Abstract
Acute myocardial infarction (MI) is a fatal event that causes a large number of deaths worldwide. MI results in pathological remodeling and decreased cardiac function, which could lead to heart failure and fatal arrhythmia. Cell therapy is a potential strategy to repair the damage through enhanced angiogenesis or by modulation of the inflammatory process via paracrine signaling. Amniotic fluid-derived progenitor cells (AFPCs) have been reported to differentiate into several lineages and can be used without ethical concerns or risk of teratoma formation. Since pigs are anatomically, physiologically, and genetically similar to humans, and pregnant pigs can be an abundant source of AFPCs, we used porcine AFPCs (pAFPCs) as our target cells. Intramyocardial injection of AFPCs has been shown to cure MI in animal models. However, intramuscular transplantation of cells has not been extensively investigated. In this study, we investigated the therapeutic potential of intramuscular injection of pAFPCs on acute MI. MI mice were divided into 1) PBS control, 2) medium cell dose (1 × 106 cells per leg; cell-M), and 3) high cell dose (4 × 106 cells per leg; cell-H) groups. Cells or PBS were directly injected into the hamstring muscle 20 min after MI surgery. Four weeks after MI surgery, the cell-M and cell-H groups exhibited significantly better ejection fraction, significantly greater wall thickness, smaller infarct scar sizes, and lower LV expansion index compared to the PBS group. Using in vivo imaging, we showed that the hamstring muscles from animals in the cell-M and cell-H groups had RFP-positive signals. In summary, intramuscular injection of porcine AFPCs reduced scar size, reduced pathological remodeling, and preserved heart function after MI.
Keywords
Introduction
Coronary artery disease (CAD) is one of the major causes of death worldwide. Acute myocardial infarction (AMI) is a CAD characterized by acute occlusion of the coronary artery. AMI often causes cell death in the ischemic area, results in pathological remodeling, decreases cardiac function, and may even lead to heart failure eventually. There has been a recent focus on the use of cell therapy to treat myocardial infarction.
Of the different cell therapy strategies used, results from a phase I clinical trial and preliminary data from a phase II trial showed that mesenchymal stem cells had a low therapeutic efficiency when used to treat MI (25). When myocardial progenitor cells were transplanted, an irregular pulse was detected (26). Although transplantation of cardiac stem cells improved cardiac performance after AMI in a phase I trial (1,2), obtaining sufficient cell numbers for transplantation was a major challenge. Induced pluripotent cells were shown to possess great differentiation potential, but carried the risk of being tumorigenic.
Progenitor cells comprise the majority of cells in human amniotic fluid after 18 weeks of gestation and are a potential source of organ-specific precursor cells for clinical trials (6). Amniotic fluid-derived progenitor cells (AFPCs) can be collected from various species including humans (17), pigs (32), and mice (20). AFPCs have been shown to express mesenchymal cell-related markers such as CD44, CD105, and CD90, but do not express hematopoietic cells markers such as CD34, CD45, and CD31 (4,17,20). AFPCs have the potential to generate several lineages (4,7,20) and have been used to ameliorate liver fibrosis (24), MI, and acute tubular necrosis (29). AFPCs possess telomerase activity, maintain telomere length after serial passaging, and do not form teratomas after transplantation (4,7,19). Furthermore, AFPCs express major histocompatibility complex (MHC) class I, do not express MHC class II (17,19), and are thought to modulate the immune system via secretion of interleukin-6 (IL-6), IL-10, monocyte chemoattractant protein-1 (MCP-1), and MCP-2 (31,27).
Studies investigating the mechanisms underlying cell therapy-mediated effects in the treatment of MI reported that donor cells differentiate into myocardial cells, endothelial cells, and smooth muscle cells to repair the lesion (1,30). However, it has been suggested that donor cells cannot reside in the heart for a long time after transplantation (9,16,21), and some studies reported a paracrine signaling mechanism by which the donor cells reduced cell apoptosis and fibrosis and increased vasculogenesis to repair the injury (8,35,36).
A number of studies have investigated the efficacy of different transplantation methods to treat MI. Although intravenous infusion of progenitor cells is a convenient approach to treat MI, only a few cells were shown to migrate to the target site, and the majority of the transplanted cells remained in the lung, causing increased risk of vascular occlusion (11,21). Infusion of the cells from the coronary artery by cardiac catheterization is a common way to treat MI. However, it causes occlusion of target vessels and increases the risk of interventions. Direct heart injection is a technique for cell transplantation that has the advantages of low numbers of cells needed and a high retention rate in the target area. However, there is a risk of cardiac perforation (23). A recent study described a noninvasive transplantation strategy using intramuscular infusion, wherein the transplanted stem cells secreted stromal cell-derived factor-1 (SDF-1), IL-6, vascular endothelial growth factor (VEGF), and hepatocyte growth factor (paracrine effect) and enhanced angiogenesis in the injured heart tissue (34).
Since pigs resemble humans in terms of anatomy, physiology, and genetics, and the pregnant pig is a rich source of AFPCs, we used pig-derived AFPCs (pAFPCs) as our target cells. In this study, we established a mouse AMI model by ligating the left anterior descending coronary artery and studied the therapeutic potential of pAFPCs delivered via intramuscular injection.
Materials and Methods
Animals
Male C57/B6 mice were acquired from the Laboratory Animal Center of National Taiwan University College of Medicine (Taipei, Taiwan). Animal maintenance and experimentation were performed under the guidelines of the Institutional Animal Care and Use Committee. Mice were fed the Laboratory Rodent Diet 5001 (PMI Nutrition Int., Los Angeles, CA, USA). The temperature in the animal facility was maintained at 21—25°C, and humidity was maintained at 50—70%. The photoperiod cycle consisted of 14 h of light and 10 h of darkness.
Establishment of Ds-Red-Harboring Pig Amniotic Fluid-Derived Progenitor Cells
Ds-red-harboring pigs were established and raised in our lab. In order to anesthetize the Ds-red-harboring pregnant (70 days) pigs (18 months old), they were treated with 10 ml Stresnil, 5 ml atropine muscularly, and 300 mg/10 ml citosol intravenously with 2 O2:1 N2 and halothane (2-bromo-2-chloro- 1,1,1-trifluoroethane contains 0.01% thymol) (all reagents: Janssen Pharmaceutica NV, CCPC, Belgium). The abdomen and uterus were dissected to derive amniotic fluid. We utilized 70-μm mesh (Miltenyi Biotec, Auburn, CA, USA) to filter the fluid and centrifuged the fluid at 1,000 × g for 10 min. The pellet was resuspended in α-MEM (Sigma-Aldrich, St. Louis, MO, USA) and at a density of 2 × 105 cells/cm2 to culture the cells in a 10-cm dish (TPP, Trasadingen, Switzerland) at 37°C in the presence of 5% CO2. Until confluence, the supernatant was discarded, and the cells were subcultured in a proportion of 1:2. These cells all expressed Ds-red protein and glowed red. Cells were stained with 0.1 g/ml 4,6 diamidino-2-phenylindole (DAPI) (Sigma-Aldrich) for 1 min, and phosphate-buffered saline (PBS) was used to rinse the cells. Cells were observed under a microscope (Leica DMi1; Leica Microsystems, Wetzlar, Germany). In order to verify if the cells obtained were progenitor cells, the cells were harvested, resuspended at a density of 5 × 105 cells/100 μl, and incubated with CD31—PE, CD4a—PE, CD44—PE, CD90—PE (1:100 for all antibiotics) (eBioscience, San Diego, CA, USA), and then subjected to flow cytometry (FACS scan flow cytometer, FACSAria; Becton-Dickinson, San Jose, CA, USA).
Establishment of Myocardial Infarction (MI) Mouse Model
We investigated whether xenogenic transplantation of pAFPCs could reduce the infarcted area while maintaining the geometry. Briefly, 8-week-old C57/B6 mice were anesthetized with an intraperitoneal injection of 2,2, 2-tribromoethanol (1 ml/10 g) (Sigma-Aldrich) (Fig. 1.1). The hair around the mouse breast was shaved, and the mouse was intubated using the pressure mode of a small animal breathing machine SAR-830/AP (CWE, Ardmore, PA, USA) to maintain breathing for the duration of surgery (Fig. 1.2). The surgery for ligation of the left descending coronary was performed as previously described (10). The left breast was dissected, the fourth rib muscle was separated, and the heart was exposed (Fig. 1.3—5). The left descending artery was ligated at a 2-mm distance from the atrium using 6-0 thread (UNIK, Taipei, Taiwan) (Fig. 1.6). A successful procedure was indicated by the blood supply turning pale.

Establishment of the mouse MI model. Mice were anesthetized with avertin, and the left chest shaved (1). The mice were then intubated and their breathing maintained with a small animal ventilator (2). The skin was severed on the left chest, and muscle layers were dissected (3, 4). We made a hole in the fourth intercostal space in order to expose the heart (5). In the MI group, the left descending coronary artery was ligated with silk suture (6). After ligation, a purse suture was made to close the skin and muscle layers (7). The operated mice were kept under a far-red lamp for recovery (8). Color change of the ventricle wall was monitored to assess if ligation of the left descending coronary artery was successful. When ligation succeeded, the wall of the infarcted region would turn pale (9, 10). Representative M-mode echocardiograph from the MI group and sham group at 4 weeks after surgery (11, 12).
The procedure is depicted in Figure 1. After ligation, a purse suture was made to close the skin and muscle layers (Fig. 1.7). The mice were placed under a red lamp for recovery (Fig. 1.8). To confirm that ligation of the left descending coronary artery was successful, the color change of the ventricle wall was monitored. When ligation was successful, the wall of the infarcted region turned pale (Fig. 1.9, 1.10). Representative M-mode echocardiographs from the MI group and the sham group at 4 weeks after surgery are shown in Figure 1.11 and 1.12, respectively.
Cell Transplantation Method and Experimental Design
Ds-red-labeled pAFPCs were cultured to seventh passage, and the cells were suspended by 2.5% trypsin-EDTA and were centrifuged at 1,000 × g for 5 min. The supernatant was discarded, and the cell pellet was dispersed by PBS. The cell concentration was diluted to 1 × 106 cells in 50 μl PBS and 4 × 106 cells in 50 μl PBS. We explored the curing efficiency of intramuscular injection of seventh passage of Ds-red-labeled pAFPCs on acute MI. Eight-week-old male C57/B6 MI mice were divided into 1) 50 μl PBS control, 2) medium cell dose (1 × 106 cells in 50 μl PBS per leg; cell-M), and 3) high cell dose (4 × 106 cells in 50 μl PBS per leg; cell-H) groups. Cells or PBS were directly injected into the hamstring muscle by a 29-gauge insulin needle (BD Biosciences, San Jose, CA, USA) 20 min after MI surgery.
Echocardiography
Mice were anesthetized by inhalation of 1% isoflurane (Sigma-Aldrich), and echocardiography was performed using Panoview β1500 (Coretronic Display Solution Corporation, Taipei, Taiwan). M-mode was used to measure end-diastolic dimension (EDD), end-systolic dimension (ESD), and thickness of the ventricular wall. The formula for ejection fraction (EF) percentage is (EDV − ESV) / EDV × 100% {end-diastolic volume (EDV): [(EDD)3 × 7 / (2.4 + EDD)] (ml), end-systolic volume (ESV): [(ESD)3 × 7 / (2.4 + ESD)] (ml)}. Left ventricle (LV) expansion index was assessed as previously described (15).
Histological Assessment
Mice were anesthetized with 2,2,2-tribromoethanol and sacrificed using KCl (Sigma-Aldrich). The heart was collected and fixed by soaking in 4% paraformaldehyde (Sigma-Aldrich) for 3 days. The tissues were embedded in methylmethacrylate (Sigma-Aldrich) and sectioned longitudinally (5-mm thickness) using a Leica/Jung 2255 microtome (Leica, Wetzlar, Germany).
Masson Trichrome Stain
Sections were dewaxed with xylene (Sigma-Aldrich) and rehydrated with different concentrations (100%, 95%, 90%, 80%, and 70%) of alcohol. The biopsy specimens were fixed for 15 min in Bouin's solution (Sigma-Aldrich) and washed with water until the yellow marks disappeared on the slice. The sections were then stained with Weigert's iron hematoxylin (Sigma-Aldrich) for 10 min, rinsed with water for 5 min, and then stained with Biebrich scarlet-acid fuchsin solution (Sigma-Aldrich) for 5 min. The sections were rinsed with water for 5 min, stained with phosphomolybdic-phosphotungstic acid solution (Sigma-Aldrich) for 5 min, washed with water, and then placed in 1% acetic acid solution (100058; Merck, Darmstadt, Germany) for a few seconds. This was followed by a dehydration step for a few seconds in 95% and 100% alcohol, respectively. The sections were eventually soaked twice for 2 min each time in xylene and then mounted for observation.
Measurement of Fibrosis Scar Size in the Left Ventricle (LV) of the Heart
We measured scar size using the formula (endodiameter of LV fibrosis scar area) + (exodiameter of LV fibrosis scar area)/(LV endodiameter + LV exodiameter) × 100%.
In Vivo Image System Detection for Ds-Red Protein
In order to investigate the donor cell fate and destiny, the hearts were evaluated for the presence of Ds-red protein using the in vivo imaging system (IVIS) Imaging System 200 (Xenogen Corporation, Alameda, CA, USA).
Statistical Analyses
Our data were presented as mean ± standard deviation. In order to measure significance, the Student's t-test was implemented. Values of p < 0.01 were regarded as significant after Bonferroni correction (SPSS version 20; IBM Corporation, Armonk, NY, USA).
Results
We successfully obtained pAFPCs, which expressed Ds-red protein (Fig. 2a—c). We also showed that pAFPCs expressed CD44 (cell migration marker) (Fig. 2d), CD90 (stem cell marker) (Fig. 2e), but did not express CD31 (endothelial marker) (Fig. 2f) or CD4a (lymphocyte marker) (Fig. 2g).

Establishment and characterization of Ds-red pAFPCs. Morphology of Ds-red pAFPCs was evaluated under fluorescent film (a), DAPI staining for nucleus (b), and merge (a) and (b) picture (c). Surface antigen markers were detected by flow cytometry. Ds-red pAFSCs expressed CD44 (cell migration marker) (d) and CD90 (stem cell marker) (e), but did not express CD4a (lymphocyte marker) (f) and CD31 (endothelial marker) (g). Scale bar: 75 μm.
We showed that MI resulted in the formation of a scar with a size of about 40% (Fig. 3a). Wall thickness of the MI and sham groups was measured, and the black bar indicates the measurement positions of LV wall thickness (Fig. 3b). There was a significant decrease in the wall thickness in the infarct region of animals in the MI group compared to the sham group (Fig. 3b). There was also a significant decrease in the LVEF of animals in the MI group compared to the sham group at 2 weeks and 4 weeks after MI. Animals in the MI group had a significantly lower EF at 2 weeks compared to the baseline EF. These animals also had a significantly lower EF at 4 weeks compared to that at 2 weeks (Fig. 3c), suggesting that after MI surgery, heart function decreased with time (Fig. 3c). Decreased wall thickness in the infarction region and formation of a fibrosis scar within injury sites indicated that a suitable MI model had been established.
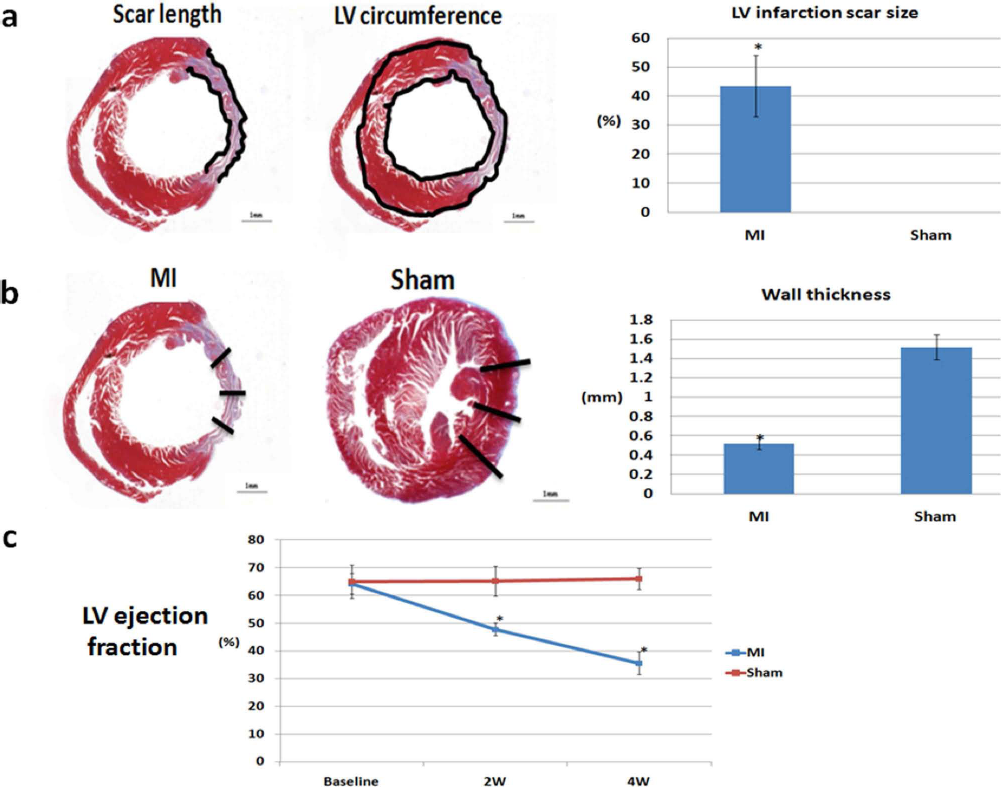
Comparison of scar size and LVEF in MI and sham control groups. LV infarction scar size was measured. The scar size was calculated as scar length divided by LV circumference and is depicted as a black line. After MI, a scar with a size about 40% formed (a). Wall thickness of MI group and sham group are shown, and the black bar indicates measurement positions of LV wall thickness (b). Wall thickness in the infarct region was significantly reduced in the MI group (b). The MI group had a significantly lower LVEF compared to the sham group at 2 weeks and 4 weeks after MI. In the MI group, the EF at 2 weeks was significantly lower than the baseline EF. The ejection fraction at 4 weeks was also significantly lower than the EF at 2 weeks. These results suggested that heart function decreased with time after MI surgery (c). Here are representative short-axis heart sections from the MI group and sham group. Sections were stained with Masson's trichrome staining. Collagen was stained with blue color. *p < 0.01 versus sham. n = 4 animals/group. Scale bars: 1 mm.
A comparison of the experimental groups at 4 weeks after MI surgery showed that the cell-M and cell-H groups had a significantly higher LVEF (43.16 ± 2.81%, 50.04 ± 5.37%) compared with the PBS group (37.54 ± 3.98%; p < 0.01) (Fig. 4a). The LVEFs of animals in the cell-M and cell-H groups at 4 weeks after MI surgery were similar to those at 2 weeks after MI (Fig. 4a). Animals in the cell-H group had a smaller LV infarction scar size compared to the PBS group (31.71 ± 1.87% vs. 39.00 ± 2.79%) (Fig. 4b). However, the animals in the cell-M and cell-H groups had a higher ventricle wall thickness at 4 weeks after MI surgery compared to animals in the PBS group (0.58 ± 0.04 mm and 0.56 ± 0.09 mm vs. 0.49 ± 0.05 mm, respectively) (Fig. 4c). LV expansion index was assessed as previously described (15). The data demonstrated that the LV expansion index was significantly lower in the cell-M and cell-H groups compared to the PBS group, indicating a lower degree of dilation (0.72 ± 0.09 and 0.63 ± 0.02 vs. 0.88 ± 0.13, p < 0.01) (Fig. 4d). As a result, MI mice in the cell-M and cell-H groups showed an amelioration in the LV expansion indexcomparedtothePBSgroupat4weeks after injection into the quadriceps. However, there was no significant difference between the cell-M and cell-H groups (Fig. 4d). Representative short axis heart sections were prepared from normal mice (Fig. 4e1), MI mice treated with PBS (Fig. 4e2), MI mice in the cell-M group (Fig. 4e3), and MI mice in the cell-H group (Fig. 4e4). They were stained with Masson's trichrome stain. There was an improvement in the MI hearts in the cell-M (B) and cell-H (C) groups (Fig. 4e1—4).

Effects of transplantation of Ds-red pAFPCs on LVEF (a), LV infarction scar size (b), wall thickness (c), and LV expansion index (d) in MI mice. Short axis of heart sections stained with Masson's trichrome from control mice (e1), MI mice treated with PBS (e2), MI mice treated with cell-M (e3), and MI mice treated with cell-H (e4). Animals in the cell-M and cell-H groups had a better LVEF compared to the PBS group, and animals in the cell-H had a better LVEF than animals in the cell-M group at 4 weeks. However, there was no significant difference between the three groups at 2 weeks (a). The LV infarction scar size was decreased in the cell-H compared to the PBS group 4 weeks after injection into MI mice quadriceps (b). Wall thickness in the cell-M and cell-H groups was thicker compared to that in the PBS group 4 weeks after injection into the quadriceps of MI mice (c). The LV expansion index was ameliorated in the cell-M and cell-H groups compared to the PBS group 4 weeks after injection into the quadriceps of MI mice. However, there was no significant difference between the cell-M and cell-H groups (d). Representative short-axis heart sections stained with Masson's trichrome staining from normal mice (e1), MI mice treated with PBS (e2), MI mice in the cell-M group (e3), and MI mice in the cell-H group (e4). There was a significant improvement seen in the cell-M (e3) and cell-H (e4) groups *p < 0.01 versus PBS group, #p < 0.01 versus cell-M group, n = 5 animals/group. Scale bars: 1 mm.
Using IVIS imaging system 200, we detected Ds-red fluorescent protein in the cell-M and cell-H groups (Fig. 5). IVIS showed that both doses (1 × 106 or 4 × 106 cells/animal) were retained in the quadriceps for at least 4 weeks after injection (Fig. 5), suggesting that the donor cells were not discarded by the host immune system.

IVIS imaging to detect red fluorescent protein signals in hearts collected from the PBS, cell-M, and cell-H groups. Two doses of AFPCs (2 × 106 and 8 × 106 cells/animal) were retained in the quadriceps for 4 weeks after injection. Scale bars: 5 mm.
Discussion
In this study, we successfully established a mouse MI model using left descending coronary artery ligation. We obtained Ds-red-harboring pAFPCs and verified their identity by flow cytometry. Porcine-derived bone marrow mononuclear cells were previously used to treat porcine MI and were shown to elevate regional blood flow fourfold and increase capillary densities threefold compared to the control group. This was shown to be affected by paracrine signaling via mediators such as interleukin-1β, tumor necrosis factor-a, basic fibroblast growth factor, VEGF, and angiopoietin 1 (18). The fact that only 10% of the donor cells were maintained for a period of 2 weeks strongly suggested that recovery of the lesion was mediated via the paracrine effect (13). Donor cells were also previously shown to stimulate the proliferation and differentiation of endogenous stem cells to myocardial cells and vessel structures (14,33). Approximately 14% of donor cells were shown to give rise to myocardial cells (30). A recent clinical trial showed recovery of heart function in 6% of study patients after infusion of donor cells to the coronary artery (12). In this study, we aimed to observe the therapeutic efficacy of a high dose or medium dose of xenogenic AFPCs injected into the leg muscle of MI mice. Our data showed that both the high dose and medium dose of donor cells ameliorated the symptoms of MI. Our results were consistent with previous data showing that injection of bone marrow-derived stem cells caused a significant increase in myocardial regeneration (28).
Although pAFPCs were previously demonstrated to be capable of giving rise to spontaneously beating myocardial cells in vitro (4), they could not differentiate to myocardial cells in vivo when directly injected into the heart (32). Interestingly, secretion of MCP-1, IL-8, SDF-1, and VEGF by AFPCs was shown to promote vasculogenesis, and injection of MI mice with conditioning medium containing these factors ameliorated the symptoms and conferred the same therapeutic effect as direct cell transplantation (3). These data suggested that AFPCs repaired the damage via paracrine signaling. It was also previously shown that AFPC-secreted cytokines injected in the leg muscles were transported to the damaged heart via the bloodstream (22). Owing to the risk of perforation when cells are directly transplanted into the heart, we injected the donor cells into the quadriceps and showed that this area provided an optimal scenario for donor cell survival.
It was previously demonstrated that xenogeneic AFPCs rescued the infarcted heart either by means of differentiating to endothelial cells, smooth muscle cells, and myocardial cells or via paracrine signaling in a rat model (3,22). Since direct transfusion of cells into the injury osite tissue presents challenges in clinical practice, a novel strategy was developed to implant the mesenchymal stem cells into the quadriceps, and the implanted cells were shown to preserve heart function in a paracrine manner (34). In the present study, we injected the Ds-red-harboring pAFPCs into mouse quadriceps and investigated whether these cells could improve the function of infarcted myocardium. We showed that Ds-redharboring pAFPCs reduced scar formation and possibly participated in remodeling after MI. Since the therapeutic efficacy of a high dosage of implanted cells was shown to be higher than the low dosage (4,17,20), we investigated if the therapeutic effect was influenced by the number of implanted cells. We injected the quadriceps of the MI mouse with a low dose (1 × 106 cells/animal) or a high dose (4 × 106 cells/animal) of Ds-red-harboring pAFPCs. We utilized echocardiography, tissue sections, and the noninvasive IVIS to evaluate therapeutic effects of the two doses and to observe whether the cells were maintained in the quadriceps.
Our echocardiography data demonstrated that cells injected into quadriceps could attenuate the reduction of LVEF. We also showed that implantation with a high cell dose was significantly more effective in the preservation of heart contractility and heart structure after MI compared to the low cell dose. Our results were consistent with a previous study where cells were directly transplanted into the lesion site of the myocardium (3). Additionally, we also used IVIS to show that the donor cells could be detected in the quadriceps for at least 4 weeks after implantation and could attenuate the deterioration of heart contractility. Our results suggest that more cells were retained at the site of implantation compared to previous studies where the cells were directly injected into the infarcted heart (5,22).
Since AFPCs were previously shown to disappear 4 weeks after transplanting into the heart (22) and kidney (29), our data suggested that the quadriceps (leg muscle) provides the optimal environment for cell survival. Furthermore, localization of the Ds-red protein in the quadriceps supports a previous study showing the role of secreted cytokines in repair of the injured site (34). However, since IVIS has a limited sensitivity for detection of the fluorescent protein, it will be informative to use more sensitive methods such as PCR to detect the localization of Ds-red-labeled cells in future studies.
Conclusion
Xenogeneic AFPCs derived from pig preserved heart function, reduced scar formation, and possibly played a role in remodeling after MI. In addition, the high dose of implanted cells showed more efficacy than the low dose. The pAFPC-transplanted group had a higher LVEF compared to the PBS control group at 4 weeks after implantation. Importantly, data from short-axis heart sections revealed that ventricle wall thickness in the cell-transplanted group was significantly thicker than in the control group, while heart expansion index and scar size in the cell-transplanted groups were significantly less than in the control group. In summary, pAFPCs improved heart function and retained structure in a mouse model of MI.
Footnotes
Acknowledgments
This research was funded by National Science Council in Taiwan (NSC102-2628-B-182A-007-MY2) and Chang Gung Memorial Hospital funding CMRPG3B1733. The authors declare no conflicts of interest.
