Abstract
Injured tendons often heal with scar tissue formation, resulting in uniformly smaller collagen fibrils and poor mechanical properties. The small leucine-rich proteoglycan decorin is well known to regulate fusion of collagen fibrils. Rat patellar tendon cells were transfected with lentiviral-encoded shRNA that specifically targets decorin. Silencing of decorin expression resulted in decreased cell growth. Three types of scaffold-free engineered tendons with different mix ratios of anti-decorin shRNA-treated cells to untreated cells at 1:0 (DCN), 1:1 (MIX), and 0:1 (CON) were utilized for repair of injured patellar tendons. Four weeks after implantation in situ, the MIX group manifested the best results (best coordination of histology, more mature collagen deposition, and larger collagen fibril diameter). Although the DCN group exhibited the largest collagen fibril diameter, this was associated with abnormal shape. Hence, regulation of decorin expression to an appropriate level is crucial for tendon repair with gene therapy.
Introduction
Tendons are systematic and densely packed connective tissues, which are composed primarily of aligned deposition of tenocytes with their associated extracellular matrix (ECM). The latter is constituted primarily of organic tendon-specific collagen fibrils (grouped together to form fibers) as well as a diverse array of other ECM proteins (17,36,37,46). The main function of tendons is to transmit mechanical forces. Therefore, they are frequently susceptible to traumatic injury arising from sports and aging (10,11). It is well documented that natural healing of injured tendons is usually accompanied with scar tissue formation, which makes the healed tendons mechanically inferior to normal tissues (12). In scarred tendons, normal large-diameter collagen fibrils are replaced by relatively small collagen fibrils (24) with decreased type I collagen content and elevated expression of types III and V collagen (17,27), together with small leucine-rich proteoglycans/proteins (SLRPs) such as decorin and biglycan (5,15,45).
Such qualitative alterations of tendon ECM are the major cause of altered tissue mechanical properties in naturally healed tendons (24).
SLRPs play important roles in collagen fibril formation and organization by binding specific sites on the collagen molecule (31). All of these are structurally related but are genetically distinct and can be divided into three classes: I, II, and III. Decorin is the prototype of class I of the SLRPs family, characterized by a protein core composed of leucine-rich repeats (LRRs), and flanked on both sides by two conserved cysteine-rich regions (12,28). The amino-terminal domain contains a negatively charged glycosaminoglycan chain that might be involved in binding to cationic domains of cell surfaces and extracellular matrix proteins. The carboxyl end domain contains two cysteine residues, and its function still remains to be understood. When interacting with their cognate ligands, two decorin molecules form a dimer and the central LRR domain is shaped like a banana (14,33,39), so that the inner concavity accommodates and provides a binding site for various ligands such as collagen type I, II, III, VI, and XIV (28,34,39).
Currently, more information on the functional role of decorin in cell biology is now available. Much scientific work has been focused on the interaction of decorin with collagen fibrils including its influence on collagen fibrillogenesis (29), collagen degradation (4), cell growth (9,16,23), extracellular signaling, and final diameter of the fibrils (35). A previous study has reported that, in the equine superficial digital flexor tendon, the distribution pattern of decorin was inversely related to the profile of collagen fibril diameter (38), suggesting that decorin is inhibitory to fibril diameter. Indeed, downregulating decorin expression with antisense RNA in vivo led to the generation of larger diameter collagen fibrils and enhanced mechanical properties in healing ligaments (24). However, complete removal of decorin expression in knockout mice generated larger fibrils (gigantic fibrils) with abnormal collagen morphology and decreased mechanical strength in mature tendons (8,43,45). Collectively, these data thus suggest that reducing decorin expression to a certain level is desired for tendon maturation and proper functional restoration of injured tendons. Gene silencing with antisense RNA is a mild technique for repressing gene expression and functions in a transient reversible manner. However, for in vivo study, a long-term stable method for gene downregulation is preferred.
Small interfering RNA (siRNA) can efficiently block the expression of target genes and was reported to be more potent and efficient than traditional antisense compounds (7,44). Short hairpin RNA (shRNA) can be cleaved into siRNA by the cellular machinery (30), and it can be stably expressed and inherited by genomic integration with a lentiviral vector. Moreover, lentiviral-mediated gene transfer is highly efficient because of its ability to stably transduce not only quiescent but also mitogenic cells, and its low immunogenicity (2,26) would be highly advantageous for in situ gene therapy. Indeed, lentiviral-mediated delivery of shRNA has been widely used for gene therapy research.
In the present study, we hypothesize that regulation of ECM components by specific lentiviral shRNA can enhance the assembly of newly synthesized collagen fibrils of tissue-engineered tendon and enhance the functional recovery of injured rat patellar tendons.
Materials and Methods
Vector Construction and Lentivirus Production
A third generation self-inactivating lentivirus vector containing a cytomegalovirus (CMV)-driven green fluorescent protein (GFP) reporter and a U6 promoter upstream of the cloning restriction sites (BclI and XhoI) was utilized (Genechem, Shanghai, China) (Fig. 1A). shRNA was designed corresponding to the 21-mer target sites using Ambion (Austin, TX, USA) criteria specifying oligo-duplexes. The target sequence of decorin is 5′-GCCTGAAAGGAATGTCTAA-3′ (nucleotides 704–722 of Rattus norvegicus decorin from GenBank accession No. BC083750) (Fig. 1A). Sense and antisense oligodeoxynucleotides contained looped overhangs for BclI and XhoI. The following oligonucleotides were used: decorin sense: 5′-TCAGCCTGAAAGGAATGTCTAATTC AAGAGATTAGACATTCCTTTCAGGCTGTTTTT TC-3′; decorin antisense: 5′-TCGAGAAAA AA CAGCC TGAAAGGAATGTCTAATCTCTTGAATTA GACATT CCTTTCAGGCTGA-3′. Additionally, scrambled shRNA, which is a random sequence and does not specifically target any gene in the cell, was used to discount any change of gene expression profile that may result from the delivery method; 100 pmol of the respective strands were annealed and phosphorylated using phage T4 polynucleotide kinase (New England Biolabs, Inc., Ipswich, MA, USA) and were ligated into the lentivirus vectors between the BclI and XhoI sites (italicized in Fig. 1A).
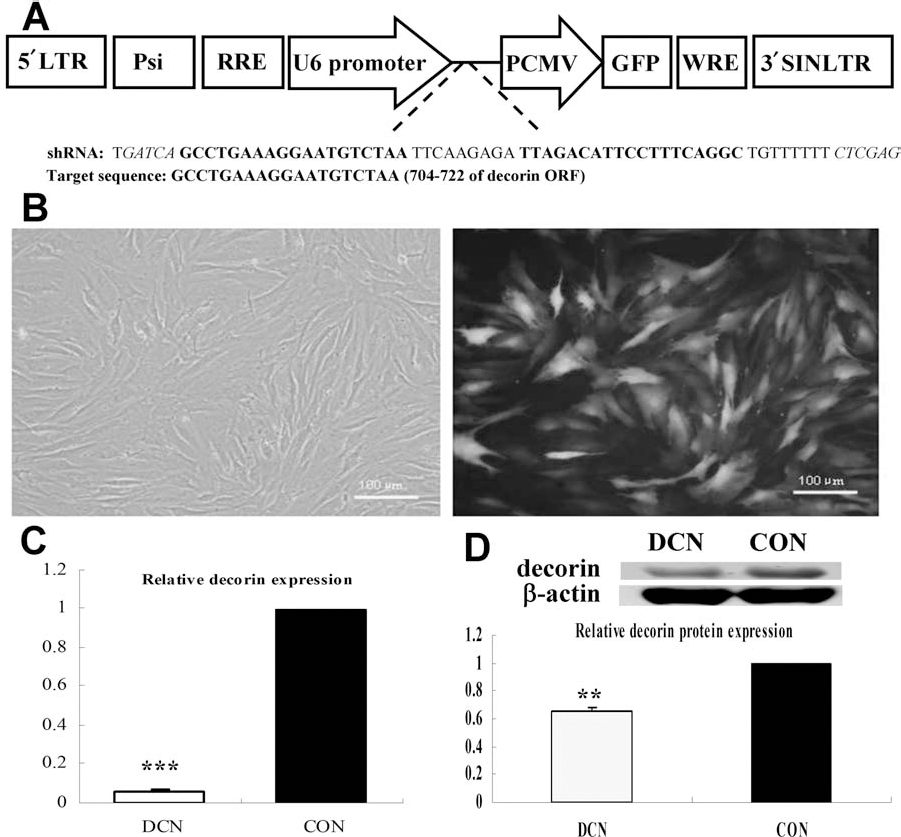
Stable decorin gene silencing in tenocytes by lentiviral-encoded shRNA. Schematic map of the lentivirus vector and the sequence of decorin short hairpin RNA (DCN shRNA) are shown in (A), the restriction enzyme site is italicized, and the target sequence is in bold fonts. High efficiency of lentivirus transfection was indicated by green fluorescent protein (GFP) coexpression (B). The expression of decorin was suppressed at both the mRNA transcription and protein translation levels (**p < 0.01, ***p < 0.001 compared with control group, n = 3) (C, D). LTR, long terminal repeat; Psi, package facilitating sequence; RRE, Rev responsive element; PCMV, plasmid cytomegalovirus; WRE, woodchuck hepatitis virus posttranscriptional response element; SINLTR, self-inactivating long terminal repeat; ORF, open reading frame.
Lentivirus was produced by transfecting a fourplasmid system comprising the constructed lentiviral vector (as described above), together with three other package plasmids (Genechem) into 293FT cells (a cell line established from primary embryonal human kidney; Invitrogen, Inc., Carlsbad, CA, USA). Transfections were performed on 293FT cells at ~95% confluence with lipofectamine (Invitrogen, Inc.) according to the manufacturer's instructions. The medium was replaced 16 h after transfection. Two days later, the virus-containing medium was collected and passed through a 0.45-μm filter (Millipore, Billerica, MA, USA) to remove cell debris and frozen at −80°C.
Cell Culture and Lentivirus Transfection
Primary tendon cells were isolated from the patellar tendon of female Sprague–Dawley (SD) rats weighing 150–170 g [Shanghai Laboratory Animal Center (SLAC), Shanghai, China]. All experimental protocols involving live animals were approved by the animal care and ethics committee of Zhejiang University (number: zju2007102001). Briefly, patellar tendons were harvested and adjacent tissues were stripped. After soaking in phosphate-buffered saline (PBS) containing penicillin (100 units/ml) and streptomycin (100 μg/ml) (Gibco, Invitrogen, Carlsbad, CA, USA) for 10 min, the tendon was cut into small fragments followed by digestion with 0.2% (w/v) mixed collagenase (Gibco) at 37°C in a humidified atmosphere of 5% CO2 and 95% air. After 4 h, an equal volume of Dulbecco's modified Eagle's medium (DMEM) containing 10% (v/v) fetal bovine serum (FBS; both Gibco) was added, and the mixture was digested overnight. The tissue residue was then removed, and the cell suspension was centrifuged at 1,200 rpm. The harvested cells were maintained in DMEM containing 10% (v/v) FBS with medium being changed every 2–3 days. When cultured primary cells reached 80% confluence, they were detached by treatment with 0.25% (w/v) trypsin and 0.1% ethylenediaminetetraacetic acid (both Gibco) and subcultured at a density of 1 × 104 cells/cm2. Cultured cells before passage 5 were used for experiments. Tendon fibroblasts were transfected with lentivirus when cells were 30–50% confluent at a multiplicity of infection (MOI) of 200. Twelve hours after infection, more than 95% of the cells were still viable and then the medium was changed. Three days later, all transfected cells showed green fluorescence and the cells were passaged for further assay and animal testing.
Quantitative Reverse Transcription-Polymerase Chain Reaction
Total cellular RNA was isolated by Trizol (Invitrogen) as described in the manufacturer's instructions. Isolated RNA (2 μg) from each sample was reverse-transcribed into cDNA using Moloney murine leukemia virus (mMLV) reverse transcriptase (Invitrogen). Quantitative real-time PCR (qRT-PCR) was employed to detect the mRNA levels of decorin, collagen α1 (I), III, α1 (V), α2 (V), transforming growth factor (TGF)-β1, and TGF-β3 (n = 3) with the ABI 7900 HT Real-Time PCR System (Applied Biosystems, Foster City, CA, USA), using SYBR Green Realtime PCR Master Mix kit (TaKaRa, Shiga, Japan) in accordance with the manufacturer's instructions. Target genes were normalized against the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Specific primers are shown in Table 1.
Specific Primers Used for Real-Time PCR

TGF, transforming growth factor; Col1α1, collagen type I, α1; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Western Blotting
Tendon cells were harvested and washed twice with cold PBS followed by lysis in M-PER [Mammalian Protein Extraction Reagent with PMSF (phenylmethanesulfonyl fluoride); Pierce Biotechnology, Inc., Rockford, IL, USA]. After 10 min on ice, lysates were centrifuged at 15,000×g for 15 min to remove insoluble material. Fifty micrograms of protein samples were resolved on 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to nitrocellulose membranes (both Millipore), followed by immunostaining with a rabbit antibody against decorin (1:500, BioVision, Milpitas, CA, USA). Protein bands were detected with horseradish-peroxidase (HRP) conjugated goat anti-rabbit IgG (H+L) (1:8,000, Promega, Madison, WI, USA) and enhanced chemiluminescent Western blot detection reagent (Pierce Biotechnology, Inc.). The blots were subsequently stripped and then reprobed for β-actin (1:5,000, Santa Cruz Biotechnology, Dallas, TX, USA). Image J software (National Institutes of Health, Bethesda, MD, USA) was utilized for densitometry measurements, and the protein level of decorin was indexed to β-actin. Untreated cells were utilized as the control group.
MTT Assay
Cell viability assay was performed to determine whether decorin knockdown could affect the proliferation and apoptosis of tendon cells. This measure was based on the metabolism of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma-Aldrich, St. Louis, MO, USA) into a blue-colored product (formazan) by the mitochondrial enzyme succinate dehydrogenase. MTT (50 μg/ml) was added and incubated at 37°C for 4 h. Subsequently, the MTT solution was discarded and 150 μl of dimethyl sulfoxide (Sigma-Aldrich) was added to dissolve the formazan crystals. The plates were then fully shaken to solubilize the formazan crystals formed. The optical density was measured at 570 nm using a Multiskan Spectrum plate reader (Thermo Labsystems, Waltham, MA, USA).
Fabrication of Tissue-Engineered Tendons
To repair tendon injury and detect collagen fibrillogenesis, scaffold-free tissue-engineered tendons were developed with tendon cell sheets. Tendon cells were cultured in high glucose DMEM with 10% (v/v) FBS and 50 μg/ml ascorbic acid (Sigma-Aldrich) and cell sheets were formed within 2 weeks. This could be detached from the substratum by applying a small roll-up force and a tissue-engineered tendon was generated. Engineered tendons generated with shRNA decorin were the experimental group (DCN; 1:0 ratio of shRNA-treated decorin tendon cells/untreated tendon cells), while the untreated tendon cell sheets served as the control (CON; 0:1 ratio of shRNA-treated decorin tendon cells/untreated tendon cells). Artificial tendon formed with a mix ratio of 1:1 treated to untreated cells was produced as MIX group, which aimed to regulate decorin expression to an intermediate level. These engineered tendons were utilized in subsequent in vitro and in vivo studies. Cultured cell sheets utilized for in situ repair were exposed to FBS-free media 24 h before detachment.
In Vivo Experiments
Animal Model
Thirty-six hind limbs of skeletally mature female Sprague–Dawley rats weighing 200–250 g were used. Under general anesthesia, a window wound model (1 mm in width and 4 mm in length) in the center of the patellar tendon was cut. The three different types of engineered tendons were used to fill up the wounds.
Determination of Collagen Content of Repaired Tendon Tissue
Four weeks after implantation in vivo, repaired tendon tissue was removed and freeze-dried, and the dry weight was measured. The amount of deposited collagen was quantified by using a collagen assay kit according to the manufacturer's protocol, which was a colorimetric-based hydroxyproline assay used to estimate total collagen content and concentration (n = 4–5, Jiancheng Ltd., Nanjing, China). The content was measured using the Multiskan Spectrum plate reader at 550 nm.
Histomorphometry
Repaired tendon tissue was removed 4 weeks after experiment in vivo and fixed in 10% neutral buffered formalin (Sigma-Aldrich), dehydrated through an alcohol gradient, cleared, and embedded in paraffin. Histological sections (10 μm) were prepared using a microtome and subsequently stained with hematoxylin–eosin and Masson trichrome (both Sigma-Aldrich) according to standard procedures to examine the general appearance of the collagen fibers (4,29). Polarizing microscopy was employed to detect mature collagen fibrils.
After Masson trichrome staining and polarizing light detection experiments, three pictures of separate view fields were randomly selected for each sample, and then Image-Pro Plus 5.1.2 (Media Cybernetics, Rockville, MD, USA) was used to quantify the quantity and degree of maturation of collagen production by calculating the related color density (20).
Transmission Electron Microscopy Imaging
To analyze the ECM deposited in repaired tendon tissue, transmission electron microscopy (TEM) was used to assess the diameter of the collagen fibrils, as well as fibril alignment. Briefly, samples were prefixed with 2.5% glutaraldehyde (Sigma-Aldrich) for 2 h at 4°C and washed twice with PBS at 4°C followed by fixation with 1% osmic acid (Sigma-Aldrich) for 2 h at 4°C. After two washes in PBS, the samples were dehydrated in an ethanol gradient and dried to a critical point. The samples were then mounted and sputter-coated with gold (Sigma-Aldrich) for viewing in a TEM (Quanta 10; FEI, Hillsboro, OR, USA) (17). About 1,000 collagen fibrils were measured in each group to obtain a true representation of the fibril diameter distribution (n = 3).
Statistical Analysis
The results are presented as mean ± standard deviation of at least three samples from each group. Differences in numerical data between groups were analyzed utilizing one-way ANOVA, followed by Tukey's honestly significant difference test if the homogeneity of variances was significant, and p < 0.05 was considered as statistically significant. The Grubbs' test was used to identify outlying values (13). Differences between the different group distributions of collagen fibers were analyzed using a Kolmogorov–Smirnov test (46).
Results
Lentiviral-Encoded shRNA Efficiently Suppressed Decorin Expression
As indicated by the green fluorescence in Figure 1B, the efficiency of lentivirus transfection is very high. To determine whether lentiviral shRNA influences decorin expression in rat patellar tendon cell, real-time PCR and Western blots were performed. RNAi led to substantial suppression of decorin transcription. Compared to the control group, there was at least 90% knockdown of decorin mRNA (p < 0.05) (Fig. 1C), and corresponding 35% downregulation of protein expression (p < 0.05) (Fig. 1D). There was no difference between the scrambled shRNA-treated tendon cells and untreated tendon cells (data not shown), so we used the untreated cells as the control group in this study. These data collectively indicated that decorin was efficiently downregulated at the mRNA and protein levels by lentiviral shRNA.
Suppression of Decorin Expression Slows Down Tendon Cell Proliferation
The MTT assay for tendon cell viability revealed that the shRNA against decorin decreased cell proliferation. The OD570 was significantly decreased at day 3 (p < 0.05) (Fig. 2A). The mean ± SD OD570 was 0.132 ± 0.010 for cultures treated with decorin shRNA, while the corresponding value for untreated control cells was 0.197 ± 0.021. These results clearly indicated that downregulation of decorin had a negative effect on the proliferation of tendon cells.

The effects of decorin gene silencing on the engineered tenocytes. The knockdown of decorin expression significantly reduced the proliferation of tenocytes (*p < 0.05 compared with control group, n = 3) (A) but had no influence on the expression of transforming growth factor (TGF)-β1 and TGF-β3 (B).
TGF-β plays major roles in inhibiting cell proliferation and decorin has been shown to bind it and neutralize its biological activity (19,22,24–26). To examine whether decorin may also transcriptionally regulate TGF-β, different isoforms of TGF-β were analyzed by qRT-PCR to compare their expression levels between cells treated with or without decorin shRNA. Despite the robust downregulation of decorin expression by shRNA (Fig. 1), there is no significant difference in TGF-β expression between the control and infected cells (TGF-β1: p = 0.199, n = 3; TGF-β3: p = 0.090, n = 3) (Fig. 2B). These results thus suggest that inhibition of tendon cell growth rate by decorin downregulation may be through released neutralization of TGF-β biological activities, but not through transcriptional upregulation of TGF-β.
Decorin Suppression Does Not Affect the Expression of Tendon Extracellular Matrix Genes
The expression levels of collagen types I, III, and V, three main structural molecules of tendons, were analyzed to examine whether suppression of decorin expression can influence basic fibril elements. The results showed that the expression levels of collagen type I, type III, type V α1 and α2 did not differ significantly (data not shown), suggesting that suppression of decorin expression does not affect tendon extracellular matrix gene.
Effects of Decorin Suppression on Collagen Deposition
Rat tendon cells proliferated rapidly and formed coherent cellular sheets within 2 weeks after attaining confluence in the presence of ascorbic acid. After the cell sheets were formed, they could be detached from the culture substratum, thus forming a relatively compact bundle of living cells embedded in a collagen matrix (Fig. 3A). A 1 mm × 4 mm window defect was created in the patellar tendon of SD rats (Fig. 3B), and the engineered tissue was placed within the tendon defect and sutured to the tendon (Fig. 3C). After 4 weeks, the regenerative tendons were slick and smooth (Fig. 3D).

Patellar tendon regeneration model in Sprague–Dawley rats and the collagen deposition of engineered tendons 4 weeks after transplantation. The engineered tendon was produced by roll-up of the tendon cell sheet 2 weeks after confluence (A), a 1 mm × 4 mm window defect was created in the patellar tendon (B), the engineered tissue was placed within the tendon defect and sutured to the tendon (C), the gross morphology of the regenerated tendon after 4 weeks, which showed slick and smooth repair (D). The amount of collagen deposited in the tissue-engineered tendon after transplantation (E). Compared with the control group (CON, 0:1 ratio of shRNA-treated decorin tendon cells/untreated tendon cells), the collagen deposition of the DCN (1:0 ratio of shRNA-treated decorin tendon cells/untreated tendon cells) group was decreased (100 ± 4.20% vs. 56.98 ± 4.87%) (**p < 0.001), while that of MIX group (1:1 ratio of shRNA-treated decorin tendon cells/untreated tendon cells) was increased by (29.22 ± 22.26%), and the difference between the MIX and DCN is significant (*p < 0.05).
The amount of collagen deposited in the tissue-engineered tendon 4 weeks after transplantation in situ was assayed to determine whether it was affected by decorin suppression. For each group, five values of collagen deposition were obtained. However in the CON group, there was a significantly deviated value from the others (the values of each were 37.52, 39.07, 13.68, 41.29, and 40.51 μg/mg). Statistical analysis of this data test by Grubbs' test (a tool for detection of outlier) indicated that the deviated value is most likely an outlier (p < 0.01). Therefore, it was excluded and the remaining four values of the CON group were used for further statistical analysis. As shown in Figure 3E, compared with the control group, the collagen deposition of the DCN group was significantly decreased (100 ± 4.20% vs. 56.98 ± 4.87%, p < 0.001), whereas that of the MIX group was increased by 29.22 ± 22.26%, and the difference between the MIX and DCN is significant (p < 0.05). These results indicated that oversuppression of decorin reduced collagen deposition, but partial inhibition led to deposition maintenance.
Suppression of Decorin Expression Leads to Formation of Large Collagen Fibrils
TEM results showed that the repaired tendon tissue had organized collagen fibrils (Fig. 4). The DCN group (Fig. 4A, D) had the largest collagen fibril diameter, but the fringe of collagen fibril was fuzzy and a little irregular. The fibrils from the MIX group (Fig. 4B, E) have a larger diameter and normal morphology similar to that of the control (Fig. 4C, F). Quantitative analyses of fibril diameters and their distribution in the treated and control groups are shown in Figure 4G–I, respectively.

Collagen fibers in tendons after remodeling in situ 4 weeks postimplantation. Ultrastructure (A–F) and histogram (G–I) of collagen fiber diameters of the DCN tendons (A, D, G), the MIX tendons (B, E, H), and the CON tendons (C, F, I) after remodeling in situ at 4 weeks postimplantation. The tendons in the DCN and MIX had a broader diameter distribution (p < 0.001), and both had a shift to larger diameter fibrils. Compared to the CON group (32.58 ± 4.9 nm, 933 fibers), the mean diameters of collagen fibrils in DCN group (37.84 ± 4.7 nm, 1,029 fibers), and MIX group (33.35 ± 5.3 nm, 1,069 fibers) were increased significantly by 16.14% (p < 0.001) and 2.36% (p < 0.05), respectively.
In comparison with CON, the tendons in the DCN and MIX had a broader diameter distribution (p < 0.001) and both had a shift to larger diameter fibrils. Compared to the CON group (32.58 ± 4.9 nm, 933 fibers), the mean diameters of collagen fibrils in DCN group (37.84 ± 4.7 nm, 1,029 fibers) and MIX group (33.35 ± 5.3 nm, 1,069 fibers) were increased significantly by 16.14% (p < 0.001) and 2.36% (p < 0.05), respectively. Hence, this indicates that suppression of decorin expression leads to the assembly of larger collagen fibrils. However, when the decorin level was over downregulated the fibrils exhibited a fuzzy appearance (Fig. 4A, D).
Suppression of Decorin Expression Enhances Injured Tendon Repair
After 4 weeks, the histology of transplanted tendons exhibited dense filling with connective tissue (Fig. 5). Hematoxylin-eosin staining of repaired tendon tissues revealed organized bundles of highly crimped fibers and cells oriented in parallel (Fig. 5A–C). However, the control group (Fig. 5C) exhibited more scar tissues compared with the DCN (Fig. 5A) and MIX (Fig. 5B) groups. Masson trichrome staining of repaired tendon indicated that there was less collagen fibrils in the DCN group (Fig. 5D) compared to the other two groups (E: MIX, F: CON). Quantification of Masson's trichrome blue staining showed that the MIX and CON groups had similar level of density, whereas the level was decreased significantly (p < 0.01) in the DCN (Fig. 5J). As shown by polarizing light microscopy, more mature collagen fibrils were developed in the DCN (Fig. 5G) and MIX (Fig. 5H) groups, which was confirmed by the polarizing calculation indicating significant augmentation of mature collagen in the MIX and DCN groups (p < 0.01) (Fig. 5K). Collectively, these data suggested that the MIX group showed the best results and implicated that maintaining the expression of decorin at a proper level would be critical for tendon regeneration.

Tendons after in situ remodeling 4 weeks postimplantation. Representative hematoxylin–eosin-stained sections (H&E), Masson-stained sections, and polarized light microscopy images of the DCN tendons (A, D, G), the MIX tendons (B, E, H), and the CON tendons (C, F, I) after remodeling in situ at 4 weeks postimplantation. Quantification of Mallory's trichrome blue staining (J) showed that the MIX and CON groups had a similar level of density, whereas in the DCN the level was decreased significantly (p < 0.01). Compared to the CON, polarizing light calculation revealed that more mature collagen fibrils were displayed in the DCN and MIX groups (K) (p < 0.01).
Discussion
In this study, we demonstrated that engineered tendon treated with lentiviral-encoded shRNA against decorin can enhance the regeneration of rat patellar tendons. Firstly, rat patellar tenocytes were genetically modified with lentiviral vectors encoding shRNA that efficiently downregulated both the mRNA and protein levels of decorin. Secondly, engineered tendons have been generated completely from shRNA treated tenocytes or a mixture of shRNA treated cells with untreated cells at a ratio of 1:1, as well as completely from untreated tenocytes. Thirdly, it was demonstrated that engineered tendons with mixture of treated and untreated cells exhibited the best results when utilized to repair injured patellar tendon. These data suggested that to some extent, downregulating the level of decorin could enhance tendon regeneration.
A previous study by Nakamura and colleagues demonstrated that downregulating decorin by injection of antisense oligodeoxynucleotides increased collagen fibril diameter (24). This is consistent with the results in this study. However, compared with local administration of oligodeoxynucleotides, gene therapy with lentiviral-mediated delivery of shRNA to tenocytes is more convenient and controllable. Moreover, it was found that aberrant giant diameter fibrils were generated when decorin was oversuppressed, characterized by abnormal morphology, which is consistent with a previous study on decorin knockout (8). Hence, a proper level of decorin expression is vital for collagen fibrillogenesis and injured tendon repair.
Collagen fibril assembly is composed of two phases. Firstly, procollagen forms intracellular, which is subsequently secreted outside the cell. The second phase takes place in the extracellular environment and involves conversion of procollagen to collagen by cleaving off the N- and C-peptides with each of the respective proteinases, followed by assembly of the fibrils through collagen fusion (6). Collagen type I is the chief component of collagen mass (97–98%) in tendon or ligament, whereas collagen type III and V are two minor components which are usually embedded within the major collagen fibrils (3,40,41). In our present study, we did not detect significant difference in the expression levels of collagen genes between control and decorin knockdown cells. This suggests that, at least at the 2D culture level, decorin suppression does not influence collagen production. Our results demonstrated that decorin downregulation improves collagen fiber assembly (the second stage, Fig. 5). Although previous study has shown that collagen type III and V play roles in regulating the diameter of fibrils by affecting the nucleation initiation of collagen assembly (41), our results imply that decorin knockdown may be directly augment collagen fiber assembly independent of regulation on collagen types III and V expression. However, it does not rule out that decorin may interact with collagen type III and V to influence their collagen fiber assembly.
Collagen deposition and its regular spatial arrangement are essential for wound tendon repair and regeneration. The poor packing and irregular collagen resulting from ablation of decorin would make it sensitive to the collagenase such as matrix metalloproteinase-13 (MMP-13) upregulated in wound tendon (8,21,25). This may explain less collagen production in the DCN group than that of the MIX group in our in vivo study. Thus, it suggests that partial inhibition of decorin expression is important for the yield of more compact collagen array yield.
It is well known that decorin not only function as an extracellular structural molecule but also to exhibit cell-signaling properties (14,19). Transforming growth factor-β (TGF-β), a general inhibitor of cell proliferation, has been reported to decrease the proliferation rate of tendon cells (18). Previous studies have reported that decorin binds TGF-β and neutralize its biological activities (18,19,22). In an attempt to examine whether decorin may regulate TGF-β transcription in tendon cells, we performed qPCR to compare the transcript levels of TGF-β between the control and decorin shRNA infected cells. No significant difference of this result suggests that decorin does not regulate the transcription of TGF-β at least in tendon cells. Based on previous evidence and our result, it thus suggests that inhibition of tendon cell growth rate by decorin downregulation may be through the release of TGF-β biological activities from neutralization by decorin but not through transcriptional upregulation of TGF-β.
While our study demonstrated the potential of decorin downregulation by shRNA-based gene therapy approach for tendon repair in a rat tendon injury model, there are limitations in the setting of the translational importance for human practice. Firstly, the isolation of low cell numbers of tenocytes requires the extended proliferation and passaging prior to transplantation in vivo. This poses a problem for tenocyte culture as tenocytes tend to lose their phenotype over passaging. However, in the case of rat patellar tendon cells, expression of ECM genes is relatively constant before passage 5 (32). So we used the cultured tenocytes from passage 0 to passage 4 in our study. Nevertheless, this may not be directly applied to human tenocytes since the phenotype of human tendon cells drifts rapidly. To circumvent the problem, human tenocytes can be cultured in a three-dimensional system to avoid dedifferentiation (1, 42). It needs further investigation to evaluate the effect of decorin downregulation on human tenocytes in the three-dimensional system. Secondly, the rat tendon injury model is relatively easy and does not always reflect the human practical injury, for instance, the injury occurred near the tendon–bone joint or the wound is not regular in shape, and so on. More clinically relevant animal models need to be created to evaluate the effect of decorin downregulation on tendon repair. Nevertheless, the limitations cannot cover the novelty of the present study, which lies in the following aspects: (1) Conceptually, our study tested the hypothesis that although downregulation of decorin is desired for tendon repair, the level of downregulation is an important parameter to achieve optimal outcome. (2) Technically, we address this using lentiviral shRNA-based stable knockdown and created the mixing strategy to control the level of decorin downregulation in a cell population. Our study paved the way for developing inducible decorin shRNA combined with the mixing strategy to further control the level and duration of decorin downregulation for optimal tendon repair. From a long-term perspective, this may lay a foundation towards developing gene therapy approach for tendon repair in the clinic.
In summary, we have demonstrated that the downregulation of decorin to a proper level by lentiviral-encoded shRNA can promote the repair and regeneration of patellar tendon. The DCN group exhibited the largest collagen fibril diameter, but associated with abnormal shape, whereas the MIX group manifested the best coordination of histology, more mature collagen deposition, and larger collagen fibril diameter. The present study may provide a new in vivo gene therapy method for tendon injury repair.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (81125014, 81071461, 31170943, 81101356, J1103603), Zhejiang Provincial Grants (Z2100086, 2009C13044, 2012C33112), Regenerative Medicine in Innovative Medical Subjects of Zhejiang Province (11-CX02), and Zijin Plan (S201100000640). The authors thank Wang Li for the TEM imaging. The authors declare no conflict of interest.
