Abstract
The epidermis is a stratified epithelium with a stem cell subpopulation in the basal layer that constantly replicates and periodically detaches from the base, undergoing a differentiation process that involves various developmental signals and regulatory pathways. During the last 10 years, a number of studies tried to elucidate the intricate scenario that maintains the epithelial shield during the entire life span. In our study, we investigated the role of Numb in the skin compartment and, in particular, its involvement in stem cell maintenance. Numb expression in the skin compartment was assessed by immunofluorescence and immunohistochemistry analysis. We evaluated Numb expression in primary epithelial cells at various differentiative stages. Moreover, we overexpressed Numb in the isolated population enriched for undifferentiated progenitors to establish its involvement in in vitro differentiation. We demonstrated that Numb in high-proliferating epithelial undifferentiated progenitors contributes to the maintenance of an undifferentiated state. This regulation involves the E3 ligases Itch binding. Moreover, the analysis of a cohort of cutaneous carcinomas showed that Numb is highly expressed in squamous cell carcinoma (SCC), where we observed a direct correlation between the expression of Numb and Ki-67. Our data indicate for the first time that Numb is involved in the maintenance of the undifferentiated proliferating stem cell pool in the epithelial basal layer and its expression could become a new marker in skin cancer.
Introduction
The epidermis is formed by several layers of cells, and it is maintained by a mitotically active basal stratum enriched in stem cells. During their life span, epithelial stem cells, in the basal layer, undergo symmetric and asymmetric division cycles, generating daughter stem cells and a dividing progenitor population, known as transiently amplifying (TA) cells. Epithelial stem cells were originally identified and isolated on the basis of their adhesive characteristics (26). In particular, the basal keratinocytes that rapidly adhere (ADH+) to collagen-treated dishes are enriched in stem cells, as indicated by their colony-forming efficiency (CFE), and have a high expression of β1 integrin, which allows a rapid attachment to collagen IV-treated plates. In contrast, basal cells, expressing a lower amount of β1 integrin, slowly adhere (ADH-) to the collagen IV and are committed to terminal differentiation after few divisions. β1 integrin is a functional marker for epithelial stem cells (8), which exploits its downstream signaling for their maintenance (32). Skin homeostasis is maintained through a complex interplay among developmental signals and other cell regulatory pathways. Expression of the transmembrane Notch ligand Δ, in the basal layer, induces commitment of neighboring epidermal stem cells through the activation of Notch, which induces β1 integrin downregulation (18) and differentiation into transit amplifying cells (12). While Notch signaling induces keratinocyte differentiation, p63 (a p53 homolog) protein is required for stem cell self-renewal in various epithelial tissues (28). p63 is essential for epithelial stratification and ectodermal development, as indicated by the rapid senescence observed after loss of p63 in conditional knockout or in heterozygosis (9). Mice lacking p63 die soon after birth, with severe limb, craniofacial, and skin defects (15,29).
There is a dual effect of p63 on the Notch pathway in keratinocytes. In mice with p63 null skin, the expression of Notch and its ligand jagged1 is downmodulated (10), while p63 overexpression in primary keratinocytes inhibits the expression of HesI, a Notch transcriptional target (16,18). Both p63 and Notch are ubiquitinated by the E3 ligase Itch and subsequently degraded at the proteasomal level (24).
The relevance of Notch signaling is well known in different cell compartments, such as the neural system, where its activity is inhibited by Numb (4). Numb influences cell fate by inhibiting the action of Notch through a not totally elucidated mechanism (6). Numb was originally described in Drosophila as a mutation that affects binary divisions in the sensory organ precursor (SOP) lineage, in which Numb is a docking protein containing a PTB (phosphotyrosine binding) domain at the N-terminus and a PRR (proline-rich region) at the C-terminal (25). The Numb mammalian homolog has been shown to be functionally related to its Drosophila counterpart. In the mouse embryo, Numb is expressed in all layers of the developing cortical plate, including the progenitor cells of the ventricular and subventricular zones. In these cells, Numb is asymmetrically localized during cell division, suggesting that it may have a role in cell fate decisions in the mammalian nervous system (30,31). Numb has been shown to influence differentiation and proliferation in different mouse neural systems (25). Numb is widely expressed in mouse embryos and in most adult tissues. Therefore, it is likely that Numb function is not restricted to neurogenesis. In our study, we evaluated the role of Numb in normal skin homeostasis and its involvement in the maintenance of keratinocytes in an undifferentiated state. We observed that Numb expression is lost during differentiation and that its exogenous expression promotes the maintenance of epithelial stem cell-specific markers. Such activity may explain why we observed increased levels of Numb in cutaneous carcinomas characterized by high proliferation rates. Our findings are supported by an observation in a recent report (21) in which Numb expression is altered in human squamous cell carcinoma (SCC).
Materials and Methods
Cell Culture, Transfection, and Infection
The isolation, culturing, and differentiation of primary keratinocytes from adult healthy skin were performed on fresh skin biopsies. The skin biopsies were obtained as part of routine surgery intervention in the surgery division at the IOM, in accordance with the institutional guidelines and after informed consent.
Skin from different origins (mostly abdominal and breast) from patients (age 30-70) was incubated overnight at 4°C in phosphate-buffered saline (PBS) w/o CaMg (Euroclone, Milan, Italy) containing antibiotics (penicillin, streptomycin, and gentamicin; Euroclone), and then transferred to a sterile Petri dish (Corning, Lowell, MA, USA) with dispase (Gibco Life Technologies, Monza, Italy) overnight at 4°C. Specimens were then washed using PBS without Ca2+ and Mg2+ and dissected horizontally to separate the upper layer containing keratinocytes from the dermal surface. The epidermal layer was subsequently incubated in Accutase (Stem Cell Technologies, Vancouver, Canada) at 37°C for 2 h. Cells were then centrifuged for 5 min at 600 × g. The supernatant was removed, and undifferentiated keratinocytes were resuspended in Epilife (Cascade Biologics, Portland, OR, USA)
Separation of subpopulations enriched in skin progenitor and stem cells was obtained by APC-conjugated anti-β1 integrin staining (#559883, 1:10; BD Pharmingen, San Diego, CA, USA) and flow cytometry sorting of weakly positive (dim) and strongly positive (bright) cells using the FacsAriaII cell sorter (BD Biosciences, San Diego, CA, USA). The selected population was maintained in Epilife medium (Cascade Biologics). Alternatively, the isolation of stem cells was carried out with the adhesion method (8). Unfractionated keratinocytes derived directly from the Accutase treatment (as described above) were plated onto type IV collagen (100 μg/ml; Sigma-Aldrich, St. Louis, MO, USA)-treated dishes. The ADH+ stem cells adhered in 20 min and were then washed to remove the ADH- cells, which were replated in another dish. For differentiation experiments, ADH+ cells on collagen-treated dishes were cultured in a medium containing 1.2 mM CaCl2 (Sigma-Aldrich) for 1 week.
Transduction of keratinocytes was done with lentiviral vectors, using pRRLsin.cPPT.hCMV.GFP.Wpre and pRRLsin.cPPT.hPGK.GFP.Wpre, new variants of previously described third-generation lentiviral vectors (5,23). To simultaneously transduce both reporter and target genes, a new lentiviral vector, Tween, was generated by engineering pRRLsin.cPPThCMV.GFP.Wpre. In this vector, the hCMV.GFP cassette was substituted with the hCMV.hPGK.GFP. A multiple cloning site was inserted downstream of hCMV, and hNumb cDNA (from Elisa Santolini-IEO-Milan, Italy) was subcloned in the XhoI site of the Tween vector. In the same way, a combination of siRNA lentiviral vectors (Thermo Fisher, Rockford, IL, USA) was used to downmodulate Numb expression in an inducible manner. The gene (RGFP or shRNA) expression was driven by tetracycline-regulated UbC promoters, and doxycycline (Sigma-Aldrich) was added to the culture medium 4 days postinfection, and was maintained at 1 μg/ml until the cells were harvested.
RT-PCR
Total RNA was extracted from cells using RNAeasy (Qiagen, Germantown, MD, USA) and 1 μg of RNA was reverse transcribed with SuperScript II (Invitrogen, Carlsbad, CA, USA). Semiquantitative PCR was performed with TaqMan validated assays (Applied Bio systems, Waltham, MA, USA) for Numb ISO1-2 (Hs01112158_m1) and Numb 3-4 isoforms (Hs00377773_m1). GAPDH (Hs99999905_m1) was chosen as reference and all analyses were carried out in triplicate. The relative intensity was compared to untreated ADH+ cell expression. The real-time data was collected using Microsoft Excel and was analyzed on DCts with the formula: expression level = 2-ΔΔCt.
Immunofluorescence and Immunhistochemistry
Cell staining was performed on cells seeded on collagen-coated multichamber slides (Nunclone; Sigma-Aldrich). Cells were washed in PIPES buffer (80 mM PIPES, pH 6.8, 5 mM EGTA, and 2 mM MgCl2; Sigma-Aldrich) and fixed with 4% paraformaldehyde/PIPES (Sigma-Aldrich) for 10 min. This step was followed by block permeabilization in PBS containing 0.2% BSA (Sigma-Aldrich) and 0.1% Triton X-100 (Sigma-Aldrich) for 10 min, followed by exposure to PBS containing 1% BSA for 1 h in order to reduce nonspecific staining. The cells were then incubated with a specific primary antibody at room temperature for 1 h. After three washes in PBS, the cells were incubated with FITC-conjugated donkey anti-mouse immunoglobulins (Jackson ImmunoResearch, Suffolk, UK) or donkey Cy3-conjugated anti-goat immunoglobulins (Jackson ImmunoResearch). Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI) (Sigma-Aldrich).
Immunohistochemical staining was done on 2-μm paraffin-embedded sections. After deparaffination–hydration, sections were permeabilized with PBS containing 0.4% Triton X-100 for 30 min and blocked with PBS containing 5% BSA for 30 min. The samples were incubated overnight at 4°C, with primary antibodies. The following day, slides were first incubated for 1 h with a fluorescence-conjugated secondary antibody (Jackson ImmunoResearch), the nuclei were stained with DAPI and the slides mounted with Mowiol (Sigma-Aldrich). Alternatively, the staining was detected using a 3,3′ diaminobenzidine (DAB)-based substrate (Sigma-Aldrich).
The primary antibodies were used at the concentration indicated from the suppliers: mouse anti-human p63 pan antibody (sc-8431, 1:100; Santa Cruz Biotechnology, Santa Cruz, CA, USA), goat anti-human Numb (sc-15590, 1:50; Santa Cruz Biotechnology), rabbit anti-human Numb (ab-14140, 1:200; Abcam, Cambridge, UK), goat anti-human Notch1 (sc-6014, 1:60; Santa Cruz Biotechnology), mouse anti-human cytokeratin 5/8 (sc-8021, 1:200; Santa Cruz Biotechnology), rabbit anti-human Ki-67 (RU, Ventana, Tucson, AZ, USA), mouse anti-human cytokeratin 5/6 (RU, Ventana), and mouse anti-Pan Keratin (RU, Ventana). When possible, immunohistochemistry was perfor med on Ventana benchmark autostainer. All microscopic images were acquired with a Leica system DM 4000B microscope (Leica Microsystems, Wetzlar, Germany), DFC 420C Camera, and Leica Application Suite V3.0.0 software, or with a Leica confocal station (Leica SP5 confocal system, mounted on a Leica DM6000 inverted microscope, equipped with an Argon-ion laser and PMT detectors.)
Ki-67 expression was scored on a scale of 0 to 4 based on the percentage of positive cells: 0 (occasional positive cells); 1 (<5% positive cells); 2 (5-20% positive cells); 3 (21-50% positive cells); 4 (>50% positive cells).
Immunoprecipitation In Vitro Binding and Immunoblotting
Cells were lysed with a buffer containing 1% Triton X-l00, 50 mM HEPES (pH 7.5), 150 mM NaC1, 10% glycerol, 1.5 mM MgCl2, 5 mM EGTA, protease inhibitors (4 mM phenyl methylsulfonylfluoride and 100 mg/ml aprotinin) (Sigma-Aldrich), and phosphatase inhibitors (10 mM sodium orthovanadate and 20 mM sodium pyrophosphate; Sigma-Aldrich) and processed. For direct immunoblot analysis, we employed 50-100 μg of total cellular proteins and 3-5 mg of total cellular proteins for immunoprecipitation (7). For immunoprecipitation, cell lysates, mock-transfected (control), or transfected with Numb FL or PTB, were incubated with 10 μl of anti-p63 antibody (sc-25040; Santa Cruz Biotechnology) or anti-Numb antibody (ab-14140; Abcam) or anti-HA (MMS-101P; Covance, Princeton, NJ, USA) and 50 μl of protein G-Sepharose beads (Sigma-Aldrich) for 1-2 h; the unbound proteins were washed six times with lysis buffer, slurry beads were resuspended with 25 μl of loading buffer, boiled for 5 min and loaded on SDS-PAGE for Western blot (WB). The antibodies for WB were used at the condition suggested from the suppliers: mouse anti-p63 (sc-8431, 1:500) or goat anti-p63 (sc-25040, 1:200), rabbit anti-p63 (sc-8343, 1:500), goat anti-human Numb (sc-15590, 1:500), and mouse anti-actin (sc-32251, 1:1,000) (Santa Cruz Biotechnology), rabbit anti-human Numb (ab-14140, 1:1,000; Abcam), mouse anti-human Itch (#611198, 1:1,000; BD Bioscences), and mouse anti-HA (MMS-101P, 1:1,000; Covance).
Results
Numb Is Expressed in the Basal Layer
To evaluate Numb expression in normal healthy skin, we performed standard immunohistochemistry (IHC) on paraffin-embedded samples. As first analysis, we evaluated, in serial sections, the presence of Numb in relation to the proliferation marker Ki-67. DAB staining revealed a stronger signal in the same high proliferative layer (Fig. 1A). Then we performed immunofluorescence (IF) staining to establish the correlation with other epithelial markers. The samples were stained for cytokeratin 5/8 and Notch, which are respectively expressed in the basal layer and in the more differentiated progenitors and precursors. The antibody against Numb revealed an intense staining in the basal layer that disappeared in the spinous layer. This expression was comparable to the p63 distribution in the basal layer (Fig. 1B). The various keratinocyte populations recognized by Numb and Notch reflect a possible difference in the activity of the two proteins. While Notch is known to promote epidermal cell proliferation and it is expressed in layers of more differentiated cells (12), Numb seems to be a marker of the basal layer, and maybe it is involved in the maintenance of a more undifferentiated state.
Numb expression in normal epidermis. Paraffin-embedded sections from normal skin biopsies were stained by immunohistochemistry (A, Numb and Ki-67) and immunofluorescence (B, cytokeratin 5/8, Notch, Numb, p63, and DAPI). One representative of five independent experiments with different samples is shown (scale bars: 50 μM).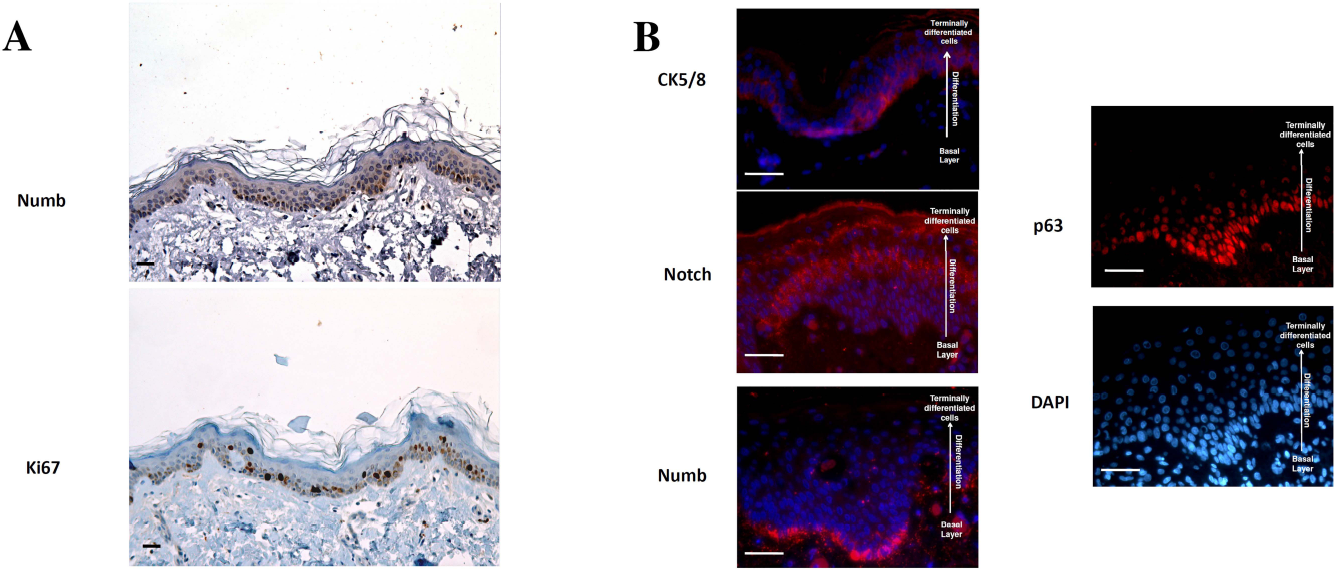
Numb Is Expressed in Undifferentiated Keratinocytes
In order to assess the presence of Numb in normal epidermis, we analyzed its expression in differentiated and undifferentiated keratinocyte subpopulations. Primary human keratinocytes were separated by substrate adhesion to obtain ADH+ and ADH- cells. The two cell populations were analyzed by flow cytometry to evaluate the relative percentage of b1 integrin bright (ADH+) versus b1 integrin dim (ADH-) cells. As expected, this analysis showed that the ADH+ cells had a higher level of b1 integrin expression (Fig. 2A). Western blot analysis indicated that p63 was expressed only in the ADH+ population. Moreover, the presence of cytokeratin 5/8 and the lower level of cytokeratin 10 evidenced the enrichment of this fraction for the undifferentiated cell population (19) (Fig. 2B). Once the two different populations of more primitive ADH+ and differentiated ADH- cells were validated, we investigated the relative expression of Numb. Western blot analysis showed that Numb was expressed at high levels in ADH+ cells, while being barely detectable in ADH- cells (Fig. 2B). Likewise, immunofluorescence staining showed that Numb and p63 were essentially coexpressed in the same cells (Fig. 2C), indicating that Numb expression is higher in the fraction enriched for undifferentiated keratinocyte progenitors.
Evaluation of Numb expression in ADH+ and ADH- keratinocytic cells. Keratinocytic cells separated by adhesion were analyzed for β1 integrin expression with an anti-β1 APC antibody by FACS (A). The corresponding cell lysates were analyzed with Western blotting with anti-Numb, p63, cytokeratin 10, cytokeratin 5/8, and actin antibodies (B). Immunofluorescence analysis on primary undifferentiated and differentiated keratinocytes. Unsorted and ADH+ primary keratinocytes were stained for DAPI (blue), Numb (red), and p63 (green). One representative of three independent experiments with cells from different donors is shown (scale bars: 20 mM) (C).
Numb Expression Is Lost During Keratinocyte Differentiation
To confirm that Numb expression is differentiation dependent, we induced in vitro keratinocyte differentiation by exposing ADH+ cells to 1.2 mM Ca2+ for 7 days (1). This procedure promotes considerable morphological modifications in differentiating ADH+ cells, with stratification and change from polygonal to a more asymmetric shape (Fig. 3A). The differentiated cells were analyzed by immunoblot for p63 and cytokeratin 5/8 expression. Ca2+ treatment resulted in a clear drop of Numb protein expression that correlated with the decrease in p63 levels (Fig. 3B), confirming that Numb expression was restricted to undifferentiated primitive cells. The analysis of Numb expression was also confirmed by TaqMan Real Time PCR, where it is possible to assess a marked reduction of Numb expression after Ca2+-induced differentiation (Fig. 3C).
Numb expression in undifferentiated and differentiated keratinocytic cells. (A) ADH+ cells were plated in collagen-treated dishes (top) and induced to differentiate for 7 days with Ca2+ (bottom) (scale bars: 50 μM). (B) Cells cultivated as in (A) were lysated and analyzed with Western blotting with anti-Numb, -p63, -cytokeratin 5/8, and -b-actin antibodies. One representative of three independent experiments is shown. (C) RT-PCR analysis of ADH+ and ADH+ Ca2+-treated cells. Numb mRNA expression is higher in ADH+ and is downregulated in Ca2+-treated cells. One representative of three independent experiments is shown.
Numb Overexpression Contributes to the Maintenance of an Undifferentiated Phenotype
To determine whether the presence of Numb in the basal layer of the epidermis is related to its possible role in the maintenance of an undifferentiated status, we cloned the Numb gene in a lentiviral vector to transduce freshly purified ADH+ cells. The percentage of cells transduced with the lentiviral vector was close to 100% after infection and in subsequent passages (data not shown). Infected cells were expanded in collagen-treated dishes in serum-free medium for a period of 10-30 days. After expansion, the cells were used in a differentiation assay to evaluate the differentiation potential in the presence or absence of exogenous Numb expression.
ADH+ cells (Numb transduced and control vector transduced) were induced to differentiate through 7 days of Ca2+ treatment and analyzed by Western blot to evaluate various stemness/differentiative markers. This analysis revealed that Numb overexpression is accompanied by the persistence of high levels of p63 and maintenance of a low level of cytokeratin 10, while control vector and untransduced ADH+ cells, incubated with Ca2+, drastically decreased the expression of p63 and increased cytokeratin 10 levels (Fig. 4A). These expressions are correlated, with the absence, in Numb-transduced cells, of major morphological change that we observed in control cells after differentiation (Fig. 4B), suggesting that Numb overexpression may prevent the Ca2+-induced differentiative cascade.
Evaluation of Numb overexpression in epithelial cells. (A) ADH+ keratinocytes infected with control empty vector and Numb-expressing vector were analyzed with Western blotting for p63, cytokeratin 10, and β-actin antibodies after 7 days of differentiation with Ca2+. (B) Morphological analysis of keratinocytes transduced as in (A) before and after 7 days of Ca2+-induced differentiation. One representative of three different experiments is shown (scale bars: 100 μM). (C) Effect of Numb overexpression on p63-Itch interaction. In the upper panel p63 Ab was used in a coimmunoprecipitation assay of HaCaT cells transduced with empty vector (Control), Numb full length (Numb FL), or the Numb phosphotyrosine binding (Numb PTB) domain. In the lower panel, the reverse assay was performed immunoprecipitating with anti-Numb (rabbit; Abcam) in cells transduced with empty vector (Control), with anti-HA in cells transduced with Numb full length (Numb FL-HA), or the Numb phosphotyrosine-binding (Numb PTB-HA) domain. It is possible to observe that Numb-PTB interferes with p63 endogenous Numb coimmunoprecipitation (upper panel, p63 IP) and immunoprecipitates endogenous p63 (lower panel, HA IP). The input is shown in the lower part. (D) Effect of Numb downmodulation on p63 expression. siRNA Dox inducible vector was used on HaCaT cells. It is possible to observe a slight reduction in p63 expression concomitant with Numb downmodulation. This reduction is abolished by MG132 (5 μM) treatment in a Numb-independent way. (E) Densitometric analysis of p63 expression on HaCaT cells with (3-4) or without Numb (1-2) downmodulation in the absence (1-3) or presence (2-4) of MG132 treatment. One representative of three independent experiments is shown.
Numb Influences Itch/p63 Binding
It is known that Numb expression regulates Notch proteosomal degradation through the E3 ligase Itch (13). Moreover, Itch is responsible for p63 degradation (24). For this reason, we evaluated the influence of Numb in Itch/p63 binding. Using an anti-p63 antibody in a coimmunoprecipitation assay, we found that Numb overexpression increases Itch/p63 binding (Fig. 4C). We also observed by coimmunoprecipitation that Numb PTB is responsible for the Itch/p63/Numb binding, where PTB alone is sufficient to coimmunoprecipitate Itch/p63 complex in HaCaT cells, and its overexpression inhibits the endogenous Numb precipitation by p63 (Fig. 4C).
Previous studies (3,20) have shown that Numb/MDM2 binding influences p53 stability. We hypothesized a scenario in which Numb/Itch binding influences p63 stability in the skin through a similar mechanism. To confirm Numb's effect on p63 stability, we downregulated Numb by SiRNA in HACaT cells, evaluating p63 expression. We found that Numb downregulation induces a slight p63 reduction, which is completely abolished by MG132 treatment (Fig. 4D, E).
Numb Expression in Skin Tumors Is Correlated with Proliferative Rate
Since Numb may support cell expansion in the basal layer by influencing the p63 stability, we investigated the Numb expression in skin tumors by immunohistochemistry. We verified that present is a diffuse Numb expression in all tumors analyzed (Fig. 5A). Moreover, we evaluated the correlation of Numb expression with the tumor proliferative rate. We analyzed 15 cutaneous squamous cell carcinomas (SCC). In all cases, we observed that Numb is expressed in these tumors with a strict correlation between Numb and Ki-67, where Numb expression is very strong in 11 high-proliferating SCC cases (>50% of cells positive for Ki-67) (Fig. 5A). In four SCC cases, where Ki-67 staining was evaluated as intermediate (21-50% of cells positive for Ki-67), the expression of Numb was rather weak (Fig. 5B), thus suggesting a role for Numb in the aggressive behavior of skin tumors.
Numb expression in cutaneous neoplastic transformation. In neoplastic epithelia, it is shown that the stain is specific for the replicating basal layer and correlates with p63, Ki-67, where cytokeratin 5/6 stain all populations (A). Squamous cell carcinoma (SCC) immunohistochemical analysis of Numb in tumors with high (B) and intermediate (C) levels of Ki-67. Magnification: 20× (left) 10× (right) are shown. Each section staining was performed in duplicate (scale bars: 50 μM).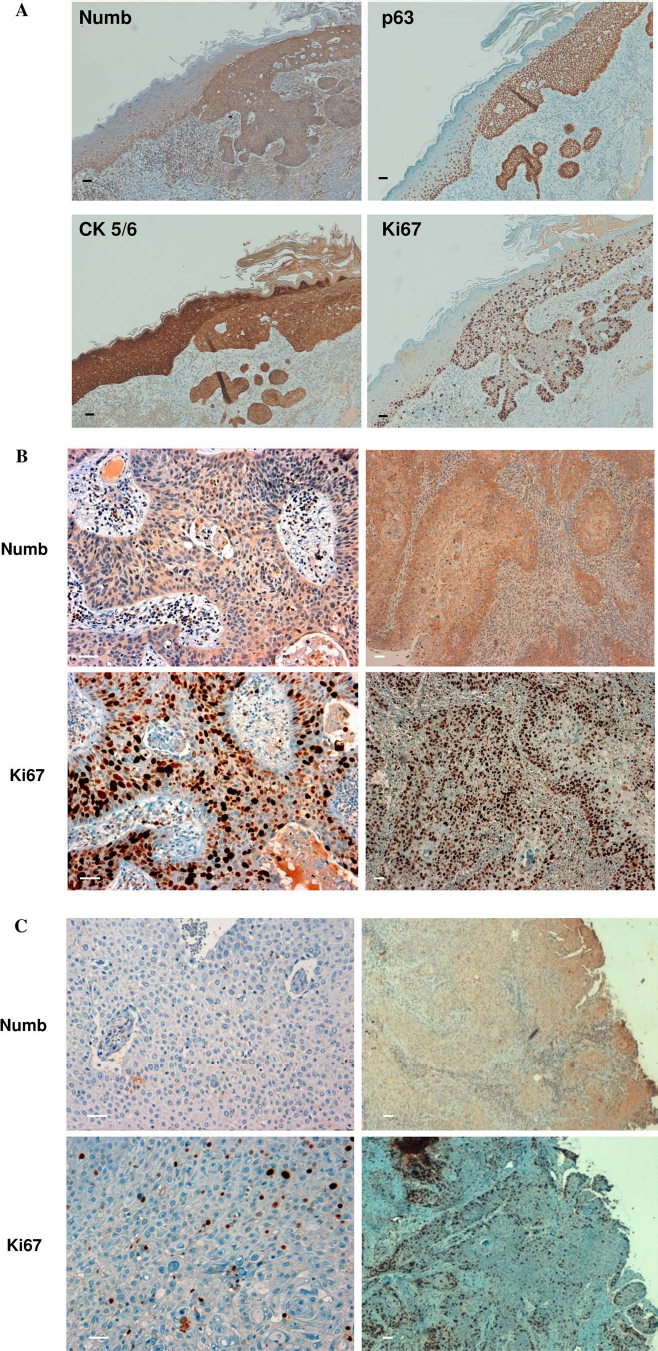
Discussion
In this study, we found that Numb is expressed in the epidermal basal layer and that its expression contributes to preserve the undifferentiated phenotype in epithelial progenitors cells. Numb is known for its ability to antagonize p53 proteosomal degradation through MDM2 binding, inhibiting its ubiquitin ligase activity (3). p63, a member of p53 family, is crucial for skin stratification and for basal layer maintenance of an undifferentiated state (9,15,28). In keratinocytic cells p63 is downmodulated through Itch-mediated ubiquitination (14). Rossi and coworkers showed that exogenous Itch mediates ubiquitination/degradation of the different p63 isoforms transduced in HEK293 cells (24). Our data indicate that Numb contributes to maintain high p63 expression in epidermal stem cells, preventing the differentiation process, and it is able to bind Itch in an in vitro binding assay (Fig. 4D). Moreover this binding prevents p63 downmodulation in a MG132-dependent way, indicating the involvement of the proteosomal apparatus. Numb effect on p63, in skin compartment, inhibits the differentiation program not only in normal but also in neoplastic tissues (19). The maintenance of p63 by Numb expression results in antagonizing Notch activity (16,18). Moreover, it can be supposed that Numb can antagonize Notch in this compartment as already demonstrated by McGill and McGlade, in other cellular backgrounds, where Numb promotes Notch proteosomal-mediated degradation through a mechanism that requires the E3 ligase Itch (13). In the epidermis, Notch signaling induces growth arrest and terminal differentiation of keratinocytic cells (22). Such inhibitory activity makes Notch a tumor suppressor in the epidermis. It is worth noting that tissue-specific ablation of Notch in mouse epidermis results in hyperplastic growth followed by development of basal cell carcinomas and enhances chemically induced skin carcinogenesis (17). Notch expression is reduced in a panel of skin and oral SCC cell lines (11). In a recent report, Pickering and colleagues (21) found in oral squamous cell carcinoma (OSCC) an amplification of numb among the alterations in Notch pathway members that accounted for 66% (23/35) of the analyzed tumors, indicating a role for Notch as an oncosuppressor in these tumors. Likewise, Numb acts as a tumor suppressor in breast cancer (3,20), while being upregulated in all grades of astrocytomas (27), in which LNX, an E3 ligase responsible for Numb degradation through proteosomal pathway, is downmodulated (2). The strict correlation between Numb and Ki-67 expression in SCC can be considered a further confirmation of the role played by Numb in the epidermal compartment. This mechanism may be particularly relevant in aggressive skin cancer, in which undifferentiated keratinocytic cells are unable to undergo terminal differentiation. In conclusion, our data suggest that Numb promotes self-renewal and proliferation in basal layer undifferentiated cells while preventing the differentiation cascade. Moreover, the role of Numb in the epidermis does not seem limited to physiological conditions, but also in skin cancer, where Numb may play a significant oncogenic role, indicating that this gene may act as oncogene or oncosuppressor, depending on the cellular background.
Footnotes
Acknowledgments
Numb Tween was a gift from Elisa Santolini, whom we all remember. This work was supported by a grant from the European Union (Epistem, Sixth Framework Program, LSHB-CT-2005-019067) and from RiMED Foundation. We thank Gianni Cesareni for his suggestions and helpful comments, IOM surgeons for samples (Marco Vieira, Mario Lipera, and Antonio Iuppa), Eleonora Aiello, Sandro Barbuscia, Maria Rita Pulvirenti, and Ismett pathology unit for IHC technical support, and Emer Tannam and Warren Blumberg for help in editing the manuscript. The authors declare no conflicts of interest.
