated mice were significantly increased compared to PBS-treated mice. ***p < 0.001; **p < 0.01 for each comparison. Data were normalized to the level (=1.0) of mRNA in control mice (PBS treated) and expressed as the means ± SEM.
Migration of AMSCs After Intraperitoneal Injection
Before investigating the possible mechanism of AMSC-mediated suppression of retinal neovascularization, we examined the migration of the intraperitoneally administered CM-DiI-labeled AMSCs in the retina. It is important to note that AMSCs were not administered intravenously, to rule out the possibility of circulating AMSCs from retinal vascular leakage. A longitudinal analysis of transplanted AMSCs in retina from P13 (postinjection day 1) to P17 (postinjection day 5) was performed. The migrated AMSCs were detected in the retina from postinjection days 1 to 5 (Fig. 5A). The injected AMSCs were located in retinal neovascularized areas, but did not integrate into the vascular network (Fig. 5B, right panel), suggesting that AMSCs did not differentiate into endothelial cells or pericytes. These results indicated that AMSC-mediated regulation of angiogenesis was conducted through a paracrine mechanism such as the release of cytokine.
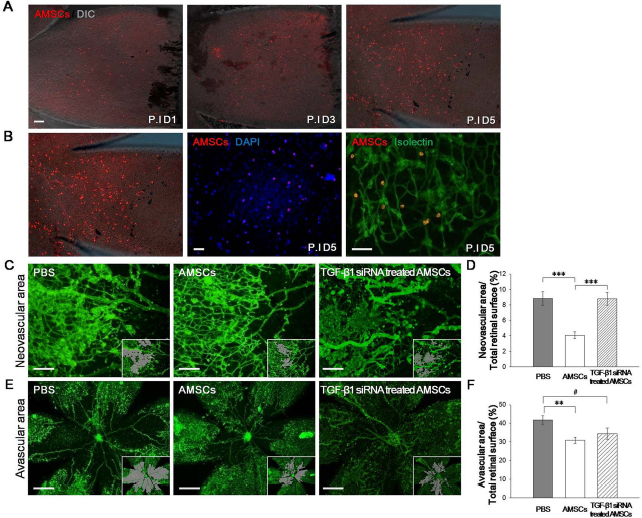
The effect of AMSCs on retinal neovascularization. (A) The longitudinal tracking of AMSCs in the retina. The migrated AMSCs were detected in the retina from 1 day to 5 days after injection. The image is a merge of DIC (gray) and DiI-labeled AMSCs (red). Scale bar: 50 μm. (B) In vivo tracking of the migrated AMSCs were detected in the retina 5 days after injection. The left panel shows a merged image of DIC (gray) and DiI-labeled AMSCs (red). The middle panel shows merged image of DAPI (blue) and DiI-labeled AMSCs (red). The right panel shows a merged image of a GS-lectin-stained vessel (green) and DiI-labeled AMSCs (red). Scale bar: 50 μm. (C-F) The effect of AMSCs on retinal neovascularization. Representative retina flat mounts were prepared at P17. Areas of retinal neovascular tuft formation (scale bar: 100 μm, C) and vaso-obliteration (scale bar: 500 μm, E) were determined by computer-assisted image analyses. Quantified areas are shown in gray (inset). Analyzed data show the percentage of areas of vaso-obliteration (D) and neovascular tuft formation (F) in the retina. Vaso-obliteration and neovascular tuft formation are reduced in the AMSC-treated group compared with the control group. TGF-β1-siRNA-treated AMSCs failed to reduce neovascular tuft formation compared with nontransfected AMSCs, but vaso-obliteration was slightly reduced in the retinas of mice treated with TGF-β1-siRNA-treated AMSCs compared with the control group. ***p < 0.001; **p < 0.01; *p < 0.05; #p < 0.06 for each comparison. Data are expressed as the means ± SEM.
The Effect of AMSCs on Retinal Neovascularization
Next, the potential of AMSCs to reduce pathological angiogenesis was examined in vivo. As shown in Figure 5C, administration of AMSCs dramatically altered the degree of pathological neovascularization induced by OIR compared with the control group. Retinal neovascular tufts, which are abundant in the central and midperipheral retina of mice in the control group, were present only at low levels in the AMSC-treated group (Fig. 5C, left and middle panels). Areas of neovascular tufts are shown in gray (Fig. 5C, inset). Quantitative analysis confirmed a reduction in neovascular tuft area of over 4.73% in the retinas of the AMSC-treated mice compared with the control mice (p < 0.004) (Fig. 5D). As expected, the retinal vasculature in the AMSC-treated group exhibited normal morphology and branching in the midperipheral and peripheral regions, with the exception of sparse avascular areas around the central retina (Fig. 5E). Areas of vascular obliteration are shown in gray (Fig. 5C, inset). Retinas of mice treated with AMSCs exhibited an approximately 10.93% reduction in avascular area compared with the retinas of mice treated with PBS (p < 0.001) (Fig. 5F). These results indicate that AMSCs suppress neovascular tuft formation and increase normal vascularized areas in proliferative retinopathy.
The capacity of TGF-β1 siRNA to nullify the beneficial effect of AMSCs in vivo was also examined. For this experiment, functional analyses were conducted using AMSCs treated with TGF-β1 siRNA (Fig. 5C, E, right panel). As expected, the TGF-β1-siRNA-treated AMSCs failed to reduce neovascular tuft formation compared to nonsilenced AMSCs (p < 0.001) (Fig. 5D); however, normal vascularized areas were marginally increased in the TGF-β1-siRNA-treated group compared with the control group (p < 0.06) (Fig. 5F), although the difference in the quantity of normal vascularized areas between untreated AMSCs and TGF-β1-siRNA-treated AMSCs was not significant (p > 0.27) (Fig. 5D). These results imply that AMSC treatment effectively suppresses retinal neovascularization through the expression of TGF-β1, while its effect on vascular obliteration is irrespective of TGF-β1.
Discussion
Stem cell therapy has the virtue of overcoming the limitations of conventional therapies, and several candidate diseases for stem cell therapy have been suggested. Recently, MSCs have received attention because of their clinical use in cell therapy for tissue regeneration and degenerative diseases. MSCs are adult self-renewing cells that play key roles in organ maintenance and repair. They can be acquired from different human tissues including bone marrow, adipose tissue, and cord blood and differentiate into specific tissues including bone, cartilage, muscle, bone marrow stroma, tendon, ligament, fat, dermis, and other connective tissues (19). Therapeutic effects of various MSCs have been reported (23). However, their applications still have obstacles in both procedures and preparation of cells: most cells transplanted into tissues are not able to survive, and it is difficult to acquire a large enough number of cells for treatments. Adipose derived (adipose)- and bone marrow (BM)-MSCs have limitations, including the requirement of an invasive surgical method to obtain them and decreasing differentiation capacity with increasing age (36,37). In contrast, placenta-derived MSCs have the strengths of high yield and sustained differentiation capacity. Moreover, placenta-derived MSCs have a powerful immunomodulatory function (17). Human placenta, which is essential in fetal development, is another important reservoir of various stem cells. Among the mixture of stem cells, AMSCs are isolated from the extraembryonic mesoderm of discarded placenta and have capabilities for self-renewal and differentiation into three mesodermal lineages: adipocytes, osteocytes, and chondrocytes; and all three germ layers: ectoderm (neural), mesoderm (skeletal muscle, cardiomyocytic, and endothelial), and endoderm (pancreatic) (26). Like other MSCs, AMSCs have immune-modulatory properties that may provide a solution for immune-related complications induced by allogeneic stem cell transplantation (44). Owing to their regenerative and immune-modulatory capacities, the efficacy of AMSCs in cell therapy has been tested in various animal disease models, including Alzheimer's disease (20,32).
In this study, we have isolated AMSCs from the amniotic membrane (fetal part) of the placenta. AMSCs produce various cytokines and growth factors, including factors necessary for self-sustenance through autocrine and/or paracrine mechanisms (4,25). In response to acute injury or inflammatory or ischemic conditions, AMSCs have been shown to contribute to repair by translocating to target areas and releasing multiple trophic factors. Consequently, these secreted factors, such as indoleaminepyrrole 2,3-dioxygenase (IDO), could recover injured retinal sites through creating an adequate environment and preventing immune rejection of injected cells (27,46). Although the paracrine effects of AMSCs have recently been suggested to play a role in many diseases, the underlying mechanism by which they influence neovascularization has not been defined in detail. Thus, we evaluated the effects of AMSCs on neovascularization.
A recent study reported that the MSCs derived from amniotic membrane exhibited strong immunosuppressive properties in inflammatory diseases via the secretion of anti-inflammatory cytokines, including TGF-β1 and prostaglandin E2 (PGE2) (24). In addition, Chang et al. demonstrated that TGF-β1-producing placenta-derived multipotent cells preferentially induced expression of Foxp3 and activation of regulatory T cells (5). On the basis of these results, we hypothesized that TGF-β1, among other paracrine factors, plays an important role in the therapeutic effects of AMSCs on inflammatory conditions. TGF-β1 functions not only as a morphogen but also as a growth factor that affects various biological processes, including immune modulation and vascular responses (1). Present research indicates that TGF-β1 is activated by contact between endothelial cells (EC) and mesenchymal cells and that it mediates a member of actions associated with the maturation of vessels and inhibition of EC proliferation (15,40). Several studies have revealed that TGF-β1 promotes EC proliferation and migration at low concentrations, whereas it exerts inhibitory effects at high concentrations (11,14,39). It has been shown that TGF-β1/ALK5 signaling inhibits EC proliferation and migration (13), and an in vitro study showed that coculture of pericytes and endothelial cells induces secretion of activated TGF-β1, which in turn promotes vascular cell differentiation and suppresses proliferation (8). Based on these functions of TGF-β1, we evaluated the effects of AMSCs on proliferative neovascularization of the ischemic retina.
To clarify the beneficial effects of AMSCs for the treatment of neovascular disorders, the soluble molecules expressed by AMSCs were characterized under normal and pathological conditions. Although recent studies have reported that the expression of immune-modulatory factors secreted by MSCs was dramatically altered in an inflammatory environment (9), none to our knowledge have assessed the angiogenic modulatory functions of MSCs under pathological conditions. Because AMSCs secrete elevated levels of TGF-β1, a well-known immune modulator in angiogenic conditions, we hypothesized that TGF-β1 secreted from AMSCs would modulate a pathological environment such as that in neovascularization. Indeed, we found that the expression of TGF-β1 was significantly increased in the presence of IFN-γ and TNF-α; however, the expression levels of novel antineovascular factors such as PEDF and ANG-1 (28,45) were not significantly affected under pathological conditions (Fig. 2). Although VEGF increased in the pathological condition, the increased level of VEGF did not affect the expression level of TGF-β1 in AMSCs (Supplementary Fig. 4; available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0). These results revealed that AMSCs respond to a pathological environment by increasing secretion of TGF-β1, and support the potential therapeutic use of AMSCs.
Proliferation of endothelial cells is an essential step in neovascularization, and we found AMSC-mediated modulation of EC propagation; the proliferation of HUVECs was significantly suppressed by coculture with AMSCs, and the AMSC-mediated inhibitory effect was enhanced when the AMSCs were stimulated with IFN-γ and TNF-α (Fig. 3). Proliferation of ECs is an essential step in neovascularization. These findings indicate that the suppressive effect of AMSCs on the proliferation of HUVECs becomes enhanced in response to pathological conditions. In addition, this suppression coincides with increased TGF-β1 activity, which has been reported to inhibit endothelial cell proliferation and induce apoptosis in a dose-dependent manner (33).
In this study, we found that TGF-β1 expression was increased in AMSCs at both the protein and mRNA levels in the pathological condition. The expression of TGF-β1 protein in IFN-γ- and TNF-α-exposed AMSCs was higher than in normal AMSCs, and suppression of HUVEC proliferation by IFN-γ- and TNF-α-exposed AMSCs was efficiently blocked by transfection of AMSCs with TGF-β1 siRNA. These results suggest that TGF-β1 is crucial for the antineovascular function of AMSCs; however, TGF-β1 siRNA cannot completely block the suppressive activity of AMSCs. As shown in Figure 3C, the expression levels of the antineovascular factors PEDF and ANG-1 were unchanged after TGF-β1 siRNA transfection. PEDF is well known to be a natural inhibitor of angiogenesis. It has been demonstrated that ischemia-induced retinal neovascularization can be dramatically inhibited by recombinant PEDF (45). ANG-1 plays a role in angiogenesis by promoting vessel maturation and stabilization and thus inhibiting neovascularization (28). The persistent expression of these factors could explain the suppressive activity of AMSCs even in the absence of TGF-β1 expression. Another possibility is that AMSCs are able to suppress angiogenesis through additional TGF-β1/VEGF-independent pathways such as HIF-1α and bFGF (Supplementary Figs. 2 and 3; available at https://www.dropbox.com/s/xoitca6m7aogocq/Kim%20et%20al._RP_Supplementary_V7.pdf?dl=0)
Our in vivo study with an OIR mouse model showed markedly increased expression of both TGF-β1 mRNA and TGF-β1 protein in the retinas of the AMSC-treated mice, which is consistent with our in vitro findings. These results indicate that TGF-β1 is secreted from AMSCs after in vivo treatment. When we evaluated the suppressive effects of AMSCs on neovascularization in OIR mice, we found that intraperitoneal injection of AMSCs led to a dramatic reduction in the formation of retinal neovascular tufts, and the antineovascular effect of AMSCs was blocked by treatment with TGF-β1 siRNA. However, normal vascularized areas were preserved in the group treated with AMSCs transfected with TGF-β1 siRNA, which may be due to the expression of other antineovascular factors, such as PEDF and ANG-1. Importantly, the occurrence of pathological neovascular tufts, a leading cause of blindness, was significantly increased in mice treated with AMSCs transfected with TGF-β1 siRNA. These results suggest that AMSCs increase TGF-β1 expression in response to retinopathic stimuli and play an antineovascular role that is mediated by TGF-β1 secretion.
Interestingly, subsequent analysis showed that a number of AMSCs existed in the ischemic retinas of the AMSC-treated group. The migratory and homing abilities of MSCs are a primary focus of stem cell trials. However, the blood–brain barrier of the central nervous system severely restricts the travel of immune cells and molecules into the brain. Although some studies have demonstrated the migration of systemically injected MSCs into the CNS, only a miniscule amount of MSCs was detected (10). Therefore, when investigating the specific effect of AMSCs on retinopathy, it is important to confirm the migration of AMSCs into CNS tissue across the blood–brain barrier (BBB). In this study, AMSCs were injected into the peritoneal cavity. In agreement with other studies, AMSCs were detected in multiple organs (e.g., lung, liver and spleen; data not shown). Among these tissues, AMSCs were scattered throughout the retina as solitary cells (Fig. 5A,B). Although disruption of the blood–retinal barrier during OIR induction caused leakage of blood components with subsequent hemorrhage into the retina, the presence of intraperitoneally injected AMSCs in the retina, especially in neovascular and avascular areas, illustrates their capacity to infiltrate peritoneal blood vessels and migrate to the injured retina.
Furthermore, we assessed the incorporation of AMSCs into the vascular network to examine the putative paracrine effects of AMSCs. Several studies, using local administration of MSCs, have reported that the therapeutic effects of AMSCs were based on direct differentiation and replacement (16). In our study, however, AMSCs migrated into the retina and repaired retinal angiogenesis without incorporating into blood vessels (Fig. 5B). Furthermore, AMSCs maintained a round shape without attaching to the vessels. These results suggest that cytokines and growth factors secreted from AMSCs regulate angiogenesis.
In summary, AMSCs exhibited strong expression of TGF-β1 in response to retinopathic conditions in vitro. Moreover, AMSCs exerted a strong suppressive effect on HUVEC proliferation only under pathological conditions in vitro. Consistent with this in vitro data, AMSCs in the retinas of mice exposed to OIR exhibited markedly increased levels of TGF-β1 and significantly reduced neovascular tuft formation. Interestingly, migrated AMSCs were clearly detected in the retina, separate from the blood vessels. These results further support the hypothesis that, in the OIR model, intraperitoneally injected AMSCs migrated to the injured tissue and reduced excessive neovascularization through the release of TGF-β1. Although further studies are needed to confirm the effect of AMSCs on neovascular disease, our results help to elucidate the effect of AMSCs in the treatment of retinal neovascular diseases.
Footnotes
Acknowledgments
This study was supported by The Korea Institute for the Advancement of Technology (KIAT, 16-006) and KOSEF (2015R1C1A2A01055144) of South Korea. The authors declare no conflicts of interest.
