Abstract
CD8+ suppressor T cells have been demonstrated to provide protection of allografts from rejection. We previously reported that soluble peptide/HLA-A2 dimer shows peptide-specific inhibitory effects on alloresponse in a coculture of peptide-pulsed T2 cells with HLA-A2 negative lymphocytes in vitro. Here we found a subset of CD8lowCD28– T cells that was induced in the dimer-treated coculture. Importantly, this population showed hyporesponsiveness to the alloantigen restimulation as well as alloantigen-specific suppression on alloreactive T cells in a cell–cell contact-dependent fashion. The suppressive mechanisms of CD8lowCD28– T cells involved an elevated expression of membrane-bound TGF-β1, but not Foxp3, CTLA-4, or IL-10. Furthermore, an over-represention of CD8lowCD28– T cells was observed in the patients after allogeneic platelet transfusion and positively correlated with the elevated concentrations of plasma HLA class I antigens. Our findings demonstrated that soluble HLA-A2 dimer could efficiently induce the tolerant CD8lowCD28– T cells with alloantigen-specific suppression on alloreactive T cells. This study might provide a new strategy for preparation of donor-specific suppressor T cells and represent an attractive alternative for induction of allograft tolerance.
Introduction
The presence of soluble HLA class I antigens in serum of healthy individuals was described approximately 40 years ago (9,51). As receptor blockage and (or) tolerogen without costimulation signal, soluble HLA class I antigens are suggested to play a role in peripheral tolerance in pathologic conditions, such as acute allograft rejection, autoimmune disease, chronic viral infections, and tumors (19,35,40,46). Direct effects on T cells mediated by soluble major histocompatibility complex–T-cell response (MHC-TCR) inter action without costimulatory signal might account for downregulation of T-cell response, like activity inhibition, apoptosis, or anergy (33,42). However, soluble MHC class II dimer is also reported to be able to induce IL-10-producing CD4+ regulatory T cells (Tregs) in antigen-specific TCR transgenic mice, suggesting Tregs may be involved in the suppressive mechanisms of soluble MHC molecules (6).
During the past decades, multiple subtypes of Tregs have been reported to be negative regulators of immune responses to self and foreign antigens, which are critical for maintaining immune tolerance (44,52). Although CD8+ suppressor T cells were first described in the early 1970s (18), extensive studies have mainly focused on the CD4+CD25+Foxp3+ Tregs in the following years (39). Recently, interest in suppressor T cells has been resurrected for their donor-specific character. A well-studied subset of suppressor T cells is CD8+CD28– T cells, which are detectable in peripheral blood of both liver and kidney allograft recipients, and increased in transplanted patients in quiescence and exhibit donor-specific suppressive effects, whereas those from patients experiencing rejection episodes are less frequent in circulation (2). The suppressive effects of CD8+CD28– T cells are reported to be mediated by regulatory molecules, such as forkhead box p3 (Foxp3), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) expression, or interleukin-10 (IL-10), transforming growth factor-β1 (TGF-β1) secretion (12,47).
Based on the peptide-specific inhibitory effects of soluble peptide/HLA-A2 dimer on alloresponse in an alloreactive coculture of the peptide-pulsed T2 cells (T2/peptide) with HLA-A2– lymphocytes in vitro (53), we explored whether suppressor T cells or Tregs were involved in the inhibition. In this study, a new subset of CD8lowCD28– suppressor T cells was observed to be induced by the dimer intervention in the same alloreactive coculture, which showed a high level of membrane-bound TGF-β1 (mTGF-β1) expression and an elevated IL-4 secretion, but no elevated Foxp3, CTLA-4 expression, or IL-10, TGF-β1 secretion. Importantly, the CD8lowCD28–T cells exhibited hyporesponsiveness to the alloantigen restimulation as well as alloantigen-specific suppression on T-cell alloreactivity in the coculture. Meanwhile, the suppression of the CD8lowCD28– T cells was cell–cell contact dependent and mediated by mTGF-β1. Although the frequencies of CD8lowCD28– T cells and the plasma HLA class I concentrations were elevated and positively correlated in patients who underwent allogeneic platelet transfusion, the significance of the T cells in graft acceptance remains to be characterized. Our findings showed that the CD8lowCD28– T cells of alloantigen-specific suppressor property were induced by the intervention of soluble HLA dimer in vitro, which might provide a practical protocol for preparation of alloantigen-specific suppressor T cells.
Materials and Methods
Human Blood Samples
Peripheral blood samples were collected from 19 healthy donors and 20 platelet transfusion patients (Table 1) hospitalized in General Hospital of Guangzhou Command, Wuhan, China. The patients with solid tumors and chemotherapy-induced thrombocytopenia were administered with prophylactic platelet transfusion to reduce the risk of hemorrhage when the platelet counts fell below a predefined threshold. No HLA typing was carried out for the recipients or donors. The blood sampling was performed within 1 week following the last transfusion. The study was approved by the Ethical Committee of General Hospital of Guangzhou Command (Wuhan, China), and informed consent was obtained from all subjects.
Patient and Control Characteristics

Cell Lines, Peptides, and HLA-A2 Dimer
T2 (CRL-1992; ATCC, Manassas, VA, USA) is a transporter associated with antigen processing (TAP)-deficient human cell line and expresses reduced amounts of HLA-A2 and no other HLA allele (24). HLA-A2-restricted peptides of tyrosinase origin Tyr368-376 (YMDGTMSQV) (48) and hepatitis B virus origin HBC18-27 (FLPSDFFPSV) (1) were synthesized and purified to >90% homogeneity by reverse-phase high-performance liquid chromatography. As stimulator cells, T2 cells were pulsed with Tyr368–376 or HBC18-27 at a final concentration of 40 mg/ml, incubated at 37°C, 5% CO2 for 3 h, and washed extensively with RPMI-1640 medium (Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (Gibco). Subsequently the cells were inactivated by irradiation at 37°C by a Co-60 source (Theratron 780; Atomic Energy of Canada Limited, Ottawa, Canada) with a dose of 200 Gy at a dose rate of 5 Gy/min. T2 cells pulsed with Tyr368–376 or HBC18-27 are named T2/Tyr and T2/HBC, respectively. E006 cell line was an Epstein–Barr virus transformed B-lymphoblastoid cell line established in our laboratory from blood sample of an HLA-A2 negative (HLA-A24, A33; male) individual (41) and used as an allogeneic stimulator. The preparation of soluble HLA-A2 dimer was described previously (53). The dimer pulsed with Tyr or HBC peptide is named Tyr/ HLA-A2 dimer and HBC/HLA-A2 dimer, respectively.
Alloreactive Coculture
Alloreactive coculture was set up with blood samples of HLA-A2– healthy individuals. HLA typing was performed by sequence-specific primers (PCR-SSP) (3). Peripheral blood mononuclear cells were isolated by density gradient centrifugation (2,000 rpm/min for 30 min) (Ficoll-Hypaque density 1.077 g/ml; Sigma-Aldrich, St. Louis, MO, USA) and incubated overnight at 37°C in complete RPMI-1640 medium containing 10% human AB serum (Sigma-Aldrich). Nonadherent cells were harvested for peripheral blood lymphocytes (PBLs). The PBLs (3 × 106 cells/well) were cocultured with the peptide-pulsed T2 cells (3 × 105 cells/well) in 24-well plates (Corning Costar, Cambridge, MA, USA), and 2 nM peptide/HLA-A2 dimer or media alone was added the next day. After coculturing at 37°C for 7 days, cells were harvested for the following experiments.
Proliferation Assays
For proliferation assays, PBLs (3 × 106 cells/well) were stained with 5 μM carboxyfluorescein diacetate succinimidyl ester (CFSE; Sigma-Aldrich) and stimulated by the irradiated T2/Tyr (3 × 105 cells/well). Tyr/HLA-A2 dimer (2 nM) or media alone was added the next day. After 7 days of coculture, cells were harvested by centrifugation (800 rpm/min for 10 min) and incubated on ice with anti-CD3-APC-cy7 (HIT3a; Biolegend, San Diego, CA, USA) and anti-CD8-PE-cy5 (HIT8a; Biolegend) (20 μl antibody in 100 μl total mixed system). After 45 min of incubation, cells were washed extensively and then fixed with PBS containing 2% formaldehyde (Sigma-Aldrich). Three-color flow cytometry analysis was performed with CFSE, anti-CD3-APC-cy7, and anti-CD8-PE-cy5 using a FACS LSRII system (Becton Dickinson, San Jose, CA, USA). Flow cytometric data files were analyzed with the “Proliferation Wizard” module in ModFit LT Macintosh software (Verity Software House Inc., Topsham, ME, USA). Proliferation index (PI) was used to quantify the proliferation magnitude of T cells.
Flow Cytometry and Intracellular Cytokine Staining
Cells were phenotypically analyzed using the FACS LSR II system. The following fluorescence-conjugated mAbs and their isotype-matched control antibodies were used; anti-IL-4-PE-cy7 (8D4-8), anti-CTLA4-PE (14D3), anti-CD28-FITC (CD28.2), and anti-IL-10-PE (JES3-9D7) (all from BD Biosciences, Pharmingen, San Diego, CA, USA); anti-CD3-APC-cy7 (HIT3a), anti-CD8-PE-cy5 (HIT8a), anti-TGF-β1-Percp-cy5.5 (BG/hLAP), anti-CD8-FITC (HIT8a), anti-Foxp3-FITC (PCH101), anti-TGF-β1-PE (TW4-2F8), anti-interferon (IFN)-γ-FITC (4S.B3), and anti-IL-17A-PE (BL168) (all from Biolegend). Twenty microliters antibody in 100 μl total mixed system was used in this study unless specifically mentioned.
For intracellular staining, cells were stimulated for 5 h with 50 ng/ml phorbol 12-myristate 13-acetate (PMA; Sigma-Aldrich) and 500 ng/ml ionomycin (Sigma-Aldrich) in the presence of brefeldin A (GolgiStop; BD Biosciences). After staining with antibodies against surface markers, cells were fixed with IC Fixation Buffer (BD Biosciences), permeabilized using 1× Permeabilization Buffer (BD Biosciences), and incubated with antibodies for intracellular staining following the manufacturer's instructions. The human Foxp3 staining kit (BD Biosciences) was used for intracellular Foxp3 staining according to the supplied protocol.
Fluorescence-Activated Cell Sorting (FACS), Restimulation, and IFN-γ ELISPOT Assay
Single-cell suspensions of the cocultural bulks were stained with anti-CD3-APC-cy7, anti-CD8-PE-cy5, and anti-CD28-FITC for 30 min at 4°C. Cell sorting was based on mean fluorescence intensity (MFI) of the stained cells using a high-speed FACS-MoFlo XDP Cell Sorter (Beckman Coulter, Inc., Miami, FL, USA). CD8high T cells were sorted for high CD8 expression (MFI of PE-cy5 > 200), CD8lowCD28– T cells for low CD8 expression (MFI of PE-cy5 ranged from 20 to 200) and negative CD28 expression (MFI of FITC < 20). The purity of both CD8high T cells and CD8lowCD28– T cells was >97% upon reanalysis.
To detect suppressive effects of the CD8lowCD28– T cells, CD8lowCD28– T cells and CD8high T cells were sorted from the T2/Tyr-primed cocultural bulks with and without the Tyr/HLA-A2 dimer intervention, respectively. Different numbers of the sorted CD8lowCD28– T cells and a fixed number of the sorted CD8high T cells (1 × 105 cells/well) were mixed at various ratios (CD8lowCD28–:CD8high) from 1:0 to 1:10 in duplicate, and restimulated by T2/Tyr (1 × 104 cells/well) in wells precoated with anti-human IFN-γ Ab (MD-1; eBioscience, San Diego, CA, USA). After incubation at 37°C for 24 h, the plates were washed and detected by IFN-γ-specific antibody (4S.B3; eBioscience). The number of IFN-γ spot-forming cells (SFCs) were visualized and counted using an automated ELISPOT plate reader (CTL-ImmunoSpot S5 Core Analyzer; Cellular Technology, Ltd., Shaker Heights, OH, USA), then analyzed with CTL Version 4.0 Academic Software (Cellular Technology Ltd.). Inhibition rate (%) was calculated according to the following method: Inhibition rate (%) = [(spot number of restimulated CD8high T cells - spot number of restimulated CD8high T cells mixed with CD8lowCD28- T cells)/spot number of restimulated CD8high T cells]
Antigen Specificity and Transwell Assays
To test alloantigen specificity of the CD8lowCD28-T cells, the Tyr/HLA-A2 or HBC/HLA-A2 dimer-induced CD8lowCD28– T cells served as suppressors; the T2/Tyr-or T2/HBC-primed CD8high T cells as responders; and T2/ Tyr, T2/HBC, or E006 as restimulators. IFN-γ ELISPOT assays and calculation of inhibition rate were performed as above.
Cell-cell contact dependence was determined by a Transwell assay; 1 × 105 sorted CD8high T cells were restimulated by the T2 cells with 5 × 104 sorted CD8lowCD28-T cells directly mixed in or physically separated in the Transwell inserts of 96-well Transwell plates (0.4 μm pore size; Corning, Corning, NY, USA). After coculturing for 24 h, IFN-γ ELISPOT assays and calculation of inhibition rate were performed as above.
Elisa
Plasma soluble HLA class I levels were measured by sandwich ELISA with W6/32 (Abcam Inc., Cambridge, UK) (34) as capture antibody and rabbit anti-β2-micro-globulin (Abcam Inc.) as detection antibody. For assays of cytokine secretion, coculture supernatants were determined for IFN-γ, IL-4, and TGF-β1 by ELISA kits (eBioscience) according to the manufacturer's instructions. The concentrations of cytokines were calculated according to the standard curve range (4-500 pg/ml for IFN-γ, 2-200 pg/ ml for IL-4, 8-1,000 pg/ml for TGF-β1). Blocking studies were performed by detecting secreted IFN-γ in the presence of neutralizing antibodies against TGF-β1 (5 μg/ml, clone: 27235; R&D Systems, Minneapolis, MN, USA) or IL-4 (0.2 μg/ml, clone: 3007; R&D Systems).
Statistical Analysis
Data were analyzed for statistical significance by ANOVA test using SPSS19.0 for Windows software (IBM, Armonk, NY, USA), and post hoc tests were applied to perform all pairwise comparisons between group means by least significant difference (LSD) t-test. Curve estimation and linear regression analyses were performed for correlation analysis. A value of p < 0.05 was considered statistically significant. The significant difference was marked as *0.01 < p < 0.05; **0.001 < p < 0.01; and ***p < 0.001, respectively.
Results
CD8lowCD28– T cells Induced by Soluble HLA-A2 Dimer
The alloreactive coculture was set up by incubating HLA-A2– PBLs and T2 cells pulsed with HLA-A2-restricted peptide Tyr368-376 (T2/Tyr) in the absence (non-dimer treated) or presence of the Tyr/HLA-A2 dimer (dimer treated). T-cell alloreactivity was revealed by an increased frequency of IFN-γ-producing CD8+ T cells after 7 days of the coculturing. Treatment with 2 nM Tyr/ HLA-A2 dimer significantly decreased the IFN-γ+ cells (frequency IFN-γ+ cells in CD8+ T cells, non-dimer-treated group vs. dimer-treated group: 9.40 ± 1.24% vs. 4.21 ± 0.55%, n ≥ 10, p < 0.001) (Fig. 1A). Notably, a subset of CD8+ T cells with significantly decreased CD8 expression emerged in the dimer-treated coculture, while their CD3 expression remained unchanged. The levels of CD8 expression regrouped CD8+ T cells into two subsets, that is, T cells with lower CD8 (CD8low) and those with higher CD8 (CD8high) expression. The gating strategy for CD8low and CD8high T cells is shown in Figure 1B.

Soluble HLA-A2 dimer intervention reduces IFN-γ-producing T cells and induces CD8low T cells in the coculture. Human leukocyte antigen A2 negative peripheral blood lymphocytes (HLA-A2– PBLs) were cocultured with T2/Tyr in the absence (non-dimer) or the presence of the soluble Tyr/HLA-A2 dimer (dimer). PBLs cultured alone were for negative control (control). After 7 days of coculturing, the cocultural bulks were tested for CD3, CD8, and interferon-γ (IFN-γ) expression. (A) The frequency of IFN-γ-producing T cells in CD8+ T cells is decreased by the dimer intervention. (a-c) Results of one representative individual are shown. (d) The mean values ± SEM are represented from three independent experiments with markers showing the difference < 0.001, by LSD i-test) (n ≥ 10). (B) The gating strategy is shown for cytometry analysis. The CD8+ T cells were divided into two subsets according to surface CD8 expression measured by the mean fluorescence intensity (MFI), that is, CD8low (MFI: 20-200) and CD8high (MFI: >200) T cells. (C) Most of the IFN-γ-producing T cells are CD8high and little is CD8low. The mean values ± SEM are represented from three independent experiments with markers showing the difference (***p < 0.001, by LSD t-test) (n ≥ 10). (D) The percentage of CD8low T cells in CD8+ T cells is increased after the dimer intervention. (a–c) Results of one representative individual are shown. (d) The mean values ± SEM are represented from three independent experiments with markers showing the difference (***p < 0.001, by LSD t-test) (n ≥ 10).
Approximately 90% of the IFN-γ-producing CD8+ T cells in alloreactive coculture were CD8high T cells, while few were CD8low T cells (Fig. 1C), suggesting CD8high T cells were the major population that were hyperresponsive to the alloantigen. However, the CD8low T-cell population was remarkably increased in the dimer-treated alloreactive coculture (CD8low cells in CD8+ T cells, dimer-treated group vs. non-dimer-treated group: 30.47 ± 3.03% vs. 17.80 ± 1.81%, n ≥ 10, p < 0.001) (Fig. 1D). CFSE dilution assays revealed CD8low T cells proliferated to a greater extent in dimer-treated cocultures compared with non-dimer-treated cocultures, resulting in the higher yield of CD8low T cells in dimer-treated cocultures [CD8low T cells number: (1.57 ± 0.16) × 105 vs. (1.08 ± 0.11) × 105, n = 5, p < 0.001, both cocultures started from 3 × 106 HLA-A2– PBLs per well] (Fig. 2A). Since a majority of the CD8low T cells were CD28– (Fig. 2B), we designated the CD8low T cells as CD8lowCD28– T cells.

Most of the dimer-induced CD8low T cells are CD28–. (A) Soluble HLA-A2 dimer-induced CD8low T cells. (a–c) The proliferation of CD8low T cells was observed after the dimer intervention. The CFSE-labeled HLA-A2– PBLs (3 × 106 cells/well) were cocultured with T2/Tyr in the absence (non-dimer) or presence of soluble Tyr/HLA-A2 dimer (dimer) for 7 days. CD8low T cells in the cocultures were gated for CFSE dilution assay. The proliferated cells were detected as the subpopulation with lower CFSE intensity. Proliferation index (PI) was calculated for the gated CD8low T cells. (d) The dimer-treated coculture yielded more CD8low T cells. The cell yields of fluorescence activated cell sorting (FACS) after 7 days of coculturing, started from 3 × 106 PBLs per well, are shown as mean values ± SEM from three independent experiments. (B) A majority of the dimer-induced CD8low T cells are CD28–. (a–c) CD28 expression of CD8high and CD8low T cells in samples of one representative individual are shown. (d) The percentage of CD28+ cells in CD8high T cells or CD8low T cells are expressed as mean values ± SEM with markers showing difference (n = 6) (***p < 0.001; **p < 0.01 by LSD t-test).
Suppressive Effects of the Dimer-Induced CD8lowCD28–T cells on Alloreactive T cells
A negative correlation between the frequencies of IFN-γ-producing CD8+ T cells and the CD8lowCD28– T cells was revealed by bivariate correlation analysis in both non-dimer-treated (R2 = 0.281, p = 0.042) and the dimer-treated (R2 = 0.314, p = 0.030) alloreactive cocultures, while no correlation was found in the negative control (R2 = 0.01, p = 0.792) (Fig. 3A). This raised the possibility that CD8lowCD28– T cells might act as suppressor cells on alloresponses.

The CD8lowCD28– T cells show hyporesponsiveness and suppression on alloreactive T cells. (A) A negative correlation between CD8lowCD28– and IFN-γ-producing T cells is unveiled by curve estimation and linear regression analyses in the coculture of HLA-A2– PBLs and T2/Tyr with the Tyr/HLA-A2 dimer (dimer) or without the Tyr/HLA-A2 dimer (non-dimer). Correlation coefficients and p values are shown for each analysis (n ≤ 10). (B) The dimer-induced CD8lowCD28– T cells show suppressive effects on alloreactive T cells. The T2/Tyr-primed CD8high were sorted from non-dimer-treated cocultures, and the Tyr/HLA-A2 dimer-induced CD8lowCD28– T cells were sorted from the dimer-treated cocultures through FACS. The CD8high T cells (1 × 105 cells/well) were mixed with the CD8lowCD28– T cells at the indicated ratios and restimulated with the T2/Tyr for 24 h. Then the IFN-γ ELISPOT assays were performed in duplicate for detecting the number of IFN-γ+ cells (n = 4). Mean values ± SEM from four independent experiments are shown (***p < 0.001, and **p < 0.01 by LSD t-test, compared with the CD8high T cells). (C) CD8lowCD28– T cells from the non-dimer-treated coculture are unable to suppress alloreactive T cells. CD8high, CD8lowCD28–, and CD8lowCD28+ T cells were sorted from different coculture bulks by FACS, mixed at ratio of 1:1, then restimulated with T2/Tyr. IFN-γ+ cells were tested for the T-cell alloreactivity, and mean values ± SEM from four independent experiments are shown (***p < 0.001 by LSD t-test, compared with the CD8high T cells).
To testify to the suppressive effects of the CD8lowCD28– T cells, the CD8high T cells were sorted from the non-dimer-treated coculture bulks, and the CD8lowCD28– T cells from the dimer-treated bulks. The freshly sorted CD8high T cells, CD8lowCD28– T cells, or the CD8high T cells mixed with various ratios of the CD8lowCD28– T cells were restimulated by the T2/Tyr, respectively. After 24 h of coculture, the numbers of IFN-γ+ cells were detected by ELISPOT assays. Significantly more spots were detected from the CD8high T cells than those from the CD8lowCD28– T cells upon T2/ Tyr restimulation (IFN-γ+ spots, CD8high vs. CD8lowCD28–: 223.83 ± 32.38/105 cells vs. 31.62 ± 7.13/105 cells; n = 4, p < 0.001) (Fig. 3B). This suggested that CD8high T cells were hyperresponsive, whereas CD8lowCD28– T cells were hyporesponsive toward alloantigens. Moreover, the responsiveness of CD8high T cells was markedly inhibited by the presence of the CD8lowCD28– T cells from dimer-treated coculture in a dose-dependent manner (Fig. 3B). In contrast, the CD8lowCD28– T cells sorted from non-dimer-treated coculture were not able to inhibit the alloresponse even at a 1:1 ratio of CD8lowCD28– to CD8high (Fig. 3C). With respect to the CD8lowCD28+ T cells, the sorted cells from the dimer-treated coculture did not show the suppressive function (IFN-γ+ cells, CD8high vs. CD8lowCD28+ with CD8high at 1:1 ratio: 223.83 ± 32.38/105 cells vs. 242.17 ± 11.45/105 cells; n = 4, p > 0.05). Collectively, these data showed that the dimer-induced CD8lowCD28– T cells were hyporesponsive and bore suppressive effects on alloreactive T cells.
Alloantigen-Specific and Cell–Cell Contact-Dependent Suppression of the CD8lowCD28– T Cells
To define antigen-specific suppression of the dimer-induced CD8lowCD28– T cells, two HLA-A2-restricted peptides, Tyr368-376 and HBC18-27, were used to pulse T2 cells and HLA-A2 dimer; then two sets of CD8high and CD8lowCD28– T cells were prepared from the non-dimer-treated and the dimer-treated cocultures. One set was the T2/Tyr-primed CD8high T cells and the Tyr/HLA-A2 dimer-induced CD8lowCD28– T cells. The other was the T2/HBC-primed CD8high T cells and the HBC/HLA-A2 dimer-induced CD8lowCD28– T cells. The CD8high and CD8lowCD28– T cells were used as responders and suppressors, respectively. T2/Tyr, T2/HBC, and E006 were used as restimulators. After cross-matching incubations of the responders, suppressors, and restimulators, inhibition rate (%) of the IFN-γ spots was calculated. The results indicated that suppression of the CD8lowCD28– T cells was alloantigen specific (Fig. 4A) because matched combinations (e.g., Tyr/HLA-A2 dimer-induced CD8lowCD28– T cells on T2/Tyr-primed CD8high T cells restimulated by T2/Tyr) showed maximum inhibition, and intermediate inhibition was observed for the partially matched combinations (e.g., Tyr/HLA-A2 dimer-induced CD8lowCD28– T cells on T2/HBC-primed CD8high T cells restimulated by T2/HBC), while mismatched combinations (e.g., Tyr/ HLA-A2 dimer-induced CD8lowCD28– T cells on T2/Tyr-primed CD8high T cells restimulated by E006) exhibited enhancement of alloreaction instead of inhibition. Since the HLA-A2 dimer-induced CD8lowCD28– T cells were able to suppress HLA-A2-restimulated alloresponse but not irrelevant alloantigen-restimulated alloresponse (E006 bearing HLA antigens other than HLA-A2), suppression of the CD8lowCD28– T cells appeared to be specific for alloantigen (Table 2). Meanwhile, the difference in inhibition rate between the matched and partially matched combinations suggested that peptide-specific suppression might also exist to some extent (Fig. 4A).

The CD8lowCD28– T cells exhibit alloantigen-specific suppression in a cell–cell contact-dependent manner. (A) Suppression of the dimer-induced CD8lowCD28– T cells is alloantigen specific. Two HLA-A2-restricted peptides, Tyr and hepatitis B virus origin (HBC), were used to pulse T2 cells and HLA-A2 dimer. Hyperresponsive T2/Tyr- or T2/HBC-primed CD8high T cells were restimulated by T2/Tyr, T2/HBC, or Epstein-Barr virus transformed B-lymphoblastoid cell line (E006), and then mixed with suppressive Tyr/HLA-A2 dimer- or HBC/HLA-A2 dimer-induced CD8lowCD28– T cells. ELISPOT assays for IFN-γ production were performed in triplicate, and inhibition rate (%) was calculated. The Tyr/HLA-A2 dimer- (left panel) or HBC/HLA-A2 dimer-induced (right panel) CD8lowCD28– T cells served as suppressors. The results are shown as mean ± SEM of three independent experiments. (B) Cell–cell contact is required for the suppression of CD8lowCD28– T cells. Freshly sorted T2/Tyr-primed CD8high T cells (R, responder), irradiated T2/Tyr (S, restimulator), and the Tyr/HLA-A2 dimer-induced CD8lowCD28– T cells were placed as indicated in a Transwell culture system. After coculturing for 24 h, ELISPOT assays for IFN-γ production were performed in triplicate. The results are shown as mean values of inhibition rate (%) ± SEM from three independent experiments with markers for significance (***p < 0.001 and **p < 0.01 by LSD t-test).
Alloreactive Coculture Assay for Alloantigen-Specific Suppression
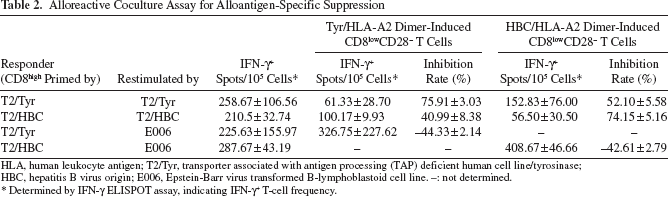
HLA, human leukocyte antigen; T2/Tyr, transporter associated with antigen processing (TAP) deficient human cell line/tyrosinase; HBC, hepatitis B virus origin; E006, Epstein-Barr virus transformed B-lymphoblastoid cell line. –: not determined.
Determined by IFN-γ ELISPOT assay, indicating IFN-γ+ T-cell frequency.
To test whether cell–cell contact was required for the suppression of CD8lowCD28– T cells, the restimulation was set up by coculturing the T2/Tyr-primed CD8high T cells with the T2/Tyr, and the Tyr/HLA-A2 dimer-induced CD8lowCD28– T cells were mixed in the coculture or placed in the Transwell inserts. The inhibition rate was significantly decreased when the CD8lowCD28– T cells were physically separated from the CD8high T cells [the inhibition rate (%) of IFN-γ spots, CD8lowCD28– cocultured with CD8high restimulated with T2/Tyr vs. CD8lowCD28– cultured separately with CD8high mixed with T2/Tyr: 63.70 ± 4.59% vs. 17.55 ± 3.20%, n = 3, p < 0.001] (Fig. 4B), indicating immunosuppression of the CD8lowCD28– T cells on the cognate CD8high T cells was cell–cell contact dependent.
mTGF-β1 Involved in Immunosuppressive Effects of the CD8lowCD28– T Cells
To further characterize the T2/Tyr-primed CD8high T cells and Tyr/HLA-A2 dimer-induced CD8lowCD28– T cells, intracellular stainings for IFN-γ, IL-17A, IL-4, Foxp3, CTLA-4, and IL-10 as well as mTGF-β1 were performed. The results unveiled dimer-induced CD8lowCD28– T cells expressed higher levels of IL-4 and mTGF-β1, but lower levels of IFN-γ than alloreactive CD8high T cells (Fig. 5A–C). No significant difference was observed between two subsets in IL-17A, Foxp3, CTLA-4, or IL-10 expression (Fig. 5A). IL-4, TGF-β1, and IFN-γ secretions were further detected by ELISA with the coculture supernatants (Fig. 5D). The results showed less IFN-γ and higher IL-4 in the supernatants after the dimer intervention (dimer-treated group vs. non-dimer-treated group, concentration of IFN-γ: 1.29 ± 0.19 vs. 1.97 ± 0.24 ng/ml, p < 0.001; concentration of IL-4: 39.15 ± 0.48 vs. 24.51 ± 0.85 pg/ml, p < 0.001; n = 8), which coincided with the results of intracellular staining (IFN-γ+ cells, CD8lowCD28– vs. CD8high: 2.52 ± 0.85% vs. 12.37 ± 2.53%, p = 0.002; IL-4+ cells, CD8lowCD28– vs. CD8high: 12.78 ± 2.81% vs. 1.12 ± 0.53%, p = 0.002; n = 6). This suggested that the CD8lowCD28– T cells might be able to induce type 2 polarization, which is important to prevent allograft from T-cell attack. Although higher mTGF-β1 expression was detected on the CD8lowCD28– T cells (mTGF-β1+ cells, CD8lowCD28– vs. CD8high: 35.50 ± 4.27% vs. 0.53 ± 0.27%, n = 6, p < 0.001), the concentration of secreted TGF-β1 in supernatants of the dimer-treated coculture was much lower (concentrations of TGF-β1, dimer-treated group vs. non-dimer-treated group: 19.54 ± 1.37 vs. 66.20 ± 1.15 pg/ml, n = 8, p < 0.001) (Fig. 5D).
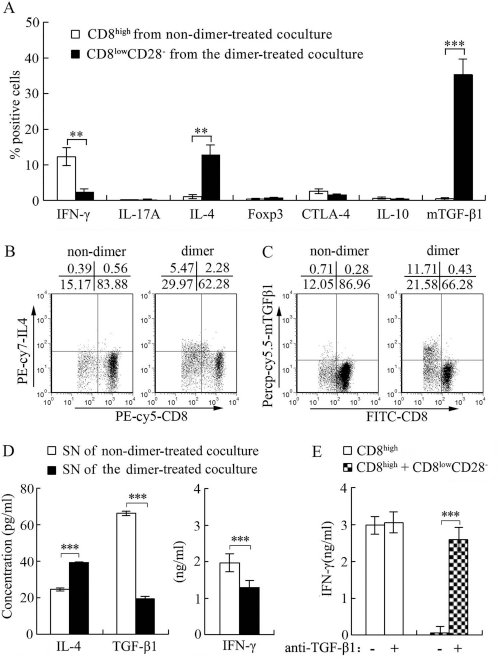
Elevated membrane-bound TGF-β1 (mTGF-β1) expression is involved in suppression of the CD8lowCD28– T cells. (A) Phenotypes of the CD8high and CD8lowCD28– T-cell subsets. The T2/Tyr-primed CD8high T cells and the Tyr/HLA-A2 dimer-induced CD8lowCD28– T cells were sorted and tested for expression of IFN-γ, interleukin (IL)-17A, IL-4, fork head box p3 (Foxp3), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), and IL-10 by intracellular staining. Surface expression of mTGF-β1 was detected using anti-human latency associated peptide (LAP) antibody, BG/hLAP, by flow cytometry. The results are shown as mean ± SEM with markers for statistical significance (n = 6). (B, C) Representative dot plots for IL-4 (B) and mTGF-β1 (C) are shown, respectively. (D) The dimer-treated coculture secretes less soluble TGF-β1. Supernatants (SN) of the alloreactive coculture with or without the Tyr/HLA-A2 dimer intervention were tested by ELISA assays for IL-4, TGF-β1, and IFN-γ secretion. Bars represent mean values of concentration ± SEM of triplicate wells from three independent experiments (n = 8). (E) Suppression of the CD8lowCD28– T cells is blocked by TGF-β1-specific antibody. The sorted CD8lowCD28– and CD8high T cells were mixed at the ratio of 1:2 and restimulated with the T2/Tyr for 24 h in the absence or presence of neutralizing antibody against TGF-β1. The data are shown as mean values of IFN-γ concentration ± SEM from three independent experiments (n = 4) (***p < 0.001 and **p < 0.01 by LSD t-test).
To explore the role of elevated mTGF-β1 level in the suppression, the sorted T2/Tyr-primed CD8high and Tyr/ HLA-A2 dimer-induced CD8lowCD28– T cells were mixed and restimulated with the T2/Tyr for 24 h in the absence or presence of anti-TGF-β1. As shown in Figure 5E, IFN-γ secretion by restimulated CD8high T cells was significantly decreased to background levels after mixing with the suppressive CD8lowCD28– T cells. The presence of neutralizing anti-TGF-β1 did not alter IFN-γ secretion by restimulated CD8high T cells, but abrogated suppressive effects of the CD8lowCD28– T cells and recovered over 80% of the IFN-γ secretion of the CD8high T cells (concentration of IFN-γ in CD8high mixed CD8lowCD28– restimulated with T2/Tyr, anti-TGF-β1 vs. none: 2.98 ± 0.31 vs. 0.05 ± 0.01 ng/ml, n = 4, p < 0.001). Taken together, these data strongly suggested that mTGF-β1 played critical role in the suppressive effect mediated by CD8lowCD28– T cells.
Overrepresented CD8lowCD28– T Cells and Elevated Levels of Plasma HLA Class I in Platelet-Transfusion Patients
The CD8lowCD28– T cells induced in vitro by the intervention of soluble HLA class I dimer exhibited an alloantigen-specific suppression similar to the well-studied CD8+CD28– suppressor T cells. To assess the clinical relevance of the CD8lowCD28– T-cell subset, samples of 20 patients that received allogeneic platelet transfusion were collected and tested for both soluble HLA I plasma levels and CD8lowCD28– T-cell frequencies. In comparison to healthy individuals (n = 19), a marked increase of CD8lowCD28– T cells was observed in the patients (CD8lowCD28– T cells, platelet transfusion patients vs. healthy donors: 10.86 ± 1.60% vs. 2.30 ± 0.35%, p < 0.001) (Fig. 6A, B). Simultaneously, the patients showed significantly higher soluble HLA class I plasma levels (concentration of soluble HLA class I plasma, platelet transfusion patients vs. healthy donors: 885.84 ± 97.71 vs. 247.42 ± 15.42 ng/ml, p < 0.001) (Fig. 6C), which positively correlated with the CD8low CD28– T-cell frequencies (R2 = 0.604, p < 0.01) (Fig. 6D).

The frequencies of CD8lowCD28– T cells and plasma levels of soluble HLA class I are positively correlated in platelet transfusion patients. Peripheral blood samples of 20 platelet transfusion patients were collected within 1 week following the last transfusion, and those of 19 healthy donors served as control. (A, B) CD8lowCD28– T cells are more frequent in platelet transfusion patients. PBLs from healthy donors and platelet transfusion patients were analyzed for surface expression of CD3, CD8, and CD28. (A) Representative dot plots for CD8lowCD28– T cells are shown. (B) The bar graph represents the mean frequency ± SEM. (C) The patients show elevated levels of soluble HLA class I antigens. Soluble HLA class I plasma levels were measured by ELISA using HLA class I-specific antibody (W6/32). The results are shown as mean values ± SEM. (D) Soluble HLA class I plasma levels are positively correlated with CD8lowCD28– T-cell frequencies in patients (R2 = 0.604, p < 0.01). (E, F) Patients with more than three times of transfusion show significantly higher plasma levels of soluble HLA class I and greater frequencies of CD8lowCD28– T cells compared to those with one or two transfusions, respectively. The results are shown as mean values ± SEM (*0.01 < p < 0.05; **0.001 < p < 0.01; and ***p < 0.001 by LSD t-test).
We also found that the number of times of platelet transfusion influenced the soluble HLA class I plasma levels and CD8lowCD28– T-cell frequencies, that is, patients with more than three times of platelet transfusions showed higher levels of soluble plasma HLA class I (concentration of soluble HLA class I, more than three times of platelet transfusion vs. one to two times of transfusion: 1,075.35 ± 159.42 vs. 696.33 ± 83.13 ng/ml, p = 0.049) (Fig. 6E) and greater frequencies of CD8lowCD28– T cells than those in patients with one or two transfusions (CD8lowCD28– T cells, more than three times of platelet transfusion vs. one to two times of transfusion: 14.82 ± 2.39% vs. 6.89 ± 1.25%, p = 0.009) (Fig. 6F). Although mechanisms need to be further clarified, our findings suggested the association of overrepresented CD8lowCD28– T cells, and high levels of plasma HLA class I existed in the platelet transfusion patients in whom alloresponse was ongoing.
Discussion
A naive CD8+ T cell has multiple fates during its primary differentiation (26,36). As an example of this diversity, a subset of T cells with downregulated CD8 expression (CD8low) is described in several situations 1(15,27,31,37,50). Although it is suggested CD8low T cells from healthy donors display increased cytotoxic effects (50), many studies show that CD8low T cells carry out suppressive function, which probably contribute to pathogen-induced immune deviation (15,21) and the generation or maintenance of transplantation tolerance (31,37). In this study, a CD8low T-cell subset with CD28– expression designated as CD8lowCD28– T cells was induced by the intervention of soluble peptide/HLA-A2 dimer in an alloreactive coculture. The CD8lowCD28– T cells were hyporesponsive to the alloantigen and possessed alloantigen-specific suppressive function on alloreactive T cells in a cell–cell contact-dependent fashion, suggesting the cells would be a subpopulation of suppressor T cells. Recent studies show that CD8+ suppressor T cells are heterogeneous and are characterized by the lack of expression of CD28 and/or the presence of regulatory molecules like Foxp3, IL-10, CTLA-4, or TGF-β1 (12,47). The well-documented CD8+CD28– suppressor T cells include CD8+CD28–Foxp3+, CD8+CD28–CTLA4+Foxp3+, and CD8+CD28– suppressor T cells with IL-10 or/and TGF-β1 secretion (8,14,17,25). The CD8lowCD28– population described in this study showed neither elevated expression of Foxp3 or CTLA-4, nor secretion of IL-10 or TGF-β1, although the regulatory factors are highly vital for the suppressive function of suppressor T cells (23,30,54). Of note, the expression of mTGF-β1 on the suppressive CD8lowCD28– T cells was significantly increased, while secreted TGF-β1 in the supernatants was markedly diminished. Like other TGF-βs, TGF-β1 is synthesized as a precursor molecule containing a latency associated peptide (LAP) in addition to the TGF-β homodi-mer. mTGF-β1 is considered to be in a latent form without bioactivity due to its noncovalent association with LAP, but recent studies suggest some regulatory T cells exhibit its suppressive effect via mTGF-β1 (30,38,43,45). In this study, the elevated mTGF-β1 expression was also revealed to play a critical role in the cell–cell contact-dependent suppression mediated by CD8lowCD28– T cells because neutralizing anti-TGF-β1 abrogated the suppressive effects (Figs. 4 and 5).
Meanwhile, immunosuppression of CD8lowCD28– T cells was alloantigen specific. Peptide-specific suppression was also observed to some extent, since the Tyr/HLA-A2 dimer-induced CD8lowCD28– T cells showed more effective inhibition on T2/Tyr-restimulated alloresponse than that on T2/HBC-restimulated alloresponse (Fig. 4A). As peptide-specific, peptide-dependent, and peptide-independent TCR– alloMHC interactions are reported to be involved in T-cell allorecognition (11), our observations suggested that these allorecognition mechanisms would also be involved in suppressive effects of the CD8lowCD28– T cells. Substantial studies reveal that CD8+CD28– suppressor T cells exhibit suppression through acting directly on antigen presenting cells (APCs) by upregulation of inhibitory receptors immunoglobulin-like transcript 3 (ILT3) and ILT4, and down-regulation of costimulatory molecules, which render the APCs tolerogenic (8,32). The cell–cell contact-dependent suppression of the CD8lowCD28– T cells suggested APCs (T2/peptide) would serve as a bridge to link the suppressive CD8lowCD28– T cells and hyperresponsive CD8high T cells.
Although the mechanisms governing the generation of CD8lowCD28– T-cell phenotype remains unknown, type 2-polarized circumstance, like high levels of IL-4, can expand dysfunctional CD8low T cells in vitro and in vivo (28,29,31). Meanwhile, since IL-4 is reported to act as a growth factor and enhance long-term survival of CD28-deficient T cells (49), the increased level of IL-4 observed in this study after HLA-A2 dimer intervention might also account for induction and survival of the CD8lowCD28– T cells. Furthermore, soluble MHC II/ Fc chimeric molecule has been shown to reduce type 1 response by negative regulation of signal transducer and activator of transcription 4 (STAT4), with compensatory augmentation of the STAT6 pathway for type 2 differentiation (7). Similar mechanisms of immune deviation might be plausible for outgrowth of the CD8lowCD28–T cells. We postulated that the HLA-A2 dimer intervention resulted in elevated IL-4 secretion via presently incompletely defined mechanisms, followed by induced type 2-polarized CD8lowCD28– T cells. This population might represent a feedback response for more IL-4 secretion and outgrowth of CD8lowCD28– T cells, as anti-IL-4 mAb was able to partially abrogate the suppressive effects mediated by soluble HLA-A2 dimer (data not shown). In addition, overrepresented CD8lowCD28– T cells and increased soluble HLA class I plasma levels were observed in platelet transfusion patients (Fig. 6). Generally, serum levels of soluble HLA class I molecules are markedly increased in immunologically active status, for example, in autoimmune diseases, graft rejection, viral infections, and cancer (4,5,10). When patients received random-donor platelet transfusions, HLA class I molecules of donor platelets impose an alloreaction to the recipient, which is often considered the reason of refractory to platelet transfusions (13,16,22). The increased levels of soluble HLA class I molecules were positively correlated with the frequencies of CD8lowCD28– T cells, suggesting the expansion mechanism(s) similar to that of our HLA-A2 dimer intervention in vitro would happen in the patients.
Our findings suggested that soluble HLA dimer-induced CD8lowCD28– T cells would be, at least, a subset of the well-documented CD8+CD28– suppressor T cells. As CD8+CD28–suppressor T cells are found to play an important role in the induction and maintenance of tolerance toward a transplanted organ in a donor-specific way, it required a practical approach in preparing a sufficient number of the T cells to find their applications. Theoretically, soluble HLA dimer-induced CD8lowCD28– T cells with alloantigen-specific suppressive activity could be prepared in large quantities in vitro, which would provide therapeutic perspectives for protecting allografts from rejection (20). Although soluble HLA dimer shows antigen-specific inhibition on alloresponse in vitro (53), injection of mismatched HLA dimer into the recipient would incur anti-HLA antibody and thus result in tissue damage in addition to diminishing the immunosuppression. Suppressive CD8lowCD28– T cells prepared in the presence of the mismatched HLA dimer in vitro and transferred back into the recipient would be expected to provide protection of HLA-mismatched allografts. However, suppressive properties of the CD8lowCD28– T cells described in this study were observed in an alloreactive coculture in vitro. Questions arising are if the T cells develop in transplanted patients with a tolerance outcome and if the T cells work in vivo. These questions need to be addressed before translation of this strategy into clinical applications.
Footnotes
Acknowledgments
The authors specially thank Xiaolin Wu, Aina Cui, Shigang Shan, and Lifeng Jin for technical assistance; Sreya Bagchi for language editing. This work was supported by grants from the National Natural Science Foundation of China (No. 30801017, No. 30972697), the “973” project of Chinese Department of Science and Technology (No. 2007CB512900), and the International Science and Technology Cooperation Program of China (2011DFA31030) and Deutsche Forschungsgemeinschaft (SFB/Transregio TRR60). Z.G.W., L.C.O.Y., X.F.W., and X.W.W. designed the research and analyzed the data; X.F.W., Z.G.W., and L.C.O.Y. performed the experiments and wrote the manuscript. Z.H.L., J.C., Q.Y., and Y.L.G. performed partial research. V.T.J., G.X.S., and X.W.W. helped revise the manuscript. All authors reviewed and approved the manuscript and final version. The authors declare no conflict of interest.
