Abstract
Digital ulcers (DUs) are a rather frequent and invalidating complication in systemic sclerosis (SSc), often showing a very slow or null tendency to heal, in spite of the commonly used systemic and local therapeutic procedures. Recently, stem cell therapy has emerged as a new approach to accelerate wound healing. In the present study, we have tentatively treated long-lasting and poorly responsive to traditional therapy SSc-related DUs by implantation of autologous adipose tissue-derived cell (ATDC) fractions. Fifteen patients with SSc having a long-lasting DU in only one fingertip who were unresponsive to intensive systemic and local treatment were enrolled in the study. The grafting procedure consisted of the injection, at the basis of the corresponding finger, of 0.5-1 ml of autologous ATDC fractions, separated by centrifugation of adipose tissue collected through liposuction from subcutaneous abdominal fat. Time to heal after the procedure was the primary end point of the study, while reduction of pain intensity and of analgesic consumption represented a secondary end point. Furthermore, the posttherapy variation of the number of capillaries, observed in the nailfold video capillaroscopy (NVC) exam and of the resistivity in the digit arteries, measured by high-resolution echocolor-Doppler, were also taken into account. A rather fast healing of the DUs was reached in all of the enrolled patients (mean time to healing 4.23 weeks; range 2-7 weeks). A significant reduction of pain intensity was observed after a few weeks (p < 0.001), while the number of capillaries was significantly increased at 3- and 6-month NVC assessment (p < 0.0001 in both cases). Finally, a significant after-treatment reduction of digit artery resistivity was also recorded (p < 0.0001). Even with the limitations related to the small number of patients included and to the open-label design of the study, the observed strongly favorable outcome suggests that local grafting with ATDCs could represent a promising option for the treatment of SSc-related DUs unresponsive to more consolidated therapies.
Introduction
Systemic sclerosis (SSc) is a connective tissue disease characterized by a complex pathological process where the main scenario is represented by progressive loss of the microvascular bed, with the consequent progressive fibrotic changes in involved organs and tissues (9,17). Raynaud's phenomenon, which occurs in around 90% of patients with SSc (8), can be considered as the clinical counterpart of the microcirculatory involvement of the fingers. Nailfold video capillaroscopy (NVC) is a simple and low-cost technique that allows one to carefully observe, just in the finger periungueal areas, these microvascular changes, starting from progressive capillary network rarefaction, with dilated or giant capillary formation, to an almost complete vascular desertification (6). The direct consequence of this process is the possible formation of painful digital ulcers (DUs) on fingertips. These events, which have been reported in 35-60% of patients (8,18,22,28), commonly lead to a relevant deterioration of the patient's quality of life, since fingertip ulcer formation and evolution is often accompanied by intense disabling pain and difficulty in performing daily living activities. Finally, their healing is frequently a long-lasting process requiring accurate and intensive topical and systemic treatment (22).
On the basis of their well-recognized regenerative and angiogenetic properties, cell therapy with autologous bone marrow-derived mesenchymal stem cells (BMMSCs) has been proposed and tested in different animal models and in some human pathological conditions characterized by peripheral ischemia and wound formation (2). Moreover, BMMSCs have also been employed with different modalities for the treatment of peripheral ischemic lesions in the course of SSc (5,10,19). The positive results of these observations have been ascribed to the multiple angiogenetic factors produced by BMMSC secretome, and to their capacity—as pluripotent cells—to differentiate into endothelial cells, or other cell types, able to somehow support the neoangiogenesis (2).
Adipose-derived stromal/stem cells (ASCs) are considered to be an attractive alternative to pluripotent cells with characteristics similar to BMMSCs. Compared with these latter cells, ASCs offer several advantages, including ease of isolation, less donor morbidity, relative abundance, and rapidity of expansion (26). Preliminary attempts with ASC cell therapy have been made to induce healing of ulcers developed in peripheral vascular diseases of some animal models and human disorders (21).
Recent data from our group have demonstrated that lip lipofilling with autologous adipose tissue-derived cell (ATDC) fractions, which are known to contain both ASCs and a stromal/vascular cell (SV) component (14), were effective in increasing the mouth opening capacity and, furthermore, in inducing local skin angiogenesis in patients with SSc (7). Starting from these encouraging results, we tried, in the present pilot study, to treat long-lasting DUs, developed in a small group of patients with SSc and having a very slow or no tendency to heal, by regional injection of autologous ATDCs. A prompt disappearance of local ischemic pain and a quite rapid DU healing was observed in all of the treated patients.
Materials and Methods
Patients
This pilot study was conducted from December 2012 to December 2013. Patients included in the study were over 18 years old, and all met the recently published classification criteria of the American College of Rheumatology for SSc (27). Furthermore, all of the patients were to have only one active DU (cardinal ulcer), over 5 months old prior to enrollment time. However, 86.7% of the selected patients had experienced other previous DUs during their disease course. DU was defined as a painful area, >2 mm in diameter, with visible depth and loss of dermis, not amenable to healing and in a site judged compatible with a vascular etiology, located at the volar surface of the digit, distal to the proximal interphalangeal digital crease. Previous long-term treatments with intravenous prostanoid, orally administered vasodilating drugs, that is, calcium channel blockers, angiotensin-converting enzyme (ACE) inhibitors, and traditional topical treatments for wound care, which included local medication and surgical removal of necrotic tissue, have been unsuccessfully carried out in all of the selected patients. Analgesics were also employed for the treatment of pain due to DUs. Exclusion criteria from the study were represented by the presence of severe extracutaneous involvement, concomitant treatment with immunosuppressive therapies (including prednisone equivalent >10 mg), and current therapy with dual and selective endothelin inhibitors. Patients suffering from diabetes and/or other vascular diseases and women currently pregnant or on lactation were also excluded from the study recruitment. All of the vasoactive therapies, that is, prostanoid infusion, ACE inhibitors, and calcium channel blockers, were discontinued at least 3 weeks before the beginning of the study. No additional therapy, with the exclusion of analgesics, was introduced after the treatment procedure and during the follow-up period.
The study was approved by the local ethical committee board and performed according to the criteria of the Helsinki declaration. A written informed consent to participate in the study was obtained from all of the enrolled patients.
Collection of Adipose Tissue
Autologous adipose tissue was harvested in each enrolled patient through liposuction from subcutaneous abdominal adipose tissue in the surgery room under local anesthesia, as previously described (7). Briefly, the collected adipose tissue was transferred into sterilized vials and processed at 920 x g for 3 min. The upper and lower phases, containing oil supernatant and mature adipocytes, and blood and plasma residuals, respectively, were then discharged, and only the intermediate layer, defined as ATDC fractions, containing both ASCs and a SV component, was used (14).
Implantation of Adipose Tissue
Under local anesthesia, a total amount of 0.5-1 ml of the autologous ATDC fraction was injected at the base of the affected finger, by sequential introduction of small aliquots in different directions from the injection site. That was done to spread the implanted ATDC fraction as widely as possible around the finger base. “Luer lock” syringes (Artsana SpA, Grandate, Como, Italy) and Coleman 18G cannulas (Mentor-Aesthetic, Santa Barbara, CA, USA) were used for the procedure. At the moment of needle removal, a small skin incision was made at the site of injection, to slightly reduce the intratissue pressure due to introduction of the additional material.
Clinical Outcome After Adipose Tissue Implantation
The primary endpoint of the study was the time to healing of the cardinal ulcers. Healing was defined as complete epithelialization of DU, regardless of residual pain.
The secondary endpoint was represented by reduction or complete disappearance of pain due to the DU. A patient's self-assessment of pain severity was performed by means of visual analog scale (VAS) (range 0-100, 100 indicating the most severe pain) and by recording the daily analgesic consumption prior to the procedure, for 3 days after the procedure, and thereafter, once a week up to complete resolution of pain or healing of cardinal ulcer. A clinical follow-up was continued for 6 months after the injection procedure, in order to verify the stability of the healing process.
Nailfold Video Capillaroscopy (NVC)
The same operator performed the NVC examination (with 200x magnification lens, by using Videocap Scalar Co. Ltd., DS MediGroup, Milan, Italy) in each patient. This was done from the second to the fifth finger of the hand involved, at baseline and 1, 3, and 6 months after the therapeutic procedure. In every finger, images from four consecutive fields, for a total extension of 1.2 mm, taken in the middle of the nailfold, were analyzed and stored by using dedicated software. The classical NVC features were analyzed, that is, the presence of enlarged and giant capillaries and hemorrhages, reduction in the number of capillaries, disorganization of the vascular array, and presence of ramified/bushy capillaries. A previously suggested classification of the different NVC patterns was adopted, and each case was defined as having an “early,” “active,” and “late” NVC pattern (6). Furthermore, by analyzing the stored images, two independent observers counted the number of capillaries present in the cuticle distal row of each finger, taking into account the stored images that included 1.2 mm of nailford capillary bed per finger. The sum of the number of capillaries counted in the four fingers constituted the reference parameter we considered in the final statistical analysis. The two observers demonstrated to have a high interrater agreement (weighted Cohen's k > 0.75). In the discordant cases, the average of the two observer counts was considered for statistical analysis.
Echocolor-Doppler Examination of the Finger Arteries
A high-resolution echocolor-Doppler examination of arterial vasculature of the hand, including the palm arch and the digital branches, was performed in each patient by the same experienced operator (G.D.L.) (Aplio SSA-700A, Toshiba Medical System Europe, Zoetermeer, The Netherlands). The insonation was obtained by using a linear probe of 12 MHz under an angle beam of 60° with respect to the major axis of the explored vessel, maintaining the same pulse repetition frequency (PRF) value and the velocity scale during all of the measurements. The examination was performed immediately before the procedure (T0) and 1, 3, and 6 months (T1, T3, T6) after the treatment. Diastolic and systolic maximum velocities (V) were recorded and used to calculate the resistivity index (RI) (peak systolic V - end diastolic V/peak systolic V).
An additional echocolor-Doppler examination was also performed immediately after the implantation procedure to check the integrity of the whole regional arterial ramification and, namely, of the digital branch of the treated finger.
Statistical Analysis
Data collected before and after the therapeutic procedure were analyzed by using paired t-tests and Wilcoxon tests for continuous and discrete variables, respectively. When multiple tests were performed, we corrected the level of significance for the number of tests, according to Bonferroni's method. Therefore, taking into account the number of tests performed in each analysis, only p < 0.008 was considered as significant.
Results
Baseline Characteristics of Patients
Fifteen patients were included in this study. Among these, eight patients had the limited and seven patients the diffuse variant of the disease, according to the currently adopted classification (17). The median age of the enrolled patients was 51.9 (range 40-66). Other main demographic and clinical characteristics of the enrolled cases, including the DU localization, diameter, and duration, are summarized in Table 1.
Clinical Characteristics and Outcome of Patients Treated With Autologous Adipose Tissue

For abbreviations, see text.
Clinical Results
Ulcer healing was observed in all of the patients of the study who underwent the treatment (Fig. 1). The mean time to healing of the cardinal ulcer was 4.23 weeks (range 2-7) (Table 1). The result was maintained during the following 6-month period, and no new DUs appeared in any one of the treated patients. One week after transplantation, pain in the treated fingers was slightly relieved, while significant pain relief occurred during the next weeks of observation (with respect to the baseline value), VAS was significantly lower at 1 month (T1) (Wilcoxon signed ranks test Z value 3.43, p < 0.001), 3-month (T3) (Z value 3.42, p < 0.001), and 6 month (T6) (Z value 3.44, p < 0.001) assessments (Fig. 2). Similarly, the amount of analgesic drugs needed to obtain pain control drastically decreased after 1 week, and none of the patients were still taking analgesics 1 month after the implantation treatment.
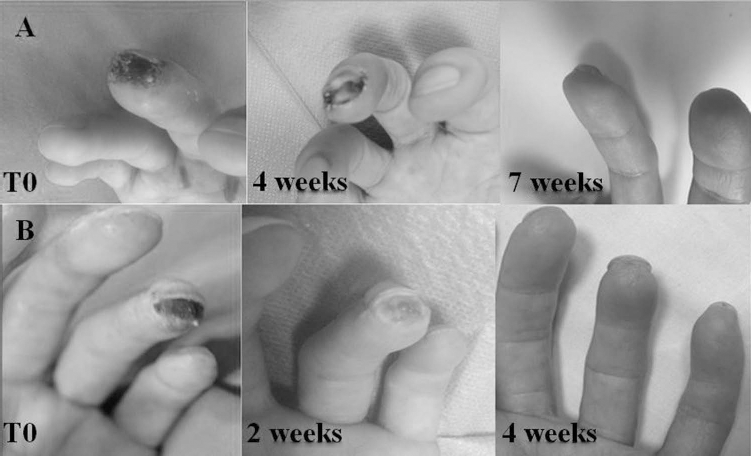
Digital ulcer trend after autologous ATDC treatment. Digital ulcer progressive healing of patient 4 (a) and patient 11 (b) after autologous ATDCs.

Changes in pain severity after autologous ATDC treatment. Changes in pain severity assessed by visual analog scale (VAS, ranging from 0 to 100) after autologous ATDCs treatment. All of the treated patients experienced a significant improvement during the weeks of observation.
No difference was observed in the disease course or in the healing time between the two patient subsets (patients with limited and patients with diffuse variants of the disease).
No adverse event associated with adipose treatment, such as local inflammatory reactions or sepsis, bleeding, and graft rejection reaction, was observed in the treated patients. No variation of digital artery structure and related perfusion was observed by the echocolor-Doppler examination after the therapeutic procedure.
NVC Features
The microvascular architecture in the treated patients was characterized at baseline by a wide vascular disorganization with abnormalities of the capillary loops that are typically associated with scleroderma pattern. Particularly, all the treated patients exhibited a capillaroscopic late pattern (6) (Fig. 3A). One month after the ATDC fraction implantation, even if the capillaroscopic features could be still defined as belonging to the “late” pattern, a statistically significant increase in the capillary count was observed (mean increase of capillaries 3.13, paired t-test value 7.81, p < 0.0001) (Fig. 3A). With respect to the baseline value (T0), the number of capillaries was also significantly higher at 3-month (T3) and 6-month (T6) assessments (mean increase of capillaries 5.06, paired t-test value 8.84, p < 0.0001, and 5.00, paired t-test value 9.05, p < 0.0001, respectively). The number of capillaries at T3 and T6 were also significantly higher compared to T1 (mean increase of capillaries 1.93, t-value 4.88, p < 0.0002, and 1.86, t-value 5.55, p < 0.0001, respectively) (Fig. 3B).

Changes in vascular features after autologous ATDC treatment evaluated by nailfold video capillaroscopy (NVC). (A) A progressive increase in the number of capillaries is shown starting from a complete capillary desertification present at baseline (patient 11 is shown). (B) The capillary count was significantly increased at 1-month (T1), 3-month (T3), and 6-month (T6) assessment with respect to baseline. The number of capillaries at T3 and T6 were also significantly higher compared to T1. The total number of capillaries that was considered for the analysis was derived by the sum of capillary loops counted in the stored NVC images taken in the four fingers considered (2-5 fingers of the treated hand). A total of four consecutive images were stored from each digit that corresponds to a cumulative area of 1.2 mm of nailfold capillary bed per finger.
As expected, most of the new capillaries we observed by NVC were constituted by the new loops that appeared in the treated finger. At the 3-month observation time, we found 41-88% new capillaries (mean 60.7%) in all the 15 patients we treated.
High-Resolution Echocolor-Doppler
No anatomical or functional abnormalities in the digit artery of the treated fingers were observed at the echocolor-Doppler examination performed immediately after the implantation of adipose tissue.
When the RI values obtained in the treated digit artery of the patients before and after the therapeutic procedure were compared, we observed a significant reduction of these values at T1, T3, and T6 in comparison with T0 (p < 0.0001 in all cases). No significant differences were noticed between values recorded at T1 with respect to T3 and T6, and at T3 with respect to T6 (Fig. 4).
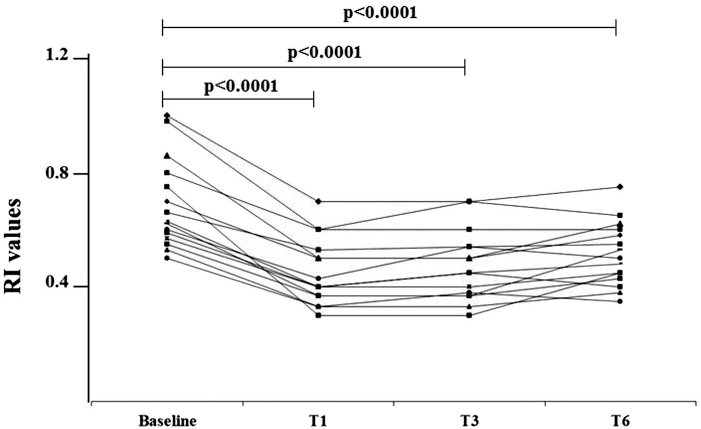
Changes in local vascularization after autologous ATDC treatment. The high-resolution echocolor-Doppler examination of the treated digit artery of the patients showed a significant reduction of RI values at T1, T3, and T6 in comparison with T0. No significant differences were noticed between values recorded at T1 with respect to T3 and T6 and at T3 with respect to T6.
Discussion
DUs are a rather frequent complication in patients with SSc, and namely in those presenting an almost complete distal capillary bed desertification, which may favor the formation of necrotic areas (8,18,22,28). These DUs are often painful, invalidating, and may lead to a severe deterioration of the quality of life, since their trend to healing is frequently very slow or null, despite the multiple local and general therapeutic measures one can adopt (22).
In the present pilot study, we successfully treated long-lasting DUs showing no tendency to improve or heal, with a totally new experimental procedure based on regional implantation of the ATDC fraction, that is, the one containing both ASCs and a SV component (14). In all of the enrolled patients, DU healing, together with a parallel rapid resolution of DU-related pain, was recorded in a few weeks. In addition, a significant increase in the number of capillaries was observed at the NVC examination in all of the posttherapy follow-up observations. Finally, the angiogenetic effect of this kind of treatment was indirectly confirmed by the reduction of RI in the digital arteries of the treated fingers after the adipose tissue implantation.
The present data confirm those obtained in our previous experience, where the perioral injection of the same ATDC fraction in patients with SSc induced not only a significant increase in mouth opening capacity, but also a histologically and NVC-proven new capillary formation in the local skin area (7).
This is, of course, an open-label pilot study, and the results herein reported involved only a small number of patients. Hence, there is a need for them to be confirmed by involving a large number of similar cases, possibly in controlled studies. However, it is worth noting that all the patients enrolled in the present study had long-lasting DUs, which did not improve or heal, despite being treated with traditional therapeutic measures from the very start onward (25).
Given the unfavorable clinical situation of the patients we treated with this new therapeutic procedure, the results, that is, DU healing and significant improvement of DU-related pain in all of the patients, can certainly be regarded as extremely positive and noteworthy.
On the other hand, our data are not completely surprising, since similar positive results have been reported in other ischemic conditions in humans and in animal models with different modalities of ATDC therapies. Bura et al. (3) recently reported a certain degree of wound improvement and healing obtained after intramuscular injection of isolated ASCs in the ischemic leg in a small group of patients with lower limb ulcers not suitable for revascularization. Lee et al. (16) obtained comparable results in another small series of patients by applying a similar protocol. Similarly, other authors observed an acceleration of excisional wound healing in diabetic and nondiabetic rats by locally administered ASCs (13,20). Furthermore, foot ulcers have been reported to have a higher prevalence of healing, and in a shorter period of time, in diabetic patients locally treated with differently processed adipose tissue cellular fractions, with respect to traditionally treated controls (3,11).
Apart from the present one, no studies have been so far reported where ATDC fractions, or purified ASC components, have been employed to tentatively treat DUs in patients with SSc.
Conversely, few studies have been performed on the therapeutic effects of BMMSC local or regional transfer in patients with SSc suffering from ischemic lesions in their fingers or limbs. Even if these studies were based on the observation of single patients, results show that the adopted procedures have constantly induced an improvement in ischemic lesions (5,10,19).
The reason why local implantation of ATDC fractions containing ASCs and SV components, as well as similar procedures with BMMSCs, may induce such positive changes in DUs of fingertips of patients with SSc remains at the moment almost completely speculative. A number of studies have shown that ASCs, as well as BMMSCs, are multipotent cells and that these cells are potentially able to differentiate themselves into other cell types, like cells of the mesoderm (condrogenic, osteogenic, myogenic), ectoderm (neurogenic), and endoderm (endothelial) lineages, under the conditioning of different stimulants (14). As demonstrated in a number of in vitro and in vivo studies [reviewed in Kokai et al. (14)], ASCs are also capable, when locally implanted, of producing a number of growth factors with proangiogenetic and proliferation-inducing factors (such as vascular endothelial growth factor and fibroblast growth factor) that may favor local angiogenesis by exerting their complex paracrine action (23). In addition, ASCs may also directly produce or stimulate the production from other resident cell types of a number of cytokines with immunomodulating action (like IL-6, IL-7, IL-8, IL-10, IL-11, TGF-β) probably capable of downregulating some pathological mechanisms in injured tissues (14,23). Furthermore, ASCs have surface receptors for growth factors and chemoattractants released by damaged tissues, like epidermal growth factor and monocyte chemoattractant I, that may induce migration and engraftment of these cells into the site of tissue injuries (1). Finally, both BMMSCs and ASCs have shown, either in in vivo or in vitro studies, to better express their angiogenetic properties when exposed to hypoxic conditions (4,29).
The SV component of the medium layer of adipose tissues separated by centrifugation contains a heterogeneous population of many cells other than ASCs that includes preadipocytes, endothelial cells and their progenitors, pericytes, hematopoietic lineage cells, and fibroblasts. Since isolation, purification, and in vitro culture of ASCs from SV fractions require a significant series of potentially dangerous cell manipulations, most of the studies conducted with ATDCs in humans have employed the whole fraction. At the present time, little information is available on the possible contribution given by the SV component in the regenerative effects that ATDCs have shown under many different conditions (15). Of particular interest is the recent observation that pericytes may give a very important contribution in tissue repair and revascularization, since these cells may work as driving stromal cells in tubuli formation during angiogenesis (24). So, one cannot exclude that SV, where pericytes are certainly present, might play an important role even in the healing of DUs in our patients. Such an assumption is further corroborated by recent data, which show the higher angiogenetic potential of freshly isolated ATDC fractions where the SV component and, namely, progenitor endothelial cells, is largely represented, when compared to cultured ATDC fraction where the SV component is reduced (12).
The multilineage differentiation potential of ASCs, the possible support to angiogenesis given by the SV component, and the local release of a number of grown factors and cytokines, able to stimulate the angiogenesis, may all have contributed to the formation of new capillary loops, and to the rapid DU healing observed in the present study. The migration capacity of ATDCs and the paracrine effects of the released angiogenetic factors may also explain why, besides in the treated digit, a significant number of new capillaries were even found in the other examined fingers.
The present study suggests that autologous transplantation of ATDC fractions may be a successful option for inducing improvement or healing in ischemic SSc-related DUs that are resistant to more traditional therapeutic approaches. The positive outcome obtained with this kind of therapeutic procedure in the limited clinical experience reported here certainly requires confirmation in larger and controlled studies.
Footnotes
Acknowledgments
The authors declare no conflicts of interest.
