Abstract
Transplantation of cryopreserved ovarian tissue is a novel technique to restore endocrine function and fertility especially for cancer patients. However, the main obstacle of the technique is massive follicle loss as a result of ischemia in the process of transplantation. Mesenchymal stem cells (MSCs) have been acknowledged to play an important role in supporting angiogenesis and stabilizing long-lasting blood vessel networks through release of angiogenic factors and differentiation into pericytes and endothelial cells. This study is aimed to investigate whether MSCs could be applied to overcome the above obstacle to support the ovarian tissue survival in the transplantation. Here we show that human MSCs could enhance the expression level of VEGF, FGF2, and especially the level of angiogenin, significantly stimulate neovascularization, and increase blood perfusion of the grafts in the cryopreserved ovarian tissue transplantation. Further studies reveal that MSCs could notably reduce the apoptotic rates of primordial follicles and decrease follicle loss in the grafted ovarian tissues. In summary, our findings demonstrate a previously unrecognized function of MSCs in improving human ovarian tissue transplantation and provide a useful strategy to optimize fertility preservation and restoration.
Keywords
Introduction
Great progress in malignant treatments, such as chemotherapy and radiotherapy, has markedly increased long-term survival rate in female patients of reproductive age (22,30). However, such treatments unavoidably lead to premature ovarian failure and infertility (4,18). As a consequence, ensuring reproductive capacity after cancer treatment has become a major concern (9,51). Several options are presently available to pre serve fertility, including the cryopreservation of embryos, oocytes, and ovarian tissues (23,32,49). Owing to the requirement for controlled ovarian hyperstimulation prior to oocyte collection, embryo or oocyte cryopreservation is not advisable in malignant cases, since it may postpone the anticancer treatments (31). Thus, ovarian tissue cryopreservation has been supplied as a promising approach to restore endocrine function and fertility, especially for cancer patients (11,37,44). Worldwide, 24 live births have been reported after transplantation of frozen/thawed ovarian tissues (11). Even though the technique is still limited, mostly in that as a result of postgrafting ischemia the graft loses more than two thirds of primordial follicles during revascularization, which compromises considerable reduction in fertility restoration and explains the occasional success of this procedure currently (7,13,41,53). Therefore, minimizing the ischemic injury of the ovarian tissues by decreasing the latency period before neovascularization establishment would maximize reproductive potential for future fertility. Previous studies demonstrated that, other than cotransplanting ovarian endothelial cells (7), the requirement for suit able grafted locations (8,48) and restriction of ovarian tissue thickness (45) or size (38) were conducive to a well-vascularized formation in ovarian tissue transplantation. In addition, angiogenic factors, such as vascular endothelial growth factor (VEGF) (16,26), granulocyte colony-stimulating factor (46), sphingosine-1-phosphate (47), fibroblast growth factor 2 (FGF2) (17), and adenosine agonist (19), were proven beneficial to the enhancement of neoangiogenesis and follicle survival in various species-derived ovarian tissue transplantation. However, because of a short half-life and easy degradation, the above factors were merely used in experimental procedures and are difficult to be practically applied (47).
In the recent years, mesenchymal stem cells (MSCs) have been extensively studied and acknowledged to play an important role in tissue regeneration (15), wound repair (3), maintenance of tissue homeostasis (52), and vascularization (24). Under appropriate conditions, MSCs could become activated, acquire endothelial phenotype (6), differentiate into pericytes (2,5,15) or endothelial cells (29), and incorporate into blood vessel walls (2,29), which support neovascularization and stabilize long-lasting blood vessels. Furthermore, a lot of research indicated that MSCs were capable of secreting various angiogenic factors, such as VEGF (20,40,42,54), angiogenin (20,40), FGF2 (20), HGF (54), and IL-6 (20,21), and induced recruitment of vascular endothelial cells to establish a microenvironment for vascular formation.
Owing to the angiogenic promotion function of MSCs, as well as their technical convenience, poor immunogenicity, genetic stabilization, and out of ethical controversy, which together contribute to the safety in clinical use (52), it may be speculated that MSCs could be applied to fertility preservation and improve the grafted tissue survival through enhancing angiogenesis and blood supply. Based on the above hypothesis, this study was aimed to investigate the role of bone marrow-derived MSCs in the ovarian cortex transplantation. Here we reported that, when transplanted with ovarian cortex tissues, MSCs could stimulate neovascularization, enhance VEGF, FGF2, and angiogenin expression, and increase functional blood perfusion of the grafts, and thus decrease apoptotic rates of primordial follicles and follicle loss. This study not only presents a novel function of MSCs in enhancing neoangiogenesis and follicle survival in human cryopreserved ovarian cortex transplantation, but translation of this work to reproductive medicine has significant implications for fertility preservation.
Materials and Methods
Isolation and Characterization of MSCs
Human MSCs were isolated and verified from the bone marrow of five individuals (three males, ages: 28, 34, and 31, and two females, ages: 39 and 27) undergoing bone marrow harvest for allogeneic bone marrow transplantation after obtaining informed consent according to institutional guidelines under the approved protocol as we described previously (55).
Ovarian Tissue Collection and Preservation
The use of human ovarian tissues was reviewed and approved by the ethics committee of Peking University (registration number: 2009005). After obtaining oral and written informed consents, ovarian tissues were obtained from a consenting 21-year-old female-to-male transsexual. Tissues were transported from the operating room to the laboratory in Leibovitz's L-15 Medium (L-15; Invitrogen, Carlsbad, CA, USA) supplemented with 1% human serum albumin (HSA; Life Global, Guilford, CT, USA), 100 IU/ml penicillin, and 100 μg/ml streptomycin (Life Technologies, Inc., Grand Island, NY, USA) soon after the surgery. After enucleating medulla with surgical scissors and scalpels, the cortices were pared down manually to ultrathin translucent shells with the thickness of ≤1 mm and cut into small pieces of 6.25 mm2 (2.5 × 2.5 mm). Then ovarian cortices were slow frozen and thawed using ethylene glycol (EG; Sigma-Aldrich, St. Louis, MO, USA) as a cryoprotectant, according to the protocol described by Andersen's group (14). Briefly, the ovarian cortices were transferred to 30 ml of 0.1 M sucrose (Sigma-Aldrich), 1.5 M EG, and 10% HSA in DPBS (Life Technologies, Inc.), and equilibrated for 30 min at 4°C. They were then stored in cryovials containing 1 ml of cryoprotectant and cooled in a programmable freezer (Kyro 360-1.7; Planer Inc., UK) with the following program: 2°C/min to -9°C, 5 min of soaking, then manual seeding for ice crystal nucleation induction, 0.3°C/min to –40°C, 10°C/min to -140°C, at which temperature the cryovials were plunged into liquid nitrogen for storage. As for thawing protocol, the cryovials were immersed in a water bath at 37°C with gentle shaking. Cortices were sequentially transferred to the following three thawing liquids: i) 10 ml of 0.25 M sucrose and 0.75 M EG in DPBS, ii) 10 ml of 0.25 M sucrose in DPBS, and iii) 10 ml DPBS, in which they were equilibrated respectively for 10 min at room temperature.
Transplantation Procedure
All animal experiments were approved by the Institutional Animal Care and Use Committee of Peking University Third Hospital. Thirty-six 8-week-old female athymic BALB/c mice (Animal Center of Medical College of Peking University) were used for the study. The mice were anesthetized by intraperitoneal injection of 2,2,2-trobromoethyl alcohol (Sigma-Aldrich) and tertamyl alcohol (Sigma-Aldrich). Two subcutaneous skin incisions were made on the lower third of abdominal wall on both sides of each mouse after sterilizing the area. Then an appropriate interspace between the skin and subcutaneous tissue was built. The prepared ovarian fragments were randomly picked and xenotransplanted into the space with the cortical side adhering to the skin and the medullar side adhering to the fascia. Immunosuppression was not performed in the study. The experiment was divided into four groups: (1) ovarian group, ovarian fragments transplanted alone; (2) ovarian (M) group, ovarian fragments transplanted with 10 μl GFR Matrigel (BD Bioscience, Bedford, MA, USA); (3) ovarian (M) + FGF2 group, ovarian fragments transplanted with 100 μg/ml FGF2 (Invitrogen) that was diluted in 10 μl GFR Matrigel; and (4) ovarian (M) + MSC group, each ovarian fragment transplanted with 5 × 105 MSCs packaged in 10 μl GFR Matrigel. The skin incisions were then closed with absorbable 5/0 Prolene (Ethicon, Somerville, NJ, USA). Each mouse received the same treatment in both sites. All procedures were performed under aseptic conditions.
For a better understanding of the study, Figure 1 shows the experimental design of the entire study.

The model of experimental design for the study. The image shows the scheme and detailed procedure of the study. MSCs were isolated from fresh human bone marrow tissue. Thawed ovarian cortex tissues obtained from a 21-year-old female-to-male transsexual were randomly picked and xenotransplanted into the subcutaneous area of the abdominal wall in female athymic mice. The experiment was divided into four groups (
In Vivo Graft Perfusion Measurement
To evaluate the blood perfusion of the grafts at indicated time points, the infrared camera (SC620; FLIR Inc., Goleta, CA, USA) with a high resolution of 640 × 480 and thermal sensitivity of 0.06°C was employed to capture both thermal responses (i.e., infrared thermography) and positions (i.e., optimal image) of the region of interest (ROI) as described previously (1). All measurements were noninvasive and conducted in a controlled room with ambient temperature of 25 ± 0.5°C and constant humidity of 50 ± 10%. The thermal imaging procedure for the steady-state thermal mapping of ROI included two primary steps: (1) keeping the ROI uncovered and resting for about 5 min and (2) then capturing the ROI's thermal and optical image. The average temperature of ROI was calculated using MATLAB 7.0 (MathWorks, Natick, MA, USA) to identify the difference among different groups. Here ROI was approximated as a circular region of each graft whose center coordinates and radius could be manually determined by the corresponding optical image. The highest temperature of each mouse's surface was set as the reference value.
Graft Recovery and Histological Evaluation
The animals were euthanized by cervical dislocation after 3, 7, and 21 days (
Immunohistochemistry
The newly formed blood vessels in grafts were investigated using anti-CD31 antibody (1:100 dilution; Abcam, Cambridge, UK). IHC staining against anti-active caspase 3 (1:200 dilution; Abcam) was performed to evaluate follicle apoptosis in the grafted tissues in different groups. The detailed procedures were previously described (17). Brown coloring of the cytoplasm/nucleus of cells was specified as positive staining (otherwise as negative staining). The positive-staining follicles and the total number of follicles were counted at high-power field magnification (400×) at five randomly selected fields in 25 different sections in each tissue sample. CD31 staining blood vessels were counted at 200-fold field at five randomly selected positions in 25 different sections in each tissue sample. Human thawed ovaries (female, age: 21) were used as a positive control for AC-3 immunostaining, and human fetal villi tissues (8-week-old fetal) were used as a positive control for CD31 immunostaining. For negative control, the primary antibody was omitted. The results of the immunostaining were evaluated by a person who was blinded to the condition.
Western Blot
Parts of the recovered grafts in each group were homogenized with lysis buffer (CW Biotech, China) and protease inhibitor cocktail (Sigma-Aldrich). The lysates were cleared by centrifugation, and the supernatants were collected. The cell extracts were resolved by 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes (Millipore, Darmstadt, Germany). The membranes were blocked by 5% nonfat milk (Life Science, BD) overnight at 4°C. Then the membranes were incubated with the indicated antibodies [rabbit monoclonal anti-VEGF antibody, 1:1,000 (Abcam); rabbit polyclonal anti-FGF2 antibody, 1:500 (Abcam); mouse monoclonal antiangiogenin antibody, 1:250 (Abcam)]. After washing in Tris-buffered saline with 0.05% Tween-20 (ZSGB-Bio Company, China), the membranes were incubated with the corresponding peroxidase-conjugated secondary antibodies (ZSGB-Bio Company) at indicated dilutions (goat anti-rabbit antibody, 1:5,000; goat anti-mouse antibody, 1:5,000). The peroxidase activity was visualized with an enhanced chemiluminescence (ECL) kit (Thermo, USA).
Statistics Analyses
All experiments were repeated three times unless otherwise specified. Analyses were carried out using the SPSS 13.0 program software (SPSS, Inc., Chicago, IL, USA). Comparison of microvascular density, temperature difference, number of primordial follicles, and percentage of AC-3-positive follicles among each group was performed by ANOVA analysis followed by pairwise comparison test using LSD method. The Pearson correlation was applied to determine the relationship between follicle number and the temperature differences. A value of
Results
MSCs Promote Neoangiogenesis in the Grafts
As was shown in Figure 2A, there was a generally time-dependent increase in microvascular formation in all of the four groups. At day 3, the microvascular density in the ovarian (M) + MSC group was 3.8 ± 0.9, which was higher than the ovarian group with 1.9 ± 0.4 (
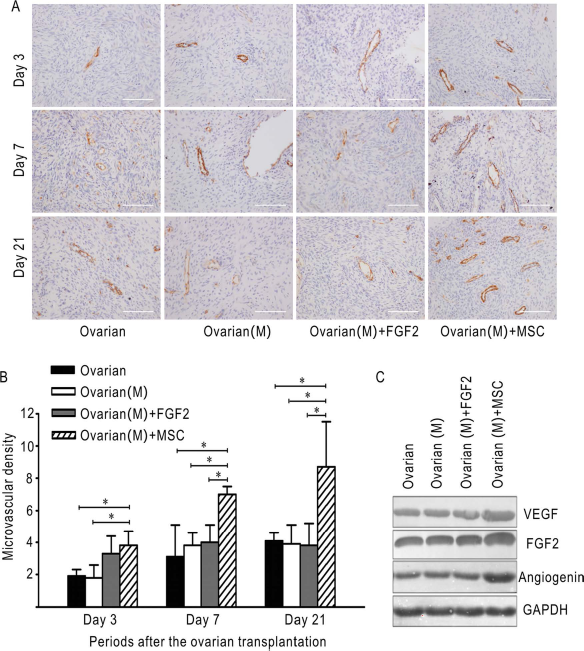
MSCs promote neoangiogenesis and enhance the expression of VEGF, FGF2, and angiogenin in the grafts. (A) Representative images of CD31 immunohistological staining of the grafts at days 3, 7, and 21 in the four groups. Brown coloring was specified as positive staining. (B) The microvascular densities as evaluated by CD31 staining in (A) at indicated time points in the four groups were expressed as mean ± SD in three independent experiments. Scale bar: 100 μm; *
MSCs Increase the Blood Perfusion of the Grafts
It is acknowledged that the temperature of the local area could be applied as an effective index for blood perfusion evaluation of the area of interest (43,50). Therefore, the in vivo infrared thermography measurement was performed to investigate the effect of MSCs on the blood perfusion of the grafts. Since several factors (e.g., the environmental temperature, humidity, and the status of each mouse) may influence the mea surement, the temperature difference between the graft (i.e., ROI), and the highest surface temperature of each mouse was calculated to represent the relative temperature of each graft. The representative results of infrared thermography are shown in Figure 3A. At day 3, there were no significant differences in the temperature among the four groups (

MSCs increase the blood perfusion of the grafts. (A) Representative images of infrared thermography at days 3, 7, and 21 in the four groups. White dotted circular lines in each mouse represent the region of each graft whose center coordinates and radius could be manually determined by the corresponding optical image. (B) The temperature differences (between the graft, i.e., ROI, and the highest surface temperature of each mouse) evaluated by infrared thermography in (A) at indicated time points in the four groups were expressed as mean ± SD in three independent experiments; *
MSCs Reduce the Primordial Follicles’ Apoptotic Rates in the Grafts
To investigate the impact of MSCs on follicle survival, active caspase 3 (AC-3) was used as a marker of follicular apoptosis. The representative images of the ovarian tissue immunohistochemistry staining at day 21 after the transplantation are shown in Figure 4A. At day 3, the percentage of apoptotic primordial follicles in the ovarian (M) + FGF2 (14.1 ± 2.4%) and ovarian (M) + MSC groups (12.6 ± 3.7%) were slightly lower than that in the ovarian (17.6 ± 4.7%) and ovarian (M) groups (16.8 ± 1.4%). However, the differences were not statistically significant among the four groups (

MSCs reduce the apoptotic rates of primordial follicles in the grafts. (A) Representative images of AC-3 immunohistological staining of primordial follicles at day 21 in the four groups. Brown coloring of the cytoplasm/nucleus of cells within follicles was specified as positive staining (white arrows). (B) Percentage of AC-3-positive staining primordial follicles at indicated time points in the four groups was expressed as mean ± SD in three independent experiments. Scale bar: 50 μm; *
MSCs Prevent the Loss of Primordial Follicles in the Grafts
The primordial follicles in the four groups were shown by H&E staining, and the representative images are shown in Figure 5A. As shown in Figure 5B, at day 3, the number of primordial follicles in the four groups were 51.3 ± 5.5, 50.3 ± 3.2, 52 ± 4.6, and 57.7 ± 8.5, respectively, which showed no significant difference among the four groups (

MSCs prevent primordial follicle loss that is negatively correlated with the temperature differences in the grafts. (A) Representative images of primordial follicles stained with H&E at days 3, 7, and 21 in the four groups. Scale bar: 50 μm. (B) The number of primordial follicles evaluated by H&E staining in (A) at indicated time points in the four groups was expressed as mean ± SD in three independent experiments; *
Discussion
Over the recent years, ovarian tissue transplantation has become a promising option to preserve fertility for patients with not only cancer but also benign diseases (12). According to the transplantation sites, there are currently two main techniques, including orthotopic (13) and heterotopic transplantation (10,37). Although the orthotopic transplantation is supposed to mimic the most favorable environment, the procedure requires abdominal surgery, which is technically complex and may increase unnecessary injuries to the body. Heterotopic transplantation is an alternative option because of the decreasing risk of surgery, rapid restoration of endocrine function, and the possibility of monitoring and retrieving oocytes (17). Of the different heterotopic sites, the subcutaneous area of the abdominal wall, which was proven to be the most preferable option for follicular development (28,37), was selected in this study.
According to the previous investigations, the initial revascularization of human ovarian xenografts occurred within 3 to 5 days after the transplantation (48). Therefore, three representative time points including days 3, 7, and 21 were selected to analyze the early, middle, and late effects of MSCs on angiogenesis. It is acknowledged that the temperature could be applied as an effective index for the functional blood perfusion of the region of interest (43,50). Therefore, the noninvasive in vivo infrared thermography was introduced to investigate the effect of MSCs on the blood perfusion of the grafts. Since several factors (e.g., the environmental temperature, humidity, and the status of each mouse) may influence the measurement, the temperature difference between the graft (i.e., ROI) and the highest surface temperature (i.e., liver area) of each mouse was calculated to represent the relative temperature of each graft. It is conceivable that a smaller temperature difference of a certain graft means it is closer to the highest surface temperature. That is to say, the smaller temperature difference of a certain graft represents higher temperature and better blood perfusion than others. In the current study, although MSCs had a significant induction in angiogenesis at day 3, blood perfusion evaluated by infrared thermography was not significantly enhanced by MSCs at this early stage. The initial increased blood perfusion by MSCs occurred at the middle stage of day 7. The most possible explanation for the delayed blood perfusion improvement by MSCs could be that the new microvascular network at the early stage did not acquire the function of blood supply. After the maturation for certain periods, the newly generated blood vessels could then support blood perfusion and thus increase the relative temperature of the grafts. Additionally, at day 7, the increase in follicle number by MSCs paralleled with the functional blood perfusion, but not with histological angiogenesis. Therefore, we speculate that MSCs improve follicle survival when the increased microvascularization only becomes functional after the graft has already recovered from ischemia. In our preliminary study of mouse fresh ovarian tissue transplantation, we found FGF2 significantly enhanced angiogenesis at day 7 (17). However, in the human cryopreserved ovarian transplantation, FGF2 induced a transient effect on angiogenesis, and the relatively increased microvascular density at day 3 was not maintained at the middle and late stages. The different species and graft size are probably the main causes that contribute to the different results in the angiogenesis by FGF2 between these two studies. Additionally, other differences between the two studies, such as the procedure of ovarian cryopreservation and thawing and grafting sites, may also account for the different responses to FGF2.
In contrast to the transient effect of FGF2, MSCs induced a long-lasting angiogenesis in the ovarian cortex transplantation. More importantly, this increased angiogenesis was followed by the persistent improvement of blood perfusion. The positive angiogenesis by MSCs in this study was consistent with the previous findings by Au et al. (2) and Koike et al. (25). One of the possible mechanisms by which MSCs promote vascular formation is that MSCs establish an angiogenic microenvironment by persistently secreting bioactive molecules to function as “drugstores” surrounding the graft. It was acknowledged that MSCs were able to secrete a series of coordinated angiogenic factors, such as VEGF (20,40,42,54), angiogenin (20,40), FGF2 (20), HGF (54), and IL-6 (20,21). In this study, the expressions of VEGF, FGF2, and especially angiogenin in the grafts were increased by MSCs. These factors could all be the vital boosters that have already proven to be conducive to angiogenesis (27,33,35). These may be interpreted as partly demonstrating the superiority of MSCs over one certain factor in promoting a long-lasting vascular formation and improving blood perfusion in human cryopreserved ovarian cortex transplantation. There are other possibilities that should be considered as well. First, following the previous findings (2,5,29), we can anticipate that MSCs may differentiate into pericytes or endothelial cells, thus providing essential cellular components to stabilize the nascent blood vessels in the transplantation. Second, MSCs may also have antiapoptotic effect on endothelial cells, which ensures the functional integrity of blood vessels. In the future, we will verify the above possibilities.
During the periods of the study, no significant toxic symptoms or signs of systemic toxicity (nausea, vomiting, fever, dyspnea, etc.) were observed after MSC treatment. Although previous studies indicated MSCs were relatively safe in clinical use and nearly no significant adverse effects had been reported (52), it was worthwhile to carry out detailed experiments to evaluate whether there was some long-term potential toxicity before its clinical application in ovarian tissue transplantation in the future.
In the present study, most follicles were identified as primordial follicles, while hardly any growing or antral follicles were observed. This phenomenon could possibly be ascribed to the following reasons. First, growing and antral follicles are more sensitive to cryoinjury, whereas primordial follicles can survive better (34). Second, according to the previous studies, there was a trend that a greater number of primordial follicles were observed in the young compared to the old counterparts (39). Therefore, the ovarian tissue we used in this study, which was obtained from the 21-year-old transsexual, exhibited mostly primordial follicles. Additionally, the structure of all grafts from the patient was relatively intact and no serious necrosis or damage was observed, and we considered that they were useful for the experiments to evaluate the effect of MSCs on angiogenesis and follicle survival. It should be pointed out that follicle development is a lengthy and complicated process, in which different stages require different gradients; for example, follicle-stimulating hormone (FSH) is necessary for the antral follicle development (36). We could therefore speculate additional substrates, multiple growth factors, and hormones, other than cotransplantation with MSCs, for sufficient periods longer than 21 days may be optimal for the long-term development of primordial follicles to antral or even preovulatory stages in the transplanted ovarian tissues. In the future, we will test the possibility of grafting human ovarian cortex with MSCs for a long period at certain conditions to acquire mature gametes. After confirming the effect of MSCs on neovascularization, blood perfusion, apoptosis rates, and follicle loss, we further investigated the correlation among the above variables. According to statistical analyses, we found that the primordial follicle number inversely correlated with the temperature difference (Spearman's rank correlation coefficient: -0.401,
Last, but not the least, neoangiogenesis was assessed by immunohistochemistry analysis for CD31, dextran, vWF, and α-SMA (26,47,48) staining in previous studies. However the amount of vascular structure could not completely represent the functional blood perfusion of the grafts. Therefore, we, for the first time, introduced a noninvasive infrared thermography technique to assess the thermal response that was indicative of the blood perfusion. Given that the infrared thermography technique is simple and noninvasive, it is worthwhile to apply it in assessing the quality of superficial ovarian transplantation for clinical patients.
Footnotes
Acknowledgments
