Abstract
The human amniotic membrane is a highly abundant and readily available tissue that may be useful for regenerative medicine and cell therapy. The amniotic membrane stem cells can differentiate into multiple cell lineages; they have low immunogenicity and anti-inflammatory functions. This research aims to examine the protocols for the isolation of human amniotic membrane stem cells, including their phenotypic characterization and in vitro potential for differentiation toward keratinocytes. Human placentas were obtained from selected cesarean-sectioned births. We isolated amniotic stem cells by trypsin and collagenase B digestion and centrifuged with Percoll. After monolayer expansion of adherent cells, the cells were characterized by immunocytology with octamer-binding transcription factor 4 and differentiated into keratinocytes by treating the cells with insulin, hydrocortisone, BMP-4, and vitamin C. Protocol for isolation of stem cells from amniotic membrane has high efficiency. Differentiation markers of stem cells into keratinocytes, such as vimentin, cytokeratin (CK) 14, and CK19, were determined by reverse transcription-polymerase chain reaction increase over time in culture. Stem cells isolated from the amniotic membrane can differentiate into keratinocytes. It has opened the prospect of using stem cells to regenerate skin and clinical applications.
Introduction
Stem cells are undifferentiated cells and have the potential growth of becoming specialized cells and having new functions. They are classified as pluripotent, multipotent, and unipotent as demonstrated through their potential to generate a variety of cell lineages. While pluripotent stem cells may give rise to all types of cells in an organism, multipotent and unipotent stem cells remain restricted to the tissue or lineages. Along with the evaluation of various molecular markers, using several functional assays can define the potency of these stem cells.
Because stem cells can differentiate into different functional cells, it has opened the prospect of using stem cells to regenerate tissues, research, drug development, and clinical applications 1 –5 . Stem cells have great potential to become important aspects of medicine. Regenerative medicine involves the use of stem cells to treat degenerative diseases or traumatic cell injuries, such as Alzheimer’s 6 , spinal cord injury, stroke, myocardial infarction, diabetes, and tissue damage due to diabetes, burns, and other diseases. Stem cells are promising candidates for use in tissue engineering and regenerative medicine applications 6,7 . However, the source of stem cells is limited, and the process of collecting cells is difficult.
Amniotic membranes, a product often discarded during childbirth, are an ideal source of stem cells 1,2,8 . Using amniotic stem cells does not have ethical and social problems. Stem cells isolated from amniotic membranes have low immunogenicity and carcinogenic risk and the ability to differentiate into various cell lineages 9 . The present study concerns the isolation and the differentiation of amniotic stem cells into keratinocytes that might be promising candidates for tissue engineering and stem cell therapy of severe skin damage.
Materials and Methods
Isolation and Cultivation of Amniotic Membrane Stem Cells
The human amnion was mechanically peeled from the chorion of a placenta obtained from an uncomplicated cesarean section with informed consent. We collected all samples from donors negative for hepatitis B virus, hepatitis C virus, and human immunodeficiency virus. The study and the use of the amnion were approved by the Ethical Committee of the Military Hospital 103, Hanoi, Vietnam.
Stem cells from the amniotic membrane were isolated by trypsin and collagenase digestion as described previously 10,11 . Briefly, the human membrane was removed and separated from amnion, washed with phosphate-buffered saline (PBS), cut into pieces around 1 cm2, and transferred with forceps to 50 ml falcon conical tubes. We added 20 ml of Dulbecco’s Modified Eagle’s Medium (DMEM; Sigma-Aldrich, Hanoi, Vietnam) with 0.25% trypsin and 0.1% collagenase B (Sigma-Aldrich) and then incubated at 37 °C with stirring at 400 to 600 rpm for 60 min. Thereafter, centrifuged at 1500 rpm for 5 min and removed the medium by aspiration. The cell pellets were resuspended in 10 ml DMEM, mixed, and filtered through a 100-µm cell strainer (Thermo Fisher Scientific, Hanoi, Vietnam). The filtrate cell number was counted by a hemocytometer.
The cells with Percoll (Sigma-Aldrich) 40.8% and 50.8% were centrifuged as described previously 11 . Briefly, 3 ml of 58.5% Percoll was added to the bottom of a 10 ml conical tube and then gently added 3 ml of 40.5% Percoll on a top of the 58.5% Percoll. Three milliliters of cell suspension was added onto the top of the Percoll solution by using a pipette leaning against the inner wall of the tube. Care must be taken not to disturb the Percoll layers. Then, the cells were centrifuged at 1500 rpm for 30 min. After centrifugation, two layers of cells will be observed: one on the top of the Percoll (fraction I) and a layer of cells at the interface of the two layers of Percoll (fraction III). Cells can also be found in the 40.5% Percoll layer (fraction II) and the 58.5% Percoll layer (fraction IV). The cells in fraction III were aspirated carefully and washed twice with PBS.
Then, amniotic stem cells were suspended in the expansion medium (DMEM supplemented with 50 U/ml penicillin, 50 µg/ml streptomycin, 0.002 M
Measurement of Cell Proliferation
Cells were grown at low density (25,000 cells/cm2 in 200 µl medium) on micro-test culture plates (96 wells). Cells were cultured at 37 °C for 1, 3, 7, 10, or 14 days. The number of cells was calculated by MTS methods (CellTiter 96 Aqueous One Solution Cell Proliferation Assay kit, Promega, Ho Chi Minh City, Vietnam). The principle of this method is the reduction of tetrazolium salts (products that absorb light at 490 nm) by living cells which will be proportional to their numbers.
Differentiation of Human Amniotic Stem Cells into Keratinocytes
The amniotic stem cell differentiations toward the keratinocytes were induced as previously described 13 . In brief, 5 × 105 cells seeded in 10-cm dishes for 7 days (D7) in the expansion medium to attain their normal morphology, and then the differentiation process began by cultivation in expansion medium supplemented with 5 µg/ml insulin, 1 µM hydrocortisone, 0.5 nM BMP-4, and 50 µg/ml of vitamin C (D0). Keratinocyte differentiation medium was replaced twice a week for 14 days. Cells grown in an expansion medium for similar time points served as control.
We harvested cells at day 1 (D1), day 7 (D7), and day 14 (D14) after the addition of the keratinocyte differentiation medium. We evaluated the differentiation from stem cells into keratinocytes by reduction of stem cell markers (octamer-binding transcription factor 4 [OCT-4]) and the appearance and increase of keratinocyte markers, including vimentin, CK-14, and CK-19 in both RNA level (reverse transcription-polymerase chain reaction [RT-PCR]) and protein (immunocytology).
RT-PCR
Total RNA was isolated from the cell by RNeasy Mini Kit (Qiagen, Hanoi, Vietnam) according to the supplier’s technical process. The concentration and quality of RNA were determined on Nanodrop and Agilent systems. The additional DNA strands synthesized by a reverse transcriptase reaction of 5 µg of total RNA using the RevertAidTM H Minus First Strand cDNA Synthesis Kit (Fermentas, Hanoi, Vietnam). Real-time-RT-PCR was performed on LightCycler 1.5 (Roche Diagnostics, Hanoi, Vietnam) using QuantiTect SYBR Green PCR (Qiagen) kit. Sequences of primers used for amplification of OCT-4, CK-14, CK-19, and 18 S rRNA cDNA were: OCT-4 forward (GAGGAGTCCCAGGACATGAA) and reverse (GTGGTCTGGCTGAACACCTT) of the human sequence (GI: 11602730), CK-19 forward (AATATGAGGTCATGGCCGAGCAGA) and reverse (TGTCTTCCAAGGCAGCTTTCATGC) of the human sequence (GI: 1519316325), CK-14 forward (TTCTGAACGAGATGCGTGAC) and reverse (GCAGCTCAATCTCCAGGTTC) of the human sequence (GI: 1519244668), 18 S forward (TGAGAAACGGCTACCACATC) and reverse (TTACAGGGCCTCGAAAGAGT) of the human sequence (GI: 1142736576). The threshold cycles (Ct) of the target mRNAs were normalized to that of 18 S rRNA. The relative target gene level was expressed to the control value (D7 or untreated cells).
After denaturation for 15 min at 95 °C, 40 to 50 PCR cycles were performed (15 s: 95 °C, 25 s: 58 °C and 20 s: 72 °C). PCR products are electrophoresis on the gel or sequenced to check the specificity of the PCR reaction.
Immunocytology
Cells on a culture dish or slide after the addition of the keratinocyte differentiation medium at D14 were fixed with absolute ethanol incubated with the first antibodies (Thermo Fisher, Ho Chi Minh City, Vietnam) against vimentin, CK19, CK14, or OCT- 4. The second antibody (Thermo Fisher) was attached to the fluorescent substance. The cell nuclei were stained with 4′,6-diamidino-2-phenylindole (Sigma Aldrich) and observed microscopically.
Protein Analysis (Western Blot)
Cells were lysed at 4 °C in a solution containing Tris–hydrochloric acid 50 mmol/l, pH 7.5, phenylmethylsulfonyl fluoride 3 mmol/l, aprotinin 10 μg/ml, pepstatin 1 μg/ml, and leupeptin 1 μg/ml. The concentration of clear fluid protein was measured using the Coomassie reagent method (Greiner, Hanoi, Vietnam). Ten microliters of a solution containing 25 µg of protein was added to each electrophoresis well. Electrophoresis well were made on 10% polyacrylamide gel (sodium dodecyl sulfate-polyacrylamide gel electrophoresis). Protein on the gel was transferred onto the nitrocellulose membrane and then incubated with mouse anti-OCT-4 and CK14 antibodies (Thermo Fisher). The protein that reacted to the first antibody will be determined by the second antibody attached to the peroxidase (Thermo Fisher). Results were expressed by the image on the film after-effects with luminescent (ECL+, Amersham Biosciences, Hanoi, Vietnam).
Results
Isolation and Cultivation of Amniotic Membrane Stem Cells
Cells from the amniotic membrane were successfully isolated. We used trypsin and collagenase B with different concentrations and incubation times (data not shown). If only trypsin is used, the amniotic membrane cleavage time is prolonged. If collagenase B is added to trypsin, the disintegration time will be faster. The optimal medium for amniotic membrane isolating is trypsin 0.25% and collagenase B 0.1%; mixing these two solutions will give the best effect and the time the enzyme dissolves the membranes is about 50 ± 10 minutes. Hyaluronidase has almost no effect on amniotic membranes, although some authors in the world have used this enzyme to isolate cells from connective tissues.
Stem cells were cultured in a plastic disc of 10 cm diameter, with an expansion medium at 37 °C, the air had 5% carbon dioxide and saturated humidity. After 24 hours of cultivation, most of the amniotic stem cells adhered to the culture plate developed into a polyhedron or rhomboid cells,and only a small amount remained as floating cells. After 7 days, it was possible to observe a homogenous cell population. These cells became the majority after the 14th day of cultivation, and this population was used for immunostaining.
Cell Proliferation
The result of cell proliferation is shown in Fig. 1a. Cell proliferation was strongest from D3 to D10, and then the proliferation decreased due to cell density increasing which inhibited not only the proliferation but also contact between cells, stimulating cell differentiation when factors that differentiated in the culture medium.

(A) Proliferative capacity of amniotic stem cells. Cells were cultured at low density on micro-test culture plates for 1, 3, 7, 10, or 14 days. The number of cells was calculated by MTS methods. The principle of this method is the reduction of tetrazolium salts (products that absorb light at 490 nm) by living cells which will be proportional to their numbers. The data are expressed as the means ± SD from five independent experiments. (B) Amniotic stem cells expressed OCT-4. Electrophoresis image of polymerase chain reaction product of OCT-4. Three different samples of amniotic membrane stem cells were put into three lanes (lanes 1, 2, and 3). OCT-4, octamer-binding transcription factor 4.
Amniotic Stem Cells Express OCT-4
PCR results showed that the stem cells expressed OCT-4 (Fig. 1b). Results consist of cells staining with antibodies from OCT-4 (Fig. 2).
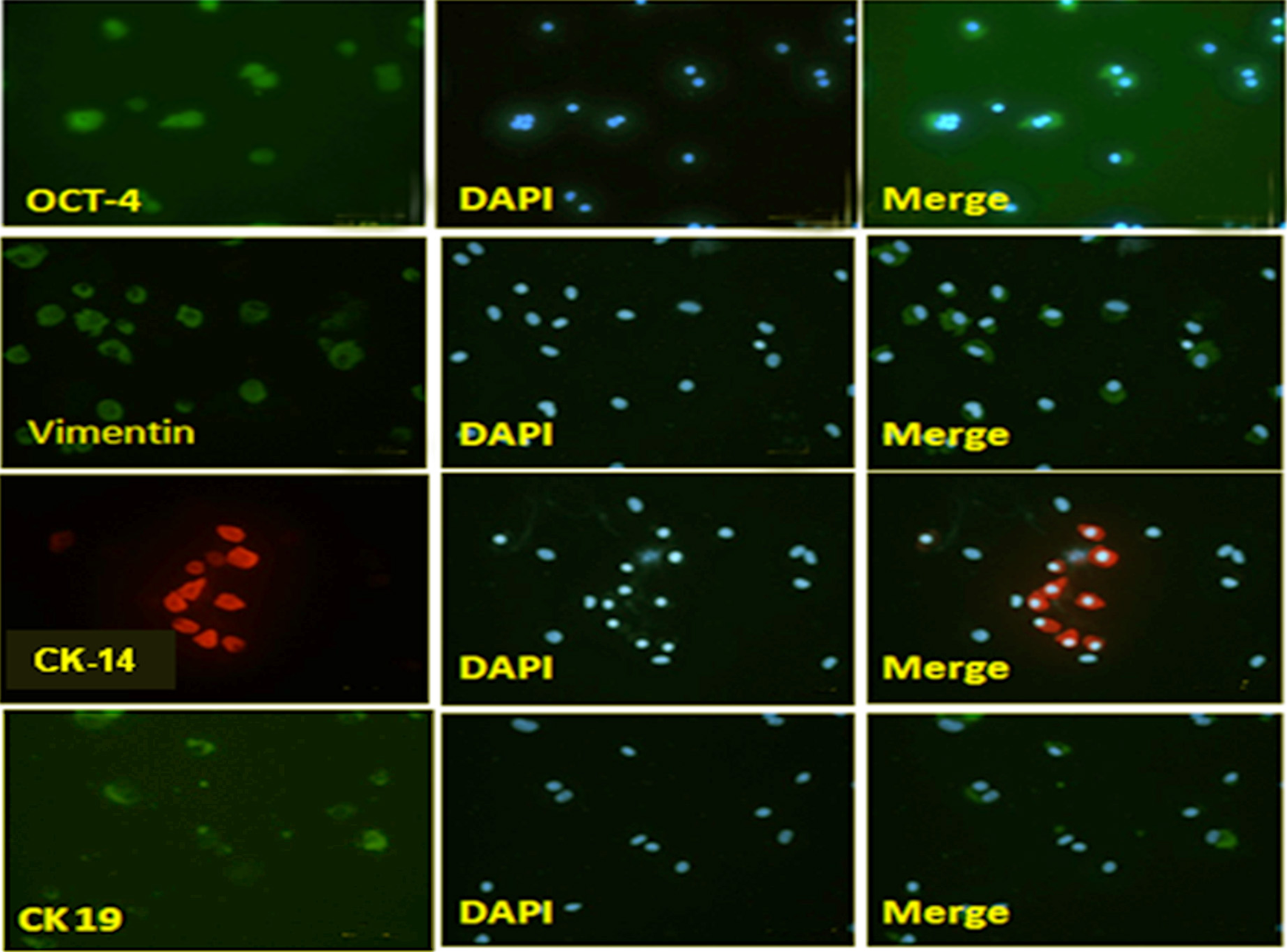
Immunohistochemistry staining of amniotic membrane stem cells after 7 days of culture with biomarkers octamer-binding transcription factor 4 (OCT-4), vimentin, cytokeratin (CK)14, CK19. 4′,6-diamidino-2-phenylindole: staining nuclei. Merge: merged images between the cell immunochemical staining image and the staining nucleus.
Differentiation of the Amniotic Membrane Stem Cells into Keratinocytes
Amniotic stem cells were induced to differentiate into keratinocytes at passage 2 by culturing cells for 14 days in the keratinocyte differentiation medium. Differentiation into keratinocytes was confirmed by immunostaining and further by RT-PCR analysis. The immunostaining results also indicated that induced cells were positive for vimentin, CK14, and CK19 (Fig. 2) specifically expressed by keratinocytes 14 . Further, the expression of keratinocyte lineage-specific genes CK14, CK19 was significantly upregulated in treated cultures as compared with control (Figs. 3a, 3b). The CK14 mRNA expression consists of the protein expression of this marker determined by Western blot (Fig. 3d). In contrast to the expression of keratinocytes, expression of OCT-4 protein decreased (Fig. 3c). The results of this study follow other published reports that indicate the upregulation of these genes in cells undergoing differentiation into keratinocytes 15 .

mRNA expression: (A) CK14 and (B) CK19. mRNA expression increased after the cells were cultured with vitamins C and BMP-4. The mRNA results were calculated in comparison with the control group (arbitrary unit). Protein expression of (C) OCT-4 and (D) CK14. OCT-4 protein decreased, while CK14 protein increased gradually with culture time. Actin was used to reference the amount of protein added to each electrophoresis well. CK, cytokeratin; OCT-4, octamer-binding transcription factor 4.
Discussion
The stem cell isolation from the amniotic membrane is performed with enzymatic digestion of the amniotic membrane. A notable variability in the purity, yield, and viability of amniotic membrane stem cell isolation using previous protocols 11,12 . These variations might be related to several factors, including mother-related parameters (e.g., age, gestational week, and delivery type), size and quality of the placenta (which is affected by the duration of the time between the delivery and stem cell isolation), type and concentration of the enzyme solution, and enzymatic digestion time 10,11,12 . A disadvantage of these methods for cell isolation is that achieving a desirable yield comes at the expense of losing cell viability and purity. In our study, the isolation of cells must be conducted within 4 h after the cesarean section. If the cells were isolated after 4 h, the cells had low viability and were susceptible to bacterial and fungal infections. We used trypsin and collagenase B combined with Percoll for isolation of the stem cell. The presented protocol in this study resulted in stem cell isolation with high yield, viability, and purity.
Amniotic cells express many stem cell markers, such as OCT-4, SSEA-4, GATA-4, hepatocyte nuclear factor-3β 16 –19 . These factors indicate that amniotic stem cells are also multipotent stem cells. We used OCT-4 only to identify amniotic stem cells. In this study, we did not aim to determine its characteristics. That is also the limitation of this study. After obtaining the cell, we used it immediately to study the possibility of proliferation and differentiation. The control group was cultured in an expansion medium. After 7 days of culture, cells grew to spread, accounting for 50% to 60% of the surface of the culture plate, and then, we added vitamin C and BMP-4 to differentiate into keratinocytes 13,14 .
The results showed that, after 2 to 3 days, the cells adhered well to the surface of the culture plate, proliferated but not differentiated. Cells still had the same circle as the original cell. At low density, when the cells had not yet contacted to each other, they had not differentiated despite the differentiation factors, such as cytokines.
We successfully differentiated the amniotic membrane stem cells into keratinocytes. The CK14 and CK19 were the first markers expressed by the cell culture on the 14th day. Our results are similar to those of other authors when differentiating stem cells into keratinocytes 15,20 . We added BMP-4 and vitamin C in the culture medium to differentiate stem cells into keratinocytes. BMP-4 has been shown experimentally to stimulate differentiation of embryonic stem cells into keratinocytes. Some authors have successfully differentiated keratinocytes into skin-like structures 20 . The results indicate that amniotic stem cells can differentiate into skin cells in vitro in which BMP-4 and vitamin C play an important role.
In our study model, cells expressed CK14 after 7 days of culture and gradually increased after 14 days. We have just stopped at the stage when cells express the markers of keratinocytes—the first stage of skin formation 21 . The process of differentiation from stem cells into keratinocytes can be divided into two stages: (1) the stem cells begin to express specific markers of keratinocytes and (2) the cell form the morphological structures of the skin 22 . Due to the condition of the study, we only identified the biomarkers of skin cells as vimentin, CK14, and CK 19. The results showed that at D7, a clear expression of differentiated cell markers at both mRNA and protein levels. We chose these markers because they appear different in both stem cells and differentiated cells. When the cell increases the expression of vimentin, CK14, and CK19, it proves that it has the function of keratinocyte 22 –24 . However, for the stem cell to completely differentiate into keratinocytes and create skin-like structures, it is necessary to have a special extracellular environment rich in collagen type IV and rich in laminin. We cannot create this environment yet.
From the results obtained in this study, we conclude that human stem cells can be isolated from human amniotic membranes with trypsin and centrifuged with Percoll, proliferated in DMEM high glucose with 10% fetal bovine serum, and differentiated into keratinocytes if added to the medium on BMP-4 and ascorbic acid. Differentiated keratinocytes express vimentin markers, CK14, and CK19.
Footnotes
Author Contributions
Dam Thi Phuong Lan and Pham Thai Binh processed the experimental data and performed the analysis. Nguyen Thi Quynh Giang, and Can Van Mao drafted the manuscript and designed the figures for the calculations. Dang Thanh Chung and Nong Van Diep collected and stored the amniotic membranes. Do Minh Trung and Pham Van Tran researched on the design, aided in interpreting the results, and worked on the manuscript. All authors discussed the results and commented on the manuscript.
Ethical Approval
The study was approved by the Ethical Committee of the Military Hospital 103, Vietnam.
Statement of Human Rights
The study was performed following the ethical standards of the responsible committee on human experimentation (institutional and national).
Statement of Informed Consent
Internet informed consent was obtained from the participants at the end of the e-questionnaire for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project received financial support from Military Hospital 103, Vietnam, and the Ministry of Science and Technology, The Vietnamese government.
