Abstract
Duchenne muscular dystrophy (DMD) is an X-linked genetic disorder caused by mutations in the dystrophin gene. Adipose-derived stem cells (ASCs) are an attractive source of cells for stem cell therapy. Losartan has been reported to improve ASC transplantation in injured mouse muscles. In the present study, we investigated whether the combined treatment of losartan and ASCs in the injured muscles of mdx mice improves regeneration. The combined treatment of ASCs and losartan remarkably improved muscle regeneration and induced muscle hypertrophy. In addition, ASCs and losartan treatment downregulated transforming growth factor-β and inhibited muscle fibrosis. We observed cells coexpressing green fluorescent protein (GFP) and dystrophin in the muscle samples of mice transplanted with GFP-positive ASCs. In the coculture in vitro experiment, we also observed that the GFP ASCs differentiated into dystrophin-expressing myotubes. The present study shows that the combination of transplanted ASCs and treatment with losartan ameliorated muscle fibrosis and improved muscle regeneration in injured mdx mice. Thus, we suggest that combined treatment with losartan and ASCs could help to improve muscle regeneration in the muscles of injured patients, including DMD patients.
Keywords
Introduction
Duchenne muscular dystrophy (DMD) is an X-linked genetic disorder characterized by progressive muscle degeneration and weakness and is caused by mutations in the dystrophin gene (3,30,31). Dystrophin is localized under the sarcolemma and forms a dystrophin–glycoprotein complex with dystroglycan, sarcoglycan, and syntrophin/dystrobrevin. These association links have mechanically stabilizing and signaling roles in mediating interactions between the intracellular cytoskeleton, plasma membrane, and extracellular matrix (ECM) (6,31). Thus, the loss of dystrophin consequently leads to a breakdown of the structural integrity of myofibers resulting in increased myofiber vulnerability, leading to muscle necrosis and regeneration. The frequent cycle of necrosis and regeneration induces chronic inflammation and fibrosis, progressively replacing the muscle tissue (27).
So far, no effective treatment for DMD has been identified. However, several studies on cell therapy have offered promising therapeutic treatments for DMD (29). Not only muscular cells, but also nonmuscular cells retain myogenic potential. For example, muscle-derived stem cells, myoblasts, mesoangioblasts, mesenchymal stem cells, and adipose-derived stem cells (ASCs) can differentiate into muscle cells upon in vitro induction or in an in vivo muscle-damaged mouse model (11,13,18,36,41). Thus, these cells were studied and used as a source for stem cell therapy. Specifically, ASCs have several advantages in stem cell therapy as well as a myogenic potential. First, ASCs can be easily obtained from adipose tissues and maintained in stable growth and proliferation (42,44). Second, they can be differentiated into adipogenic, osteogenic, chondrogenic, neurogenic, or myogenic lineages (21,38,39,44). Third, ASCs secrete various cytokines such as interleukin-6 and growth factors such as basic fibroblast growth factor (bFGF), hepatocyte growth factor, and insulin-like growth factor (IGF), which are involved in muscle satellite cell activation and regeneration (7). Finally, allogenic ASCs injected intravenously or locally induce no signs of immune rejection (13,44). Thus, ASCs seem to be an ideal type of cell for allotransplantation. Based on previous studies, ASCs can be used as a promising candidate for stem cell therapy in skeletal muscle regenerative diseases including DMD.
For successful cell transplantation, it is very important to make a good stem cell niche for settlement and differentiation of these cells. As mentioned above, DMD shows chronic injuries and inflammation leading to muscular fibrosis in muscles. Generally, fibrosis shows excessive deposition of ECM components mainly composed of collagen fibers and various other fibrous proteins. In a recent report, a network of dense collagen and fibrous proteins significantly decreased the distribution and efficacy of nanotherapeutics (12). In addition, transforming growth factor (TGF)-β, a profibrotic cytokine that is elevated in DMD (8), induces bad conditions for successful cell transplantation and muscle regeneration. Persistent overexpression of proteins, such as TGF-β, promotes the formation of fibrotic tissues to replace damaged myofibers and inhibits satellite cell activation, impairs myocyte differentiation, and results in the failure of skeletal muscle regeneration (4).
Losartan is a potent inhibitor of angiotensin II type I receptor and is used to manage blood pressure. Currently, DMD patients are being treated with losartan for its presumptive cardiac benefits and fibrosis reduction. Its effect on skeletal muscle strength and function made it highly anticipated for patient enrollment in 2011 (18). In addition to blocking TGF-β activation, losartan not only restored the regenerative capacity and improved muscle function in mouse models of DMD and congenital muscular dystrophy (10,15) but also improved the stem cell niche by inhibiting fibrosis in skeletal muscle injury (35).
Mdx mice are an animal model for the human disease DMD and exhibit high levels of TGF-β at an early age (32,45). We induced muscle damage in the mdx mice through laceration, and these mice were treated with ASCs and losartan. In the present study, we investigated whether combined treatment of ASCs and losartan could overcome the bad conditions, such as the high-level TGF-β, and improve muscle regeneration after injury.
Materials and Methods
Isolation of Mouse ASCs
C57BL/6J mice (male, 14 weeks old) and green fluorescent protein (GFP) transgenic C57BL/6J mice (male, 13 weeks old) were used for isolation of mouse ASCs. C57BL/6J mice were obtained from Japan SLC, Inc. (Hamamatsu, Japan). GFP transgenic mice, under the control of a chicken β-actin promoter and cytomegalovirus enhancer, were a gift from Dr. Masaru Okabe (Osaka University, Suita, Osaka, Japan) (33). ASCs were isolated as previously described (35). All mice were anesthetized, and abdominal fat was harvested and washed with Dulbecco's phosphate-buffered saline (PBS; PAA, Linz, Austria) and 70% ethanol (Duksan Pure Chemicals, Ansan, Korea). The adipose tissues were then finely minced and digested with 0.2% type I collagenase (Worthington Biochemical, Lakewood, NJ, USA) in PBS in a 37°C shaking incubator for 10 min. The digested tissue was passed through a 70-μm nylon cell strainer (SPL Life Sciences, Pocheon, Korea) to remove debris and then centrifuged at 200 × g for 3 min to collect the cell pellet. The pellet was resuspended and washed twice with PBS.
After isolation, the cells were plated onto low-glucose Dulbecco's modified Eagle medium (DMEM) (Gibco, Carlsbad, CA, USA) containing 10% fetal bovine serum and 1% penicillin/streptomycin solution (WelGENE, Daegu, Korea) and incubated at 37°C in a humidified environment containing 5% CO2. Passage 4 ASCs were used for transplantation and coculture.
Animals
Eight- to 12-week-old, male mdx mice (C57BL/ 10ScSn-Dmdmdx/J, an animal model of DMD) were used in this study. Mdx mice were kindly provided by Dr. Jacques P. Tremblay (CHUQ Research Center, Quebec City, Canada). All mice were maintained in a room at 22 ± 2°C with a relative humidity of 50 ± 10% and a 12-h light–dark cycle and fed standard food pellets and water ad libitum. All handling of the animals and experimental procedures were performed in accordance with NIH (Bethesda, MD, USA) guidelines and approved by the Kyungpook National University Institutional Animal Care and Use Committee (Approval No. KNU 2011 – 61).
Animal Experimental Design
The mdx recipient mice were divided into four groups: control (Con) group (n = 7), losartan (Los) group (n = 7), ASC group (n = 7), and ASC + losartan (ASC + Los) group (n = 7). The recipient mice were anesthetized with zoletil (Virbac, Carros, France) and xylazine (Bayer, Leverkusen, Germany), and then local muscle lacerations were performed in the gastrocnemius and soleus muscles of all the mice as previously described (35). Part of the muscle was removed with a 4-mm biopsy punch (Integra Miltex, Plainsboro, NJ, USA); ASCs (5 × 105 cells/PBS 10 μl per animal, ASC and ASC + Los groups) or the same volume of PBS without cells (Con and Los groups) were then directly injected into the damaged area using a micropipette (Eppendorf, Hamburg, Germany). Five mice per group were transplanted with wild-type ASCs, and the remaining two were transplanted with GFP-positive ASCs to visualize the regenerated myofibers. The skin was closed using a 3/0 black silk suture (Ethicon, Livingston, Scotland, UK). The Con and ASC groups were fed tap water, and the Los group and ASC + Los groups were fed losartan (Merck & Co., Readington Township, NJ, USA) dissolved in tap water (0.6 g/L) (10). After 3 weeks, all mice were sacrificed, and the injured muscles were collected.
Gross Examination of the Injured Muscles
After 3 weeks, the injured muscles were exposed and then evaluated by gross examination. The injured muscles were collected, and the weight of each skeletal muscle was immediately measured.
Measurement of Serum TGF-β Levels
Blood samples were collected at the time the mice were sacrificed, and the serum was isolated. The serum TGF-β level was measured with an enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Minneapolis, MN, USA).
Histopathology
For histopathological analysis, samples of the muscle tissues were collected from all the mice, fixed in 10% neutral-buffered formalin (DC Chemical Co., Seoul, Korea), processed by standard methods, and embedded in paraffin wax. Tissues sectioned at 4 μm were deparaffinized in toluene (Duksan Pure Chemicals) and rehydrated in a graded alcohol series. Hematoxylin (Sigma-Aldrich, St. Louis, MO, USA) and eosin (Junsei Chemicals, Tokyo, Japan) (H&E) staining and Masson's trichrome (MT; Sigma-Aldrich) staining were performed.
After H&E staining, the areas of all the myofibers visible at the original magnification of 400× in the injured portions were measured with the Leica Application Suite program (Leica Microsystems, Wetzlar, Germany). The average area of the myofibers was then calculated.
The number of centrally located nuclei, a hallmark of skeletal muscle regeneration, was also counted. Five areas were randomly selected at the original magnification of 400× per section, and the number of central nucleic (CN) myofibers and peripheral nucleic myofibers were counted, respectively. The percentage of CN myofibers was then calculated.
Isolation of Muscle-Derived Stem Cells (MDSCs) From MDX Mice
MDSCs were isolated with the preplate technique as previously described (19). Skeletal muscle biopsies were obtained from the hindlimbs of 4-week-old mdx mice and washed with PBS and 70% ethanol. The isolated muscle tissues were then finely minced and digested with 0.2% type I collagenase for 1 h, 0.2% dispase (Roche, Mannheim, Germany) for 45 min, and 0.1% trypsin-EDTA (Gibco) for 30 min in a 37°C shaking incubator. The digested tissue was passed through a 70-μm nylon cell strainer and centrifuged at 200 × g for 3 min to collect the cell pellet. The pellet was resuspended and plated onto growth medium [high-glucose DMEM (PAA), 10% fetal bovine serum (PAA), 10% horse serum (HS), 1% penicillin–streptomycin, 1 ng/ml bFGF (PeproTech, Rocky Hill, NJ, USA)]. Several hours after plating, the supernatant containing nonadherent cells was transferred into new tissue culture dishes. This step was repeated, and slowly adhering cells were used for the coculture.
Coculture with GFP-Positive ASCs and MDX-Derived MDSCs
For the coculture, the mdx-derived MDSCs were plated onto six-well plates (SPL Life Sciences) at a density of 1 × 105 cells/well in growth medium. After 24 h, GFP-positive ASCs were seeded onto the same well at a density of 5 × 104 cells/well in growth medium. After 24 h, the medium was changed to a differentiation medium (high-glucose DMEM, 5% HS, 1% penicillin–streptomycin). The cocultured cells were treated with losartan (10 μM) every other day for up to 15 days.
Quantitative Reverse Transcriptase-Polymerase Chain Reaction (qRT-PCR) Analysis
Total RNA was extracted from frozen muscle tissues and cells with Trizol reagent (Invitrogen, Carlsbad, CA, USA) following the manufacturer's instructions. The concentration of the isolated RNA was measured with the Quanti-iT RNA assay kit (Invitrogen) and a Qubit fluorometer (Invitrogen). cDNA was synthesized from the extracted RNA using the RT&GO mastermix (MP Biomedicals, Santa Ana, CA, USA). To analyze the mRNA expression levels, Rotor-Gene Q (Qiagen, Hilden, Germany) was used. The expression of the following genes was analyzed: mTOR, raptor, Pax7, myogenin, and type I collagen. The expression of the target genes was normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH). The ΔΔ threshold cycle (ΔΔCt) method was used to analyze the relative expression. Primers for mTOR (QT00118734), raptor (QT00145502), Pax7 (QT00147728), myogenin (QT00112378), and GAPDH (QT01658692) were obtained from Qiagen. Type I collagen was amplified with the following primers: 5′-TGCAACATGGAGACAGGTCA GA-3′ (forward) and 5′-GAACGGGAATCCATCGGTC AT-3′ (reverse) (18).
Immunoblotting Analysis
Snap-frozen muscle tissues were homogenized in RIPA buffer containing 1 mM sodium orthovandadate (Na3VO4), 50 mM NaF, and protease inhibitor cocktail tablets (Roche). The lysate was centrifuged at 3,000 × g for 10 min at 4°C to remove solid tissue and debris. Subsequently, the supernatant was centrifuged at 12,000 × g for 20 min at 4°C to obtain soluble cytosolic protein. The protein concentration was determined by the Bradford method (Bio-Rad, Hercules, CA, USA). Protein samples were separated by 10–12% SDS-polyacrylamide gel and Criterion XT 3–8% tris-acetate gel (Bio-Rad) electrophoresis. For the immunoblot, proteins were electrotransferred to a polyvinylidene fluoride membrane (Schleiche & Schuell Bioscience, Dassel, Germany) and blocked with a 3% bovine serum albumin (Sigma-Aldrich) in Tris-buffered saline (Sigma-Aldrich). The membrane was immunoblotted with the following primary antibodies: anti-FRAP (H-266) [=Mammalian target of rapamycin (mTOR), 1:2,000 dilution; Santa Cruz Biotechnology, Santa Cruz, CA, USA], anti-Raptor (1:2,000 dilution; Santa Cruz Biotechnology), anti-Pax7 (1:2,000 dilution; Santa Cruz Biotechnology), anti-myogenin (1:1,000 dilution; Santa Cruz Biotechnology), anti-4E-BP1 (1:2,000 dilution; Santa Cruz Biotechnology), and anti-phospho-4E-BP1 (1:1,000 dilution; Cell Signaling Technology, Danvers, MA, USA). Equal quantities of protein loadings were confirmed by detecting anti-GAPDH (Cell Signaling Technology). The primary antibodies were detected using horseradish peroxidase-conjugated anti-mouse or rabbit IgG (1:2,000 dilution; Santa Cruz Biotechnology). The specific binding was detected by using the Super Signal West Dura Extended Duration Substrate (Pierce, Rockford, IL, USA) and exposure to medical X-ray film (Kodak, Tokyo, Japan).
Immunofluorescent Studies
Two mice per group, transplanted with the GFP-positive ASCs, were transcardially perfused with 4% paraformaldehyde (Yukari Pure Chemicals, Tokyo, Japan). The collected muscle tissues from these mice were embedded in medium, and 8-μm-thick sections were then obtained. The tissue sections were immunostained with the primary antibody anti-dystrophin (1:100 dilution; Abcam, Cambridge, MA, USA) as previously described (25). Tetramethylrhodamine-5-(and-6)-isothiocyanate [5(6)-TRITC]-conjugated anti-mouse IgG (1:100 dilution) for dystrophin was used as the secondary antibody (Invitrogen). The nuclei of the tissues were counterstained with 4′,6-diamidino-2-phenylindole (DAPI, dihydrochloride) (Molecular Probes, Leiden, the Netherlands).
The cultured cells from all the groups were fixed in 4% paraformaldehyde at room temperature for 15 min and immunostained with the following primary antibodies: anti-dystrophin (1:100 dilution; Leica Microsystems) and anti-troponin I (1:200 dilution; Santa Cruz Biotechnology). 5(6)-TRITC-conjugated anti-mouse IgG (1:200 dilution) was used as the secondary antibody (Invitrogen). DAPI was used as a nuclear counterstain.
The fusion index of the stained cells with troponin I was calculated as follows: (number of myogenic nuclei of multinucleated cell/total number of nuclei present in the observed field) × 100. Fifteen different microscopic fields were chosen randomly to calculate the fusion index.
Statistical Analysis
All results taken from each group were shown as the mean ± SEM. Statistical analyses were performed by one-way ANOVA with Tukey's HSD tests for multiple comparisons post hoc using IBM SPSS Statistics (IBM Corporation, Armonk, NY, USA). The statistical significance value was set at p < 0.05.
Results
Combined Treatment of Losartan and ASCs Increased the Muscle Wet Weight and Area of Myofibers In Vivo
Three weeks after injury, the muscles were removed and then grossly examined. The injured wounds were almost healed in all groups. Grossly, the ASC + Los group had a larger muscle size compared to the Los group (Fig. 1A). The wet muscles of the ASC + Los group weighed more than those of the Los group (p = 0.032) (Fig. 1B).

Gross findings and histopathological evaluation of injured muscles 3 weeks after muscle laceration. (A) The ASC + Los group had a larger muscle size compared to the Los group. (B) Wet muscle weight of the ASC + Los group had a higher value than that of the Los group. (C) Histological findings of the regeneration region. The myofiber area of the Los, ASC, and ASC + Los groups was significantly larger than that of the Con group. Moreover, the Los, ASC, and ASC + Los groups showed an increased number of internal nuclei compared to the Con group (original magnification 400×, scale bar: 50 μm). (D) The ASC transplanted groups (i.e., ASC and ASC + Los groups) had a significantly higher value for the myofiber area than that of the control group. (E) The Los, ASC, and ASC + Los groups had an increased percentage of CN myofibers, compared to the control group. Values represent the mean ± SEM (*p < 0.05).
In the H&E staining, inflammatory cells were quite widely infiltrated in all groups, but this phenomenon is a normal healing reaction. The area of the myofibers for the Los, ASC, and ASC + Los groups was significantly larger than that of the Con group. In addition, the Los, ASC, and ASC + Los groups showed enhanced muscle regeneration compared to the Con group (Fig. 1C). Based on the result of the H&E staining, the area of the myofibers and the percentage of CN myofibers were measured. The ASC transplanted groups (ASC and ASC + Los groups) had a significantly larger area of myofibers than that of the Con group. This was confirmed by the histological findings with the H&E staining (Fig. 1D). The Los, ASC, and ASC + Los groups had increased percentages of CN myofibers, a hallmark of skeletal muscle regeneration, compared to the control group (p = 0.0077, 0.0005, and 0.0002, respectively) (Fig. 1E).
Combined Treatment of Losartan and ASCs Significantly Increased mTOR Signal-Associated Factors
mTOR signaling has been known to regulate muscle growth through the activation of mTOR-complex1 (mTORC1). mTORC1 is composed of mTOR, mammalian lethal with Sec13 protein 8 (mLST8), and raptor (43). qRT-PCR and immunoblotting analysis for mTOR and raptor were done to determine the mechanism responsible for the differences in muscle size, wet weight, and myofiber area between the groups. As shown in Figure 2, the expression of both mTOR and raptor was significantly upregulated in the ASC + Los group compared with the Con group (p = 0.0018 and 0.0151, respectively). The ASC alone group also showed a significant increase in the mRNA expression of mTOR and raptor (p = 0.0138 and 0.0189, respectively), whereas the Los group did not show an increase in mTOR and raptor expression (Fig. 2A, B).
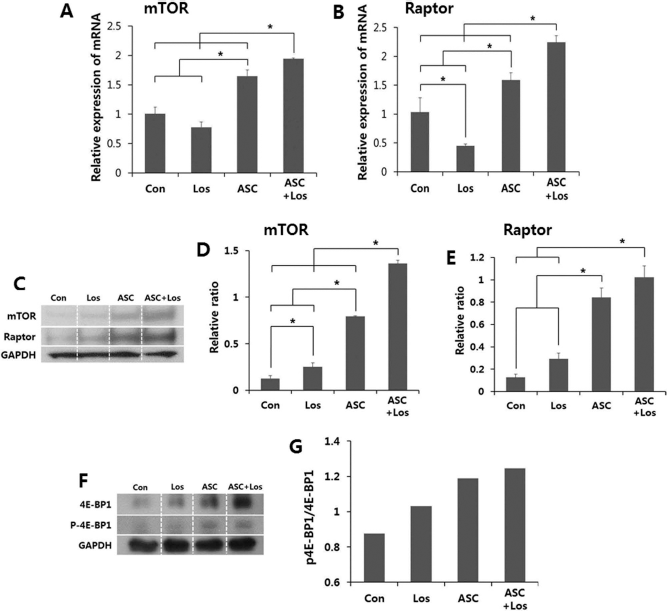
qRT-PCR analysis and immunoblotting analysis for mTOR, raptor, and 4E-BP1 and p-4E-BP1 expression levels in the injured muscles. (A) The relative mRNA expression of mTOR was significantly upregulated in the ASC-transplanted groups (i.e., ASC and ASC + Los groups) compared to the Con group and Los group. (B) The raptor mRNA expression level also was significantly upregulated in the ASC-transplanted groups, and the ASC + Los group also showed a higher expression of raptor than that of the ASC group. (C–E) From the protein expression analysis, the relative expression of mTOR methodically increased in the Los, ASC, and ASC + Los groups with respect to control group. In the case of raptor, the expression level of the ASC and ASC + Los groups was dramatically higher than that of the ASC nontransplanted groups (Con and Los groups). (F, G) In the analysis to demonstrate the activity of mTOR and raptor, the relative ratios of p-4E-BP1/4E-BP1 had similar patterns with the results from the mTOR and raptor expression analyses. Values represent the mean ± SEM (*p < 0.05).
In the protein expression analysis, the relative expression of mTOR methodically increased in the Con, Los, ASC, and ASC + Los groups (Fig. 2C, D). Raptor expression had similar patterns with mTOR expression. A significant difference was seen between the ASC-transplanted groups (ASC and ASC + Los) and the ASC nontransplanted groups (Con and Los) (p = 0.0000008 and 0.0000011, respectively) (Fig. 2C, E).
To analyze the activities of mTOR and raptor, immunoblotting was done for 4E-BP1 and phosphor-4E-BP1 (p-4E-BP1), which are their target factors. The relative ratios of p-4E-BP1/4E-BP1, with similar patterns for that of the mTOR and raptor expression patterns, were methodically increased in the Con, Los, ASC, and ASC + Los groups (Fig. 2F, G).
Pax7 Expression Was Significantly Upregulated by Losartan Treatment
To evaluate the effect of the combined treatment on the self-renewal of skeletal progenitor cells, Pax7 expression was measured by qRT-PCR in vivo. An increase in Pax7 mRNA expression was not detected in the ASC + Los group compared to the Con group. However, the Los group showed an elevated Pax7 expression compared with the other three groups (p = 0.0620, 0.0071, and 0.0002, respectively) (Fig. 3A).
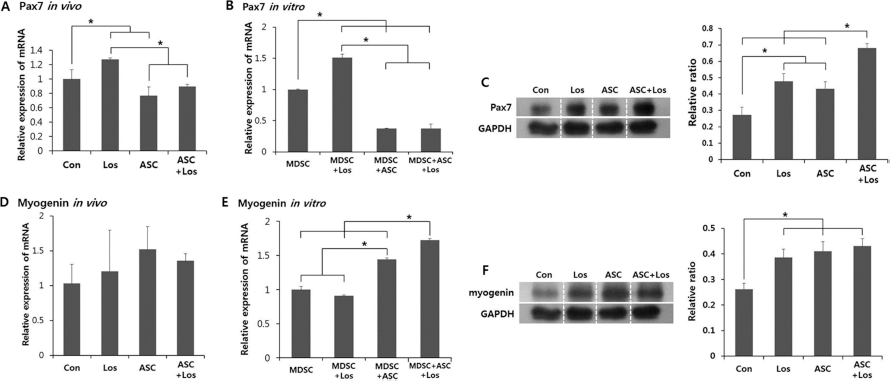
qRT-PCR and/or immunoblotting analysis for Pax7 and myogenin expression levels in injured muscles and cocultured cells. (A) Increase in Pax7 mRNA expression in injured muscle was not detected in the ASC + Los group compared to the Con group. However, the Los group showed elevated Pax7 expression compared with the other three groups. (B) In cocultured cells, Pax7 mRNA in the losartan-treated MDSCs was significantly higher than in the cells without losartan treatment. Losartan did not increase the Pax7 mRNA in the ASCs cocultured with MDSCs. (C) The Los group had significantly increased levels compared to the Con group, and the ASC + Los group had drastically increased levels compared to the ASC group. (D) Although the results did not reach a significant level, the average for the myogenin expression level was higher in the injured muscles of the ASC-transplanted group compared to the Con and Los groups; the ASC group had a higher value than that of the Con group, and the ASC + Los group had a higher value than that of the Los group. (E) In cocultured cells, the relative expression of myogenin calculated in proportion to the number of cultured mdx-derived MDSCs significantly increased in the ASC coculture. (F) In the myogenin protein expression analysis, all three groups (Los, ASC, and ASC + Los groups) showed higher expression than that of the Con group. Values represent the mean ± SEM (*p < 0.05).
To confirm the results of the in vivo experiments, the Pax7 levels were measured by qRT-PCR in cells cocultured and calculated in proportion to the number of cultured mdxderived MDSCs. The Pax7 expression patterns were similar to those observed in the in vivo experiment (Fig. 3B).
Although immunoblotting analysis for Pax7 in the injured muscles had slightly different results, it showed that losartan treatment increased Pax7 protein expression. The Los group had significantly increased levels compared to the Con group (p = 0.0148), and the ASC + Los group had drastically increased levels compared to the ASC group (p = 0.0054) (Fig. 3C).
Transplantation or Coculture of ASCs Increase Myogenin In Vivo and In Vitro
To evaluate the effect of losartan and/or ASC treatment on myogenic differentiation, myogenin expression was quantified by qRT-PCR. Although the results did not reach a significant level, the average of the myogenin expression level was higher in the injured muscles of the ASC-transplanted groups compared to that of the ASC nontransplanted groups; the ASC group had a higher value than that of the Con group, and the ASC + Los group had a higher value than that of the Los group (Fig. 3D).
A similar result pattern was obtained in our in vitro experiments. The myogenin relative expression calculated in proportion to the number of cultured mdx-derived MDSCs significantly increased in the ASC (p = 0.0212 and 0.0016, respectively) (Fig. 3E).
For the immunoblots of myogenin in injured muscle, all three treatment groups (Los, ASC, ASC + Los groups) were significantly upregulated compared to the Con group (p = 0.0092, 0.0060, and 0.0007, respectively) (Fig. 3F).
Combination of Losartan and ASCs Promotes Downregulation of Serum TGF-β Level
ELISA tests were used to evaluate serum TGF-β1 levels. The serum TGF-β1 level significantly decreased in all three treatment groups (Los, ASC, ASC + Los) compared to the Con group. The losartan and ASC cotreated group (ASC + Los group) had a dramatically decreased TGF-β1 level compared with the other groups, that is, the Con, Los, and ASC groups (p = 0.0000002, 0.00009, and 0.0016, respectively) (Fig. 4A).

Evaluation of serum TGF-β levels and muscle fibrosis. (A) The serum TGF-β level was measured with an ELISA kit. All three groups (Los, ASC, and ASC + Los groups) showed significantly decreased serum TGF-β levels, especially the ASC + Los group. (B) H&E and Masson's trichrome staining showed that muscle fibrosis induced by injury was reduced in all groups, especially in the ASC and ASC + Los groups compared to the Con group (original magnification 200× and 400×, scale bar: 100 and 50 μam). (C) The mRNA of collagen type I was quantified by qRT-PCR. The mRNA expression of collagen type I was dramatically decreased by treatment with losartan and/or ASCs. Values represent the mean ± SEM (*p < 0.05).
Combined Treatment with Losartan and ASCs Dramatically Ameliorated Muscle Fibrosis
The H&E and MT staining clearly showed large decreases in fibrosis in the injured muscle of all three treatment groups compared to that of the Con group (Fig. 4B). The Los, ASC, and ASC + Los groups showed dramatic decreases in the mRNA expression levels of collagen type I compared to the control group (p = 0.000047, 0.000002, and 0.000005, respectively). The lowest expression of mRNA for collagen type I was observed in the ASC and ASC + Los groups (Fig. 4C). These results had a similar pattern to the one observed with the serum TGF-β1 levels (Fig. 4A).
ASCs Restored Dystrophin Expression In Vivo and In Vitro
To confirm whether differentiated GFP-positive myotubes could express dystrophin in vivo, immunofluorescent staining of the injured muscle samples was done with an antibody against dystrophin. While the expressions of both dystrophin and GFP were not observed in the Con group, GFP-positive myofibers in the ASC-transplanted group expressed dystrophin (Fig. 5A).
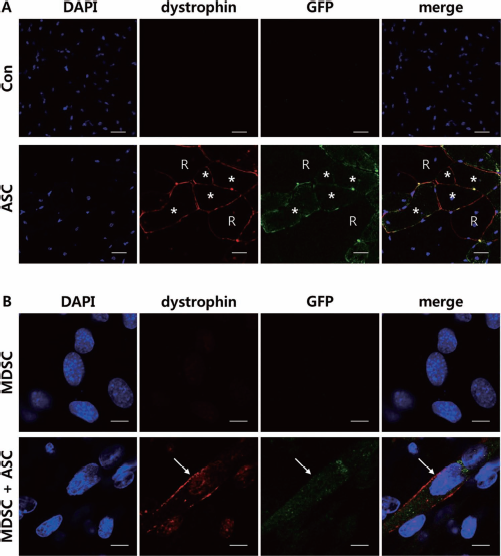
Dystrophin expression in vivo (A) and in vitro (B). (A) While both dystrophin and GFP expression were not observed in the Con group, GFP-positive myofibers (white stars) in the ASC-transplanted groups expressed dystrophin. The presence of revertant dystrophin-expressing myofibers, which do not express GFP, is marked with R (original magnification 400×, scale bar: 20 μm). (B) While mdx-derived MDSCs did not express dystrophin, GFP-positive cells differentiated into myotubes expressing dystrophin (white arrows) (scale bar: 10 μm).
To confirm the results of the in vivo experiments, the cells cocultured for 15 days were fixed, and immunofluorescent staining was done with an antibody against dystrophin. While mdx-derived MDSCs did not express dystrophin, the GFP-positive cells differentiated into myotubes expressing dystrophin (Fig. 5B).
ASCs Differentiated Myotubes Expressing Troponin I In Vitro and Promoting Myogenic Fusion
The isolated MDSCs from the mdx mice and the ASCs from the GFP-positive mice were cocultured. To confirm whether the GFP-positive ASCs could differentiate into muscle fibers, the cells, cocultured for 15 days, were fixed and immunofluorescent stained with an antibody against troponin I, a marker of myogenic differentiation, and visualized with confocal microscopy. Of course, the mdxderived MDSCs differentiated into troponin I-expressing myotubes. Interestingly, the GFP-positive ASC-derived myotubes double stained for both GFP and troponin I. These double-stained myotubes were multinucleated (Fig. 6A). When ASCs were cultured alone, these cells did not differentiate into myotubes (data not shown).

Troponin I expression and fusion index in vitro. (A) Mdx-derived MDSCs differentiated into troponin I-expressing myotubes. Interestingly, GFP-positive ASCs differentiated into myotubes double stained for both GFP and troponin I (white arrows). The double-stained myotubes were multinucleated myotubes (original magnification 400×, scale bar: 20 μam). (B) Coculture with MDSCs and ASCs (MDSCs + ASCs and MDSCs + ASCs + Los) had higher fusion indexes than the single culture of MDSCs (MDSCs and MDSCs + Los). Values represent the mean ± SEM (*p < 0.05).
Cocultured ASCs not only differentiated into myotubes by themselves, but also promoted myogenic fusion. Coculture with MDSCs and ASCs (MDSCs + ASCs and MDSCs + ASCs + Los) had a higher fusion index than a single culture of MDSCs (MDSCs and MDSC + Los) (p = 0.0371 and 0.000007, respectively) (Fig. 6B).
Discussion
We studied the effects of the combined treatment of ASCs and losartan on muscle regeneration in injured mdx mice, an animal model of DMD. Roughly, the ASC + Los group had increased muscle size, and the Los group had decreased muscle size compared to the Con group (Fig. 1A). When the wet weight of the isolated muscles was measured, the measured values also were significantly higher in the ASC + Los group and lower in the Los group than in the Con group (Fig. 1B). Histopathological findings and the measured results from the area of the muscle fibers produced similar patterns as the gross findings and as the measured results of the muscle wet weights (Fig. 1C, D). To identify the mechanism that increased the size and weight of the muscles and the area of the myofibers, qRT-PCR and immunoblotting were done for mTOR and raptor. The mTOR signaling pathway controls muscle growth through mTOR and FoxO (43). Binding of IGF1 to its receptor leads to the activation of Akt. It inhibits protein degradation by repressing the transcription factors of the FoxO family and stimulates protein synthesis through mTORC1 (28,43). In addition, it has emerged that mTORC1 is a key regulator of skeletal muscle differentiation and remodeling. mTORC1 regulated myoblast differentiation at different stages. mTOR promotes nascent myotube formation in the initial stage of differentiation (16,17) and regulates myotube fusion and maturation in the late stage (34). mTOR forms two different protein complexes: the rapamycin-sensitive mTORC1 when bound to raptor and the rapamycin-insensitive mTORC2 when bound to rictor (43,47). The qRT-PCR and immunoblotting results clearly show that the mTOR and raptor mRNA and protein expression levels were dramatically increased in the ASC transplanted group (e.g., ASC and ASC + Los groups) (Fig. 2A–E). In addition, to show mTOR and raptor activities, immunoblotting for 4E-BP1 and p-4E-BP1 was done. mTORC1-mediated phosphorylation of 4E-BP1 releases the 4E-BP1 from eIF4E, resulting in translation initiation (26). The expression ratio patterns of p-4E-BP1 to 4E-BP1 were similar to the mTOR and raptor expression (Fig. 2F, G). These results suggest that the size and wet weight of the muscles and area of the myofibers in the ASC-transplanted groups increased through mTORC1-mediated phosphorylation of 4E-BP1. mTORC1 integrates growth factor signaling with a variety of signals from nutrients and the energy status of the cell. The activity of the mTOR signaling pathway can be modulated by a variety of factors and pathways acting on different steps, and mTORC1 was activated by the binding of IGF1 to its receptor (40,43). After transplantation, ASCs remained viable at the injured site and secreted growth factors in a continuous and regulated manner in response to the environmental cues, including IGF (2,5,9,14,22–24,37,44,46). We thus concluded that the increased muscle size, wet weight, and area of the myofibers in the ASC-transplanted groups are due to the following mechanism: The transplanted ASCs secrete growth factors; those stimulate the mTOR signaling pathway, which activates the synthesis of protein and muscle hypertrophy.
Pax7 is a specific marker for muscle satellite cells, a muscle progenitor cell. Its mRNA was significantly upregulated in the Los group compared to the other groups (Fig. 3A) and in the cells that were cultured in the presence of losartan (Fig. 3B). Additionally, from the protein expression analysis, Pax7 was upregulated by losartan treatment (Fig. 3C). During the course of muscle regeneration, a subset of myoblasts reenters the quiescent state to replenish the satellite cell pool for the subsequent muscle repairs (7). TGF-β inhibits myoblast reentry into this quiescent state. We suggest that losartan restores the self-renewal of satellite cells, which was inhibited by TGF-β, thus increasing Pax7 expression in the Los group. The lower Pax7 expression in the ASC transplanted groups might be due to the presence of ASCs, while the relatively low expression levels of Pax7 might dilute the mRNA of the ASC-transplanted group. We verified that the muscular differentiated GFP ASCs had quite a lower expression of Pax7 compared to the MDSCs that were differentiated alone (data not shown).
The expression average of myogenin, which is expressed in terminally differentiated myocytes (7), was higher in the injured muscles of the ASC transplanted groups compared to the ASC nontransplanted groups, though these increases did not reach a significant level (Fig. 3D). However, in the immunoblotting analysis of the injured muscle and the cell culture experiment, the relative expression level of myogenin significantly increased in the ASC transplanted groups and ASC cocultured cells (Fig. 3E, F). ASCs remained viable after transplantation and secreted a variety of growth factors and cytokines, which activated the differentiation of satellite cells to myoblasts and from myoblasts to myotubes at the injured site (2,5,9,14,22–24,37,44,46). The myogenic effect of these growth factors and cytokines might increase the number of myogenin-expressing myocytes.
Acute injury of healthy muscles produces rapid and controlled inflammation that removes dead and damaged myofibers and promotes replacement of the injured fibers. However, in conditions of chronic injury, as occurs in muscular dystrophy, chronic inflammatory events result in the excessive accumulation of ECM components, which inhibit myogenic repair and lead to muscles being replaced with fibrotic/scar tissue. The key cytokine responsible for this aberrant regeneration is TGF-β, which activates fibroblasts to produce ECM components and ECM-remodeling factors, including the autocrine production of TGF-β, collagen, fibronectin, serine proteases (such as uPA/plasmin), metalloproteinases, and tissue inhibitor of metalloproteinases (27). TGF-β protein and connective tissue proteins, including hydroxyproline and procollagen, are significantly upregulated in mdx mice compared with the C57BL/6 controls (1,20). The serum TGF-β1 concentration, evaluated by ELISA, was significantly downregulated by losartan treatment; the Los group had a lower level than that of the Con group, and the ASC + Los group had a lower level than that of the ASC group (Fig. 4A). Large decreases in muscle fibrosis were observed with H&E and MT staining in all three treatment groups compared to the control group (Fig. 4B). Significant decreased expression of collagen type I mRNA was observed in the Los, ASC, and ASC + Los groups compared to the Con group (Fig. 4C). This mRNA reduction pattern was similar to the serum TGF-β1 reduction pattern. Moreover, the lowest expression of collagen type I mRNA was observed in the ASC and ASC + Los groups. TGF-β expression, histological fibrosis, and collagen type I expression were lower in the ASC + Los group, indicating better muscle regeneration in that group.
Dystrophin immunofluorescent staining was done to verify whether differentiated GFP-positive myotubes expressed this protein. While the myofibers of the control mdx group in vivo and mdx-derived MDSCs in vitro did not express dystrophin, the GFP-positive cells differentiated into myotubes in vitro, and myofibers in vivo expressed dystrophin (Fig. 5A, B). Immunofluorescent staining for troponin I, a marker of myogenic differentiation, was used to verify that the GFP-positive ASCs did differentiate into myotubes in vitro. We indeed observed that multinucleated myotubes were derived from ASCs, which were doubly fluorescent for GFP and troponin I (Fig. 6A). We also confirmed that GFP-positive ASCs differentiated into mature multinucleated myotubes, which expressed both dystrophin and troponin I.
Coculture with MDSCs and ASCs showed increased multinucleated myotubes compared to MDSCs cultured alone (Fig. 6B). These results were commensurate with the results from the myogenin expression analysis. In addition, cocultured ASCs not only differentiated into myotubes, but also seemed to promote myogenic differentiation. Park et al. reported that mTOR signaling is required for the late-stage fusion process during skeletal myotube maturation (34). In our study, mTOR signaling-associated factors (mTOR, raptor, and p-4E-BP1/4E-BP1) were upregulated by ASCs. We suggest that the myogenic differentiation is promoted by ASCs through mTOR signaling.
Because we used differentiation medium that can only differentiate myogenic cells (e.g., MDSCs) into myotubes, when ASCs were cultured alone these cells did not form myotubes (data not shown). It was only when ASCs were cocultured with MDSCs that they differentiated and interacted with the MDSCs to form myotubes. We suggest that the cocultured ASCs and MDSCs formed proper muscle niches, which caused ASCs to differentiate into myotubes or fuse with MDSCs. This might be critical in the therapeutic potential for the treatment of muscle dystrophy with ASCs in vivo. When ASCs are transplanted into injured muscles or dystrophic muscles, endogenous MDSCs interact with the transplanted ASCs and make muscle niches. In this environment, ASCs differentiate into muscle to contribute to muscle regeneration.
In muscular dystrophies, frequent damage to myofibers leads to chronic inflammation mediated by TGF-β, resulting in an excessive accumulation of ECM components, which inhibit myogenic repair and lead to muscle fiber replacement by fibrotic/scar tissue (27). Our experimental results show that losartan downregulated TGF-β, inhibited muscle fibrosis, and restored the self-renewal of satellite cells. Moreover, ASCs transplanted into the injured muscles of mdx mice directly differentiated in myofibers and secreted a variety of growth factors, indirectly inducing muscle hypertrophy and muscle differentiation of satellite cells. To summarize, we conclude that the combined treatment of losartan and ASCs effectively improves muscle regeneration in injured mdx mice (Fig. 7).
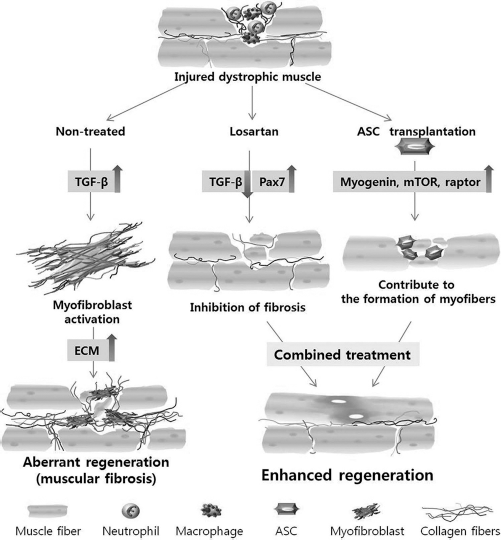
Scheme for the mechanism of therapeutic effects of ASCs and losartan in injured dystrophic muscles. Losartan downregulated TGF-β and inhibited muscle fibrosis. ASCs transplanted into the injured muscles of mdx mice directly differentiated into myofibers and upregulated myogenin. mTOR and raptor induced muscle hypertrophy and muscle differentiation of satellite cells.
Conclusion
In this study, we showed the therapeutic effects of ASCs and losartan in the injured skeletal muscle of mdx mice. Losartan downregulated TGF-β and inhibited muscle fibrosis. ASCs transplanted into the injured muscles of mdx mice directly differentiated into myofibers and upregulated myogenin, mTOR, and raptor, inducing muscle hypertrophy and muscle differentiation of satellite cells. Thus, we suggest that combined treatment of losartan and ASCs could help to improve muscle regeneration in patients with muscle injuries, including DMD patients.
Footnotes
Acknowledgments
This research was supported by the Bio-industry Technology Development Program, Korea Institute of Planning & Evaluation for Technology in Food, Agriculture, Forestry & Fisheries (312062-5) and the Korea Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (A111551). The authors declare no conflicts of interest.
