Abstract
Recent studies have demonstrated that transplantation of induced pluripotent stem cell-derived neurospheres can promote functional recovery after spinal cord injury in rodents, as well as in nonhuman primates. However, the potential tumorigenicity of the transplanted cells remains a matter of apprehension prior to clinical applications. As a first step to overcome this concern, this study established a glioblastoma multiforme xenograft model mouse. The feasibility of controlling immune suppression to ablate the grafted cells was then investigated. The human glioblastoma multiforme cell line U251 MG was transplanted into the intact spinal cords of immunodeficient NOD/SCID mice or into those of immunocompetent C57BL/6J H-2kb mice treated with or without immunosuppressants [FK506 plus anticluster of differentiation (CD) 4 antibody (Ab), or FK506 alone]. In vivo bioluminescent imaging was used to evaluate the chronological survival of the transplanted cells. The graft survival rate was 100% (n = 9/9) in NOD/SCID mice, 0% (n = 6/6) in C57BL/6J mice without immunosuppressant treatment, and 100% (n = 37/37) in C57BL6/J mice with immunosuppressant treatment. After confirming the growth of the grafted cells in the C57/BL6J mice treated with immunosuppressants, immune suppression was discontinued. The grafted cells were subsequently rejected within 3 days in C57BL/6J mice treated with FK506 alone, as opposed to 26 days in C57BL/6J mice treated with FK506 plus anti-CD4 Ab. Histological evaluation confirmed the ablation of the grafted cells. Although this work describes a xenograft setting, the results suggest that this immunomodulatory strategy could provide a safety lock against tumor formation stemming from transplanted cells.
Introduction
The central nervous system (CNS), including the brain and the spinal cord, has traditionally been considered a representative example of an organ system in which regeneration is difficult. However, this view is being overturned by recent progress in stem cell biology. Several studies recently demonstrated the effectiveness of neural stem/progenitor cell (NS/PC) transplantation for the repair of spinal cord injury (SCI) (14, 20, 28). However, clinical application of NS/PCs has not been realized in Japan due to ethical issues related to the use of cells derived from aborted fetal tissues. Under such circumstances, Yamanaka et al. (37) introduced several genes into somatic cells to create induced pluripotent stem cells (iPSCs) with embryonic stem cell (ESC)-like pluripotency and proliferative capacity. Recently, we showed the reparative efficacy of iPSC-derived NS/PCs in mouse and nonhuman primate SCI models (18, 27, 42).
Nonetheless, before clinical application of such cells, several concerns must be addressed. Perhaps the most important is the potential tumorigenicity of iPSC-derived NS/PCs. Genetic and epigenetic abnormalities acquired from their origin, in addition to reprogramming stress and the culture environment, all increase the tumorigenicity of human iPSC-derived cells. Moreover, a number of investigations reported the increasing incidence of teratomas and gliomas due to transplantation of “dangerous” ESC- and iPSC-derived NS/PC clones (4, 9, 17, 21, 44). To overcome the risk of tumor formation, safety screening of iPSC-derived NS/PCs prior to transplantation is essential. However, despite such screenings, the risk of tumorigenicity after transplantation is not eliminated. A recently developed ganciclovir (GCV)-mediated cell ablation system employing herpes simplex virus type 1 thymidine kinase (TK) (the delta TK/GCV system) shows the potential to reduce the cancer risk (6). On the other hand, this system could still augment the risk of tumorigenesis resulting from gene transfer and/or insertional mutagenesis, and it has not yet been used in clinical practice. Furthermore, a spinal cord tumor model to evaluate tumorigenesis has not been established.
From the viewpoints of the timing of the transplantation after the injury, safety concerns, and cost performance, we assume allogeneic iPSC-NSC transplantation into the injured spinal cord in first-in man study rather than autologous transplantation. Therefore, we hypothesized that allografted iPSC-NSC-derived tumors could be eliminated by inducing immunorejection without any adverse events. As a first step, to confirm the feasibility of this concept, we used human glioblastomamultiforme (GBM) U251 MG cells in this study because GBM is the most aggressive and toughest tumor against immunorejection. The purpose of this study is to determine whether xenografted GBM could be eliminated by discontinuing immunosuppressant without any adverse events such as deterioration of motor function. Here we developed a novel spinal cord tumor model mouse by implanting U251 cells, followed by bioluminescence imaging (BLI). BLI accurately monitored grafted tumor growth, in addition to immunorejection. We transplanted the cells into the intact spinal cords of nonobese diabetic/severe combined immunodeficient (NOD/SCID) mice, as well as those of immunocompetent C57BL/6J H-2kb mice with or without immunosuppressant treatment [FK506 plus anticluster of differentiation (CD4) antibody (Ab) or FK506 alone]. We successfully induced the immunorejection of the grafted U251 cells in the xenograft setting of the C57BL/6J mouse solely by discontinuing immunosuppressant treatment without any adverse events, suggesting that tumors arising from currently employed cell transplantation practices for the management of SCI could be ablated by withdrawing immunosuppressants.
Materials and Methods
Cell Culture and Lentivirus Transduction
Human male GBM U251 MG cells (kindly gifted from Professor Ikeda, Yamaguchi University Graduate School of Medicine, Yamaguchi, Japan) were cultured in Dulbecco's modified Eagle's medium (Sigma-Aldrich, St. Louis, MO, USA) containing 10% fetal bovine serum (Life Technologies, Carlsbad, CA, USA) and 5% penicillin/streptomycin (Nacalai Tesque Inc., Kyoto, Japan.), passaged using trypsin-EDTA, and then seeded into fresh culture medium. For transplantation data about human iPSCs-NS/PCs (hiPSCs-NS/PCs), cell culture and neural differentiation of hiPSCs (clone 253G1, Caucasian, 36 years old, female, human dermal fibroblast) were performed as previously described (18, 21, 27). The lentivirus was prepared and transduced into U251 cells as described previously (22, 46). Briefly, a fusion HIV-1-based lentiviral vector (kindly gifted from Dr. Hara, Brain Science Institute, RIKEN, Japan) expressing Venus fluorescent and luminescent protein fused to firefly luciferase (ffLuc) under the control of the elongation factor promoter was used (10). This vector enabled grafted cells to be detected as strong bioluminescent ffLuc signals in live mice and also as green fluorescent protein (GFP)-positive cells by using an anti-GFP Ab in fixed spinal cord sections because the Venus protein was originally modified from GFP (25). The concentrated virus was added to the culture medium to infect U251 cells and hiPSCs-NS/PCs.
Cell Transplantation
Adult female NOD/SCID (n = 9), C57BL/6J H-2kb (n = 54) mice, and BALB/cA H-2kd (n = 5) mice (8–9 weeks old, 18–22 g; Clea Japan Inc., Tokyo, Japan) were anesthetized with an intraperitoneal (IP) injection of ketamine (100 mg/kg body weight; Sankyo Life Tech Co., Ltd., Tokyo, Japan) and xylazine (10 mg/kg body weight; Bayer, Leverkusen, Germany). After laminectomy at the 10th thoracic spinal vertebra, the dorsal surface of the dura mater was exposed. U251 cells and hiPSCs-NS/PCs with phosphate-buffered saline (PBS) for vehicle (5 × 105 cells/2 μl) were injected with a glass micropipette at a rate of 1 μl/min with a Hamilton syringe (25 μl) and a stereotaxic microinjector (KDS 310; Muromachikikai Co., Ltd., Tokyo, Japan). All experiments were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals of Keio University (Assurance No. 13020) and the NIH Guide for the Care and Use of Laboratory Animals (National Institutes of Health, Bethesda, MD, USA).
BLI
The Xenogen-IVIS spectrum-cooled charge-coupled device optical macroscopic imaging system (Summit Pharmaceuticals International Corporation, Tokyo, Japan) was used for in vitro BLI, as well as for in vivo BLI to confirm the survival of transplanted cells. Monitoring was performed one to two times/week beginning 1 week after transplantation. Briefly, D-luciferin (Promega KK Corporation, Tokyo, Japan) was administered by IP injection at a dose of 300 mg/kg body weight. Animals were placed in a light-tight chamber, and photons emitted from luciferase-expressing cells were collected. The integration time was fixed at 1 min for each image. All images were analyzed with Living Image software (Caliper Life-Sciences, Hopkinton, MA, USA). The BLI signal was quantified in maximum radiance units [photons/s/cm2/steradian (p/s/cm2/sr)] and presented as log10 (photons/s).
Immunosuppressive Therapy
For the immunosuppressive therapy protocol, female C57BL/6J H-2kb mice were randomized to receive FK506 (Prograf®; Astellas Pharma Inc., Tokyo, Japan) plus anti-CD4 Ab (n = 37; BioXcell, West Lebanon, NH, USA), or FK506 alone (n = 11). FK506 was administered at a dose of 5 mg/kg once daily by subcutaneous injection, and anti-CD4 Ab was administered at a dose of 10 mg/kg by IP injection on day -2 before transplantation and day 7 after transplantation. For hiPSC-NS/PC transplantation study, female BALB/cA H-2kd mice (n = 5) were randomized to receive FK506 and anti-CD4 Ab by the same protocol and continuing once per week until 100 days after transplantation.
Motor Function Analysis
Hindlimb motor function was evaluated using the locomotor rating test of the Basso Mouse Scale (3). Well-trained investigators, blinded to the treatments, performed the behavioral analysis, and the Basso Mouse Scale scores were determined at the same time each day.
Histological Analysis
Animals were anesthetized and transcardially perfused with 0.1 M PBS (Nacalai Tesque Inc.) containing 4% paraformaldehyde (Nacalai Tesque Inc.). The spinal cords were removed, embedded in Optimal Cutting Temperature compound (Sakura Fine Technical Co., Ltd., Tokyo, Japan), and sectioned in the sagittal plane on a cryostat (CM3050; Leica Microsystems, Wetzlar, Germany). Sections were stained with hematoxylin and eosin (H&E) and the following primary Abs: anti-GFPAb (rabbit IgG, 1:200; Frontier Institute Co., Ltd., Hokkaido, Japan), anti-human nuclear antigen (HNA) Ab (mouse IgG, 1:200; Chemicon, Temecula, CA, USA), anti-Ki-67 Ab (rabbit IgG, 1:200; Novocastra, Newcastle upon Tyne, UK), anti-CD11b Ab (rat IgG2b 1:200; BD Pharmingen, Franklin Lakes, NJ, USA), anti-natural killer p46 (NKp46)Ab (rat IgG2a, k, 1:500; BioLegend, San Diego, CA, USA), anti-terminal deoxynucleotidyl transferase (TdT) Ab (Apop Tag plus Fluorescein in situ apoptosis detection kit; Chemicon), and anti-CD3 Ab (rat IgG1, 1:100; AbD Serotec, Kidlington, Oxfordshire, UK). The samples were incubated with primary Abs at 4°C overnight. They were then incubated with secondary Abs conjugated to Alexa 488, Alexa 555, or Alexa 633 (Invitrogen, Carlsbad, CA, USA) as appropriate for the primary Ab host species at a 1:500 dilution for 1 h at room temperature. Nuclei were stained with Hoechst 33258 (10 μg/ml; Sigma-Aldrich). Samples were examined on an inverted fluorescence microscope (BZ 9000; Keyence Co., Osaka, Japan) or a confocal laser-scanning microscope (LSM 700; Carl Zeiss, Oberkochen, Germany).
Flow Cytometry
Isolated peripheral blood leukocytes were analyzed by triple immunofluorescence staining followed by flow cytometry. The following Abs were all purchased from eBiosciences (1:100; San Diego, CA, USA): anti-CD3-allophycocyanine (APC)Ab (clone 145-2C11), anti-CD4-fluorescein isothiocyanate (FITC)Ab (clone GK 1.5), and anti-CD8-phycoerythrin (PE)Ab (clone 53-6.7), Armenian hamster IgG isotype control APC (clone eBio299Arm), rat IgG2b K isotype control FITC (eB149/10H5), rat IgG2a K isotype control PE (eBR2a). The cells were stained with a mixture of these Abs at 4°C for 30 min. Flow cytometric analysis was performed on a fluorescence-activated cell sorting (FACS) Calibur (BD Biosciences, San Jose, CA, USA).
Statistical Analysis
All data are presented as the mean ± the standard error of the mean (SEM). Repeated-measures ANOVA followed by Bonferroni post hoc test was used to determine significant differences in the BLI analysis (Statcel; OMS Ltd., Saitama, Japan). For all statistical analyses, the significance level was set at p < 0.05.
Results
In Vitro BLI of Lentivirally Transduced Human GBM U251 Cells
U251 cells were cultured and labeled with ffLuc genes via lentiviral transduction, as reported previously (10) (Fig. 1A). Signals detected from these cells were sufficient for qualitative analysis by fluorescence microscopy (Fig. 1B, C). To examine the sensitivity of the BLI technique, the Xenogen-IVIS system was used to detect the luminescence intensity of various numbers of cells (5 × 105–3.5 × 106 cells per well) in the presence of D- luciferin, as reported previously (29). Quantitative analysis clearly revealed that the luminescence intensity in vitro was directly proportional to the cell number (Fig. 1D, E). The minimum number of cells that could be measured by their photon counts with in vitro BLI was approximately 1,000. FACS caliber analysis revealed that >90% of these cells were GFP positive (data not shown).

Luminescence and fluorescence in lentivirally transfected U251 cells. Phase (A) and fluorescent (B) microscopic images of well-dispersed transfected U251 MG cells. (C) BLI detected luminescence in different cell number cultures (0, 2 × 105, 7.5 × 105, 1.5 × 106, 3 × 106 cells). (D) Correlation between photon counts and cell numbers. (E) One thousand cells were sufficient to elicit a BLI signal in vitro. Scale bars: 200 μm (A and B).
Survival of Human GBM Cells Grafted Into the Spinal Cords of Immunodeficient and Immunocompetent Mice
Transduced U251 cells (5 × 105) were transplanted into the intact spinal cords of immunodeficient NOD/SCID (n = 9) and immunocompetent C57BL/6J H-2kb (n = 6) mice. The photon counts of the U251 cells grafted into C57BL/6J mice without immunosuppressant treatment decreased to background levels at 15 days after transplantation, suggesting that immunorejection of the cells occurred within 2 weeks in all six C57BL/6J mice. By contrast, the photon counts of the U251 cells grafted into the NOD/SCID mice sharply increased at 2 weeks after transplantation, suggestive of the survival and growth of the grafted cells in each of the immunodeficient mice (p < 0.05) (Fig. 2). These findings indicate that even in the immune-privileged CNS of C57BL/6J mice, xenografted glioma cells were rejected at about 2 weeks without immunosuppressant treatment. Consistently, histological analysis revealed that U251 cells grafted into NOD/SCID mice survived well and migrated into the host spinal cord at 28 days after transplantation. The resulting tumor showed nuclear atypia, microvascular proliferation, necrosis, and Ki-67 index was 18% (Fig. 3A, B).

BLI images and photon counts over time after transplantation of U251 cells into NOD/SCID mice, as well as C57BL/6J mice with and without immunosuppressant treatment. (A) BLI images of representative mice (upper: NOD/SCID mouse, middle: C57BL/6J mouse with immunosuppressant treatment, lower: C57BL/6J mouse without immunosuppressant treatment) at 0, 18, and 28 days after GBM U251 cell transplantation. Even with a xenogeneic transplantation, the transplanted tumor engrafted and grew the same as in the NOD/SCID mice group, by using two immunosuppressants. (B) Quantitative analysis of the photon counts derived from grafted cells. Graft survival rate at day 14 was 100% (n = 9/9; 2/9 animals were dead after day 14) in NOD/SCID mice, 100% (n = 10/10) in C57BL/6J mice with immunosuppressant treatment, and 0% (n = 0/6) in C57BL/6J mice without immunosuppressant treatment at 14 days. Data represent the mean ± SEM (*p < 0.05; repeated-measures ANOVA followed by Bonferroni post hoc test.)
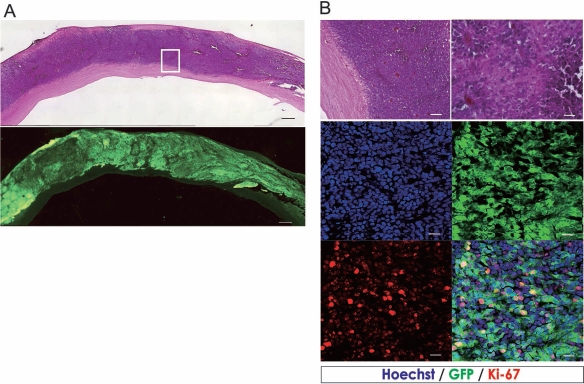
Histological analysis of grafted U251 cells in NOD/SCID mice. (A) Low magnification H&E staining (top) and GFP staining (bottom). Scale bar: 500 μm. (B) High magnification H&E staining (top), and Hoechst (blue), GFP (green), and Ki-67(red) (bottom) staining. Grafted GBM cells showed good survival and migrated into the host spinal cord. The tumor showed nuclear atypia, microvascular proliferation, and necrosis. Ki-67 index was 18%. Scale bars: 500 μm (A) and 25 μm (B).
Immunorejection of Human GBM U251 Cells Grafted Into C57BL/6J Mice After Discontinuation of Immunosuppressant Treatment, Resulting in Functional Recovery
To avoid the immunorejection of the xenografts, human GBM U251 cells were transplanted into C57BL/6J mice treated with immunosuppressants. Long-term survival of the grafted cells was achieved, similar to the situation in NOD/SCID mice. The graft survival rate was 82% (n = 9/11) in the FK506 group, 100% (n = 8/8) in the FK506/anti-CD4 Ab group, and 100% (n = 7/7) in NOD/SCID mice at 28 days (Table 1). With the increase in the size of the tumor, almost all mice showed complete paralysis of the hindlimbs at 21–28 days after transplantation.
Graft Survival for the Different Treatment Groups
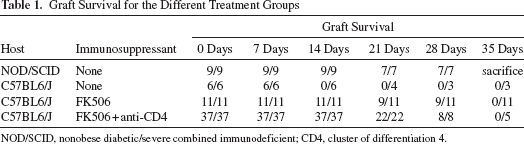
NOD/SCID, nonobese diabetic/severe combined immunodeficient; CD4, cluster of differentiation 4.
To induce the immunorejection of the xenografted tumor cells, the administration of immunosuppressants was discontinued at different time points after transplantation. In the FK506 group, the immunosuppressive therapy was discontinued at 28 days after cell transplantation, which induced the immunorejection of grafted U251 cells within 1 week (Fig. 4A). By contrast, in the FK506/anti-CD4 Ab group, immunosuppressive therapy was discontinued at 7 or 14 days after transplantation. In 5 of 17 mice in the FK506/anti-CD4 Ab group, immunorejection of grafted U251 cells was achieved by discontinuing immunosuppressants at 7 days after transplantation (Fig. 4A), resulting in motor functional recovery of the hindlimbs (Fig. 4B). On the other hand, all of the mice (n = 20/20) died before tumor rejection when immunosuppressant treatment was discontinued at 14 days after transplantation.
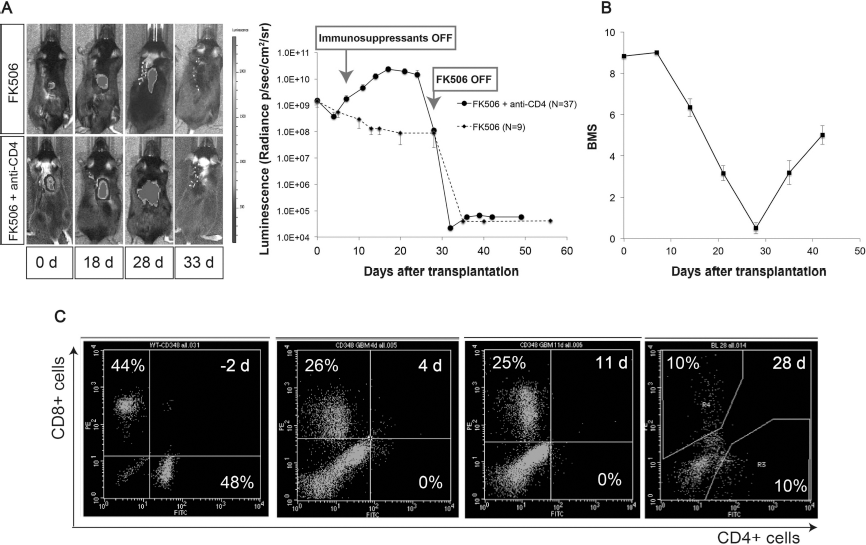
Regulation of the fate of grafted U251 cells by immunomodulation. (A) Representative BLI images and quantitative analysis of the photon counts over time after transplantation of U251 cells into C57BL/6J mice with immunosuppressant treatment. Graft survival rate was 82% (n = 9/11) in the FK506 treatment group and 100% (n = 22/22; 15/37 mice died between 14 and 21 days) in the FK506/anti-CD4Ab treatment group at 21 days. Immunorejection of all the grafted U251 cells was achieved by discontinuing immunosuppressants. In the FK506 group, the immunosuppressive therapy was discontinued at 28 days after cell transplantation. By contrast, in the FK506/anti-CD4 Ab group, immunosuppressive therapy was discontinued at 7 days after cell transplantation. (B) Hindlimb motor function was assessed by the Basso Mouse Scale. Hindlimb motor function was lost to the level of complete paralysis with increasing tumor size in some mice. After discontinuation of immunosuppressants, grafted mice showed functional recoveries with the disappearance of the tumor in the surviving group (n= 14/48). (C) Lymphocyte monitoring in C57BL/6J mice with immunosuppressant treatment. Lymphocyte subsets were observed by FACS with anti-CD4 and anti-CD8 Abs. CD4+ T-cells were immediately depleted following administration of immunosuppressants and recovered after immunosuppressant treatment was discontinued.
Lymphocyte subsets in the peripheral blood were then examined by using FACS with anti-CD4 and anti-CD8 Abs. CD4+ T-cells were immediately depleted with administration of immunosuppressants, but recovered after immunosuppressant treatment was discontinued (Fig. 4C).
Histological Analysis of U251 Cells in C57BL/6J Mice with Immunosuppressant Treatment
Histological analysis revealed that xenografted human GBM U251 cells survived in C57BL/6J mice with immunosuppressive therapy. In some mice, the tumor protruded to the outside from the posterior median sulcus. However, the grown tumors were rejected after discontinuation of immunosuppressants (Fig. 5). The graft rejection status is shown at various posttransplantation time points in Figure 6A. Infiltration of inflammatory cells into the tumor, such as CD11b-, NKp46-, and CD3-positive cells, as well as an increase in the number of TdT-positive cells were observed during the process of tumor rejection. These cells became prominent by 39 days after discontinuation of immunosuppressants (Fig. 6B, C).

Histological analysis of grafted U251 cells in C57BL/6J mice with and without immunosuppressant treatment. Consistent with the BLI results, grafted U251 cells survived in C57BL/6J mice with immunosuppressant treatment. The tumors were rejected after discontinuation of immunosuppressant treatment. Scale bar: 500 μm (A) and 100 μm (B).

Immunohistochemistry analysis during the immunorejection of grafted U251 cells. (A) Rejection graft status. (B) Quantitative analyses of the numbers of HNA-, CD3-, CD11b-, NKp46-, and TdT-positive cells (detected by the TdT-mediated dUTP nick-end labeling assay) cells after discontinuing immunosuppressant treatment. (C) HNA staining clearly shows the gradual reduction of the tumors. (D) CD11b, CD3, NKp46, and TdT staining in inflammatory cells. The numbers of CD11b-, CD3-, and NKp46-positive cells increased up to 32 days after transplantation and then gradually decreased. TdT-positive cells also became more prominent following transplantation. Scale bars: 100 μm (C and D).
Immunorejection of hiPSC-NS/PCs Grafted Into BALB/cA Mice After Discontinuation of Immunosuppressant Treatment
We also performed xenotransplantation of hiPSC-NS/PCs into the intact spinal cord of BALB/cA mice (n = 5) and observed tumor formation. This tumor showed biphasic pattern with areas of high and low cell density. The high cell density areas contained compact bipolar cells with Rosenthal fibers, whereas the low cell density areas contained loose-textured multipolar cells with microcysts. Histologically, these tumors resembled low-grade gliomas rather than teratomas. After confirming the tumor growth, we discontinued the immune suppression at 100 days after transplantation. Thereafter, we confirmed complete tumor rejection by BLI and histopathology (Fig. 7).

Representative quantitative analysis of photon counts derived from grafted hiPS-NS/PCs and pathological analysis of iPSC-NS/PC derived tumor. (A) Quantitative analysis of photon counts derived from grafted hiPSC-NS/PCs. The graft survival rate was 100% (n = 5/5) in the BALB/cA mice given immunosuppressant treatment (FK506 plus anti-CD4 mAb). After discontinuing the administration of FK506 and anti-CD4 mAb, all the grafted cells were rejected, and drastic reductions in the signal intensity were observed. (B, C) Representative H&E-stained sagittal images of the mouse spinal cord after cell transplantation. H&E staining revealed a biphasic tumor pattern with areas of high and low cell density. The high cell density areas contained compact bipolar cells with Rosenthal fibers, whereas the low cell density areas contained loose-textured multipolar cells with microcysts. The image within the square box is shown at higher magnification. After discontinuing the administration of immunosuppressants, the tumor was rejected completely. Scale bars: 500 μm in (B1, C1), 50 μm in (B2, C2), and 25 μm in (B3, C3).
Discussion
In terms of the clinical application of iPSC-based cell therapy, the most serious issue is the prospective tumorigenicity of the transplanted cells (21, 30, 44). Indeed, there is no report of the formation of tumors other than teratomas after transplantation of iPSC derivatives. However, here, we observed formation of a glioma-like tumor after the transplantation of NS/PCs derived from an inappropriate iPSC line that was established using a retroviral vector (Nori et al., submitted). Based on this result, in regard to potential tumorigenesis after iPSC-NSC transplantation, we have to consider not only teratoma formation but also tumors derived from cells of the neural lineage. In fact, we plan allogeneic iPSC-NSC transplantation into the injured spinal cord in a first-in human study from the viewpoints of the timing of transplantation after injury, safety, and cost performance. To minimize the risk of the therapy using iPSCs-NSCs, it will be important to examine whether the allografted iPSC-NSC-derived tumors could be eliminated without any adverse events by inducing immunorejection. As a first step, to confirm the feasibility of this concept, we used human GBM because GBM is the most aggressive and toughest growing tumor against immunorejection. To this end, we established a human GBM-grafted mouse model with or without immunosuppressant therapy. This system allowed the course of the tumor fate to be observed in real time using BLI under immunodeficient and immunocompetent conditions. We also utilized the same approach against the glioma-like tumor that developed after the transplantation of NS/PCs derived from inappropriate iPSCs. We propose that our model may serve as a valuable tool to perform translational research of iPSC-derived NS/PC transplantation for SCI.
Human Glioma-Grafted Model
Many brain tumor models using rat glioma cell lines have been reported (5, 15, 16, 19, 45). However, there are several limitations to the use of these rodent tumor models, such as the different response of rodent versus human glioma cell lines to treatment with chemotherapy agents (34, 40, 41). Therefore, xenotransplantation models of human glioma cell lines into animal models have begun to be investigated (31–33). In the present study, we developed a novel mouse model of an intramedullary spinal cord tumor by using the human GBM U251 MG cell line. We also refined the human GBM xenograft model by using BLI, which is a reliable method for live monitoring of tumor growth in vivo (24, 26, 29, 39). The advantage of BLI is that tumor cells are tagged with the luciferase gene, and only living tumor cells are visualized, while GFP-based imaging can also detect dead cells. Furthermore, in contrast to magnetic resonance imaging, BLI detects only implanted cells without the inclusion of inflammatory regions (11, 29, 36). By measuring this luminescence, we can quantitatively evaluate the survival, growth, and rejection of grafted tumor cells over time.
Immunodeficient Mouse Model
The work described herein made use of the NOD/SCID mouse, which is an immunodeficient mouse lacking both T- and B-lymphocytes. The transplantation of luciferase-transfected U251 cells into this mouse strain produces enormous tumors with a consistent rate of growth, permitting the use of BLI to quantitatively measure the extent and duration of the tumor response to immunosuppressive therapy. For animal subjects receiving an injection of U251 cells, histopathological analysis revealed that tumor growth had progressed to the medulla, and our approach allowed anatomically accurate modeling of this type of brain tumor. Furthermore, histological assessment showed that this spinal cord tumor had cells characterized by nuclear atypia, microvascular proliferation, necrosis, and a high Ki-67 index (18%), similar to the findings of human grade IV GBM. Our model is simple and reproducible and may be useful to evaluate the effectiveness of new treatments for spinal cord GBM.
Immunocompetent Mouse Model
The CNS, comprising the brain and the spinal cord, has long been assumed to exhibit higher immunological tolerance than other organs due to the following anatomical and physiological characteristics: 1) The CNS is in a state of immunological neglect because the brain and spinal cord are isolated from the rest of the body by the blood–brain barrier; 2) the brain and spinal cord contain a paucity of lymphocytes; 3) expression of major histocompatibility complex is low in the CNS; 4) antigen- presenting cells do not function well in the CNS; and 5) T-cell apoptosis is induced by Fas/FasL in the CNS (23, 35). We also transplanted U251 cells into the spinal cord of immunocompetent C57BL6/J mice without immunosuppressant administration. Consequently, immunorejection of the grafted cells was precipitated, even though the CNS is a so-called immune-privileged site. These observations may be due to the invasion of activated T-cells and NK cells into the CNS beyond the blood–brain barrier (12). To avoid this immunorejection, we administered immunosuppressants (FK506 and anti-CD4 Ab) to the immunocompetent mice. FK506 treatment prevented immunorejection in 80% of mice, whereas the combination of FK506 and anti-CD4 Ab treatment resulted in the complete engraftment of xenografted cells in all mice. These findings suggest that our human GBM model has great merit for the study of the immune dynamics of xenotransplantation.
Immunorejection of the Grafted Tumor
To obtain a proof of concept regarding the immunorejection of graft-derived tumor cells by immunomodulation, we first confirmed their growth in the presence of immunosuppressants via BLI. Next, we discontinued the administration of FK506 and anti-CD4 Ab to induce rejection of the tumor cells. Our GBM model showed hindlimb paralysis and eventual death at 4 weeks after transplantation under immunosuppressant administration. Given that the halflife of FK506 in the blood is 12 h and that of mouse anti-CD4 Ab is 9 days, the recovery of CD4-positive cells in the blood took about 14 days after the discontinuation of the immunosuppressants. At the beginning of this study, we discontinued administration of the immunosuppressants at 2 weeks after transplantation. Although CD4-positive cells in the peripheral blood were recovered at this timepoint, all the mice died due to tumor growth. Therefore, we discontinued administration of the immunosuppressants on day 7 after U251 MG cell transplantation, when the tumors were rejected in all the mice. Based on this result, we considered that we might take advantage of immunorejection to control the survival of grafted highly malignant tumors such as GBM. In general, in the immunorejection of a xenograft, the problem is hyperacute rejection by NK cells to α-gal (13). However, this mechanism does not occur in xenografts of human cells in mice. In the present study, it is likely that the immunorejection occurred mainly through T-cells as well as by an interaction with APCs, rather than via NK cells (Fig. 6), which is supported by several previous reports (1, 2, 8, 38). Although it is well known that GBM can avoid immune attack through tumor immunity (43), transplantation immunity caused rejection through T-cells, resulting in complete eradication of the grafted GBM without any adverse events such as neurological deterioration in this study. We also performed xenotransplantation of NS/PCs derived from an inappropriate hiPSC line into the intact spinal cord of BALB/cA mice and observed tumor formation. After discontinuation of immune suppression, we observed complete tumor rejection by BLI and histopathology. Finally, we confirmed the effectiveness of this paradigm against the tumor derived from the iPSCs-NS/PCs. These findings suggest that it is possible to induce immunorejection of any type of xenografted tumor cells by immunomodulation. However, the tumorigenic mechanisms of iPSCs-NS/PCs are still to be elucidated. Therefore there may be various differences between iPSC-NS/PC-derived tumor and glioblastoma, which are thought to arise from intrinsic glial cells with genetic mutations, abnormal epigenetic modifications and altered cellular metabolisms (7). These issues should be addressed in the future study.
Conclusions
In conclusion, the present study demonstrated that a mouse xenograft model of U251 glioma might be a reliable tool to target human spinal cord tumors in preclinical studies. Using this model, we were able to manipulate the survival of the grafted cells, even high-grade glioma cells, by immunorejection. This model may also be useful for the study of the therapeutic effect of anticancer drugs against malignant tumors. We are currently developing an iPSC-derived NS/PC transplantation therapy for SCI in animal models (18, 27). The issue of the tumorigenicity of iPSC-derived NS/PCs remains a major concern toward clinical application of cell therapy in the near future. By applying the results of this study, we can evaluate whether the allografted cells can be eliminated by immunorejection in the case of tumors developing after transplantation of iPSC-derived NS/PCs for SCI.
Footnotes
Acknowledgments
We appreciate the help of Dr. K. Fujiyoshi, Dr. O. Tsuji, Dr. Y. Takahashi, Dr. S. Kaneko, Dr. A. Yasuda, Dr. S. Nori, Dr. T. Konomi, Dr. S. Shibata, Dr. M. Shinozaki, Dr. R. Zhang, Dr. S. Tashiro, Dr. S. Kawabata, Dr. Y. Nishiyama, Dr. K. Hori, Dr. M. Ozaki, Dr. T. Iida, Dr. K. Matsubayashi, and Dr. S. Suzuki, who are all members of the spinal cord research team at the Department of Physiology, Orthopaedic Surgery and Rehabilitation Medicine, Keio University School of Medicine. We also thank Ms. T. Harada, Ms. S. Miyao, Ms. M. Mizutani, Ms. Y. Fukaya, Ms. R. Yoshida, and Ms. H. Shimada for their assistance with experiments and animal care. This work was supported by grants from the Japan Science and Technology (JST)-California Institute for Regenerative Medicine (CIRM) collaborative program to M.N.; a grant-in-aid for Research Center Network for Realization of Regenerative Medicine, Centers for Clinical Application Research on Specific Disease/Organ, and the Ministry of Education, Culture, Sports, Science and Technology (MEXT) from the JST to H.O.; grants-in-aid for Scientific Research from the Japan Society for the Promotion of Science (JSPS) and the MEXT; Keio Gijuku Academic Development Funds; a grant-in-aid for the Global Center of Excellence (COE) program from the MEXT to Keio University; and by a grant-in-aid for Scientific Research on Innovative Areas (Comprehensive Brain Science Network) from the MEXT. H.O. is a scientific consultant for San Bio, Inc., Eisai Co., Ltd., and Daiichi Sankyo Co., Ltd. The other authors declare no conflict of interests.
