Abstract
Neural restoration has proven to be difficult after brain stroke, especially in its chronic stage. This is mainly due to the generation of an unpropitious niche in the injured area, including loss of vascular support but production of numerous inhibitors against neuronal regeneration. Reconstruction of a proper niche for promoting local angiogenesis, therefore, should be a key approach for neural restoration after stroke. In the present study, a new biomaterial composite that could be implanted in the injured area of the brain was created for experimental therapy of brain ischemia in the mouse. This composite was made using a hyaluronic acid (HA)-based biodegradable hydrogel scaffold, mixed with poly(lactic-co-glycolic acid) (PLGA) microspheres containing vascular endothelial growth factor (VEGF) and angiopoietin-1 (Ang1), two factors that stimulate angiogenesis. In addition, the antibody of Nogo receptor (NgR-Ab), which can bind to multiple inhibitory myelin proteins and promote neural regeneration, was covalently attached to the hydrogel, making the hydrogel more bioactive and suitable for neural survival. This composite (HA–PLGA) was implanted into the mouse model with middle cerebral artery occlusion (MCAO) to explore a new approach for restoration of brain function after ischemia. A good survival and proliferation of human umbilical artery endothelial cells (HUAECs) and neural stem cells (NSCs) were seen on the HA hydrogel with PLGA microspheres in vitro. This new material was shown to have good compatibility with the brain tissue and inhibition to gliosis and inflammation after its implantation in the normal or ischemic brain of mice. Particularly, good angiogenesis was found around the implanted HA–PLGA hydrogel, and the mouse models clearly showed a behavioral improvement. The results in this present study indicate, therefore, that the HA–PLGA hydrogel is a promising material, which is able to induce angiogenesis in the ischemic region by releasing VEGF and Ang1, thus creating a suitable niche for neural restoration in later stages of stroke. This manuscript is published as part of the International Association of Neurorestoratology (IANR) special issue of Cell Transplantation.
Introduction
Stroke, especially cerebral ischemic infarction, is a serious brain disease, which causes extensive cellular loss, leading to permanent and incurable paralysis for the patients. So far, no effective treatment is available to reverse the brain damage caused by stroke since the matured neuron is difficult to regenerate.
Recently, many experimental researches in stroke models using various types of stem cells and their derivatives showed neurological improvement, providing a promising therapeutic approach for the patients (1,26). The therapeutic benefit of cell therapy has been shown to be mediated by trophic support and immunomodulation, instead of cell differentiation and replacement (19). This may impact the therapeutic effect, since replacement of lost neurons would essentially help neural restoration after brain injury. An unfavorable niche for grafted stem cells in the infarct brain after stroke may be a possible reason for this limitation.
It is known that an abounding blood supply or a complete vascular network in the local area is a key element of the niche for neurogenesis (28,49). This has already been demonstrated within the subventricular zone (SVZ) of the rodent, where the stem cells attach closely on the endothelial cells of the vessel networks, forming a special niche, and thus support proliferation and differentiation of the stem cells (6,36). Within the vascular niche or microenvironment, therefore, the blood vessel endothelial cells (ECs) and neural stem cells (NSCs) are ideally located for interaction for performing neurogenesis and angiogenesis (27,44). There has been evidence that cotransplantation of ECs and NSCs can increase survival, proliferation, and differentiation of ischemia-induced NSCs (27). Vascular ECs are also known to secrete certain soluble factors that act on the adult NSCs and stimulate neurogenesis (32,35). It is clear, therefore, that reconstruction of the microenvironment to help angiogenesis in the local area is a better foundation for neurogenesis, which leads to restoration of brain ischemia. Moreover, setting up a proper niche for promoting angiogenesis helps in the promotion of the local blood supply, which is a direct key element against cerebral stroke (7,49).
It is known that two important proteins, vascular endothelial growth factor (VEGF) and angiopoietin-1 (Ang1), are key factors for promoting angiogenesis (2). VEGF has been demonstrated to increase the number of capillary ECs in a given network, while the angiopoietins, especially Ang1, are required for the formation of mature blood vessels (33). Actually, there is a complementary functional relationship between VEGF and Ang1 (31), and they have been codelivered previously for cardiovascular diseases by gene therapy (34,38,43), as well as being used just as soluble growth factors (3,18).
Although angiogenic VEGF and Ang1 have shown good potential as therapeutic factors, their short half-lives following direct delivery in vivo presents a practical challenge (10). A vehicle is therefore needed to carry the VEGF and Ang1 into the brain lesion and stay there for a longer time. Fortunately, in our previous studies, a hyaluronic acid (HA)-based biodegradable hydrogel scaffold was created to mimic the extracellular matrix (ECM) in the brain (15,29,48). This HA hydrogel was modified with a polyclonal anti-Nogo-66 receptor antibody in order to promote regeneration in the injured central nervous system (CNS) (46). The gel could deliver antibodies slowly at a long term and also serve as a scaffold for neural regeneration following its implantation into injured tissue. The results have shown that HA hydrogels modified with anti-Nogo-66 receptor can support neural cell attachment and survival both in vitro and in vivo, indicating that these modified hydrogels have provided a proper niche for repair of injury in the CNS (46). In addition, our previous study provided a delivery system based on HA hydrogels mixed with brain-derived neurotrophic factor (BDNF) and VEGF-loaded poly(lactic-co-glycolic acid) (PLGA) microspheres for better controlling of the delivery of growth factors and supporting NSCs in the brain (48).
In the present study, we therefore propose to use a new material system, that is, the HA hydrogel modified with anti-Nogo receptor antibody and poly-l-lysine, embedded with PLGA microspheres containing VEGF and Ang1 (HA–PLGA hydrogel composite), for implantation in ischemic brain to improve the vascular niche for neural regeneration. The results of our study indicate that the newly created HA–PLGA hydrogel composite has a potential for neurorestoration via angiogenesis in situ after brain stroke.
Materials and Methods
Synthesis of VEGF- or Ang1-Loaded PLGA Microspheres
Microspheres of PLGA (Sigma, St. Louis, MO, USA) were fabricated using the water-in-oil-in-water (w/o/w) emulsion technique to encapsulate VEGF (PROSPEC, Ness-Ziona, Israel) or Ang1 (R&D Systems, Minneapolis, MN, USA) in a similar fashion to that we previously described (48). Briefly, 250 mg of PLGA polymer was dissolved in 2 ml dichloromethane (Sinopharm Chemical Reagent, Shanghai, China); 20 μg VEGF was dissolved in 1 ml bovine serum albumin (BSA, 0.1% wt; Sigma) and added into the PLGA (50:50, Mw 5–15 kDa) solution; or 20 μg Ang1 was dissolved in 1 ml BSA (0.1% wt; Sigma) and added into the PLGA (50:50, Mw 38–54 kDa) solution. Since, in the physiological process of blood vessel formation, the bioactivity effect of VEGF is prior to Ang1, and biodegradation of PLGA slows down with increasing molecular weight, we simulated the process of angiogenesis in which the release time of Ang1 should be longer than VEGF. Afterwards, 10 ml of a prefiltered 1% (w/v) polyvinyl alcohol (PVA, Alfa Aesar, Ward Hill, MA, USA) solution was then slowly added to the mixture. An ultrasonic cell crusher instrument (Xinyi Ultrasonic Equipment, Ningbo, China) was used to mix PLGA microsphere solutions (60 W, 90 s) with the ultrasonic horn until the sample became milky colored. This primary w/o/w emulsion was then immediately poured into a beaker containing 300 ml of the prefiltered 0.3% (w/v) PVA solution and stirred constantly for at least 12 h, in a fume hood to remove the dichloromethane. The final emulsion was washed twice with distilled water and centrifuged at 1,557 × g for 15 min. Finally, the deposits were resuspended in a minimal amount of deionized water and freeze-dried overnight to obtain PLGA microspheres.
Preparation of HA–PLGA Hydrogel Composite
The PLGA microspheres were embedded in the HA hydrogel, forming a delivery system as follows (48). HA sodium salt (Mw 2.6–2.7 kDa, Fureda Biochem Co., Ltd., Jinan, China) was dissolved in distilled water to a concentration of 10 mg/ml, mixed with 0.03 g poly-l-lysine (PLL, Sigma), then VEGF- and Ang1-loaded PLGA microspheres (0.04 g for each) were added into the HA solution successively. Adipic dihydrazide (ADH, Sigma) 1.5 g and carbodiimide reagent (EDC, Sigma) 0.3 g were dissolved in water and added to the HA–PLGA mixture solution. The reaction solution was allowed to become a hydrogel at room temperature overnight. The hydrogel was washed with deionized water five times, and an HA hydrogel without VEGF- and Ang1-loaded PLGA micro-spheres was used as the control.
HA and HA–PLGA Hydrogels Modified with Antibody of Nogo Receptor
The HA and HA–PLGA gels were modified with polyclonal anti-Nogo-66 receptor for the repair of defects and the support of neural regeneration in the brain. The preparation of polyclonal antibodies and immobilization of antibodies on the backbone of HA and HA–PLGA hydrogels followed our previous study (23). Ten milligrams of antibody (from Beijing Key Laboratory for Brain Aging, Xuanwu Hospital, Capital Medical University, China) was dissolved in 1 ml phosphate buffer (ZSGB-BIO, Beijing, China) and added with NaIO4 (Sigma) to reach the final concentration of 0.02 M. This antibody solution was mixed with HA and HA–PLGA hydrogels for 24 h, respectively.
Scanning Electron Microscopy for PLGA Microspheres and Hydrogels
In order to observe the morphology of the lyophilized PLGA microspheres and hydrogel composites by scanning electron microscope (SEM; JEOL 6460LV, Tokyo, Japan), they were dried under a vacuum, coated with gold by a sputter coater (JEOL, JFC-1600, Tokyo, Japan) for 60 s, and then observed at 25 kV accelerating voltage.
Detecting Releasing Kinetics of PLGA Microspheres with BCA
To test the releasing kinetics of VEGF or Ang1 in vitro, 30 mg VEGF- or Ang1-loaded microspheres were disrupted with PBS (pH = 7.4) in a shaking incubator (25°C), and the supernatants of 300 μl were harvested at time points from 1 h to 13 days. All samples were replenished with PBS using the same volume. The amounts of released VEGF and Ang1 were quantified using bicinchoninic acid (BCA) Protein Assay kits (Thermo Fisher Scientific, Rockford, IL, USA).
Culture of HUAECs in HA/HA–PLGA Hydrogel Composites
To investigate the angiogenic potential of HA–PLGA hydrogels in vitro, human umbilical artery endothelial cells (HUAECs, from National Platform of Experimental Cell Resources for Sci-Tech, Beijing, China) were cultured in Dulbecco's modified Eagle's medium (DMEM)/F12 (Gibco, Grand Island, NY, USA), 10% fetal bovine serum (FBS, Gibco). Both HA hydrogels with and without PLGA microspheres were prepared in 24-well plates (Corning Incorporated Life Sciences, Tewksbury, MA, USA). All the hydrogels were sterilized by ethylene oxide (Yongding disinfection equipment, Beijing, China) before use. The HUAECs (in 100 μl DMEM/F12, 1 × 105 cells/μl) were seeded, respectively, into the hydrogels of HA and HA–PLGA using a syringe. Over 5 days in culture, half of the cells adhered and were distributed on the hydrogels and immunofluorescently stained using von Willebrand factor antibody (vWF; Abcam, Cambridge, MA, USA). The other half of the cells were stained with acridine orange (AO; AppliChem, Boca Raton, FL, USA) for visualization of cell proliferation on the hydrogels. The images were obtained by using a laser scanning confocal microscope (Leica Microsystems LAS AF-TCS SP5, Tokyo, Japan).
Culture of NSCs in HA/HA–PLGA Hydrogel Composites
The NSCs were obtained from enhanced green fluorescent protein (EGFP) transgenic mice (provided by Institute of Laboratory Animal Science, Peking Union Medical College, C57BL/6J-TgN, n = 8) (21). In brief, the cells were isolated from the forebrain of E12.5 mice and gently triturated without trypsinization. Dissociated cells were grown as neurospheres in DMEM/F12. Following 7 days of primary culture, the neurospheres were dissociated into a single cell suspension, seeded (in 100 μl DMEM/F12, 1 × 105 cells/μl) into hydrogels using a syringe and cultured in 24-well plates for 5 days. The hydrogels of HA and hydrogel mixed with PLGA microspheres (HA–PLGA), together with the cultured cells, underwent immunofluorescent staining for nestin, a marker of NSCs (1:1,000, Abcam). The cellular nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Sigma). The proliferation of NSCs was determined using a laser scanning confocal microscope and Image-Pro Plus software (Media Cybernetics, Rockville, MD, USA).
Determination of Brain Compatibility of HA/HA–PLGA Hydrogel Composites
The brain compatibility of HA and HA–PLGA hydro-gel composites was determined by checking the extent of gliosis and inflammation after its implantation into the normal mouse brain. The experimental procedure was according to our previous approach (16). The C57BL/6J mice were provided by the Center of Experimental Animals, Capital Medical University, China. All procedures were approved by the Institutional Animal Care and Use Committee of Capital Medical University (2010-X-016). Male C57BL/6J mice (4 weeks old) were divided into two groups (n = 5 for each) and were used for intracortical implantation of the HA hydrogel and HA–PLGA hydrogel composite, respectively. An additional group of mice (n = 5) acted as a control with no implantation after lesion. The brain lesion for all mice was made by opening the right side of the skull and a block of cortical tissue (1.5 mm × 1.5 mm × 1.0 mm) mechanically removed from the parietal lobe using a dental excavator (Dental Materials Factory of Shanghai Medical Instruments, Shanghai, China) under deep anesthesia with intraperitoneal injection of 6% chloral hydrate solution (6 ml/kg, Sinopharm Chemical Reagent, Shanghai, China). The cortical cavity from the lesion was filled by the hydrogels of the same volume with the cavity before the wound was closed. Ionized calcium-binding adapter molecule 1 (Iba1) and glial fibrillary acidic protein (GFAP) immunocytochemistry was used to show the extent of gliosis and inflammation after implantation. After 2 weeks of survival, all the mice were perfused through the aorta with 100 ml of 4% paraformaldehyde fixative (Sinopharm Chemical Reagent, Shanghai, China) under the same anesthesia, and their brains were removed. The frontal series of 20-μm-thick sections was cut using a frozen slicer (Leica CM1900, Wetzlar, Germany) and incubated with the appropriate dilutions of antibody against Iba1 (1:1,000; Wako, Chuo-Ku, Japan) and GFAP (1:1,000; Abcam), respectively, for 12 h at 4°C. After washing with 0.5 M PBS, the sections were exposed to a solution of Dylight 594 fluorescein isothiocyanate-conjugated anti-rabbit IgG (EarthOx Life Science, Millbrae, CA, USA) for 2 h at room temperature. The sections were then observed using a laser scanning confocal microscope system.
Surgical Operation for MCAO Model
Thirty-two male C57BL/6J mice (4 weeks old), weighing from 23 to 28 g, were used for the surgery. A permanent focal cerebral ischemia was made by coagulation of the right middle cerebral artery (MCA) as described in detail elsewhere (24,40–42,47). The animals were anesthetized by an intraperitoneal injection of 6% chloral hydrate and placed in a stereotactic frame (Benchmark, Neurolab, St. Louis, MO, USA). After thorough cleaning, a skin incision was made between the lateral part of the orbit and the external auditory meatus, followed by a partial resection of the upper part of the temporal muscle. The right zygoma was dissected, and a hole (diameter 1.5 mm) was made using a dental drill (Saeshin Precision Ind. Co., Daegu, Korea) in the bone to allow for exposure of the MCA. The MCA was occluded by electrocoagulation with a general metallic heat applicator (Zhongxiyuanda Technology Co., Ltd., Beijing, China) at a proximal location. The hole in the skull was afterwards sealed with dental cement (Dental Materials Factory of Shanghai Medical Instruments, Shanghai, China) (37). After suturing (Medical Suture Needle Co., Ltd., Shanghai, China), mice were returned to their cages in a controlled environment of 27–28°C. Two days later, the brains were removed from two animals after perfusion (as described above), and coronal slices were made for staining with 2% 2, 3, 5-tripenyltetrazolium chloride (TTC, Sigma), to determine the extent of the brain infarct.
Forelimb Use Asymmetry Test
Forelimb use of the mice during exploratory activity was analyzed by the cylinder test, which is designed to evaluate movement asymmetry in rodent models after CNS disorders (17,30). The recorded mice were put in a beaker (BOMEX, Beijing, China) as a transparent cylinder (9 cm in diameter and 15 cm in height) for 10 min, depending on the degree of activity during the trial. Scoring was recorded by an experimenter during a 10-min testing session. A single overall limb use asymmetry score was calculated as follows: Limb Use Asymmetry Score = (ipsilateral –- contralateral)/(contralateral + ipsilateral + both).
Intracerebral Implantation of HA/HA–PLGA Hydrogel Composites
All mice treated with MCAO were divided into three groups: one group for implantation of HA hydrogel mixed with two kinds of PLGA microspheres (HA–PLGA, n = 10), another group for implantation of HA hydrogel only (HA, n = 10), and the third group with opening of the cranial bone but without implantation as a blank control (MCAO, n = 10). Two additional mice were sacrificed for localizing the MCAO lesion in the cortex, in order to determinate the location of hydrogel implantation.
Two weeks after MCAO, the animals were anesthetized again by intraperitoneal injection of chloral hydrate solution. The skull was opened, and a lacunar infarct in the frontal–parietal cortex was exposed, the infarct cavity being about 1.5 mm × 2.0 mm × 1.0 mm, a piece of HA–PLGA or HA hydrogel sized to the cavity was placed into the lesion site. Thrombin (Sinopharm Chemical Reagent, Shanghai, China)-soaked gel foam (Zhonghua Pharmaceutical, Shanghai, China) was used for bone errhysis, and the skin was sutured. Three groups of mice were sacrificed at 6 and 10 weeks after MCAO, respectively (n = 5).
Immunocytochemical Analysis for Angiogenesis
The mice were perfused, and their brains were removed. Coronal brain sections (20 μm) were cut by the frozen slicer (Leica CM1900, Germany) for further immunocytochemistry. To observe blood vessel regeneration, the sections were incubated with the appropriate dilutions of laminin antibodies (1:1,000; Abcam) for 12 h at 4°C. After washing with 0.5 M PBS, the sections were exposed to a solution of Dylight 594 fluorescein isothiocyanateconjugated anti-rabbit IgG for 2 h at room temperature. The sections were stained with DAPI and studied by laser scanning confocal microscope using the PerkinElmer Confocal System (Akron, OH, USA). A vascular area of around 1.5 mm of the peri-infarct region for each animal was selected, according to the immunohistochemical staining with laminin. This laminin-positive area in periinfarct region of the ischemic hemisphere was measured with the Image-Pro Plus software to determine the status of vascular regeneration in the area.
Statistics
The data from all studies were analyzed using one-way ANOVA. LSD test was used to separate treatment means if the main effect was found to contribute significantly (SPSS, IBM, Armonk, NY, USA). A value of p < 0.05 indicates a significant difference between the groups. All results were presented as mean ± standard deviation.
Results
SEM Observation and Releasing Kinetics of PLGA Microspheres
Under the SEM, the appearance of PLGA ***micro-spheres containing VEGF and Ang1 was similar to that we described before (48), showing homogeneity with an average diameter of 1~5 μm (Fig. 1A, B). The releasing kinetics of VEGF and Ang1 in vitro was shown by BCA assay and revealed a slow release over time (Fig. 1C, D).
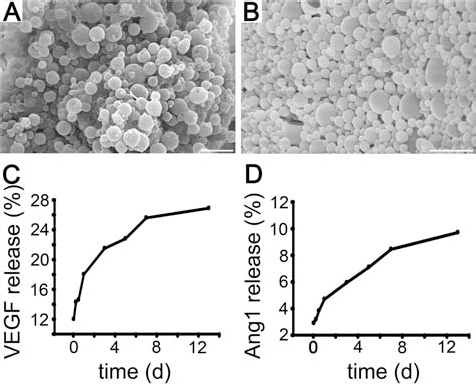
SEM of PLGA microspheres and factor release curves. (A, B) SEM photographs showing lyophilized VEGF-loaded (50:50, Mw 5–15 kDa) and angiopoietin 1 (Ang1)-loaded (50:50, Mw 38–54 kDa) PLGA microspheres under the SEM (A, B, scale bar: 5 μm). Diagrams showing releasing rate curves of the two microspheres in PBS at pH 7.4 and 25°C (C, D).
The cumulated amount of VEGF accounted for 26.9% of the total released quantity at the 13th day, and that of Ang1 was 9.71%, indicating that the VEGF was released from the microspheres more quickly. Thus, by using different molecular weights of PLGA, we obtained two types of PLGA microspheres and made the release time for Ang1 longer than that of VEGF.
Characterization of HA/HA–PLGA Hydrogel Composites
Hydrogels prepared with ADH derivatives of HA were shown to be stable. After lyophilizing, the appearance of the HA hydrogel changed to a white color from being colorless and transparent (Fig. 2A, B). Under SEM, the hydrogels demonstrated a macrophase separation structure with a three-dimensional network of flakes with 5–60 μm width, forming an open interconnected porous system (Fig. 2C, D), as described in our previous studies (15,16). When the HA hydrogel was mixed with PLGA microspheres, the color of the composite (HA–PLGA) became milky (Fig. 2A, E), and no visual difference from HA hydrogel after lyophilizing was evident (Fig. 2B, F). Under the SEM, both hydrogels presented the same porous structure (Fig. 2C, D, G, H).

Macroscopic and SEM visualization of HA/HA–PLGA hydrogels. HA hydrogel without mixture of PLGA microspheres (A–D) and HA hydrogel mixed with PLGA microspheres (E–H). A, B, E, and F show naked-eye observation of the hydrogels before (A, E) and after lyophilizing (B, F). C, D, G, and H indicate microstructure of the hydrogels under SEM, (D) shows higher magnification of the picture (C), and (H) shows higher magnification of the picture (G). (A, B, E, F scale bar: 10 mm; C, D, G, H scale bar: 250 μm).
HUAEC and NSC Proliferation in HA/HA–PLGA Hydrogel Composites (Fig. 3)

The proliferation of HUAECs and NSCs on HA/HA–PLGA hydrogels. Photographs showing growth of HUAECs for 5 days in HA (A–D) and HA–PLGA (A′–D′) hydrogels. Many cells with staining in the hydrogels are vWF-positive HUAECs. NSC growth in HA (E–I) and HA–PLGA (E′–I′) hydrogels after 5 days of seeding. Histograms showing HUAEC proliferation in the two hydrogels, respectively (J), and the proliferation of HUAECs in the HA–PLGA composite was faster than that in the HA hydrogel (K) at the same time points (3 days, 5 days, and 7 days) (*p < 0.05). NSC proliferation was also significantly increased over time in the two hydrogels (L) (*p < 0.05). Scale bar: 100 μm. DAPI, 4′,6-diamidino-2-phenylindole.
After being seeded in the two hydrogels for 5 days, the HUAECs were fluorescently stained with vWF and were shown to have good adherence to both HA (Fig. 3A–D) and HA–PLGA hydrogels (Fig. 3A′–D′) in vitro. A large number of cells survived and grew in good condition, indicating the two hydrogels were suitable for HUAEC adherence and survival. After HUAECs (100 μl; 1 × 105 cells/μl) were seeded on HA and HA–PLGA hydrogels, respectively, over 7 days, the HUAECs proliferated rapidly in both hydrogels by a random cell count (Fig 3J). As shown in Figure 3K, cells in HA–PLGA hydrogel showed a faster proliferation, compared with that in HA hydrogel.
The survival of NSCs in the hydrogels was observed in vitro with fluorescent staining of nestin, after being seeded for 5 days. It could be seen that the NSCs adhered and survived well on HA (Fig. 3E–I) and HA–PLGA (Fig. 3E′–I′) hydrogels. Also the cells were shown to have favorable proliferation within both hydrogels over time, but no significant difference was seen between the two hydrogels (Fig 3L).
Inhibition of Brain Inflammation and Gliosis by Implantation of HA/HA–PLGA Hydrogel Composites
After 2 weeks of implantation, much less Iba1-positive microglia, caused by the cortical lesion, were seen in the brain area around the graft in two hydrogel implantation groups (Fig. 4B, C), compared with that in the control group (*p < 0.01, n = 5) (Fig 4A).
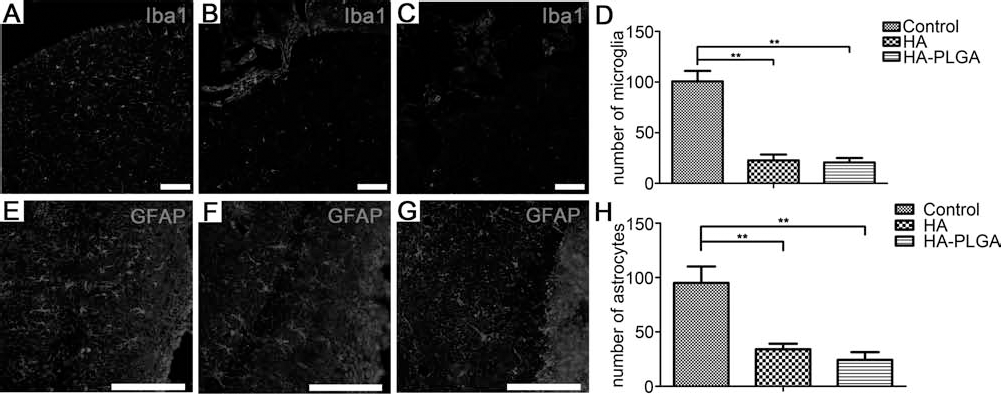
Inhibition of brain inflammation and gliosis after implantation of HA/HA–PLGA hydrogels in brain. Photographs showing reactive microglia after implantation of HA and HA–PLGA hydrogels at 2 weeks after cortical implantation (A–D). Very few microglia are revealed around the implants of HA hydrogel (B) and HA–PLGA composite (C), while many more microglia were observed in the brain tissue with no implant (control) (A). Statistical analysis (D) indicating the significant difference among these three groups (**p < 0.01, n = 5). Implantation of HA or HA–PLGA hydrogels inhibits reactive astrocytes caused by brain lesion after 2 weeks (E–H). Relatively fewer astrocytes are revealed around the lesion by implantation of HA hydrogel (F) or HA–PLGA composite (G), with no intense immunostaining in the tissue area around the entire perimeter of the implants. The intense GFAP immunostaining is just seen in the control brain without implantation (E). Statistical analysis (H) indicating that the numbers of astrocytes in these three groups have a significant difference (**p < 0.01, n = 5). Scale bar: 100 μm. Iba1, ionized calcium-binding adapter molecule 1.
With regard to GFAP-positive astrocytes, which reflect gliosis by the lesion, large numbers of GFAP-positive cells were observed in the control mice (Fig 4E). However, very few such astrocytes were observed in the brain tissue along the perimeter of the implants in the groups of HA hydrogel (Fig 4F) and HA–PLGA composite (Fig 4G) after 2 weeks of implantation (*p < 0.01, n = 5).
Preparation of MCAO Mice and Implantation of Hydrogels
For testing mouse stroke models, the TTC method reliably delineated the infarct core at 2 days after MCAO (Fig 5A). After 2 weeks of MCAO, there was a focal lesion cavity in the cortex of about 1.5 mm × 2.0 mm × 1.0 mm (Fig 5B), which was the location for hydrogel implantation. The hydrogels of either HA or HA–PLGA, sized to the dimensions and shapes of the cavities, were implanted into the lesion sites. One group of MCAO mice without implantation was taken as control. It was clear that both HA and HA–PLGA hydrogels properly filled the defect areas of the cortex and closely merged with the brain tissue after 10 weeks of MCAO (Fig. 5C, D).
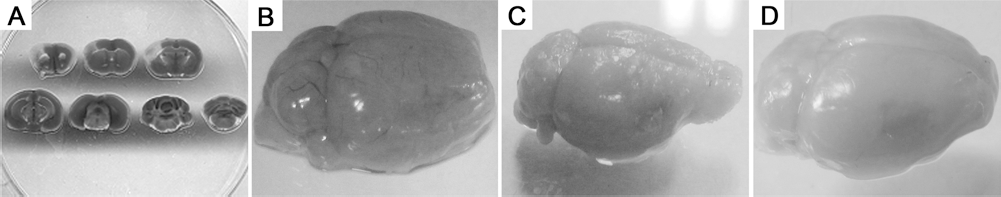
Ischemic model of MCAO mice and HA/HA–PLGA hydrogel implantation. Two days after MCAO, brain slices were stained with 2, 3, 5-triphenyl tetrazolium chloride (TTC) to visualize the ischemic lesions (A). After 2 weeks of MCAO, brain infarction atrophy and defect was seen in the right hemisphere (B). After 10 weeks of MCAO, the defect areas remained filled by the HA hydrogel (C) and HA–PLGA hydrogel (D), and showed good compatibility with the brain tissue.
Behavior Improvement Recorded by Cylinder Testing
The forelimb asymmetries of the mice were recorded before and 2, 6, and 10 weeks after MCAO, through the cylinder test. As seen in Figure 6, the movements of the forelimb, contralateral to the brain ischemia, were significantly reduced after MCAO at all of the time points. After implantation of HA hydrogel alone with no PLGA micro-spheres, the movements of the impaired forelimb showed some improvement by 10 weeks (Fig. 6). After implantation of HA hydrogel together with PLGA microspheres, however, a significant recovery of forelimb movements could be seen clearly through the behavior testing by 6 weeks (Fig. 6).

Cylinder text of ischemic mice following implantation of HA/HA–PLGA hydrogels. Asymmetry of forelimb use was recorded before and 2, 6, and 10 weeks after MCAO. The asymmetry of forelimb use was scored as [ipsilateral (I) – contralateral (C)]/[C + I + both (B)], with higher scores reflecting preferential use of the unaffected limb and, therefore, more severe impairment. Ischemic animals showed long-term improvement of function after treatment with hydrogels. In the HA hydrogel group, there was a decreased score between 2 weeks and 10 weeks (*p < 0.05, n = 10); and in the HA–PLGA hydrogel group, there was a decreased score for 2 weeks compared to 6 weeks and 10 weeks (**p < 0.01, n = 10), indicating an earlier improvement by implantation of HA–PLGA composites, compared with that by implantation of HA hydrogel alone.
HA–PLGA Enhanced Angiogenesis in MCAO Mice
Immunohistochemical staining with anti-laminin antibody, revealing the basement membrane of vessels, was used to observe angiogenesis in ischemic brain after the 6th and 10th week of implantation of HA–PLGA hydrogel (Fig. 7). If the MCAO mice received no implantation, a great reduction in the number of blood vessels was seen in the peri-infarct area at the 6th week (Fig. 7A, B, H). At this time, quantitative analysis (Fig 7I) of blood vessels in peri-infarct area showed that the density of blood vessels in the HA hydrogel group (Fig 7D) was higher than that with no implantation group (Fig 7A). The density of blood vessels in the HA–PLGA hydrogel group, however (Fig 7I), was much higher (Fig 7F) than that in the HA hydrogel group (Fig 7D). This disparity was even bigger at the 10th week after MCAO (Fig. 7G compared with E and C). These data proved that the HA hydrogel mixed with PLGA microspheres releasing VEGF and Ang1 was a promising angiogenesis inducer in the ischemic region.
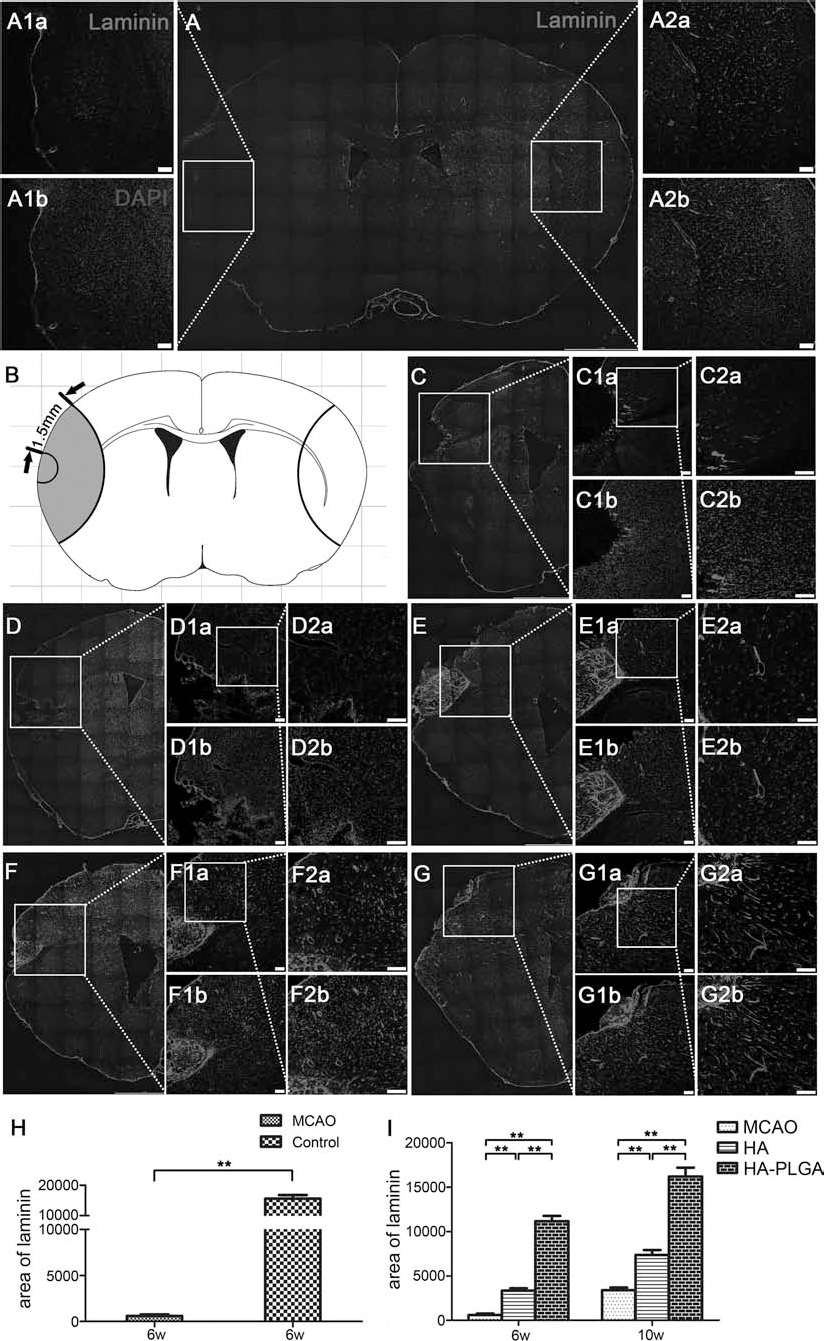
The enhanced angiogenesis by HA–PLGA hydrogels in MCAO mice. Photographs (A–G) showing angiogenesis in the ischemic area after implantation of HA and HA–PLGA hydrogels. The basal laminin of blood vessels was immunohistochemically shown in the peri-infarct area after 6 weeks (A, D, F) and 10 weeks (C, E, G) of MCAO, and cellular nuclei were stained by DAPI. The densities of blood vessels at peri-infarct area (A1) and corresponding area contralateral to the ischemia (A2), after 6 weeks of MCAO treatment but without implantation, are compared in (A). (A1a, A1b) show higher magnification of the peri-infarct area in picture (A), indicated by a white square. (A2a) and (A2b) show higher magnification of the contralateral nonischemic area in (A) from a corresponding white square. This comparison of the blood vessels was quantitatively analyzed and is shown in the histogram of (H), indicating a significant decrease in peri-infarct area [1.5 mm from the core, see diagram (B); **p < 0.01]. The density of blood vessels at peri-infarct area after 10 weeks of operation is shown in picture (C). (C1a) and (C1b) show higher magnification of (C) indicated by a white square, and (C2a) and (C2b) show a higher magnification from (C1a). After 6 weeks of MCAO, the laminin-immunopositive blood vessels were increased both in the HA (D) and the HA–PLGA implanted groups (F), with a more significant increase in the HA–PLGA group (F). The phenomenon of increased blood vessel number was more obvious at 10 weeks (E, G, magnified in E1, E2, G1, and G2, respectively), in particular in the HA–PLGA group (G, G1, and G2). Quantitative comparison of the number of blood vessels within the peri-infarct region in each group is shown in (I) (**p < 0.01, n = 5). Scale bar: 200 μm.
Discussion
The results of the present study show a clear improvement in the behavior of stroke mice, after intracerebral implantation of a modified HA hydrogel mixed with PLGA microspheres, which are loaded by molecules of VEGF and Ang1. Meanwhile, the phenomena of angiogenesis appeared at the implanted brain area, which 2 weeks previously was infarcted by MCAO. Our results therefore provide essential evidence that promotion of local angiogenesis is a useful strategy for treating stroke, especially in its later stages.
Actually, the correlation between angiogenesis and functional improvement after ischemic stroke has been observed in both animal models and patients with stroke (13,20,22). Based on this, there have been a few studies in which the utilization of promoting angiogenesis for treating stroke is efficacious (12). It was reported that increased angiogenesis in cerebral ischemia could stimulate endogenous recovery mechanisms, including neurogenesis, synaptogenesis, and neuronal and synaptic plasticity (32). The angiogenic state may create a “vascular niche” in which NSCs are generated and promoted to migrate (19).
In many studies, VEGF has proven to be a suitable molecule for stimulating survival, migration, and proliferation of neurons, while Ang1 could also act directly on neurons and enhance neural repair (12,50). The two molecules, therefore, have effects on both the neural and vascular compartments impacted by stroke. However, studies with therapeutic VEGF application revealed quite different effects depending on the route of delivery. Local application (i.e., ICV or intraparenchymal) of VEGF, VEGF-secreting cells, or vectors into the injured area revealed a reduction of ischemic damage and a functional improvement (14). By contrast, certain studies with systemic application (i.e., IA, IV, or IP) of VEGF exhibited harmful effects to the brain, like an increase in hemorrhagic transformation, microvascular injury, and inflammation (52). In addition to route, the time point of delivery seemed to play a role in the actions of VEGF in the ischemic brain, since the majority of unfavorable effects of VEGF used acute application strategies (39), in which the factor was delivered 1 h after the stroke. Differently, administration of VEGF using a postacute delivery strategy (as late as 2 or 3 days after stroke) exhibited a beneficial effect, even though it was a systemic delivery (IA or IV) (4,50,51,53). These results have clearly indicated that the approach for delivery of angiogenic factors is essential for treating stroke.
It has been known that brain tissue engineering in the postinjury brain represents a promising option for cellular replacement and rescue, providing a cell scaffold for either transplanted or resident cells (5). This is because a biocompatible scaffold may provide the necessary structural support for the cells in defective tissue and also artificially create a microenvironment suitable for cell growth and integration. Meanwhile, a biocompatible scaffold may serve as a source of biochemical signals that influence cell behaviors and functions (11). A HA–poly-d-lysine (PDL) copolymer hydrogel with an open porous structure and viscoelastic properties similar to brain tissue was shown to have good compatibility with brain tissue in our previous studies, demonstrating that the HA–PDL hydrogel has promise as a suitable scaffold material for repair of defects in the brain, including those caused by a stroke (45). Technologies to encourage angiogenesis in a controlled manner by using tissue engineering, that is, the usage of PLGA nanoparticles to encapsulate VEGF, have also been applied. This injectable system can control VEGF release for a longer term both in vitro and in vivo, since the PLGA microsphere is a copolymer, which is an efficient drug-delivery assembly approved therapeutic device owing to its biodegradability and biocompatibility (9). Therefore, a suitable niche for angiogenesis and repair of brain tissue damage could be constructed if a combination of HA scaffold with PLGA microspheres is induced for treating stroke.
It was shown in our former data that neurotrophic factors could be encapsulated in PLGA microspheres with about 20~30% releasing rate on the 6th day, and the bioactivity of the released factors was maintained and was helpful in increasing the survival rate of neurons in vitro (48). In addition, PLGA microspheres could be combined with a hydrogel to form hybrid systems to deliver VEGF and Ang1 intramuscularly to the ischemic site in the gracilis muscle. The results showed that codelivery of VEGF and Ang1 was advantageous to retaining muscle tissues and significantly induced vessel enlargement at the ischemic site, and they believed the microsphere/hydrogel hybrid systems may be a promising vehicle for delivery of multiple drugs for many therapeutic applications (37). In the present study, we used a lower molecular weight of PLGA to encapsulate VEGF, and a higher weight to encapsulate Ang1, to prioritize their releasing rates. This prioritization was confirmed by the releasing rate curves of the two microspheres in our in vitro study (see Fig. 1C, D), and this endorses the fact that selecting different molecular weights of microspherical vehicles can determine the sequence of released drugs brought by the vehicles (25).
It is thus reasonable to propose that filling up of the injured cavity using the HA hydrogel scaffold and enhancing angiogenesis using a PLGA microsphere delivery system did increase the therapeutic effect of tissue engineering and benefit functional recovery after stroke in the present study. By implanting this composite into the ischemic area, the angiogenesis in situ occurred clearly when it was evaluated at the 6th and 10th week after MCAO and implantation. This angiogenesis was proved by laminin immunohistochemistry, which was considered to be a simple, useful, and specific method to study vascular structures in the CNS (8). Correspondingly, a strong trend toward better behavior performance was observed in the HA–PLGA hydrogel composite-treated mice, indicating this material enhanced peri-infarct blood vessel density and thus improved behavior function. In addition, the HA–PLGA hydrogel composite showed a good support for NSC survival and growth in vitro in our study. This means that the implanted composite might create a suitable niche for survival of either endogenous or exogenous NSCs, which would further help in the improvement of ischemic injury. This needs to be verified, however, by a future in vivo study that will use the combined implantation of HA–PLGA composite with NSCs. It should not be doubted anyway that the composite of HA hydrogel and VEGF- and Ang1-loaded PLGA microspheres has provided an effective approach for further study on therapy of brain ischemia.
Footnotes
Acknowledgments
This work was supported by National Nature Science Foundation of China (No. 81070977, No. 81200931, No. 81271388), Capital Medical University Key Laboratory Research (No. 2011SJZS05, No. 2012SJZS03), and National Basic Research Program of China (No. 2012CBA01307). The authors declare no conflicts of interest.
