Abstract
The aim of this study was to investigate the effects of elevated donor HbA1c levels (type 2 diabetes, T2D) on the islet yield and functionality postisolation. In this retrospective analysis, donors for islet isolations were classified into two groups: T2D group (HbA1c ≥ 6.5%, n = 18) and normal group (HbA1c < 6.5%, n = 308). Optimum pancreas digestion time (switch time) was significantly higher in the T2D group compared to the normal group (13.7 ± 1.2 vs. 11.7 ± 0.1 min, respectively, p = 0.005). Islet yields were significantly lower in the T2D group compared to the control (T2D vs. control): islet equivalent (IEQ)/g (prepurification 2,318 ± 195 vs. 3,713 ± 114, p = 0.003; postpurification 1,735 ± 175 vs. 2,663 ± 89, p = 0.013) and islet particle number (IPN)/g (prepurification, 2,519 ± 336 vs. 4,433 ± 143, p = 0.001; postpurification, 1,760 ± 229 vs. 2,715 ± 85, p = 0.007). Islets from T2D pancreata had significantly lower viability (T2D vs. control: 91.9 ± 1.6 vs. 94.4 ± 0.3%, p = 0.004) and decreased oxygen consumption rate (DOCR) (T2D vs. control: 0.09 ± 0.01 and 0.21 ± 0.03 nmol O2 100 islets−1 min−1, p = 0.049). The islets isolated from T2D donor pancreata reversed diabetes in NOD-SCID mice in 9% (2/22) compared to islets from control donor pancreata, which reversed diabetes in 67% (175/260, p < 0.001). In conclusion, this study demonstrates that elevated HbA1c (≥6.5%) is associated with impairment of islet function and lower islet yield; however, these islets could not be suitable for clinical applications.
Introduction
The advancement of islet isolation technology enables islet transplantation to be a promising treatment for type 1 diabetes (4,29). Recently, a review article outlined factors influencing islet isolation outcomes (14). These factors include general donor information (22,24), pancreas characteristics (8,19), selection of enzyme during processing (1,6,16), and key parameters directly associated with isolation procedure (3,11,13). Reviewing and screening general donor information, such as age, BMI, medical history, and chemistry panel, is a critical step toward successful islet isolation. Based on widely acknowledged islet isolation protocols, glycated hemoglobin (HbA1c) levels are used to exclude prediabetic and diabetic donors for islet isolations intended for clinical application. According to the Clinical Islet Transplantation (CIT) Consortium, donor HbA1c that exceed the threshold levels of 6.1% do not meet the criteria for isolation and transplantation (9). HbA1c reflects the average blood glucose levels over the past 2–3 months (25) and is recommended by the American Diabetes Association (ADA) and the World Health Organization (WHO) as a criteria to diagnose diabetes (18). Currently, an individual with ≥6.5% HbA1c levels is diagnosed with type 2 diabetes (T2D); therefore, in this investigation, we categorized donors with HbA1c levels ≥6.5% as T2D group (18).
In 2004, Deng et al. reported a study in which pancreatic islets were isolated from patients with T2D (10). Structural and functional abnormalities of islets isolated from donors with T2D were analyzed and compared to that from non-T2D donors. They reported a reduction of islet yield from donors with T2D and also provided useful information to understand the pathological mechanisms of T2D. Recently, there has been an increasing demand for pancreatic islets from diabetic donors for research purposes (17); islets from donors with elevated HbA1c have also been transplanted into type 1 diabetic patients (20). However, studies regarding the characterization of isolated islets from patients with elevated HbA1c or T2D are rarely published. In this study, a retrospective analysis on the islet isolation results from normal control and T2D donors was conducted. The islet yield, size distribution characteristics, and islet functionality were compared and analyzed between the two donor groups.
Materials and Methods
Donor Groups
Pancreas donation consent was obtained from donor families for islet transplantation and research. All tissue utilized for assessment also had institutional review board approval from the Beckman Research Institute of City of Hope. Donor pancreata were procured from heart-beating brain-dead donors according to the established protocol at the City of Hope (16). In this study, two groups were assigned according to HbA1c levels documented. Donor HbA1c levels were quantified using the D-10″ Hemoglobin Analyzer (Bio-Rad Laboratories, Hercules, CA, USA) according to the manufacturer's instructions using whole blood samples. The analyzer is designed for determining the percentage of HbA1c (100 × HbA1c/total Hb) using ion-exchange high-performance liquid chromatography. The donors with HbA1c levels of ≥6.5% were characterized as the T2D (n = 18) and <6.5% as the control group (n = 308) in conformity with WHO and ADA criteria (25).
Islet Isolation
Islet isolations were performed using an automated method initially described by Ricordi et al. (29) and with modifications to accommodate the setup of the Current Good Manufacturing Practice (cGMP) facility at the City of Hope. Briefly, the pancreata were trimmed, cut, and cannulated prior to perfusing the enzyme solution through the head and body/tail using an automated perfusion apparatus (BioRep Technologies, Miami, FL, USA). The enzymes used for isolations were Liberase” HI (Roche Diagnostics, Roche Applied Science, Indianapolis, IN, USA), Liberase MTF C/T (Roche Diagnostics), SERVA Collagenase NB1 with neutral protease (SERVA Electrophoresis GmbH, Heidelberg, Germany), and VitaCyte (VitaCyte, LLC, Indianapolis, IN, USA) reconstituted in Hank's balanced salt solution (Mediatech, Manassas, VA, USA). Digestion of the pancreas was done by recirculating the enzyme and utilizing a gentle mechanical dissociation of the pancreatic tissue. Switch time, defined as the optimal end point for enzymatic digestion, was initiated when >50% of islets were free. The islets were identified by dithizone (DTZ; Sigma-Aldrich, St. Louis, MO, USA) staining. Digestion efficacy was determined by digested pancreatic tissue weight/ initial pancreatic tissue weight before digestion × 100. After digestion, collected tissue was washed to remove traces of the digestive enzyme. All digested tissues were combined and purified with Biocoll Separation Solution (Biochrom AG, Cedarlane Laboratories, Burlington, Ontario, Canada) using continuous density gradients (16). The isolated islets were then cultured in Connaught Medical Research Laboratories (CMRL) 1066-based media (Mediatech) for 24–72 h prior to assessment.
Islet Yield and Islet Size Distribution
Islets were analyzed using standard quality assessments currently performed at City of Hope (16). Islet yields were measured and recorded both as islet equivalent (IEQ) and islet particle number (IPN) at prepurification and postpurification. Islet yields were normalized per gram of pancreatic tissue by dividing the total IEQ or IPN by the pancreas weight (g) prior to digestion.
Islets of 150 μm in diameter were conventionally used to standardize the islet count to IEQ using the established counting method previously described (28). Final islet counts were then categorized into three size ranges based on islet diameter: small (50–150 μm), medium (150–300 μm), and large (≥300 μm) islets. Percentages of islet number in each size range were calculated by the following: percentage of islet number in each size range (%) = (IPN of each size range/total IPN of sample taken from the same islet preparation) × 100. Islet viability was determined using fluorescent microscopy (Olympus, Center Valley, PA, USA) and fluorescent dyes: fluorescein diacetate and propidium iodide (Sigma-Aldrich) (2).
Glucose-Stimulated Oxygen Consumption Rate (ΔOCR)
Glucose-stimulated oxygen consumption (ΔOCR) rate of islets was measured to evaluate the amount of oxygen consumed during metabolism. In this assay, 750 IEQ islets were sampled from each islet preparation, and ΔOCR was measured following the method previously described (30). ΔOCR was defined as the difference in measured OCR of islets exposed to 3 mmol/L glucose (Hospira, Inc., Lake Forest, IL, USA) for 15 min, followed by exposure to 20 mmol/L glucose for 30 to 45 min. Results were expressed in nmol O2 100 islets−1 min−1.
Islet Transplantation in Diabetic Mice
Male nonobese diabetic/severe combined immunodeficient (NOD-SCID) mice (NOD CB17-Prkdcscid), 10–12 weeks of age (Jackson Laboratory, Bar Harbor, ME, USA), were used for in vivo assessment. The animals were cared for in accordance with the Guidelines and Principles of Laboratory Animal Care. All the procedures involving animals were approved by the Institutional Animal Care and Use Committee of the City of Hope. Mice were rendered diabetic by intraperitoneal injections of 50 mg/kg streptozotocin (STZ; Sigma-Aldrich) for 3 consecutive days. Mice that exhibited hyperglycemia (>350 mg/dl) for 2 consecutive days were used as recipients. Human islets (1,200 IEQs) from the same donor were transplanted under the left kidney capsule of two to three diabetic mice. Blood glucose levels were measured using a glucometer (LifeScan, Inc., Milpitas, CA, USA) starting 24 h posttransplantation, two to three times per week for 4 weeks. Recipient mice that maintained blood glucose levels <200 mg/dl throughout 4 weeks were considered to have reversed diabetes. At the end of each experimental period, a nephrectomy was performed to confirm graft dependence.
The percentage of reversal rate of diabetes in NODSCID mice was calculated based on the ratio of the number of mice that reversed diabetes to that of the total number of mice transplanted.
Statistical Analysis
GraphPad Prism (GraphPad Software 6.0, La Jolla, CA, USA) was used to analyze the data and generate the figures. Normal distribution of the data was confirmed by Kolmogorov–Smirnov test. For parametric tests, an unpaired Student's t-test (two-tailed) or nonparametric chi-square test was used to compare the control and diabetic groups. Values were expressed as mean ± standard error of mean (SEM). For nonparametric data, the Wilcoxon rank-sum test was used to compare the two donor groups. Receiver operating characteristic (ROC) curves, plots of the sensitivity [true positives/(true positives + false negatives)] versus 1- specificity [true negatives/(true negatives + false positives)], were generated as previously described (30). For all the tests used, a value of p < 0.05 was considered significant.
Results
Donor Information
As shown in Figure 1, documented HbA1c levels of the T2D (n = 18) donors were significantly higher than that of the controls (n = 308) (7.6 ± 0.4 vs. 5.4 ± 0.1%, p < 0.001) (Fig. 1A). Blood glucose levels at admission for T2D group were significantly higher than that for control group (231.4 ± 19.4 vs. 178.1 ± 3.9 mg/dl, p = 0.001) (Fig. 1B). There were no significant differences between the two donor groups with respect to age (49.3 ± 2.3 vs. 43.9 ± 0.7 years, p = 0.070), gender (male 33%/female 67% vs. male 46%/female 54%, p = 0.082), body mass index (BMI; 33.0 ± 1.8 vs. 30.1 ± 0.3 kg/m2, p = 0.063), and pancreas cold ischemic time (7.6 ± 0.6 vs. 8.1 ± 0.2 h, p = 0.508). There were also no significant differences between two subgroups in each assessment in terms of age, gender, BMI, and pancreas cold ischemic time.

HbA1c (A) and blood glucose levels at admission (B) of two donor groups. Glycated hemoglobin (HbA1c) levels were significantly higher in type 2 diabetes (T2D) donors (n = 18) compared to control donors (n = 308) (***p < 0.001) (A). Blood glucose levels at admission were significantly higher in T2D (n = 18) versus control (n = 308) donors (**p = 0.001) (B).
Islet Isolation Results
Digestion switch time for pancreata in the T2D group (n = 18) was significantly higher than that in control group (n = 308; 13.7 ± 1.2 vs. 11.7 ± 0.1 min, p = 0.005) (Fig. 2A). In the T2D group, one isolation had a longer digestion switch time (28 min) compared to mean value (13 min) of the same group. Excluding this case, digestion switch time was not significantly different between the two groups (T2D vs. control: 12.9 ± 0.8 vs. 11.7 ± 0.1 min, p = 0.105). The digestion efficiency was very similar between the groups (63.6 ± 4.6 vs. 67.7 ± 1.0, p = 0.361) (Fig. 2B).

Switch time of pancreas digestion (A) and digestion efficacy (B) of two donor groups. Switch time for pancreas digestion was significantly higher in T2D groups (n = 18) compared to control groups (n = 308) (**p = 0.005) (A). No significant difference in digestion efficacy was observed between the two groups (p = 0.361) (B).
The IEQ per gram of pancreatic tissue digested was significantly lower in the T2D group (n = 18) compared to the control group (n = 308) (T2D group vs. control group) at prepurification (2,318 ± 195 vs. 3,713 ± 114 IEQ/g, p = 0.003) and at postpurification (1,735 ± 175 vs. 2,663 ± 89 IEQ/g, p = 0.013) (Fig. 3A). Similarly, significantly lower islet yields were also observed in the T2D group compared to the control group (T2D group vs. control group) regarding the IPN per gram of pancreatic tissue digested (prepurification: 2,519 ± 336 vs. 4,433 ± 143 IPN/g, p = 0.001; postpurification: 1,760 ± 229 vs. 2,715 ± 85 IPN/g, p = 0.007) (Fig. 3B).

Islet yields in IEQ/g (A) and IPN/g (B) pancreatic tissue of two donor groups. Significantly lower islet yields/g pancreatic tissue were recovered from T2D pancreata (n = 18) compared to the control pancreata (n = 308): islet equivalents/gram (IEQ/g) tissue at pre- (**p = 0.003) and postpurification (*p = 0.013) (A); islet particle number/gram (IPN/g) tissue at pre- (**p = 0.001) and postpurification (**p = 0.007) (B).
Representative images of the DTZ-stained purified human islets are shown in Figure 4. Donut-shaped and faintly DTZ-stained degranulated islets were seen from T2D donors (HbA1c ≥ 6.5%) (Fig. 4A, B). Islets from normal donors (HbA1c < 6.5%) were well granulated and stained densely with DTZ (Fig. 4C, D).

Representative images of isolated and purified human islets stained with DTZ. Islets from T2D donors are donut-shaped, faintly stained with dithizone (DTZ), and degranulated, which are indicated by white arrows (A, B). Islets from normal donors are well granulated and stained densely with DTZ (C, D).
The islet size distributions are depicted in Figure 5. There were no significant differences between the two donor groups (T2D, n = 18; control, n = 29) at postpurification in terms of islet size ranges of 50–150 μm (p = 0.484), 150–300 μm (p = 0.377), and >300 μm (p = 0.665).
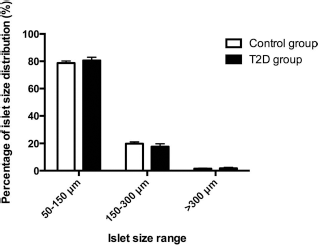
Distribution of islet size was measured at postpurification and categorized based on diameter: small (50–150 um), medium (15–300 um), and large (≥300 μm). No significant differences in islet size distribution were observed between the T2D (n = 18) and control (n = 29) groups across all three size categories, small (p = 0.484), medium (p = 0.377), and large (p = 0.665).
Islet Viability and ΔOCR
Figure 6A delineates that islet viability in the T2D group (n = 18) was significantly lower than that in the control group (n = 290) (91.9 ± 1.6 vs. 94.4 ± 0.3%, p = 0.004). ΔOCR results showed that the T2D group (n = 10) had a significantly lower oxygen consumption rate than the control group (n = 133), with values of 0.09 ± 0.06 and 0.21 ± 0.03 nmol O2 100 islets−1 min−1, p = 0.049 (Fig. 6B).

Islet viability (A) and function (B) of two donor groups. Islet viability was significantly lower in T2D group (n = 18) compared to the control group (n = 290) (*p = 0.004) (A). Islets from T2D donor pancreata (n = 10) showed significantly lower oxygen consumption rate (ΔOCR) compared to islets from the control group (n = 133) (*p = 0.049) (B).
Islet Transplantation in Diabetic Mice
Figure 7 shows the results of the in vivo assessment of islets transplanted into STZ-induced diabetic NOD-SCID mice. The islets isolated from T2D donor pancreata (n = 9) reversed diabetes in 9% (2/22) compared to islets from control donor pancreata (n = 114), which reversed diabetes in 67% (175/260), p < 0.001 (Fig. 7A). The ability of predicting the reversal rate of diabetes in NOD-SCID mice using donor HbA1c levels was highly significant in both the diabetic and the control group: the area under the curve (AUC) was 0.596 (95% CI: 0.528–0.664, p = 0.006, ROC analysis) (Fig. 7B) (AUC will be 1 if the correct classification is 100% and 0.5 if there is no predictive relationship).
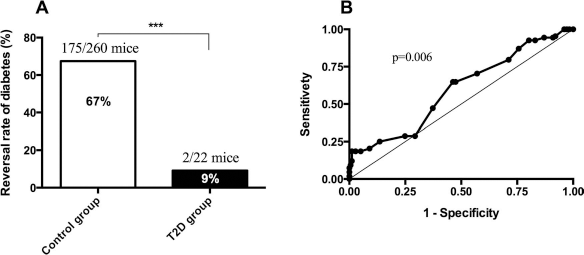
Reversal of diabetes in NOD-SCID mice posttransplantation. The reversal of STZ-induced diabetes was significantly lower in nonobese diabetic severe combined immunodeficient (NOD-SCID) mice that were transplanted with islets from the T2D group (n = 9) compared to the control group (n = 114) [9% (2/22) vs. 67% (175/260), ***p < 0.001] (A). The ability of HbA1c level to predict reversal rate of diabetes in NOD-SCID mice was highly significant in both the diabetic and control groups: the area under the curve (AUC) was 0.596 (95% CI: 0.528–0.664, p = 0.006) (AUC will be 1 if the correct classification is 100% and 0.5 if there is no predictive relationship) (B).
Discussion
This study demonstrates that lower yield and poorly functional islets were isolated from donors with elevated HbA1c levels (≥6.5%). Chronic shortage of donor pancreata represents a major problem for isolating islets for research and clinical application. The restriction of donor pancreata with elevated HbA1c may further limit organs for islet isolations for research and clinical applications. Therefore, it is imperative to utilize every organ for potential islet isolation, especially those from donors with suboptimal or elevated HbA1c. In this study, the control group had normal blood glucose at the time of admission, whereas the T2D group had markedly increased blood glucose levels, though no significant differences in age, gender, and BMI were observed between the two groups.
The process of islet isolation from both groups was identical. However, the switch time for one pancreas, which was fibrotic, from the diabetic group was exceptionally longer (28 min). Statistically, there were no significant differences between the switch times for the control and diabetic groups should this pancreas be excluded from this study. It is worth noting that this donor pancreas in the T2D group had significantly elevated HbA1c (11.4%) despite not having any documented medication(s). Our results indicated that the islet yields both in IEQ and IPN per gram of pancreatic tissue at pre- and postpurification were significantly higher in the control donors compared to the T2D group, which was also observed in a previously published study (10). Islet morphologies from diabetic pancreata were donut-shaped, mosaic, and degranulated, particularly from donors with elevated HbA1c levels (≥6.5%).
Previous studies have reported that islets from T2D donors tend to have diminished insulin secretion, which may be attributed to lower β-cell and higher α-cell composition resulting in failure to reverse diabetes in transplanted mice (10,20,27). In the early stages of diabetes, islets are often larger in size, and thus high islet yield may be expected (unpublished observation). It has also been suggested that higher islet numbers have a higher success rate in reversing induced diabetes (5,7,10). The systematic analysis of islet size distribution indicated that there was no difference in terms of the percentage of islet size distribution at postpurification. It has been reported that the mean size of the islets in native pancreata of diabetic patients is similar to that of nondiabetic patients (7). Another study has also shown that the decrease in β-cell mass in diabetic patients was marginal (23), signifying that islet number and size may be similar between normal and T2D donors. Furthermore, a recently published study showed that islet density in pancreas, as well as β-cell mass, but not mean islet size, negatively correlated with HbA1c levels (21). Altogether, our data further signify that the severity and length of T2D are associated with lower islet yield and not attributed to smaller islet size, confirming that islet size distribution was not a factor in a diminished islet yield.
Islets isolated from control donors maintained significantly better viability compared to T2D donors. Moreover, ΔOCR, an indicator for metabolic activity of islets (31), was lower in the T2D group than the control group with comparable stimulation indices (data not shown), further indicating that elevated levels of HbA1c negatively impact islet quality as well.
The islets from T2D donors failed to consistently reverse diabetes when transplanted into diabetic NOD-SCID mice, confirming previously reported studies (10,20). Therefore, it is critically important to carefully assess islets from donors with elevated HbA1c (≥6.5%) for transplantation into patients with type 1 diabetes. It is noteworthy that the Edmonton group reported two cases of clinical islet transplantation with islets from donors with elevated HbA1c; however, the islets did not reverse diabetes in NOD-SCID mice (20). One of the two patients achieved insulin independence for 4 years after receiving islets from a donor with HbA1c level of 6.3%. Another patient could not achieve insulin independence but had approximately 50% reduction of exogenous insulin requirement when transplanted with islets from a donor with elevated HbA1c of 7.9% (20). The Edmonton group reported that islets from higher HbA1c (7.9%) demonstrated marginal function when transplanted into diabetic patients in comparison with islets from donors with HbA1c < 6.5% (6.3%). Thus, the level of HbA1c is critical to determine the outcome of islet function and survival posttransplantation. In this study, the diabetes reversal rate was considerably lower in mice transplanted with islets from diabetic donors (HbA1c > 6.5%). Furthermore, the ROC analysis revealed that the predictive power of HbA1c level to the reversal rate of diabetes in mice was statistically significant in both groups. Our study also supports the results of the aforementioned Edmonton study (20) and another published study by Deng et al. (10).
The limitations of this retrospective analysis are lack of histology, gene expression, and biochemical studies for islets from donors with elevated HbA1c. In particular, analysis of islet cell composition from T2D and normal control donors would be useful using previously published methods (15,32). Comprehensive studies regarding the above features of islets from T2D could be expected from investigators within the IIDP network (17). The results of viability, ΔOCR, and transplant in NOD-SCID mice indicated that the mice in the T2D group may have received less functional islets despite having an equal number of initial islets transplanted (1,200 IEQ) between T2D and control groups. In this regard, Friberg et al. reported in their study that functional islet mass is an influential factor for graft survival after islet transplantation (12). The transplant results in NOD-SCID mice provide strong evidence that islets from the donors with elevated HbA1c should be evaluated carefully before being considered for clinical transplantation. Thus, research on islets from both normal and T2D donors to study pathophysiology of β-cell function and survival is imperative. Indeed, islet distribution through a website-based automatic matching algorithm has been established so that investigators could receive islets for in vitro and in vivo studies (17,26).
In conclusion, elevated levels of HbA1c negatively affect islet function and yield; however, islets from donors with elevated HbA1c (≥6.5%) could not be suitable for clinical applications.
Footnotes
Acknowledgments
The authors thank Randall Heyn-Lamb and Karen Ramos for providing and interpreting donor information. We extend a special word of thanks and appreciation to Dr. David Smith for his advice and statistical analysis. We also thank Chris Orr and Henry Lin for reviewing the statistical advice and proofreading the manuscript. This work was financially supported by the Department of Diabetes and Metabolic Diseases Research, Beckman Research Institute of the City of Hope. Human pancreatic islets were provided by the NIDDK-funded Integrated Islet Distribution Program (IIDP) at the City of Hope. The authors declare no conflict of interest.
