Abstract
Adult stem cell therapy for the treatment of tendon injuries is a growing area of research. This study is aimed to investigate the efficacy of human adipose-derived stem cell (hADSC) injection on the tendon during its healing process in a rat model of rotator cuff injury. hADSCs were injected 3 days after collagenase-induced rotator cuff injuries in experimental groups, while the control group received saline as a placebo. Histological and biomechanical analyses were performed 7, 14, 21, and 28 days after collagenase injection. Compared to the control group, it was found that inflammatory cells were significantly decreased in the hADSC-treated group after collagenase injection for 7 and 14 days. In the hADSC-injected group, the fiber arrangement and tendon organization had also been improved. On the seventh day after collagenase injection, the load to failure of the hADSC-injected group (15.87 ± 2.20 N) was notably higher than that of the saline-injected group (11.20 ± 1.35 N). It is suggested that the tensile strength of the supraspinatus tendon was significantly enhanced. Local administration of hADSCs might have the possibility to restore the tensile strength and attenuate the progression of tendinitis. Taken together, these findings demonstrate that the recovery processes in damaged tendons can be facilitated architecturally and functionally after hADSC injection.
Introduction
The human shoulder muscle has the ability to maintain shoulder joint stability in the glenohumeral joint, the sternoclavicular joint, and the acromioclavicular joint (39). Rotator cuff muscles include the subscapularis, the supraspinatus, the infraspinatus, and the teres minor. These have been found to facilitate shoulder movement as well as maintain the glenohumeral joint stability. Tendons bind rotator cuff muscles to bones and allow the arm to move, slide, rotate, and stretch through muscle contractions (39). The architecture of the tendon is organized from collagen fibrils, fibers, and fascicles to the intact tendon (18). Tendons are structured by high-density fibers and extracellular matrix proteins to form fibrous connective tissue. Macromolecules of extracellular matrix provide structural and functional regulation of tendon organization. The dry mass is approximately 30% of tendon weight and is comprised of 60–80% of type I collagen, 2% of elastin, proteoglycan (1–5%), and others (14,18,30,35). Rotator cuff muscles and their tendons are crucially important to the function of the shoulder, such as stabilizing shoulder joint actions (3).
Shoulders can support a wide range of movement, which may create vulnerable pull to the tendon. Most tendon injuries are caused by an extruding structure around the surrounding bones and ligaments. Damage to the rotator cuff is the most common form of soft tissue injury in the musculoskeletal system, often manifesting as tendinitis or tearing (1). These conditions can eventually lead to tendon degeneration (2). Risk factors for rotator cuff injuries include abnormal shoulder structure, glenohumeral joint dynamic instability, and joint abnormality. Elderly and obese patients also have a higher risk of tendon damage, and their tendons become increasingly prone to microtears, calcification, and fibrovascular proliferation. Over half of the cases of rotator cuff injury occur in individuals above the age of 60 years (24,27). Tendinopathy is characterized by different conditions of tendon injuries. Specifically, tendinitis is acute tendon damage resulting from single intrinsic or extrinsic stimulation, whereas tendinosis is chronic inflammation of the tendon resulting from multiple minor injuries (12,21, 33,34). Current treatments for rotator cuff tendon injuries involve physiotherapy, nonsteroidal anti-inflammatory drugs, steroid injections, surgery, etc. (2). However, the consequence after tendon repair often results in ischemic change and partially irreversible damage, for which no existing drug or clinical procedure is effective. It is important to develop treatment methods to improve the regeneration of damaged rotator cuff tendons.
The improvement of healing damaged tendons and ligaments with stem cells has been investigated in many animal models (9). Stem cells are primitive cells with the potential to differentiate into various lineages for the regeneration of muscles, bones, and organs (23). Adipose-derived stem cells (ADSCs) are one type of mesenchymal stem cell (MSC), and they are found in the white adipose tissue. These adult stem cells have demonstrated their therapeutic potential in regenerative medicine (7,25). ADSCs have been shown to have the same multipotent capability as other MSCs (38). ADSCs demonstrate the ability to differentiate into various lineages, such as tenocytes, fat cells, neurons, endothelial cells, skeletal muscle cells, smooth muscle cells, and cardiac myocytes. Cytokines and growth factors released by ADSCs have also been shown to have modulatory activity in tissue remodeling (6,19,20). Studies on ADSCs have provided evidence for restoration of damaged chondrocytes and injured cartilage (15). In addition, ADSCs have also been indicated to suppress inflammation, promote angiogenesis and enhance fibroblast proliferation (5,26). Human ADSCs isolated by using minimally invasive procedures could illustrate tremendous potential in tissue repair and tissue regeneration applications (13,16,40).
Previous studies have established a rat model of rotator cuff injury by collagenase injection by surgically detaching skin (27,33). The idea of using rats as an appropriate animal model is due to the fact that the supraspinatus tendon in a rat shoulder is structurally similar to the human coracoacromial arch. This rotator cuff injury model can mimic human disease syndromes and provide reproducible results. In the present study, we successfully created a rat model of rotator cuff tendon injury with direct collagenase injection. Furthermore, tensile tests and histological analyses were investigated for treatment with hADSCs in the current rat model. It was revealed that the tensile strength of the supraspinatus tendon was significantly improved. Local administration of hADSCs was able to recover the tensile strength and attenuate the progression of tendinitis. Taken together with current findings, local injection of hADSCs might have a beneficial effect on treating injured rotator cuff tendons and reveal supportive foundations for future clinical applications in tendon therapy.
Materials and Methods
Animals and Experimental Design
Specific pathogen-free, female Sprague–Dawley (SD) rats, aged 12.5 weeks, weighing 250 ± 20 g, were purchased from the National Laboratory Animal Center (Taipei, Taiwan). All rats were housed in a laboratory room at controlled temperature (22 ± 2°C) and appropriate humidity (50–70%) and were allowed adequate light and food. The protocols were conducted in accordance with the Guidance Suggestions for the Care and Use of Laboratory Animals and were evaluated by the Animal Board Committee of China Medical University. The experimental design in the study is depicted in Figure 1. In brief, 120 SD rats were grouped with or without hADSC treatment on different postinjection days. The supraspinatus tendon in the left forelimb was subjected to biomechanical tests, and another supraspinatus tendon in the right foreleg of the same rat was subjected to histological analyses.

Schematic flow chart of experimental design and groups in the study of rotator cuff tendon injury. SD, Sprague–Dawley; hADSC, human adipose-derived stem cell.
Collagenase-Induced Tendinitis in a Rat Model of Rotator Cuff Injury
A dose of 0.01 μl/kg chloral hydrate (15307; Riedel-de Haen, Seelze, Saxony, Germany) was administered intraperitoneally for anesthetization. Rotator cuff injury was produced by collagenase injection (33) with some modifications to a previous study. In summary, (I) fur on the neck and shoulder of rats was shaved, (II) the shoulder cheek bone was fixed and pulled by hands, (III) the location of acromion (located in the vertex of the shoulder) was identified, and (IV) type II collagenase (80 U/8 μl/1 min) (C6885; Sigma, St. Louis, MO, USA) was then injected near the supraspinatus tendon between the coracoids and the clavicle by using a 27-gauge needle (NN**2713R9; Terumo, Tokyo, Japan). After injection, the rats were returned to the original cages until further treatment was continued on the third day.
Biomechanical Evaluation
The rats were fully anesthetized and euthanized with chloral hydrate, and the entire supraspinatus muscle and tendon of the shoulder along with the humeral head of treated rats were isolated and collected. The mode of tear and the load to failure at a rate of 10 mm/min with a preload of 5 N (8,11) was evaluated by a materials testing machine (JSVH1000; Japan Instrumentation System, Nara, Japan). The harvested tendons were then placed on a horizontal rack thrust apparatus by using a customized fixtureclamping system, and deep-freeze spray (–40°C) was used to anchor the muscle in one side of the machine. The tendon was loaded until it pulled apart from the bone or ruptured at its midsubstance. The supraspinatus tendon was fixed to this system along its anatomic direction to allow the tensile loading and tendon-to-bone interface to form a right angle. The maximum tensile strength was recorded for later statistical analysis.
Tissue Preparation and Histological Analysis
At the scheduled time points, rats were fully anesthetized and euthanized with chloral hydrate (15307; Riedelde Haen), and the entire supraspinatus muscle and tendon were obtained. The specimens were fixed in 4% paraformaldehyde (16005; Sigma) for 24 h and embedded in paraffin by an enclosed tissue processor (ASP300 S; Leica, Nussloch, Baden Württemberg, Germany). Tissue sections of 2 mm were sliced by using a tissue slicer (RM2125 RTS; Leica). The slides were stained with hematoxylin (GHS116; Sigma) and eosin (HT110216; Sigma) following a standard procedure, and all the samples were surveyed using a light microscope (ZEISS, Jena, Thuringia Land, Germany). The histological images of supraspinatus tendons were independently examined in 10 different microscopic fields (200×) of one sample and were scored by two independent pathologists with experience in musculoskeletal pathology for elimination of bias; “-” was defined as a total count of less than 20 mononuclear inflammatory cells under 10 different microscopic fields, “-/+” was defined as a total count of 20~50 mononuclear inflammatory cells under 10 different microscopic fields, “+” was defined as a total count of 50~70 mononuclear inflammatory cells under 10 different microscopic fields,“++” was defined as a total count of 70~100 mononuclear inflammatory cells under 10 different microscopic fields, “++/+++” was defined as a total count of 100~150 mononuclear inflammatory cells under 10 different microscopic fields, and “+++” was defined as a total count of greater than 150 mononuclear inflammatory cells under 10 different microscopic fields.
Isolation and Amplification of hADSCs
hADSCs were provided by GWOXI Applied Technology Co., Ltd. (Hsinchu, Taiwan). The study protocol was approved by the Scientific Review Committee and the Institutional Review Board of the China Medical University Hospital. The procedures were summarized as follows. One Asian male, aged 46, with local anesthesia had an abdominal 0.3-cm incision below the navel (approximately 3 cm). Three grams of fat tissue were collected, and the wounded area was sutured after treatment had been finished. Separation and purification of stem cells was performed in the Good Tissue Practice Certification laboratory. Isolated hADSCs were cultured in Keratinocyte-SFM (10724; Gibco, Grand Island, NY, USA), 10% fetal bovine serum (SH30071.03; Hyclone, UT, USA), 50 μg/ml bovine pituitary extract (13028–01; Gibco), 5 ng/ml human recombinant epidermal growth factor (10450; Gibco), 2 mM N-acetyl-l-cysteine (A9165; Sigma), and 0.2 mM l-ascorbic acid 2-phosphate (A8960; Sigma) under an atmosphere of 5% CO2 at a temperature of 37°C.
Characterization of hADSC Surface Proteins
Specific cell markers expressed in hADSCs were detected for purity. To identify surface proteins, hADSCs were collected and adjusted to 2 × 105 cells/ml. After washing with phosphate-buffered saline (PBS, 14200–075; Gibco) once, cells were resuspended in 50 μl PBS and stained by 1 μl of surface protein-specific antibodies conjugated with fluorescent dyes in a dark environment for 30 min. Prepared cell samples were analyzed by LRS II flow cytometry (BD Pharmingen™, Franklin Lakes, NJ, USA) for expression levels of cell surface proteins. Surface proteins in hADSCs possessed the following surface proteins: cluster of differentiation 29 (CD29), CD44, CD73, CD90, CD105, and CD166 (BD Pharmingen™, San Jose, CA, USA). In the meantime, hADSCs tested negative for CD14, CD45, and CD31 (AbD Serotec, Raleigh, NC, USA).
hADSC Injection Into the Area of Treated Rotator Cuff in Rats
Cultured hADSCs were trypsinized (25300–054; Invitrogen, Carlsbad, CA, USA) and collected. Cell concentration was adjusted to 3 × 108 cells/ml in PBS, and 10 μl from prepared cell solution was delivered by a microsyringe at collagenase-treated sites. Procedures for cell injection into sites of rotator cuff injuries in rats were according to the tendinitis model of collagenase injection described above. The control group was injected with 10 μl of PBS.
RNA Isolation
hADSCs at 2 × 105 were seeded into a 6-cm Petri dish (403166; Corning, Darmstadt, Hessen, Germany) within 5 ml culture medium and harvested after culture for 1, 2, and 4 days. hADSCs were collected and pelleted in a microcentrifuge at 60 × g, and the culture medium supernatant removed. Pelleted were lysed with Trizol reagent (15596–018; Invitrogen) and extracted with chloroform (102445; Merck Millipore, Darmstadt, Hessen, Germany). The upper aqueous phase was collected by centrifugation at 4°C, 18,000 × g for 15 min, and total RNA was precipitated from solution by the addition of an equal volume of isopropanol (9084–03; J.T. Baker, Center Valley, PA, USA). RNA pellets were washed twice with 75% ethanol (459844; Sigma) and resuspended in diethylpyrocarbonatetreated water (10977–015; Invitrogen). Concentration and quality of the RNA samples were analyzed by absorbance at 260/280 nm, before they were stored at −80°C.
Quantitative RT-PCR
cDNA was generated using Superscript III (18080085; Invitrogen), and quantitative PCR was carried out using the StepOnePlus™ Real-Time PCR System (Life Technologies Inc., Grand Island, NY, USA). Data presented here are means of biological duplicates. Samples were normalized using β-actin primers. Primers were designed using Primer Express® Software Version 3.0 (Applied Biosystems, Foster City, CA, USA). Type I collagen (Col1a1) F: 5′-CCCT GGAAAGAATGGAGATG and R: 5′-CCACTGAAACC TCTGTGTCC; tenascin C (TNC) F: 5′-GGGTCCTCAA GAAAGTCATCCG and R: 5′-CTGACTCCAGATCCAC CGAAC; decorin (DCN) F: 5′-CCAGAAAAAATGCCC AAAACTC and R: 5′-TCGCACTTTGGTGATCTCATTC; β-actin F: 5′-ATCGTCCACCGCAAATGC and R: 5′-TCA AGAAAGGGTGTAACGCAACT.
Statistical Analysis
Values of ultimate load failure were measured by a materials testing machine and were summarized by the means along with standard errors. Statistical significance of collagenase-impairing tension or comparison between hADSC treatment and control group was assessed by oneway ANOVA followed by Tukey's multiple comparison test using Prism 6.0d (GraphPad Software, Inc., La Jolla, CA, USA). The expression ratios of different genes by using quantitative RT-PCR analysis were represented by the means along with standard errors. Statistical significance was assessed by the Student's t-test using Prism 6.0d. Differences were considered statistically significant when values of p < 0.01.
Results
Modification of Acute Rotator Cuff Injury in Rat Models
Assessment in Appearance of the Supraspinatus Tendon
We assessed the appearance of the supraspinatus tendon at 3, 7, 14, 21, and 28 days after collagenase injection. At day 3, severe bleeding and inflammation were noticed in the supraspinatus tendon. Despite bleeding and swelling around tendon muscle areas, there was no obvious impairment found. Muscles and tendons were gradually recovered with a layer of translucent tissue similar to that of normal tendon on day 14 (Fig. 2).

Visual observation of injured supraspinatus tendon. Collagenase was injected into the shoulder once for impairing supraspinatus tendon in rats. Appearance was evaluated at days 3, 7, 14, 21, and 28. Tissue at day 0 acted as a control group and contains normal tendons.
Histological Analysis in Injured Supraspinatus Tendon
A visual assessment indicated that the injured tendon had been healed between days 14 and 28. Therefore, we conducted pathological analysis to assess the arrangement of fibrous collagen as well as the status of injured cells and the general condition of the tendon. Results were represented for 3, 7, 14, 21, and 28 days post-collagenase injection, as shown in Figure 3. At day 0, tenocytes in the control group displayed a normal morphology and staggered arrangement with collagen fibers that were neat and parallel. At days 3 and 7, tendon tissues showed a disordered arrangement of collagen fibers due to tearing and severe inflammation. At day 14 and 21, the number of inflammatory cells had been reduced, and the arrangement of collagen fibers had largely improved in tendon tissues. At day 28, inflammation was no longer observed, and the arrangement of collagen fibers showed structural integrity, compared with observations on day 7.

Histological analysis of the supraspinatus tendon treated with or without collagenase at 3, 7, 14, 21, and 28 days by H&E stain. The control group (untreated tendons; day 0) displayed normal cell morphology. Supraspinatus tendon injuries were most severe on day 3, and then began to recover gradually. Complete recovery was achieved during 21–28 days. Scale bar as represented.
Biomechanical Test of the Supraspinatus Tendon
Biomechanical tests can be applied to evaluate the tensile strength of tendons after impairment. The load to failure measured by biomechanical test were 5.19 ± 1.09 N, 11.20 ± 1.35 N, 20.63 ± 2.42 N, 23.80 ± 3.78 N, and 28.65 ± 2.41 N at 3, 7, 14, 21, and 28 days, respectively, after tendon impairment (Fig. 4). The mean tensile strength of normal tendons was 32.32 ± 2.24 N. The tensile strength had been decreased by 84% on day 3, which indicates that collagenase destroyed the tendon of the rotator cuff. On the contrary, at day 28, the tensile strength of the injured tendons was equal to that of normal tendons, which indicates full recovery of injured tendons. In summary, instead of using surgical methods, we used a modified animal model (9,22,32) of acute rotator cuff injuries to inflict wounds in the supraspinatus tendon.

Biomechanical test of the supraspinatus tendon. Results of the tensile strength analyses at 3, 7, 14, 21, and 28 days postinjury; n = 5, one-way ANOVA (**p < 0.01; ***p < 0.001).
Effect of hADSC Injection on Collagenase-Induced Injuries of Rotator Cuff in Rats
Characterization of hADSC Surface Markers
To test the potential therapeutic capability of hADSCs in the treatment of rotator cuff injuries, we first analyzed the purity of isolated hADSCs by using flow cytometry analysis. The profiling of hADSCs identified the highly positive expression of CD29, CD44, CD73, CD90, CD105, and CD166, but little CD14, CD31, and CD45 expression (Fig. 5A). Surface markers of cultured hADSCs at different passages (p7, p10, and p13) were characterized. Results suggested that the properties of hADSCs had little or no change after multiple generations of culturing (Fig. 5B). hADSCs were utilized after the seventh passage for the treatment of the rotator cuff injury model.
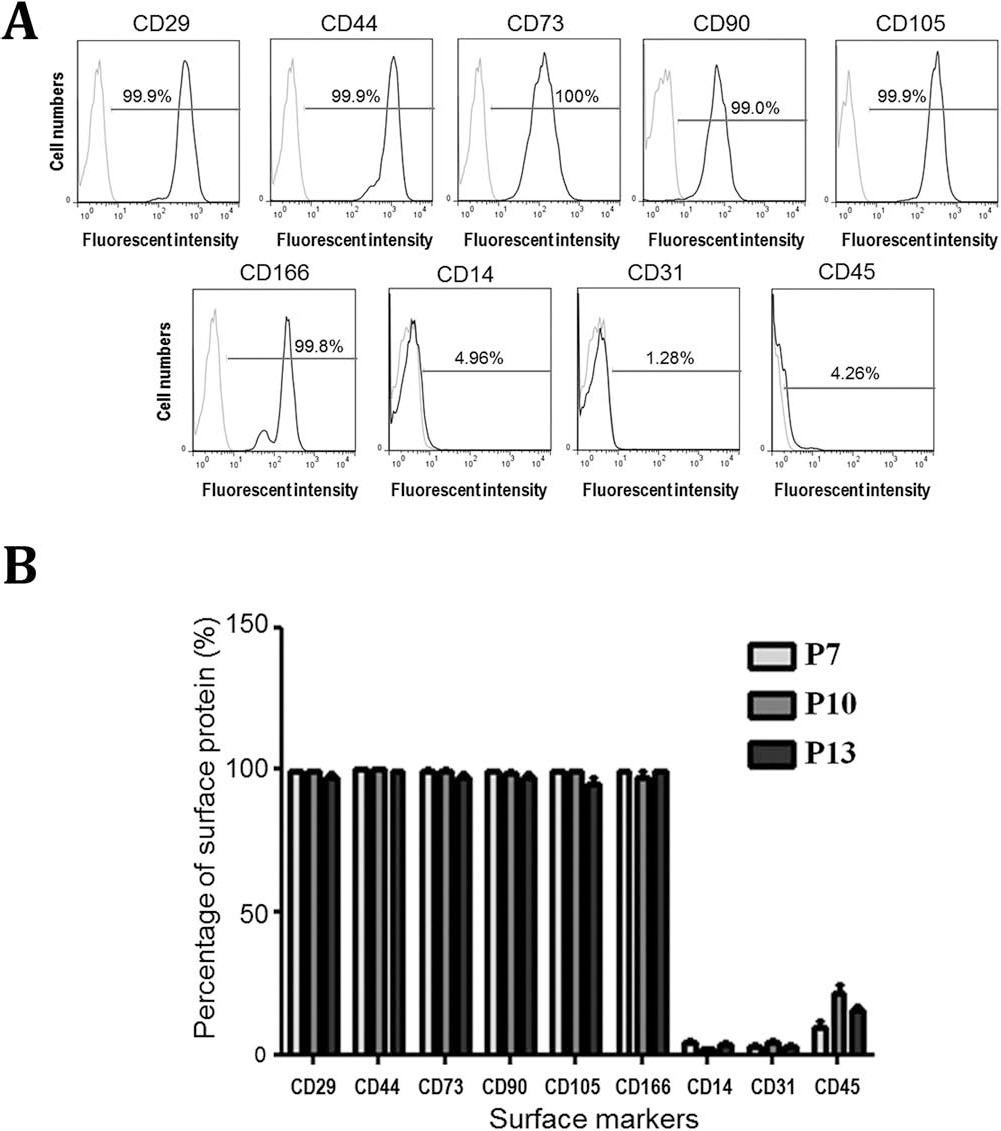
Characterization of hADSC surface markers by flow cytometry analysis. (A) Surface proteins on hADSCs were detected by fluorescent antibodies (CD29, CD44, CD73, CD105, and CD166). CD14, CD31, and CD45 were not detected on hADSCs. The gray lines represent the control group, and the black lines represent the experimental group. (B) Percentage of different surface proteins was characterized from three passages of hADSC in vitro culture.
Assessment in Appearance of Supraspinatus Tendon After hADSC Injection
To evaluate the therapeutic activity of hADSCs, we assessed the appearance of the supraspinatus tendon at days 7, 14, 21, and 28 post-collagenase treatment. At day 7, rats that received hADSC injection demonstrated a significant reduction in bleeding and swelling. In addition, muscles and tendons were covered with a layer of translucent tissue (Fig. 6, circle). On day 14, this coating had turned milky white and opaque. On day 21, the milky white tissue had disappeared. Precisely, the layer of translucent tissue surrounding muscles and tendons did not appear until day 14 in the group of rats that did not receive hADSCs, and that coating only turned milky white and opaque on day 21. Acute tendon injures showed significant recovery in both groups at day 28 (Fig. 6).
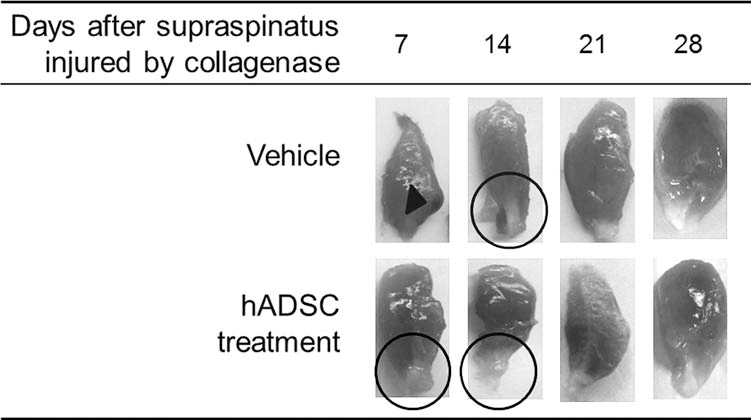
Visual observation of injured supraspinatus tendon treated with or without hADSCs. Collagenase was used to induce impairment of the supraspinatus tendon. Three days later, hADSCs were injected into the injured area. Visual observation and assessment of supraspinatus tendons at days 7, 14, 21, and 28 were presented. Arrowheads indicate severe bleeding and swelling areas. Translucent newborn layers (circle) were found around the muscle–tendon areas.
Histological Analysis in Injured Supraspinatus Tendon Treated with hADSCs In Vivo
Results were represented for 7, 14, 21, and 28 days after hADSC injection on day 3, as shown in Figure 7. At day 7 in the control group, tendons and muscle tissues showed significant aggregation, severe cracking, and a messy arrangement of collagen fibers. Conversely, collagen fibers were neater and more parallel in the hADSC-injected rats at day 7. These data indicate the beneficial effect of hADSCs on tendon recovery. At day 14, the hADSC-treated rats continued to show gradual restoration in tendons and fibers. Collagen fiber formation increased, the number of inflammatory cells (as judged by H&E staining) was significantly reduced, and the morphology of the tendons had become round and flat (Table 1). These observations provide evidence that supports the therapeutic value of hADSCs. On days 21 and 28, we did not observe inflammatory cells, and tendon cell morphology was round and flat in both hADSC-treated rats and untreated rats (Fig. 7).

Histological analysis of injured supraspinatus tendon after hADSC injection. Collagen fibers were cleaved by collagenase, and hADSC treatment greatly improved the arrangement of collagen fibers by day 7. The overall morphology was analogous to normal tendon cells in the group of acute injury with/without injection of hADSCs at days 21 and 28.
Histological Grade of Abnormality in Injured Supraspinatus Tendons Treated With or Without hADSCs

Abnormal level: – (0%); –/+ (17%); + (34%); +/++ (50%); ++ (66%); ++/+++ (82%); +++ (100%).
hADSCs, human adipose-derived stem cells.
Using Biomechanical Tests to Evaluate the Ability of hADSCs to Treat Supraspinatus Tendon Injuries
The results of the biomechanical test for hADSC-injected supraspinatus tendon were summarized as follows. The tensile strength of untreated supraspinatus tendons were 5.19 ± 1.09 N, 11.20 ± 1.35 N, 20.63 ± 2.42 N, 23.80 ± 3.78 N, and 28.65 ± 2.41 N on days 3, 7, 14, 21, and 28, respectively. The tensile strength of the supraspinatus tendon treated with hADSCs was 15.87 ± 2.20 N and was significantly higher than that without hADSCs on day 7 (Fig. 8). On day 14, the tensile strength of the supraspinatus tendon treated with hADSCs was slightly but nonsignificantly reduced to 19.21 ± 3.68 N compared to the vehicle control. On days 21 and 28, the tensile strength of the supraspinatus tendon treated with hADSCs was 23.39 ± 0.51 N and 28.96 ± 4.88 N, respectively (Fig. 8). Taken together, the restoration of injured supraspinatus tendon was facilitated by hADSC injection at the beginning.

Biomechanical test in injured supraspinatus tendons treated with or without hADSCs. Load to failure was measured for tensile strength in injured supraspinatus tendons with or without hADSC treatment. Day 0 was represented as the normal tensile strength. Data from each group (n = 5) was presented as an average and standard deviation. Statistical analyses were conducted through t-tests. (**p < 0.01).
mRNA Expression of Extracellular Matrix Proteins in hADSCs In Vitro
To provide an explanation for restoration of injured supraspinatus tendons treated with hADSCs, the same lot of hADSCs were cultured, and we examined the level of mRNA expression of Col1a1, TNC, and DCN, which are extracellular matrix proteins that play pivotal roles in tenogenesis. The present data indicated that the trend of expression levels of three different genes was elevated after hADSCs were cultured for 4 days in vitro. mRNA levels of Col1a1 and TNC were significantly upregulated on day 4 (Fig. 9).

mRNA expression of extracellular matrix proteins in hADSCs in vitro. Expression of the mRNA of three genes (Col1a1, TNC, and DCN) was analyzed by quantitative RT-PCR, and an elevated mRNA expression level from the three extracellular matrix genes was observed (**p < 0.01).
Discussion
The objective of the present study was to determine if the application of hADSCs in a rat model of rotator cuff repair would improve the biomechanical characteristics of the repair of the tendon and alter the histological findings during the healing process. Here we have shown that the healing process of injured supraspinatus tendons could be facilitated and improved by hADSC injection at 7 days, although we were unable to show any difference in the biomechanics or the histology between hADSC-treated and vehicle control groups at 28 days. In the meantime, the newly formed tissue in the tendon-to-muscle interface was more elastic and eventually less scarred following treatment with hADSCs compared with no treatment in the injured supraspinatus tendon. Furthermore, re-creating the appropriate microenvironment could be the key to improve the healing of the supraspinatus tendon treated with hADSCs, which might provide immunomodulatory activities.
Rotator cuff lesions are the most common type of shoulder injury, and these injuries increase in prevalence with age (31). In patients with severe disability of the shoulder, physicians often consider surgically repairing rotator cuff injuries even with unreliable improvement (29). Based on our study, stem cell therapy presents a promising strategy for the treatment of rotator cuff lesions. Additionally, local administration of ADSCs into the injured tendon could improve the healing process and reduce fatty infiltration in rabbit subscapularis (24). Uysal et al. demonstrated that ADSCs can enhance primary tendon repair using biomechanical and immunohistochemistry analysis (38). Moreover, other recent evidences have suggested that ADSCs are an appropriate resource of stem cells for tendon repair and regeneration (10,36,37). Therefore, ADSCs have great potential to improve treatments for rotator cuff lesions.
Animal models of rotator cuff lesions have been used to investigate the ability of MSCs (28), ADSCs (17), and tendon-derived stem cells (4) for the treatment of tendon injuries. However, few suitable animal models have been developed to assess the regeneration of rotator cuff tears using hADSC therapy. This article reports an improved model of rotator cuff tendinitis, which simulates tendon tearing through the injection of type II collagenase into the joint to induce a supraspinatus tendon injury. We then observed and evaluated the appearance of the tendon for 28 days (Fig. 2). Pathology data clearly showed tears and an aggregation of inflammatory cells in tendons injected with type II collagenase (Fig. 3). Following hADSC treatment, results from pathology and telescopic strength analyses showed significant recovery in the supraspinatus tendon.
Footnotes
Acknowledgments
We appreciate that GWOXI Applied Technology Co., Ltd. (Hsinchu, Taiwan) kindly provided hADSCs for the study. The authors of this article thank Prof. Chang-Zern Hong for his contributions and English editing of this article. This study was supported by the Ministry of Science and Technology (MOST 103–2633-B-039–002 and NSC 102–2314-B-039–021-MY3), Ministry of Health and Welfare (MOHW104-TDU-B-212–124–002), Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence (MOHW104-TDU-B-212–113002), and China Medical University Beigang Hospital (CMUBH R102–012) in Taiwan. This study was also supported in part by China Medical University and Hospital (DMR-104–053 and 10242644) awarded to Dr. Shao-Chih Chiu. The authors declare no conflicts of interest.
