Abstract
It has been reported that rapamycin (RPM) can induce de novo conversion of the conventional CD4+Foxp3-T cells into CD4+Foxp3+ regulatory T cells (iTregs) in transplantation setting. It is not clear whether RPM can similarly generate suppressor CD8+ T cells to facilitate graft acceptance. In this study, we investigated the ability of short-term RPM treatment in promoting long-term acceptance (LTA) of MHC-mismatched skin allografts by generating a CD8+ suppressor T-cell population. We found that CD4 knockout (KO) mice (in C57BL/6 background, H-2b) can promptly reject DBA/2 (H-2d) skin allografts with mean survival time (MST) being 13 days (p < 0.01). However, a short course RPM treatment in these animals induced LTA with graft MST longer than 100 days. Adoptive transfer of CD8+ T cells from LTA group into recombination-activating gene 1 (Rag-1)-deficient mice provided donor-specific protection of DBA/2 skin grafts against cotransferred conventional CD8+ T cells. Functionally active immunoregulatory CD8+ T cells also resided in donor skin allografts. Eighteen percent of CD8+ suppressor T cells expressed CD28 as measured by flow cytometry, and produced reduced levels of IFN-γ, IL-2, and IL-10 in comparison to CD8+ effector T cells as measured by ELISA. It is unlikely that CD8+ suppressor T cells mediated graft protection via IL-10, as IL-10/Fc fusion protein impaired RPM-induced LTA in CD4 KO mice. Our data supported the notion that RPM-induced suppressor CD8+ T cells home to the allograft and exert donor-specific graft protection.
Introduction
Induction of immunological tolerance to transplanted grafts remains an elusive goal. Recent advances in characterizing CD4+Foxp3+ regulatory T cells (Treg), a subset of CD4+ T cells known to be essential for creation and maintenance of self-tolerance (34,36,38,43), have led to a renaissance of this field, and efforts have been focused on reinvigorating the traditional immunosuppressive agents with novel applications in promoting Tregs. Rapamycin (RPM) has been shown to enable the expansion of natural Tregs and induce the de novo generation of Tregs in murine models (2,44,45). More recently, RPM has been reported to elevate Treg proportions in renal transplant recipients (18). It is not clear whether RPM can similarly generate suppressor CD8+ T cells to facilitate acceptance.
It is generally taken that there exist multiple types of regulatory T cells that may exert suppressive functions under specific conditions (22,37,48). Therefore, thorough investigation of suppressor T cells under each unique condition is necessary before considering T-cell manipulation or enrichment therapies (23,37,41,42). Recent evidence suggests that certain CD8+ T lymphocytes can act as regulatory cells in suppressing immune responses (15). These CD8+ Treg cells can suppress autoimmune experimental encephalomyelitis and play a role in oral tolerance (21,24,30). Nevertheless, the precise identity of these cells and the molecular basis for their immunosuppressive properties has not been fully characterized, particularly in transplant models. Several cell surface markers are reported to be associated with CD8+ immunoregulatory T cells, including CD25, CD122, Qa-1, and lack of CD28 (3,12,19,28). Herein, we describe the effect of RPM on the development of CD8+ T-cell-driven immunoregulation in a fully major histocompatibility complex (MHC)-mismatched skin transplant model using CD4 knockout (KO, H-2b) recipients, in the absence of the dominant CD4+Foxp3+ Treg population.
Materials and Methods
Animals
Male C57BL/6 CD4KO (H-2b) (CD4tm1Mak; CD4 antigen; targeted mutation 1, Tak Mak), C57BL/6 CD8KO (H-2b) (CD8atm1Mak; CD8 antigen, alpha chain; targeted mutation 1, Tak Mak), DBA/2 (H-2d), C3H/He (H-2k), and C57BL/6J-Rag KO (H-2b) (Rag1tm1Mom, recombination activating gene 1; targeted mutation 1, Peter Mombaerts) mice were purchased from the Jackson Laboratory (Bar Harbor, ME). Animal use and care was in compliance with guidelines established by the animal care committee at Beth Israel Deaconess Medical Center (Boston, MA).
Reagents
The following anti-mouse monoclonal antibodies (mAbs) were purchased from BD Pharmingen (San Diego, CA) and used for cell surface and intracellular staining: fluorescein isothiocyanate (FITC)-labeled anti-mouse CD4 (clone RM4–5), anti-mouse CD8 (53–6.7) and isotype control Abs; phycoerythrin (PE)-labeled anti-CD25 (3C7), anti-CD122 (TM-β1), anti-CD28 (37.51), anti-CD44 (IM7), anti-IL-10 (interleukin-10; JES5–16E3), anti-IFN-γ (interferon-γ; XMG1.2), anti-Foxp3 (forkhead box P3; MF23), anti-CD11b (M1/70), anti-GR1 (granulocyte differentiation antigen; RB6 8C5), anti-CD4 (GK1.5), anti-CD45R/B220 (RA3 6B2), anti-erythrocytes (TER-119), anti-Fcγ-RII/III (2.4G2), purified rat anti-mouse CD4 (H129.19), anti-CD8a (53-6.7), anti-CD3 (145-2C11), and anti-CD28 (37.51); and CyChrome anti-mouse CD8 (53-6.7). Other reagents and their purveyors include: anti-rat IgG (Dynal Biotech Inc., Carlsbad, CA), magnetic activated cell sorter (MACS) columns (Miltenyi Biotic Inc., Auburn, CA), mitomycin C (Sigma-Aldrich, St. Louis, MO), [3H]methylthymidine (NEN Research Products, Boston, MA), Super Frost Plus glass slides (Fisher Scientific, Pittsburgh, PA), 96-well U-bottom plates from Becton Dickinson (Franklin Lakes, NJ), and nylon cell strainer (BD, Bedford, MA). RPM (Wyeth-Ayerst, Princeton, NJ) was prepared in carboxymethyl cellulose for IP injections. IL-10/Fc was produced and purified as previously reported (49).
Skin Transplant and Immunosuppression Protocol
Full-thickness tail skin grafts (1 cm2) from donor mice were transplanted onto the thoracic wall of recipient mice. The skin grafts were secured with an adhesive bandage for 7 days posttransplantation. One group of recipient mice was treated with RPM at 3 mg/kg/day IP on days 0, 1, and 2 followed by treatment every other day for 2 weeks, as previously reported (26), while the second group was not treated. IL-10/Fc, a long-lived form of IL-10 (49), was administered as a monotherapy or in combination with RPM at 5 μg (IP) every other day for 5 days in the designated study groups (49). Graft survival was assessed by daily visual inspection. Rejection was defined as the complete necrosis and loss of viable skin tissue.
Preparation of Purified CD8+ T Cells
CD8+ T cells were purified as previously described (29). After red blood cells (RBCs) were lysed by hypotonic shock, lymph node and spleen cells were depleted of macrophages, granulocytes, B cells, and erythrocytes by incubating them first with anti-CD11b (Mac-1) mAb, anti-GR1 (8C5) mAb, anti-CD4 (GK1.5) mAb, anti-CD45R/B220, and anti-erythrocytes mAbs and then treated with magnetic beads coupled to anti-rat Ig. Purified CD8+ T cells were negatively selected using MACS columns. The purity of the resultant CD8+ T-cell populations as determined by flow cytometry was greater than 95% in all experiments. CD8+ T cells purified from the naive CD4 KO mice were used and defined as effectors, while the CD8+ T cells purified from the long-term acceptance (LTA) RPM-treated CD4 KO recipients were used and defined as Tregs.
Adoptive Cell Transfer
CD8+ T cells were obtained from CD4 KO naive mice or LTA RPM-treated hosts >100 days post-skin grafting, and were adoptively transferred in various mixtures into Rag-/- hosts via tail vein injection. Mice were transplanted 24 h later with skin allografts.
Cell Staining and Flow Cytometry
Pooled lymph nodes and spleens were homogenized in PBS [supplemented with 0.5% fetal calf serum (FCS) and 0.2% NaN3] with a nylon cell strainer, and distributed in 96-well U-bottom plates (4 × 106 cells per well). Blood samples were drawn from the tail of control Rag-/- mice, or from the Rag-/- recipient mice 30 days after retransplantation of RPM-treated LTA skin or fresh DBA/2 grafts. RBCs were lysed by hypotonic shock. Immunostaining of these cells was performed on ice for 30 min per step. To prevent nonspecific binding of mAb, all samples were preincubated with blocking anti-Fcγ-RII/III mAb (2.4G2). The following Abs combinations were used for surface phenotype analysis: CyChrome anti-CD8, FITC anti-CD4, PE anti-CD25/anti-CD28/anti-CD122/anti-CD44. For intracellular staining of Foxp3, cells were first surface stained with FITC anti-CD8, fixed, permeabilized, and then stained with PE-anti-Foxp3, anti-IFN-γ (XMG1.2), or anti-IL-10 (JES5?16E3). All samples were analyzed using a FACSort equipped with CellQuest software (BD Biosciences, Mountain View, CA). Data were collected and analyzed by gating on CD8+ populations.
In Vitro Proliferation and Cytokine Production
CD8+ T cells were purified from lymph nodes and spleen of naive CD4 KO mice or from LTA RPM-treated hosts. CD8+ T cells were then cultured in 96-well U-bottom plates coated with anti-CD3 (1.0 mg/ml) and anti-CD28 mAbs (0.5 mg/ml) and culture supernatants at day 3, 5, and 7 were collected. IL-2, IL-4, IL-5, IL-6, IL-10, tumor necrosis factor-α (TNF-α) and IFN-γ production was measured by ELISA assays. Data are representative of three separate experiments.
In Vitro Suppression Assay
Spleen cells from both DBA/2 and C3H/He mice were depleted of T cells with anti-CD4/CD8-coated beads, treated with mitomycin C at 50 μg/ml for 30 min, and used as stimulators (8 × 104) in U-bottom 96-well plates. CD8+ T cells from both naive and RPM-treated LTA CD4 KO recipient mice were purified and used as responders in mixed lymphocyte reaction (MLR). Varying numbers of CD8+ T cells (0, 1 × 105, and 2 × 105) were added to the MLR culture for 4 days. Cells were pulsed with [3H]methylthymidine (0.5 μCi/well) in triplicate for the last 12 h of culture before harvesting and incorporated radioactivity was counted. Data are representative of three separate experiments.
Histopathology and Immunohistochemistry
Skin grafts from LTA RPM-treated CD4 KO mice 100 days posttransplantation and tail skins from naive DBA/2 mice were removed, snap frozen, and stored at −80°C. The snap frozen tissues were cut into 5-μm sections, mounted on Super Frost Plus glass slides, fixed in formalin for 5 min, and stained in H&E for histological evaluation. Alternatively, the tissue sections were fixed in ice-cold acetone, rinsed in PBS, blocked with 7% horse serum for 30 min, and stained with 1 μg/ml anti-CD4 and anti-CD8 primary mAb overnight at 4°C. The tissue sections were then blocked with 1% hydrogen peroxide and incubated with 2 μg/ml biotinylated rabbit anti-rat secondary antibody for 60 min at room temperature. Tissue sections were then treated with avidin-biotin complex-HRP for 30 min and the signal was visualized with 3,3-diaminobenzidine.
Statistical Analyses
Statistical analysis was performed using GraphPad Prism 5 software. Graft survival times were analyzed by Kaplan-Meier survival analysis and the statistical difference were measured using the log rank test. In the MLR, comparison of the counts per minute (cpm) between the outcome of the three compared types of CD8+ T cells (donor-specific or donor-nonspecific stimulator cells mixed with tolerant CD8+ T cells vs. donor-specific stimulator cells mixed with naive control CD8+ T cells) in the main two groups (1 × 105 and 2 × 105 cells) was performed using the two-way ANOVA method, followed by Bonferroni posttest. To compare FACS analysis data from CD8+ T cells isolated from naive versus LTA mice, we used unpaired t-test. And for ELISA cytokine assay, we used chi-square test to compare naive versus tolerant groups (day 5 and day 7). A value of p < 0.05 was considered statistically significant.
Results
Short-Term Treatment of Rapamycin Induces Long-Term Acceptance and Generation of CD8+ Suppressor T Cells
CD4 KO mice were transplanted with DBA/2 skin allografts and were treated with or without RPM as described elsewhere (26). Long-term skin allograft survival (>100 days) was achieved in all RPM-treated CD4 KO mice (n = 6), whereas rapid rejection (MST = 13 days) was noted in untreated controls (n = 6, p = 0.004) (Fig. 1a).
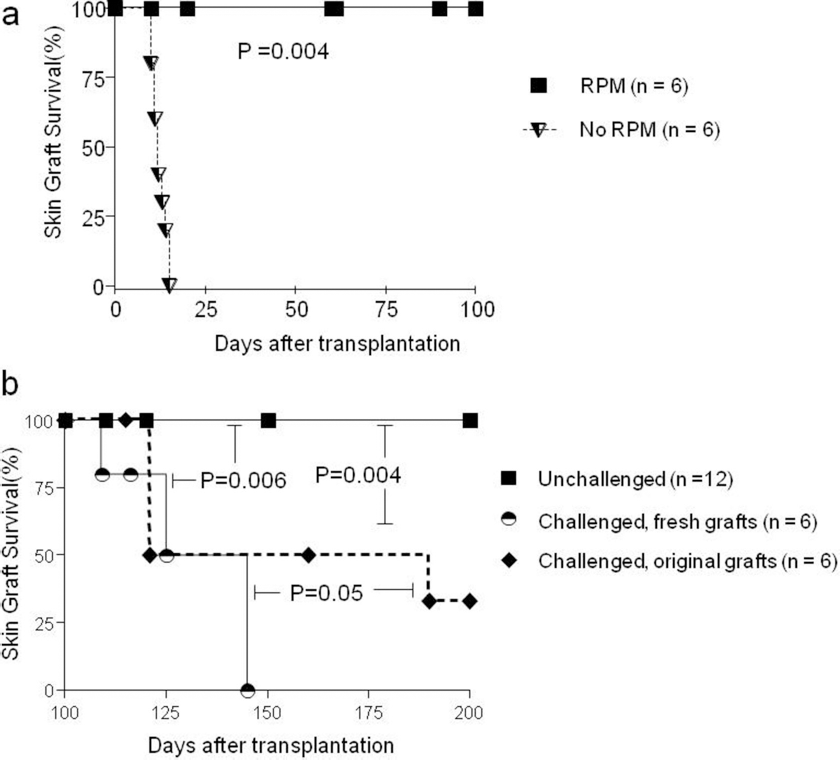
Short-term treatment of rapamycin (RPM) induces long-term acceptance and generation of CD8+ suppressor T cells. (a) C57BL/6 CD4 knock out (KO) mice were transplanted with DBA/2 tail skin allografts, and were either left untreated or treated with RPM as described in Materials and Methods. RPM induced universal acceptance of the grafts for >100 days, whereas untreated grafts were promptly rejected with mean survival time (MST) at 13 days. (b). After 100 days of acceptance, mice bearing the surviving grafts were either unchallenged, or challenged with the fresh second DBA/2 skin grafts. Rechallenging induced graft rejection, yet the original grafts were rejected slower than the fresh grafts.
To determine whether LTA skin grafts from RPM-treated recipients were protected by CD8+ suppressor T cells, we did the following experiment. We challenged RPM-treated hosts (n = 6) with a second DBA/2 skin graft at 100 days posttransplantation of the first graft, long after the cessation of RPM treatment. Both the first and the second DBA/2 grafts were rejected, but the first grafts were rejected slower than the newly transplanted second grafts (p = 0.05) (Fig. 1b), suggesting that the first graft might be protected by infiltrated suppressor-type T cells.
CD8+ Suppressor T Cells Exhibit Donor-Specific Hyporesponsiveness and Exert Strain-Specific Graft Protection Upon Transfer
We assessed the alloantigen specificity of the CD8+ suppressor T cells in MLR in vitro. CD8+ T cells from naive CD4 KO mice or from LTA RPM-treated hosts were stimulated with mitomycin C-treated splenocytes from donor DBA/2 (H-2d) or third-party C3H/He (H-2k) mice. Compared to CD8+ T cells of naive CD4 KO mice, CD8+ T cells isolated from lymph nodes and spleen of LTA mice were hyporesponsive to DBA/2 donor strain stimulator cells (p = 0.04), but responded well to the third-party C3H/He stimulator cells (Fig. 2a).
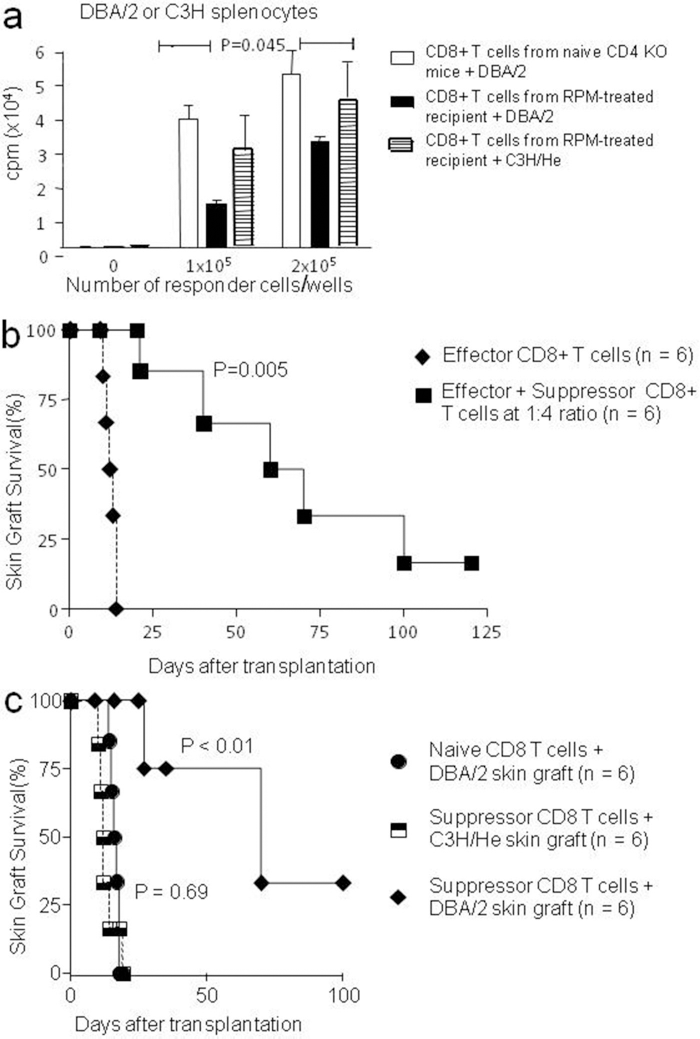
CD8+ suppressor T cells exhibit donor-specific hyporesponsiveness and exert strain-specific graft protection upon transfer. (a). CD8+ T cells (1–2 × 105) from naive CD4 KO mice or from long-term acceptance (LTA) RPM-treated CD4 KO mice were stimulated with 4 × 105 mitomycin C-treated DBA/2 (H-2d) or C3H (H-2k) splenocytes in 96-well U-bottom plate for 3 days. T-cell proliferation in these cultures was measured by incorporated [3H]thymidine of triplicate wells. Although this hyporesponsivenes of CD8+ T cells isolated from LTA mice compared to CD8+ T cells of naive CD4 KO mice was statistically significant as measured by two-way ANOVA (p = 0.045), Bonferroni posttest showed no statistical significance. (b). Naive CD8+ T cells (0.2 × 106) alone or in mixture with suppressor CD8+ T cells (0.8 × 106) at 1:4 ratio were transferred into recombination activating gene KO (Rag-1-/-) mice, followed by transplanting DBA/2 skin allografts 24 h later. (c) CD8+ T cells (0.2 × 106) from naive CD4 KO mice or from LTA RPM-treated CD4 KO were adoptively transferred into Rag-1-/- mice, followed by transplanting DBA/2 (H-2d) or C3H/He (H-2k) skin allografts 24 h later.
Next, we tested the strain-specific graft protection by these CD8+ T cells in vivo. We transferred a mixture of CD8+ suppressor T cells from LTA mice plus CD8+ effector T cells from naive CD4 KO mice at 4:1 ratio into Rag-1-/- mice. As a control, Rag-1-/- mice were transferred with CD8+ effector T cells alone. Twenty-four hours later, both groups were transplanted with DBA/2 skin grafts. Transfer of 0.8 × 106 CD8+ suppressor T cells powerfully suppressed the ability of 0.2 × 106 CD8+ effector T cells in rejecting the DBA/2 skin (p = 0.005) (Fig. 2b). Then, we transferred 2 × 105 CD8+ T cells from naive mice or from LTA RPM-treated hosts into Rag-1-/- mice (n = 6 for each group), followed by transplanting DBA/2 (H-2d, n = 6) or C3H/He (H-2k, n = 6) skin grafts 24 h later. Compared to CD8+ effector T cells from naive mice, CD8+ T cells from the LTA RPM-treated mice had a much delayed rejection tempo of DBA/2 skins (p < 0.01), with about 30% of the grafts surviving long term (>100 days). In sharp contrast, CD8+ T cells from the LTA RPM-treated mice promptly rejected the third-party C3H/He skins at a tempo no different from that of CD8+ T effector cells from naive mice (p = 0.69) (Fig. 2c, and data not shown).
Functionally Active Immunoregulatory CD8+ T Cells Reside in Donor Skin Allografts and Facilitate LTA in RPM-Treated CD4 KO Mice
After we demonstrated in the aforementioned experiments that CD8+ T cells present in the secondary lymphoid organs of LTA RPM-treated mice were suppressing donor-specific alloimmune response, we next investigated whether these CD8+ Treg cells home to the LTA DBA/2 skin allografts. For this, we performed histopathology and immunohistochemistry analysis of skin allografts (n = 3) from RPM-treated mice after 100 days of acceptance. The results revealed that DBA/2 grafts from LTA RPM-treated hosts, but not the freshly isolated DBA/2 tail skins, harbored CD8+ T cells (Fig. 3a, b).

Functionally active immunoregulatory CD8+ T cells reside in donor skin allografts and facilitate LTA in RPM-treated CD4 KO mice. Immunohistochemistry was performed in (a) the skin allografts after 100 days of transplantation from LTA RPM-treated mice or in (b) the normal DBA/2 tail skin. Numerous CD8+ T cells (arrows) can be detected in the LTA RPM-treated grafts, but not in the normal skins (magnification 40x; scale bar 0.1 μM). (c) Graft-infiltrating CD8+ T cells exited from the tolerant graft into the circulation of lymphopenic hosts. LTA DBA/2 allografts (100 days posttransplantation, n = 5) or syngeneic DBA/2 grafts (30 days post autotransplantation, n = 5) were transplanted onto Rag-1-/- hosts. As another control group, fresh DBA/2 tail skin allografts were transplanted directly onto Rag-1-/- hosts (n = 6). Peripheral blood lymphocytes (PBL) samples were collected 30 days post-skin transplantation. (d). RPM-treated major histocompatibility complex (MHC) fully mismatched skin allografts in CD4 KO recipients are protected from rejection by graft-infiltrating and functionally active CD8+ Treg cells. CD8+ T cells (0.2 × 106) from naive B6 CD4KO mice were adoptively transferred into Rag-1-/- hosts bearing either LTA DBA/2 allografts, DBA/2 allografts retransplanted 30 days after syngeneic transplantation, or freshly transplanted DBA/2 allografts. Under such rechallenge by CD8+ effector T cells, the survival of LTA DBA/2 skin allografts was significantly prolonged compared to that of the control DBA/2 grafts that have not been treated with RPM (p = 0.02).
Then, we investigated if the graft-infiltrating CD8+ T cells were functionally active in protecting the allografts from rejection. Due to their limited number, we used the following method to expand the graft-infiltrating CD8+ suppressor T cells: DBA/2 skin allografts (n = 5) harvested 100 days posttransplantation from LTA RPM-treated CD4 KO mice were retransplanted onto C57BL/6 Rag-1-/- mice. As a control for any resident CD8+ T cells from the skin and for the transplantation process, we transplanted DBA/2 skin onto syngeneic DBA/2 hosts. Thirty days after syngeneic transplantation, we retransplanted DBA/2 syngeneic grafts (n = 55) onto C57BL/6 Rag-1-/- mice as allografts that have not been treated with RPM. As another control, freshly prepared DBA/2 skin allografts were transplanted directly onto Rag-1-/- hosts (n = 56). After a month, any graft-residing T cells will migrate out from the graft, repopulate the lymphopenic host, and can be detected in the circulation. FACS analysis showed that CD8+ T cells could not be detected in peripheral blood lymphocytes (PBL) from Rag-1-/- mice of the two control groups, whereas a significant population (4.6% of PBL) of CD8+ T cells was present in PBL of Rag-1-/- recipients of LTA allografts (p < 0.01) (Fig. 3c). To test for the protective function of graft-homing CD8+ T cells (from LTA RPM-treated mice) that expanded by homeostatic proliferation in Rag-1-/- mice, we adoptively transferred 0.2 × 106 CD8+ effector T cells from naive mice into Rag-1-/-hosts bearing either LTA DBA/2 allografts, DBA/2 allografts retransplanted 30 days after syngeneic transplantation, or freshly transplanted DBA/2 allografts. Under such rechallenge by CD8+ effector T cells, the survival of LTA DBA/2 skin allografts was significantly prolonged as compared to that of the controls DBA/2 grafts that have not been treated with RPM (p = 50.02) (Fig. 3d). This indicates that RPM-treated MHC fully mismatched skin grafts in CD4 KO recipients are protected from rejection by graft-infiltrating and functionally active CD8+ Treg cells.
Molecular Profiles of RPM-Induced CD8+ Suppressor T Cells
Based on previous reports (3,12,28), we studied the expression of surface markers CD25, CD28, CD44, CD122, as well as intracellular cytokine IFN-γ and IL-10 in CD8+ suppressor T cells. We found that about 18% of CD8+ suppressor T cells from LTA RPM-treated mice expressed CD28, compared to 3.5% in CD8+ effector T cells from control mice (p < 0.01) (Fig. 4a). There were no significant differences of other surface markers such as CD25, CD44, and CD122 between the two types of CD8+ T cells. In addition, while >10% of cells in the CD8+ effector T cell population expressed IFN-γ, only 2.3% of cells in the CD8+ suppressor T-cell population expressed this cytokine (p < 0.01) (Fig. 4a). There was no difference in IL-10+ cells between the two groups.

RPM-induced CD8+ suppressor T cells do not produce interferon-γ (IFN-γ). (a). CD8+ T cells from either naive or LTA RPM-treated CD4 KO mice were gated for flow cytometric analysis of cell surface markers and intracellular cytokines. Results are representative of three independent experiments. (b). Purified CD8+ T cells (2 × 105 cells per well) from either naive or LTA RPM-treated CD4 KO mice were stimulated with anti-CD3 (1.0 mg/ml) and anti-CD28 (0.5 mg/ml) in 96-well U-bottom plate and the supernatants were collected on day 5 and 7 for ELISA. Data are presented as mean ± SD values of triplicate wells. Results are representative of three independent experiments.
Next, we stimulated CD8+ T cells from LTA RPM-treated mice or from naive CD4 KO mice with plate-bound anti-CD3 and soluble anti-CD28 mAb. ELISA analysis of day 5 and day 7 supernatants revealed that IFN-γ (p < 0.01), IL-2 (p < 0.05), and IL-10 (p < 0.05) were more robustly expressed by CD8+ T cells from naive mice, compared to those from the LTA RPM-treated CD4 KO mice (Fig. 4b). Although CD8+ T cells from naive mice produced elevated levels of IL-5, IL-6, and TNF-α compared to those from the LTA RPM-treated CD4 KO mice, the difference did not reach statistical significance.
IL-10/Fc Does Not Potentiate RPM to Induce CD8+ Suppressor T Cells
Although RPM-induced CD8+ suppressor T cells did not seem to express IL-10, it is possible that RPM could induce the expression of this cytokine from antigen-presenting cells to potentiate the generation of CD8+ suppressor T cells. To test this possibility, CD4 KO or CD8 KO mice (only bearing CD8+ or CD4+ T cells, respectively) were transplanted with DBA/2 skin grafts, and were either nontreated or treated with RPM alone, IL-10/Fc protein alone (IP 5 μg every other day for 5 days), or RPM + IL-10/Fc in combination. IL-10/Fc alone was ineffective and even impaired, not improved, the graft survival in CD4 KO mice, regardless of RPM treatment (Fig. 5a). IL-10/Fc also failed to improve graft survival in CD8 KO mice (Fig. 5b). Thus, IL-10 is unlikely to mediate the beneficial effect of RPM in promoting CD8+ suppressor T-cell generation.

Interleukin 10 fusion protein (IL-10/Fc) does not potentiate RPM to induce CD8+ suppressor T cells. (a) Four groups of C57BL/6 CD4 KO mice (n = 6 per group) that do not have CD4+ T cells were transplanted with DBA/2 skins, and were either untreated or treated with RPM or IL-10/Fc protein alone or in combination. (b) Similar to (a) except that the recipients were C57BL/6 CD8 KO mice (n = 4 per group) that do not have CD8+ T cells. Graft survival was recorded as described in Materials and Methods.
Discussion
Rapamycin (RPM) is widely used in the clinic to prevent transplant rejection. This drug is a potent and reasonably well-tolerated immunosuppressive agent. In collaboration with the Turka laboratory, we have previously reported that RPM promotes the apoptotic death of many alloreactive T cells, thereby promoting tolerance (26,47). In several other models, RPM enhances the efficacy of costimulatory blockade-based therapy to induce graft tolerance (4,27,39,40), while calcineurin inhibitors block tolerance induction in these models (14,26,47). We previously demonstrated that RPM promotes and synergizes with anti-CD154 to convert peripheral alloreactive CD4+Foxp3- T cells into apoptosis-resistant Foxp3+ iTreg cells that can mediate donor-specific skin graft protection upon transfer (14). In a companion study using a pretransplant conditioning regimen of donor-specific transfusion plus anti-CD154 mAb, the enhanced donor-directed Treg activity in the CD4+CD25+ T-cell pool could be further strengthened by addition of RPM but not calcineurin inhibitors (14).
In this study, we probed for the presence of the CD8+ T cells with donor specific regulatory function. For clarity we utilized CD4 KO mice as recipients. CD4 KO mice vigorously reject skin allografts (46). The mammalian target of rapamycin (mTOR)-mediated signaling events seem absolutely essential for CD8+ T cell-mediated rejection, as RPM, an mTOR inhibitor, preferentially inhibits the in vivo expansion of alloactivated CD8+ T cells and uniformly produces LTA in CD4 KO mice (46). Herein we have corroborated the ability of short-term RPM therapy to promote LTA of allogeneic skin transplants in CD4 KO mice (Fig. 1a). In these hosts, allospecific CD8+ suppressor T cells are present and these cells can cause donor-specific hyporesponsiveness in vitro (Fig. 2a) and protection of fully mismatched allogeneic skin grafts in vivo (Fig. 2b).
Following retransplantation of skin allografts harvested from stable LTA RPM-treated CD4 KO mice onto Rag-1-/- mice, graft-infiltrating CD8+ suppressor T cells migrated from the graft site, expanded in the new host, and protected test allografts from acute rejection after transfer of challenging CD8+ effector T cells (Fig. 3). A similar finding concerning the CD4+ Treg has been reported by Graca et al. (16) in a model of peripheral tolerance to minor histocompatibility antigen (Ag)-mismatched skin allografts and by our laboratory (11) in a model in which skin allograft tolerance is achieved via bone marrow transplantation, RPM, and costimulation blockade. Thus, in this model CD8+ Treg are present in both secondary lymphoid organs and reside in the allograft itself. These graft-homing CD8+ Treg cell are functionally active in protecting the grafts from rejection.
Cosmi et al. (7) reported the existence of human CD8+CD25+ thymocytes with regulatory activity but it is not known whether such a subset exists in the human periphery. Bienvenu et al. have shown that CD8+CD25+ T cells from MHC class II-deficient mice exert regulatory function (3). Endharti et al. have reported that CD8+CD122+ T cells are regulatory and perform its regulatory function through production of IL-10 (12). Najafian et al. have reported in an autoimmune disease model the existence of functionally protective CD8+ Treg cells that are CD8+CD28- (32). But, when they tested it in a heart transplant model, they described novel CD8+ programmed death receptor 1 (PD1+) Treg cells induced by the blockade of the inducible costimulatory molecule and its ligand (ICOS-B7h) pathway (20). Our experiment demonstrated that the CD8+ suppressive T cells are CD28+ (Fig. 4a). In line with new information concerning the diversity of CD4+ Tregs, there is a potential need for characterizing unique subpopulations based upon expression of homing molecules of CD8+ Treg cells that infiltrate certain microenvironments (6).
IL-10 is a well-documented immunosuppressant that inhibits macrophage-dependent antigen presentation and CD4+ T-cell proliferation in vitro (8,9,13). Other studies suggested that CD8+ suppressor T cells induce their suppressive effects via induction of IL-10 (19,31,33). However, our current data were not able to prove the assumed hypothesis that coadministration of RPM in combination with a recombinant IL-10/Fc of a long in vivo half-life may exert synergistic effect and possibly expand CD8+ suppressor T cells (Fig. 4c). Moreover, the RPM-induced CD8+ suppressor T cells secreted less IL-10 upon restimulation (Fig. 4b) with similar IL-10 intracellular secretion compared to CD8+ effector T cells (Fig. 4a). RPM is a potent anti-inflammatory agent (5). This would explain the reduced secretion of all the tested inflammatory cytokines by RPM-induced CD8+ suppressor T cells in our study.
IL-10 inhibits alloantigen-specific proliferative responses and induces a long-lasting anergic state in human purified CD8+ T cells when added concomitantly with the antigen in the presence of antigen presenting cells (APC). Moreover, the generation of allospecific cytotoxic activity is inhibited by IL-10. These effects are indirect and are mediated through inhibition of the costimulatory functions of APC (17). In contrast, IL-10 has no direct inhibitory effects on the proliferation of purified CD8+ T cells activated by anti-CD3 mAb and promotes the growth of activated CD8+ T cells in combination with low doses of IL-2 (35).
Taken together, these results indicate that IL-10 has differing effects on CD8+ T cells depending on their state of activation. This result may help to explain the paradox of both enhancing and inhibitory effects observed after IL-10 treatment in different in vivo experimental models (17). In the system studied in this report, the use of IL-10/Fc as an adjunct to RPM resulted in results inferior to RPM monotherapy with respect to graft survival in CD4 KO mice with no positive effect on CD8 KO mice.
We think that blocking the mTOR-mediated signaling not only causes apoptotic death of the alloreactive CD8+ T cells in vivo (24,45), but could also lead to generation of the CD8+ Tregs. These cells are capable of suppressing the function of newly generated alloreactive CD8+ T cells and thereby help promote the LTA. These CD8+ Tregs probably function through inhibition of certain inflammatory and proliferation-inducing cytokines like IFN-γ and IL-2 (Fig. 4b). Ablamunits et al. reported that administration of a humanized monoclonal anti-CD3 antibody to patients with type 1 diabetes increases their C-peptide responses and the CD8/CD4 ratio (1). This effect was mostly due to generation of CD8+ Tregs. They have reported that incubation of human peripheral blood mononuclear cells with mAb in vitro induces CD8+ Treg cells that are capable of inhibiting proliferation of CD4+ T cells through secretion of certain cytokines (1). Konya et al. have suggested the importance of generating CD8+ Treg cells in vitro or in vivo for therapeutic purposes (25). Here we show generation of CD8+ Treg cells in vivo by RPM treatment that enhances acceptance of allograft. We conclude that RPM can induce CD8+ suppressor T cells that may act independently of IL-10. These cells are CD28+ and produce less inflammatory cytokines upon restimulation than classical alloreactive CD8+ T cells. Furthermore, these cells apparently home to the graft site where they may express donor-specific graft protecting activity. Further studies are needed to characterize these cells and the immunostimulatory potential of IL-10/Fc should be taken into account for designing further therapies, possibly by generating an IL-10 mutant fusion protein devoid of its immunostimulatory function (10).
Footnotes
Acknowledgments
This work was supported by Grants JDRF 1-2007-551 (to W.G) and P01 AI041521 (to T.B.S) from Juvenile Diabetes Research Foundation and National Institute of Health. We thank Eva Csizmadia for her excellent technical help with IHC in this study. The authors declare no conflicts of interest.
