Abstract
In our previous study, intracerebral implantation of peripheral blood stem cells (PBSCs) improved functional outcome in rats with chronic cerebral infarction. Based on this finding, a randomized, single blind controlled study was conducted in 30 patients [PBSC group (n = 15) and control group (n = 15)] with middle cerebral artery infarction confirmed on a T2-weighted MRI 6 months to 5 years after a stroke. Only subjects with neurological deficits of intermediate severity based on the National Institute of Health Stroke Scale (NIHSS; range: 9–20) that had been stable for at least 3 months were enrolled. Those in the PBSC group received subcutaneous G-CSF injections (15 μg/kg/day) for 5 consecutive days, and then stereotaxic implantation of 3–8 × 106 CD34+ immunosorted PBSCs. All 30 patients completed the 12-month follow-up. No serious adverse events were noted during study period. Improvements in stroke scales (NIHSS, ESS, and EMS) and functional outcomes (mRS) from baseline to the end of the 12-month follow-up period were significantly greater in the PBSC than the control group. The fiber numbers asymmetry (FNA) scores based on diffusion tensor image (DTI) tractography were reduced in every PBSC-treated subject, but not in the control group. Reduction in the FNA scores correlated well with the improvement in NIHSS. Furthermore, a positive motor-evoked potential (MEP) response by transcranial magnetic stimulation (TMS) appeared in 9 of the 15 subjects in the PBSC group. This phase II study demonstrated that implantation of autologous CD34+ PBSC was safe, feasible, and effective in improving functional outcome.
Introduction
Little advance has been made to restore neurological function following ischemic stroke, and thrombolytic therapy continues to be the only proven therapy that improves functional outcomes despite its short therapeutic window (31). Given that neuronal death is a primary mechanism that limits functional recovery, stem cell therapy has been proposed as a potentially effective “regenerative” approach (23). However, though implantation of embryonic grafts has shown promising benefits, as shown in previous clinical trials (4), it was hampered by logistical and ethical considerations. Although adult human bone marrow and human umbilical cord blood (hUCB) contain mesenchymal stem cells (MSCs) and hematopoietic stem cells (HSCs) that have the potential to repair brain injuries (through intracerebral or intravenous route) (6), the complex culturing conditions for in vitro expansion of MSCs, as well as the required human leukocyte antigen (HLA) matching for hUCB implantation between donor and recipient, may limit the clinical application of these cell types. Comparatively, cell therapy using autologous stem cells without in vitro expansion is both technically less demanding and clinically more feasible for restoring neurological function after stroke.
Persistent proliferation of resident neural stem cells (NSCs) in the striatal wall of the lateral ventricle suggests their potential utility in restoring lost neural cell populations after brain injury. Many studies have described neuronal recruitment from endogenous NSCs into target sites in neurodegenerative diseases such as stroke (3,29), Huntington's disease (8), and Alzheimer's disease (17). Taken together, these studies have suggested the potential for recruiting new neurons from resident NSCs as a therapeutic strategy. Though several groups have used chemotaxic or trophic factors, delivered into the brain either by protein infusion or viral vectors (1,5) to mobilize endogenous NSCs, these therapeutic methods are still clinically unapplicable. Importantly, exogenous transplanted stem cells could persistently secrete trophic factors and chemotaxic factors, such as brain-derived neurotrophic factor (BDNF), stromal cell-derived factor 1 (SDF-1), and vascular endothelial growth factor (VEGF). We found in our previous preclinical animal study (33) that these factors stimulate endogenous NSC proliferation and angiogenesis (30,37).
Peripheral blood hematopoietic stem cells (PBSCs) are increasingly used as an autologous source for cell therapies, including those for the regeneration of non-hematopoietic tissues including skeletal muscle (11), heart (26), and neurons (35). Notably, the yield of PBSCs may be amplified by the use of granulocyte-colonystimulating factor (G-CSF), which mobilizes HSCs from the bone marrow to the peripheral blood (9). In order to prove the hypothesis that stereotaxic transplantation of PBSCs might induce the resident NSCs to repair the damaged corticospinal tract (CST), we aimed to evaluate the safety, feasibility, and efficacy of PBSC implantation in improving functional outcomes in stroke patients with stable, neurological deficits. Based on promising results in a preclinical animal study (33) and human phase I studies, we anticipated autologous PBSCs to be a viable source for cell therapy that could facilitate functional recovery after ischemic stroke.
Materials and Methods
Study Design and Patients
This study was a randomized, single blind controlled phase II clinical trial. Patients' ages were between 35 and 75, with ischemic stroke in the middle cerebral artery (MCA) territory, confirmed by magnetic resonance imaging (MRI). Patients with an onset of between 6 months to 5 years before enrollment were eligible for this trial and were randomized into the PBSC or the control group with a sample size of 15 patients in each group. The qualifying NIHSS was set at 9–20 with motor deficits that had to be stable for at least 3 months prior to enrollment. Patients with confounding disorders such as cancer, infection, increased bleeding tendency, leukopenia, thrombocytopenia, hepatic/ renal dysfunction, or pregnancy were excluded.
This trial was approved by the Institutional Review Board (IRB) of the China Medical University Hospital (Taichung, Taiwan) and registered at the ClinicalTrials. gov site (#NCT00950521). Written informed consent was obtained from each patient prior to enrollment.
Initial assessment upon enrollment included medical history and current medications, physical and neurological examination, a stroke symptom questionnaire, a neurological deficit examination based on National Institutes of Health Stroke Scale (NIHSS), European Stroke Scale (ESS), ESS Motor Subscale (EMS) (10), and functional status based on modified Rankin Scale (mRS) (7). Blood samples for checking the complete blood counts (CBCs), biochemistry, and serum cytokine levels were also taken for all registered patients and examined in the Department of Laboratory Medicine at China Medical University Hospital. A brain MRI was conducted to confirm prior ischemic stroke in the MCA territory.
Procedures
The trial protocol is summarized in a scheme in Figure 1. After baseline data had been documented to qualify for enrollment (153 patients were assessed for eligibility), 30 patients were assigned randomly (1:1) via SAS software (Cary, NC, USA) to either the PBSC or the control groups. Current medications such as antiplatelet or antihypertensive medications and rehabilitation programs were allowed in both the PBSC and the control groups. Those in the PBSC group were given 15 μg/kg G-CSF (FilgrastimR, Kirin, Tokyo, Japan) subcutaneously for five consecutive days (36) with CBCs checked daily and NIHSS, ESS, EMS, mRS recorded. In the control group, they only received the former medications (antiplatelet and antihypertensive drugs) and maintained the original rehabilitation programs without any G-CSF treatment and cells transplantation. They all agreed to be recruited into the control group.
In the PBSC group, an 18-gauge double-lumen catheter was placed into the femoral vein for leukapheresis (Cobe Spectra, Terumo BCT, Lakewood, CO, USA) to collect white blood cells (WBCs) the morning after the last G-CSF administration. The whole procedure of cell sorting was performed under the manufacturer's instruction (Miltenyi, Bergisch Gladbach, Germany). In brief, aphresed leukocytes (60 ml) were mixed gently with the cell sorting kit containing anti-cluster of differentiation 34 (CD34) antibody conjugated with nano-iron beads (CD34-nano-iron complex; Miltenyi) for 30 min in a certified Good Tissue Practice (GTP) laboratory. After washing procedures, CD34+ cells were immunosorted from aphresed WBCs labeled with CD34–nano-iron complex using a cell separator (CliniMACS, Miltenyi) per the manufacturer's protocol and then resuspended in 750 μl normal saline for transplantation. Percent of CD34+ cells in each preparation was estimated by flow cytometry with a FACScalibur flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA) using an anti-human CD34 antibody (Becton Dickinson). The supramagnetic nano-iron beads (50-nm dextran iron bead, Miltenyi) for CD34+ cell sorting have been approved as a magnetic cell label for in vivo cell visualization under MRI (16).

Study protocol. NIHSS, National Institute of Health Stroke Scale; ESS, European Stroke Scale; EMS, ESS-Motor Subscale; mRS, modified Rankin Scale; MRI-DTI, magnetic resonance imaging-diffusion tensor imaging; TMS-MEP, transcranial magnetic stimulation-motor-evoked potential; PBSC, peripheral blood stem cell; G-CSF, granulocyte-colony-stimulating factor; CD34+, cluster of differentiation 34-positive.
Before PBSC implantation, a stereotaxic frame (LEKSELL model G, Elekta, Sunnyvale, CA, USA) for targeting in the brain with MRI-guided computerized software (BrainLab, Westchester, IL, USA) was placed on the patients' head under local anesthesia and mild sedation. After a burr hole was created, a 1.5-mm inner diameter needle (LEKSELL Sterotaxic System) equipped with Hamilton syringe (Reno, N V, USA) was stereotaxically inserted down to the deepest target point as the first implantation site. The second and third implants were delivered as the needle was withdrawn along the same trajectory at two different depths, appropriately spaced adjacent to the infarct. The second and third trajectories with three implants each were completed in a similar manner in sequence. These three injection targets were principally set around the infarction cavity rim (5 mm to 10 mm beneath the rim) from the peripheral subcortical brain parenchyma to the deeper site along the course of the estimated corticospinal tract (CST). Based on our previous preclinical animal study (33) and two clinical trials (18,19), we implanted a total of about 3–8 × 106 CD34-immunosorted cells (in 750 μl normal saline) stereotaxically for each patient of the PBSC group. Following the operations, MRIs were performed to trace the presence of PBSCs labeled with supermagnetic nanoiron particles. The subjects were kept for 1 day for close observation and then discharged.
Patients in the PBSC and control groups were followed every 1–3 months in the clinic for 12 months. An independent safety committee monitored the results of the trial including frequency of adverse events (AEs). The primary end points (NIHSS, ESS, EMS, and mRS) were determined at baseline and at 6 and 12 months after PBSC implantation. Investigators responsible for rating NIHSS had to be certified by the Taiwan Stroke Society. In this trial, all clinical information and trial data from each patient were assessed by the data recorders and clinician raters in a singly blinded fashion. In order to avoid the subjective assessment by the recorders or the clinicians, each patient took a hat to mask the surgical scar in their skull. In addition, diffusion tensor imaging (DTI) and motor-evoked potentials (MEPs) induced by transcranial magnetic stimulation (TMS) were performed as secondary endpoints during each visit. The study protocol of this trial is summarized in Figure 1.
The predefined primary endpoints were improvements in neurological deficits based on NIHSS, ESS, EMS, and mRS from baseline to the end of the 12-month follow-up. Secondary endpoints were the extent of reduction in fiber number asymmetry (FNA) scores (22) on MRI-DTI, and restoration of MEP response [“MEP(+)”] (21) on TMS-MEP.
Magnetic Resonance Imaging Applying DTI and T2-STAR (MRI-T2*)
MRI including axial T2-weighted (T2WI), T2-star (T2*), and DTI tractography was conducted using a 3.0 Tesla system (General Electric, Wauwatosa, WI, USA). The MRI protocols were as previously described with modifications (22). In brief, the patients' heads were placed in a human birdcage head coil with a 15-cm outer diameter. The acquisition parameters were repetition time/echo time (TR/TE), 6795/72 ms; field of view (FOV), 23 × 23 cm; and matrix size, 112 × 112. DTI data were acquired by using single-shot echo planar imaging (EPI) sequences with the sensitivity encoding (SENSE) parallel imaging scheme. In DTI data processing, diffusion-encoded fractional anisotropy (FA)-weighted images were elaborated by a calculation scheme as previously described (22). In brief, regions of interest (ROI) in the posterior limb of the internal capsule (PLIC) were first drawn on an axial slice of color-coded FA images. The second ROI was plotted at a pontine level below the superior cerebellar peduncles. A third ROI consisted of the precentral gyrus and its underlying white matter. We then counted the total number of fibers for each tract of the PBSC and control group after rebuilding the fiber tracts through outlining these ROIs. According to neuroanatomic and DTI-derived atlases (40), DTI data were obtained by placing ROIs on the entire CST area at the level of the rostral pons on axial sections (left and right sides) on the basis of the T2-weighted image using the image display software (NordicNeuroLab, Bergen, Norway). An analytic equation [(valuenoninfarct – valueinfarct)/(valuenoninfarct + valueinfarct)] for measuring the fiber numbers was used to calculate FNA scores (22). The final scores ranged from −1 to 1 with positive values showing a reduced fiber count in the infarct hemisphere and a value of 0 indicating an equal fiber number between the two hemispheres.
Transcranial Magnetic Stimulation and Motor-Evoked Potential
The neurophysiological basis for assessing the extent of recovery of the CST system applied qualitative and quantitative assessment of MEPs elicited by TMS as previously described (38). TMS was performed using a figure-of-eight-shaped coil of an outer winding diameter of 70 mm connected with a Magstim 200 magnetic stimulator (Magstim, Carmarthenshire, UK). Threshold was defined as the stimulus intensity required to produce an MEP of at least 100 μV amplitude in 5 of 10 consecutive trials. For the stimulation of the lesioned hemisphere, a maximum of 100% output intensity was delivered if no evoked potential wave (EPW) can be provoked by the three intensity levels set above threshold or if threshold could not be determined appropriately. Each patient was examined at three time points: baseline, 6, and 12 months after PBSC implantation to examine the possible reorganization of the CST after treatment. Reproducible motor responses with minimal peak-to-peak amplitude of 200 μV in at least 5 of 10 consecutive trials were defined as a positive MEP response (MEP+).
Statistical Analysis
We estimated the real differences of clinical stroke scale score including the NIHSS, ESS, EMS, and mRS between two groups at each time point, including the baseline, at 6 months, and 12 months. The sample size (15 treated patients and 15 control patients) was decided in consultation with the Taiwan Department of Health (DOH). Wilcoxon two-sample test was used to assess the differences between the PBSC and control group. A chi-square test was used to assess the categories for clinical characteristics between the PBSC and control group. The trend test and interaction between each time point and group for each clinical stroke scale score at all three time points was performed using generalized estimating equation (GEE) model after controlling for age and sex. A simple linear correlation analysis was used to investigate the relationship between FNA scores in DTI of MRI and NIHSS scores relative to baseline. The correlations between these variables were determined by the Pearson correlation coefficient. The differences of FNA scores at three time points (at baseline, 6 months, and 12 months) in PBSC or control group was performed with Bonferroni-corrected one-way ANOVA. Statistical significance was assumed with a value of p < 0.05. All analyses were done with SAS version 9.0.
Results
Patients' Characteristics
The demographics and clinical characteristics were comparable between groups (Table 1).
PBSC Implantation
In the PBSC group, 3–8 × 106 CD34+ cells (6.6 ± 1.8 × 106) immunosorted from the aphresed leukocytes by CliniMACS were implanted through three trajectories into the peri-infarcted area (Fig. 2A, B). The purity of CD34 immunoreactivity examined by flow cytometry was from 87% to 97% (91.5 ± 4.5%) after CD34+ immunoselection (Fig. 2A, B). In MRI-T2*, the cells implanted were clearly visible 24 h after grafting as hypointense areas at three injection sites in each PBSC-treated patient (Fig. 2A, B). The hypointense signal induced by the supramagnetic iron bound to the cell surface persisted for more than 1 month around the infarcted area and were then gradually quenched (Fig. 2C).
Safety
Safety results were assessed during the whole course of the study. No serious adverse events (AEs) related to PBSC implantation were noted during the follow-up period. Subjective complaints of headache, bone pain, and the other symptoms during G-CSF treatment were symptomatically relieved and resolved after G-CSF injection. In summary, there were no deaths, serious AEs, or other unfavorable symptoms in the follow-up period after G-CSF treatment in the PBSC group or the control group.
Clinical Outcomes
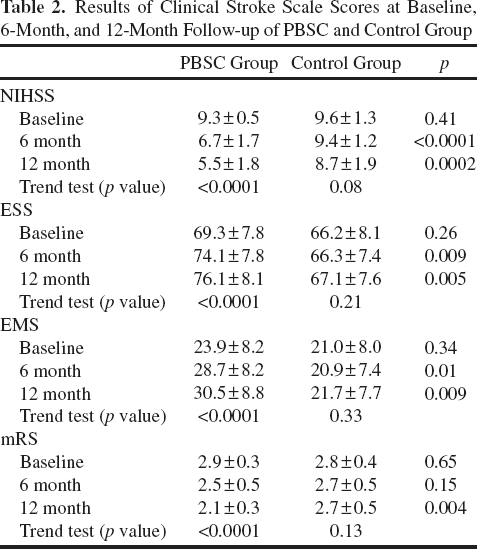
Changes in NIHSS, ESS, EMS, and mRS from baseline to 12 months were analyzed and compared between the PBSC and the control group (Table 2). The PBSC-treated group showed a significant time-dependent improvement in NIHSS ESS, and EMS scores from baseline to the 6-month mark and the 12-month mark, compared to the control group (Table 2). In addition, the other stroke outcome measure of mRS showed improvement (from mRS grade 2.9 ± 0.3 to grade 2.1 ± 0.3) in PBSC-treated patients (Table 2). However, little change of mRS (from mRS grade 2.8 ± 0.4 to grade 2.7 ± 0.5) was detected in the control group (Table 2). Lastly, in the PBSC group, the NIHSS and the mRS decreased, and the EMS and the ESS increased over the 12-month trial period, suggesting that the patients receiving the treatments recovered promptly over time (Table 2, trend test, p < 0.0001).
Neuroradiological assessment by MRI showed subcortical infarction or cortical combined with subcortical infarction over middle cerebral artery (MCA) territory, which evenly distributed in the PBSC group (seven subcortical infarction and eight cortical with subcortical infarction) and control group (nine subcortical infarction and six cortical with subcortical infarction). Serial MRI examinations revealed no anatomic or structural changes, including cerebral hemorrhage or microbleeds in the brains of patients after PBSC treatment (Fig. 2A, B). In MRI-DTI, a prominent increase in the number of CSTs was found even in patients with large infarction sizes after PBSC treatment (Fig. 3A). There were significant differences between the FNA scores for the CST at 6 months and 12 months in the PBSC-treated group (p < 0.01) compared to the control group (Fig. 3A, B, D–G). Semiquantitatively, FNA scores for the CST in patients with PBSC treatment were lower in the 6-month follow-up and 12-month follow-up than that at the baseline (Fig. 3B). In contrast, no significant differences were found among the FNA scores in the control group (Fig. 3B). In order to correlate improvements in clinical stroke scale scores with lower FNA scores, a simple linear correlation analysis was calculated from the data derived from the PBSC-treated group. The percentage change of the NIHSS measured at 12 months after implantation strongly correlated with the FNA scores in the PBSC-treated group (Fig. 3C; correlation coefficient: r = −0.96; p < 0.001), but not in the control group.
Clinical Characteristics of Recruited Patients in Pretreatment Stage (Mean ± SD)

PBSC, peripheral blood stem cell; NIHSS, National Institutes of Health Stroke Scale; ESS, European Stroke Scale; EMS, ESS-Motor Subscale; mRS, modified Rankin Scale.

Representative figure of MRI-T2* image. MRI-T2* tracking of PBSCs after transplantation. (A, B) Summarized characteristics of patients in PBSC group. (C) The hypointense signal (arrows) in the peri-infarct area 1, 14, and 28 days after implantation indicating location of PBSCs labeled with nano-iron particles. S/S, symptoms and signs; MRI-T2*, T2* image in MRI; No, number; Lt, left limbs; Rt, right limbs; y, years; IC, intracerebral.
Results of Clinical Stroke Scale Scores at Baseline, 6-Month, and 12-Month Follow-up of PBSC and Control Group
In TMS-MEP analysis, a positive MEP response (MEP+) was found to be significantly associated with a better recovery in the PBSC-treated patients, compared to the corresponding negative responses from the control patients (Fig. 4A). Nine of the 15 PBSC-treated patients had an MEP+ response after the 12-month follow-up (Fig. 4B, C). Conversely, none of the control patients demonstrated an MEP+ response (Fig. 4D, E).
Discussion
Our study demonstrated that the therapeutic strategy using stereotaxic autologous PBSC implantation was feasible and safe in elderly ischemic stroke patients within 5 years from the onset of symptoms. Review of the laboratory data revealed no consistent or clinically significant changes in hematology, biochemistry, or urinalysis. Further, this study also provided important evidence on the efficacy of PBSC implantation in improving stroke-related motor deficits. After 12-month follow-up, significant improvements in stroke severity scores of the NIHSS, ESS, EMS, and mRS were found in PBSC-transplanted patients compared to the control group. Neuroimaging investigation of MRI-DTI revealed lower FNA scores of CST in the PBSC-treated subjects than the control group. In the functional analysis of CST integrity evaluated by TMS-MEP, an MEP+ response after TMS was noted only in PBSC-treated subjects. All measurements were consistent with identifying a trend toward improved neurological and functional recovery in the PBSC-treated group.
In previous clinical trials, autologous MSCs and allogenic human neuronal cells (NT2/D1 human precursor cell line, Layton Bioscience, Sunnyvale, CA, USA) have been demonstrated to improve the motor deficits in the stroke patients (18–20). According to the successful clinical trial (18,19), they injected 5–10 million cells stereotaxically, which is similar to our cell number of about 3–8 × 106 CD34-immunosorted cells for each patient of the PBSC group. In this study, in line with mounting evidence of neural repair after adult stem cells treatment, stereotaxic injection of autologous stem cells into stroke patients resulted in the reconstruction of injured CST and the rebuilding of the electrophysiological activity from the brain to the limbs. Given that G-CSF may be neuroprotective, improvement due to the G-CSF/PBSC cocktail may, in part, come from G-CSF-mediated neuroprotection, as shown in a previous study (34). Nevertheless, some clinical trial results seemed to conclude that no significant efficacy of G-CSF treatment was detected in improving either subacute or chronic stroke-related neurological deficits (13,36). This indicated that if the neurological improvement has been found in the PBSC group, the therapeutic effect might be contributed mainly from PBSC transplantation. Taken together, we provide here the first evidence that intracerebral implantation of autologous stem cells could improve the motor function of hemiplegic limbs in stroke patients.
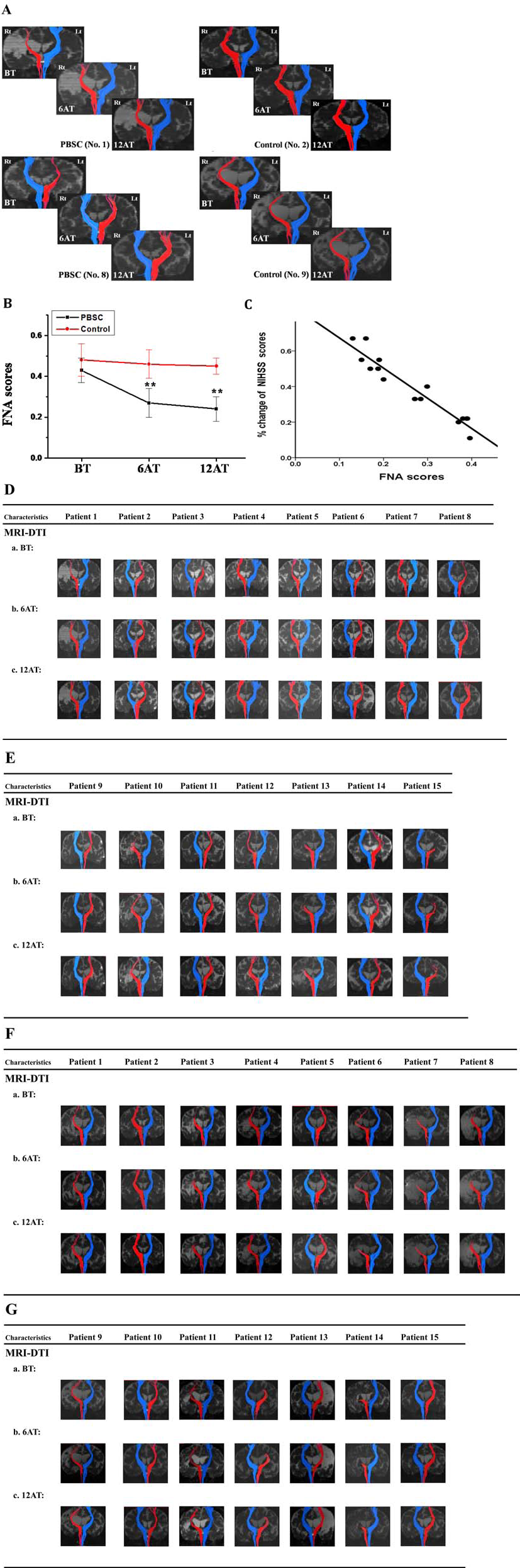
Representative temporal changes of tractography in MRI-DTI. (A) Representative CST tractography of infarcted side (red) or normal side (blue) in MRI-DTI [PBSCs group (case No. 1 and 8), left panel; control group (case No. 2 and 9), right panel] showed red-colored fibers or blue-colored fibers, respectively. (B) Changes in fiber numbers asymmetry (FNA) scores in the PBSC and control groups among baseline, 6 months, and 12 months. Values are mean ± SD, **p < 0.01 versus control. (C) At 12 months after implantation, FNA scores showed a strong correlation with percent change of NIHSS scores in the PBSC-treated group (r = −0.96, p < 0.001). (D–G) Summarized temporal changes of MRI-DTI in PBSC group (D, E) and control group (F, G). MRI-DTI, diffusion tensor image of MRI; Rt, right; Lt, left, BT, before implantation; 6AT, 6 months after implantation; 12AT, 12 months after implantation.

Representative temporal change of MEP responses in TMS-MEP. (A) A positive MEP response [MEP (+)] was present in the PBSC-treated patient (left panel), but not in the control patient (right panel). (B–E) temporal change of TMS-MEP in PBSC group (B, C) and control group (D, E). TMS-MEP, motor evoked potential by transcranial magnetic stimulation; MEP(+), a positive MEP response; BT, before implantation; 6AT, 6 months after implantation; 12AT, 12 months after implantation.
Exogenous stem cell transplantation stimulates the endogenous NSCs to proliferate (30,37) and induce axonal regeneration (2) in the brains of a rat model of stroke. Further, several studies have shown that MSC transplantation enhances the axonal reconnection and remodeling in the ischemic hemisphere (24,25). Here we demonstrate the improvement of CST integrity, using imaging and neurophysiological techniques to evaluate the therapeutic efficacy of PBSC implantation in chronic stroke patients. Imaging by MRI-DTI tractography provides quantitative measurement of CST fiber tracts and their integrity so that the topographic relationship between lesion location and CST can be evaluated (39). FNA is predictive of motor impairment when all motor tracts, including traceable fibers descending in the posterior pons (so-called “alternate motor fibers”), are examined, and this finding can be compared to predictions from CST (22). Twelve months after PBSC implantation, we found lower FNA scores in the treatment group than in the control group, indicating possible new genesis and/or reconnection of descending motor fibers in the PBSC-treated patients. Taken together, DTI-derived measures of CST might be a valid structural marker of motor improvement. The integrity of all descending motor tracts, not only the corticospinal tract, appears to account for stroke recovery after stem cell implantation.
MEP by TMS is a useful and objective means of assessing the CST functional integrity and thus has a prognostic value in predicting poststroke motor recovery (21). An MEP+ response within 72 h of stroke onset predicts a good chance for much functional recovery (15). Furthermore, recovery of muscle strength in stroke patients correlates strongly with the amplitude of MEP (27). In this study, we used MEP to evaluate the treatment efficacy of PBSC implantation in chronic stroke patients. PBSC-treated patients with neurological functional recovery exhibited a reappearance of MEP+ responses (12). Despite a strong presence of MEP+ responses in many treated patients, it is intriguing to notice that no MEPs could be elicited in 6 of the 15 PBSC-treated patients, despite their functional amelioration. TMS preferentially stimulates the deep tangential fibers of the gray matter, leading to an indirect (trans- synaptic) stimulation of neurons of the corticospinal tract (32). These fibers, in turn, project via monosynaptic pathways to a motor neurons innervating intrinsic hand muscles (28). Thus, any perturbation within this route may interfere with the generation of MEPs. We are uncertain of the true reason(s) for the heterogeneous MEP manifestations in the current treatment group. It is possible that reappearance of a positive MEP response may show later than the 12-month follow-up period, and this warrants long-term surveillance of these patients to answer this question.
In this study, we used the CliniMACS system (Miltenyi) to select CD34+ human peripheral blood progenitor cell concentrates by means of the immunomagnetic method. Although these microbeads applied for sorting specific stem cells have been safely transplanted into the patients with hematological disease (14), we first demonstrated that immunosorted CD34+ PBSCs could not only be applied for treating central nervous system disorders but also could be used as a tracking strategy for monitoring the fate of the grafted stem cells in the brain on MRI. With regard to the tracking time of magnetic-labeled cells, previous investigations have shown that the hypointense spot on T2-weighted images persisted for about 3 to 4 weeks in the implanted rat brain (16). In this clinical trial, we also found that the hypointensity of the grafted cells showed prominently at 24 h after implantation and became smaller gradually in the following 4 weeks. In order to disclose whether the transplanted stem cells were entrapped by infiltrating macrophages, further animal studies will be done to examine the mechanism.
In summary, we demonstrated promising improvements in PBSC-treated patients, as evidenced by the primary trial endpoint of increased scores of clinical stroke scales. In addition, the secondary endpoint of reduction in FNA scores for the CST in MRI-DTI and a positive MEP response of TMS-MEP were also present in the patients with PBSC implantation. Despite this success, it should be noted that this was a preliminary study and, due to the small number of participating patients, any inferences are tentative. In the future, we plan to conduct multicenter, large-scale, double-blinded, placebo-controlled, randomized studies to better evaluate the effect of PBSC implantation in patients suffering from chronic stroke.
Footnotes
Acknowledgments
This article is presented in memory of Dr. Hung Li in the Institute of Molecular Biology, Academia Sinica, Taiwan, who was the former principal investigator of this project. The Taiwan Department of Health Clinical Trial Center of Excellence grant# DOH-102-TD-B-111–004 provided funding required for patient recruitment, data collection, and data analysis. This work was also supported by research grants from Taiwan's National Science Council (NSC97–2314-B-039–036-MY3; NSC100–2314-B-039–002-MY3; NSC100–2321-B-039–006; NSC100–2321-B-039–005; NSC100–2632-B-039–001-MY3; NSC102–2325-B-039–006; NSC101–2321-B-039–001; NSC102–2325-B-039–001). The authors declare no conflicts of interest.
