Abstract
The first microRNA, let-7, and its family were discovered in Caenorhabditis elegans and are functionally conserved from worms to humans in the regulation of embryonic development and stemness. The let-7 family has been shown to have an essential role in stem cell differentiation and tumor-suppressive activity. Deregulating expression of let-7 is commonly reported in many human cancers. Emerging evidence has accumulated and suggests that reestablishment of let-7 in tumor cells is a valuable therapeutic strategy. However, findings reach beyond tumor therapeutics and may impinge on stemness and differentiation of stem cells. In this review, we discuss the role of let-7 in development and differentiation of normal adult stem/progenitor cells and offer a viewpoint of the association between deregulated let-7 expression and tumorigenesis. The regulation of let-7 expression, cancer-relevant let-7 targets, and the application of let-7 are highlighted.
Introduction
The earliest found microRNA (miRNA), lethal-7 (let-7), has been shown to possess an essential role in the development of Caenorhabditis elegans and Drosophila melanogaster (73,78), and its DNA sequence and function is highly conserved across different species. During embryo development, accumulation of let-7 can activate some pluripotent factors, such as LIN28, for determination of differentiation. Current opinions reveal that let-7 controls “stemness” by repressing self-renewal and promoting differentiation in normal development for many kinds of stem cells/progenitors.
Recently, there are a number of studies of let-7 focused on fundamental roles in cell proliferation, reprogramming, development, differentiation, and tumorigenesis [reviewed in (9,71,79)]. Although let-7 is known to be functionally involved in regulating the differentiation of stem cells/progenitors and reprogramming of cell fates, the physiological or biological activities of the let-7 family are still elusive. Recently, growing evidence revealed that let-7 acts as a tumor suppressor by targeting numerous genes. Most of the studies suggest that the level of let-7 expressed in various tumor stem-like cells is lower than that in differentiated counterpart cells. Examining how to regulate and process the expression of let-7 in tumor cells could be valuable in helping us to use let-7 as a tool for cancer treatment. Furthermore, critical discussion about normal adult stem cells and tumor cells should be examined for the further development of let-7-based therapy.
The Role of LET-7 in Normal Stem/Progenitor Cells
Let-7 expression is absent in embryonic stem cells (ESCs) or adult progenitors (17,56), and then increasing levels of functional let-7 upon differentiation appears to be a common scenario (Fig. 1). These properties are also observed in tumor-initiating cells; loss of let-7 switches normal somatic cells into tumor-initiating cells. Such loss might be caused by chromosomal abnormalities, epigenetic alterations, or impairment of the process of let-7 at the posttranscriptional level (7,90). Otherwise, normal stem cells can be transformed directly to cancer-initiating cells under high self-renewing rates without normal surveillance (Fig. 1). Low levels of let-7 expression are a prominent feature in certain stem cells.

Differential level of let-7 expression in normal progenitors and cancer-initiating cells. During the differentiation process, progenitors become more restricted to specific cell lineages. Broken arrows indicate proposed sources of cancer stem cells: loss of lethal-7 (let-7) could transform normal proliferating or differentiated progenitors into dedifferentiated cancer stem cells.
Stem/progenitor cells have unlimited proliferating activities for self-renewal, yet they are capable of differentiating into various cell types (pluripotency) by certain stimuli. However, before stem cell therapies are applied, a full understanding of the timing that schedules multipotent and pluripotent stem/progenitor cells to differentiate would be extremely meritorious. Recent work has demonstrated that the let-7 family members play key roles in controlling the timing of differentiation or reprograming transition of various cells via complex regulatory mechanisms or targets (5,59). Furthermore, the let-7 family has also been shown to govern endogenous activities in various different adult cells (Table 1).
Let-7 Expressed in Somatic and Stem/Progenitor Cells

Let-7, lethal 7; HMGA2, high-mobility group AT-hook 2; LRRK2, leucine-rich repeat kinase 2; DP, differentiation-regulated transcription factor protein; ES, embryonic stem; EB, embryoid body; Bach1, BTB and CNC homology 1, basic leucine zipper transcription factor 1; TGF, transforming growth factor; β-R1, chemokine C-X-C motif ligand 11; IL-6, interleukin 6; HNF4A, hepatocyte nuclear factor 4A; ascl1a, achaete–scute complex homolog 1a; hspd1, heat shock 60-kDA protein 1; oct4, octamer-binding transcription factor 4; pax6b, paired box 6b; SOX-2, sex-determining region Y box 2; N/A, not applicable; Hbf, fetal hemoglobin; CDK6, cyclin-dependent kinase 6; CDC25A, Cell division cycle 25a; Igf2, insulin-like growth factor 2; IMP, Insulin-like growth factor 2 mRNA-binding proteins.
In Drosophila, let-7 determines the appropriate timing for cell cycle exit, metamorphosis, neuromusculature remodeling, juvenile-to-adult-stage transition, and adult behavior (13,87). As found in the Drosophila model of Parkinson's disease, the increasing level of let-7 attenuates the pathogenic leucine-rich repeat kinase 2 (LRRK2) effect by targeting mRNAs of E2F1 and differentiationregulated transcription factor protein (DP) (29). The zebra fish ortholog of let-7 exhibits basal expression in the uninjured retina to suppress the expression of regeneration-associated genes and to block the premature Müller glia dedifferentiation (77). In the adult newt, let-7 controls the transdifferentiation and regeneration of lens and pigment epithelial cells (64,95).
Numerous experiments from mammals also investigated the activity of let-7 in physiological and development function (Table 1). In mice, let-7 has been shown to regulate the neural lineage specificity in ES cells and affect the brain development (17,56,111). Furthermore, epithelial progenitors in mammary glands were detected with no or little levels of let-7, and this suggests that progenitors retain the status of self-renewing and reconstitute the mammary gland by modulation of lin28/let-7 loop (35,108). The expression of the let-7 family triggers the formation of the embryoid body, and differentiation of three germ layers by multiple mechanisms in human (38,98) and mouse (56,93,111) ESCs. The let-7 family has been shown to play an essential role in the generation and development of many adult tissue cells, including adipogenesis in the 3-day transfer, inoculum 3 × 105 cells (3T3)-L1 mouse cell line and adipocytes (50,88), cardiac hypertrophy by cardiomyocytes (20,109), angiogenesis of endothelial cells (15,46), liver development (33,96), manipulation of body size and puberty (33,96), and newborn ovarian development (1,42). In the circulating blood system, let-7 is shown to be involved in not only the developmental control of reticulocytes (49,69) and T-cells (4,89) but also in the increasing sensitivity of Fas-mediated apoptosis in peripheral blood mononuclear cells (100,104).
The regulatory mechanism of let-7 is quite essential for the maintenance of stem/progenitor cell pluripotency as well as for reprogramming somatic cells into induced pluripotent stem cells (Fig. 2). Lin28, sustaining the stemness state of various stem cells, is an important regulator of let-7 and is also downregulated by let-7 during differentiation (18,90). Recently, lin28 and let-7 have been shown to have opposing expression patterns for the reciprocal regulation and functions in development and cell fate transition. Lin28 modulates the production of endogenous primary (pri)-let-7 and precursor (pre)-let-7 transcripts in ESCs. Although the suppressor, lin28, induces inhibitory oligouridylation in ESCs, monouridylation occurs in somatic cells without lin28 expression to promote let-7 biogenesis (30). Lin28 could block the function of either Drosha or Dicer in the process of let-7 and act as a posttranscriptional repressor for the let-7 biogenesis (31,86,101). A recent study showed that another key gene, high mobility group AT-hook 2 (HMGA2), for the self-renewal and the maintenance of stemness in adult stem cells, is highly expressed in hematopoietic, adipotic, and neural progenitors. During differentiation, the increasing levels of let-7 directly target HMGA2 to reduce the expression, following the inhibition of cell proliferation (11,18). Recently, proteins related to the function of cell cycle, cell proliferation, and apoptosis have been shown to be interrupted by let-7. Insulin-like growth factor 2 mRNA-binding proteins (IMP1), an oncofetal gene expressed during early fetal life, regulates proliferation in stem cells and is targeted by let-7 for the blockade of Myc and K-Ras expression (55,60). Genome-wide studies by microarray analysis in cancer cells revealed that let-7 inhibits multiple cell cycle-associated genes, including cell division cycle 25 A (CDC25A); cyclin D1, D3, and A; and cyclin-dependent kinase 4 (CDK4) and 6 (51). Interestingly, the expression of let-7a can suppress the doxorubicin- and paclitaxel-induced apoptosis by targeting caspase-3 in the cancer cell lines, A431 (female epidermoid carcinoma), and HepG2 (male hepatocellular carcinoma) (94). In summary, the let-7 family inhibits many key genes associated with cell cycle regulators, such as CDC25A and CDK6, early expressed embryonic genes including LIN28, HMGA2, mouse Lin41, and IMP-1 and growth and proliferation-related genes including RAS and c-MYC. The effects of let-7 in ES cells has been shown to be an inhibitory effect on the expression of lin28, c-Myc, spalt-like transcription factor 4 (Sall4), and downstream genes of pluripotency factors, in particular, sex-determining region Y box 2 (Sox2), octamer-binding transcription factor 4 (Oct4), and Nanog. Moreover, wild-type ESCs transfected with let-7 are not able to undergo differentiation due to the antagonizing effects of other miRNAs, such as ESC-specific cell cycle-regulating miRNAs (ESCC) in ESCs (16,56).
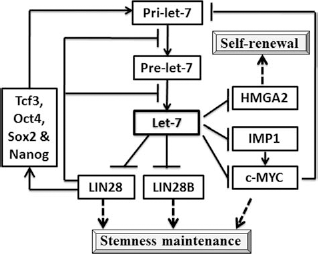
Regulation of let-7 activities on self-renewal and stemness maintenance. HMGA2, high-mobility group AT-hook 2; IMP1, insulin-like growth factor 2 mRNA-binding proteins; Pre, precursor; Pri, primary; oct4, octamer-binding transcription factor 4; Sox2, sex-determining region Y box 2; tcf3, transcription factor 3.
Deregulation of LET-7 in Human Cancers
Expression profiling of miRNA revealed that levels of miRNAs are differentially expressed in many human cancers, suggesting that miRNA profiling has diagnostic and/or prognostic potential. The let-7 family has been known to regulate proliferation, apoptosis, and differentiation in tumor cells and is thus suggested as a potent target for cancer therapies. Controversial expressing levels of let-7 in different cancer cells have been shown and indeed require further examination. Let-7 contains nine family members (let-7a, let-7b, let-7c, let-7d, let-7e, let-7f, let-7g, let-7i, and miR-98) in humans and is highly conserved across animal species in sequence. The transcriptional and posttranscriptional regulation of let-7 is still unclear, and let-7 targeting on multiple genes reveals that the specificity of let-7 still needs to be addressed. Deregulation of let-7 could involve etiological processes by targeting apoptotic or cell cycle-related genes in a tissue or stage-dependent manner. Tumor-associated let-7 family members can be acquired from the PhenomiR database (http://mips.helmholtz-muenchen.de/phenomir/). The PhenomiR database affords information about the differential expression of the let-7 family in human cancers.
Here we have summarized the clinical results describing the expression of let-7 family members from papers studying human cancers (Table 2) and suggest the further investigation of the deregulation of let-7 family in various human cancers. Let-7 is generally believed to be a tumor suppressor. Loss of the let-7 family also has prognostic value, while it indicates poor survival in many human cancers (19,81,103). However, upregulation of certain let-7 family has been demonstrated in several human cancers. The upregulation of let-7a, b, c, d, e, and f was associated with a high grade of leukemia and prostate cancer (19,28,54). The conflicting results of deregulation of let-7 in various cancers suggest that let-7 may have different functions in different diseases or in different cell contexts.
Deregulation of Let-7 in Human Cancers

In human malignant cholangiocytes, increasing levels of let-7a contribute to the survival effects of interleukin (IL)-6 activity and the constitutively increased phosphorylation of signal transducer and activator of transcription 3 (Stat-3) through the mechanism involved in the neurofibromatosis (58). The highly expressed let-7 has been shown to increase the chemoresistance of hepatocellular cancer stem cells through a novel regulatory mechanism of let-7/miR-181s (57). In hepatocellular carcinoma HepG2 and squamous carcinoma A431 cells, let-7a targets caspase-3 for increasing the activity of chemoresistance to apoptosis (94). Let-7b, expressed in diffuse large B-cell lymphomas, epigenetically downregulates the tumor suppressor gene, positive regulatory domain I/B-lymphocyte-induced maturation protein 1 (PRDM1/Blimp-1) (68). In leukemia cells, let-7 isoforms a, b, c, and d are expressed at higher levels than in normal peripheral blood mononuclear cells (100). The let-7 family is selectively secreted into the extracellular environment via exosomes in a metastatic gastric cancer cell line and, thus, may explain its oncogenic characteristics including tumorigenesis and metastasis (70). Further investigation of the role of let-7 and its regulation in tumorigenesis is necessary and urgent for miRNA-based cancer therapy.
As accordant with the physiological function of the let-7 family, miRNA-based cancer gene therapy offers the theoretical theme of targeting multiple genes and interfering with networks controlled by let-7. Reconstitution of the tumor-suppressive activity of let-7 has produced favorable antitumor outcomes in experimental models. Pending subjects need to be discussed and resolved prior to the consideration of let-7-based cancer gene therapy. It contains several issues including the need for the specificity of mRNA targeted by let-7 in various cancers, the incomplete knowledge of the regulatory mechanism and biogenesis of let-7 that affect the therapeutic efficiency, the possibility for nonspecific immune activation, and the lack of a defined, optimal system of delivery. The limitation to tumor-suppressor let-7 therapy is the paucity of let-7-targeted genes known to induce or preserve malignant phenotypes; moreover, more than one genetic alterations are necessary for carcinogenesis. Many differentiated tumors with average or high expression of let-7 are reported. Also, as it is true for many gene therapeutics, the eradication of treated tumors is rare even in experimental systems because of the technical difficulty of transducing sufficiently large proportions of cells in the tumors.
Complexity and Uncertainty of LET-7 Biology and its Regulatory Mechanism
Although restoration of normal let-7 expression provides the possibility for the future cancer therapy, limited knowledge concerning its transcriptional, posttranscriptional, and processing control during biogenesis of let-7 and tumorigenesis makes it difficult to directly apply let-7 as a therapeutic strategy. It is necessary to confirm that the downregulation of let-7 in tumors is a primarily pathogenic factor during tumorigenesis. Supporting the hypothesis for cancer stem cells, many studies convey the opinion that the epigenetic downregulation of let-7 in cancer stem/progenitor cells is common and leads to the upregulation of oncofetal genes (HMGA2, lin28, Ras, Myc, etc.) and, thereby, to the progression of stemness activity and tumorigenesis. Nevertheless, many data have shown a tumor-suppressive role for let-7; high expression of let-7 in tumors is still observed and would be beneficial to survival signals (57,68,100).
Generation and maturation of miRNAs is governed by various regulatory and transport machinery at different stages of primary, precursor, and mature miRNAs. Finally, the miRNA-associated protein-RNA-induced silencing complex (miRISC) interacts with miRNAs and performs endogenous functions (12,14,44). Therefore, genetic and epigenetic defects likely affect both coding and noncoding RNA transcription, contributing independently or in concert with the altered expression of miRNAs. In ESCs, the core transcription factors, Oct4/Sox2/Nanog/transcription factor 3 (Tcf3), promote the transcription of both pri-Let-7g and Lin28. Appropriated expression of pri-Let-7g can be detected, but the mature form of let-7g is missing after the blockade of lin28 (111). In this posttranscriptional regulation, two major factors, Drosha and Dicer, are shown to be mediated by lin28, resulting in the inhibition of let-7 maturation (30,53). It is suggested that the inhibition of maturation process of pri-Let-7 transcripts plays an important role in the maintenance of the pluripotent state. It has also been shown that there is a feedback loop between lin28 and let-7 for regulating pre-let-7 maturation during neural stem cell commitment (74,80). Thus, the negative lin28/let-7 circuit loop has a major influence over ESC fate. Although there are many antitumor effects by use of let-7 for the treatment of cancer cells in various experimental models, let-7 is still involved in a complex and unclear regulatory network of miRNA that governs the self-renewal or differentiation of stem/progenitor cells. Further studies are required.
Current Approach and Delivery System for the LET-7-Based Gene Therapy and Their Limitations
Most recently, many researchers have used a number of strategies to deliver let-7 into different cancer cells in experimental models for the purpose of gene therapy. However, clinically applicable tools for gene delivery are limited and currently are focused on retroviral-based or adenoviral-based vectors. New delivery methods are required for improving the safety and efficacy of let-7 gene-based therapy. The first case of let-7 functioning as a tumor suppressor in vivo was shown to suppress non-small cell lung tumorigenesis (47). Although the sustained expression of let-7 is sufficient to inhibit tumorigenesis, the authors infer that let-7g could be present and active in escaping tumors (47). It is suggested that let-7-resistant tumors might eventually relapse. It is important to understand the effect of long-term expressed let-7 on tumors or other normal cells or the use of let-7 miRNAs as a therapeutic agent. A general drawback for current gene therapy strategies is the lack of specific markers for targeting tumor cells while systematically administrating the viral vector carrying the gene of interest. This is critical for minimizing unexpected side effects. The alternative way for most clinical trials has been to rely on gene delivery directly into accessible tumors.
In addition to viral vector-based gene therapy, synthetic miRNA has also been used in gain-of-function assays. These miRNA mimics are small, chemically modified RNA molecules that mimic endogenous mature miRNA molecules and are commercially available (3). By the use of formulated synthetic miRNA, the therapeutic potential of synthetic miR-34a against human multiple myeloma cells in vitro and in vivo has been successful in preclinical models. MiR-34a-based treatment strategies provide a proof of concept that formulated synthetic miRNA can support a framework for the development of miR-34a-based treatment strategies in patients (22). Since miRNA mimics have no vector-based toxicity, if their delivery agents do not cause side effects over long-term use, they can be a promising therapeutic approach for tumors.
Alternative Ways to Bypass the Disadvantages of LET-7-Based Therapy
Although it is understandable that let-7 plays a critical role in regulating the self-renewal and pluripotency of ESCs, much less is known about the molecular mechanisms for regulating let-7 expression to maintain pluripotency in stem/progenitor cells. It is necessary to perform the systematic examination and study of both functional trans-molecules and cis-regulatory elements involved in the regulation of let-7 expression between the transition of stem/progenitor and differentiated cells. A recent study involving large-scale identification of let-7 promoters in both human and mouse cells was an excellent starting point for characterizing individual miRNA promoters (16,18). Studies in cluster promoters of miRNAs have demonstrated the binding of Sox2, Oct4, and Nanog in the let-7 promoter region. This suggests that the different hierarchical regulation of let-7 via biogenesis, integrated circuits with let-7, and other factors forms a regulatory feedback loop. As noted above, c-Myc has been shown to transcriptionally repress the expression of let-7 in human liver cells (104). Unexpectedly, although mature let-7 family members are depleted in undifferentiated cells, the primary let-7 transcripts and the hairpin precursors can be detected in these cells in vitro, as well as during early developmental stages of mice in vivo (32,101). The accumulation of mature let-7 miRNA thus appears to be regulated, at least in part, posttranscriptionally. Recently, LIN28 and LIN28B, two homologs of the C. elegans heterochronic gene lin28, were shown to inhibit the processing of let-7 family members (101). Additionally, mouse lin41 has been shown to suppress let-7 activity, at least in part, by antagonizing Argonaute 2 (80). Taken together, studies on the expression of let-7 have shown a role in inhibiting the induction of multi-potency in progenitor cells. It will be important to study whether depletion of MYC or LIN28 can substitute for the antisense knockdown of let-7. Evidence for the limitations of let-7 include a study in which forced expression of let-7 (by cotransfection of mature let-7 or use of transgenic let-7 precursors not subject to LIN28 regulation) can interfere with reprogramming of fibroblasts into induced pluripotent stem cells (iPSCs) (105).
LIN28, with Multifunctions, Provides an Alternative Approach to Block LET-7-Associated Tumorigenesis
There is a tight link between inflammation and cancer in epidemiological and clinical aspects. Inappropriate resolution of inflammatory responses often leads to various chronic diseases including cancer (92). In the tumor transformation of breast cells, the transient activation of Src triggers an inflammatory response mediated by nuclear factor of κ light polypeptide gene enhancer in B-cells (NF-κB) that directly activates lin28 transcription and rapidly reduces let-7 levels (36,102). NF-κB is important in regulating the normal innate and adaptive immune responses seen in states of inflammation. However, evidence revealing particular mechanisms by which NF-κB influences cancer initiation, promotion, and progression is fascinating but still vague (25,66). It suggests that the activity of let-7 is obstructed by activation of lin28 through the epigenetic signaling molecules, such as NF-κB. Although let-7 targets lin28 expression to block oncogenetic activity, lin28 is also shown to be in a feedback loop to downregulate the expression of let-7 by interfering with pri- and pre-let-7 processing. Directly elevating let-7 in the treatment of cancer therapy is possibly inefficient since lin28 is continuously expressed under the inflammatory microenvironment of a tumor site. Since the expression of lin28 is reciprocal to the maturation of mature let-7, lin28 is abundant in early developmental stages and declines upon differentiation (18,74).
While using let-7 as a target for cancer therapy, there is still a possibility to impair the replenishment of normal stem/progenitor cells owing to no specific tumor-targeting strategy. Suppression of lin28 may provide an appropriate strategy for further cancer gene therapy. Although lin28 is found to be highly expressed in maintaining the pluripotency of ESCs by the blockade of let-7 expression, lin28 has also been shown to differentially promote and inhibit specific differentiation during neurogliogenesis (90,97). This suggests that lin28 does not function exclusively through blocking the let-7 family. Moreover, in neural stem/progenitor cells, the mechanism for keeping stemness is associated with the regulation of Musashi1 (Msi1) in concert with lin28 (43). This study revealed that Msi1 acts as a novel factor that can influence stem cell maintenance by controlling synergistically with lin28 blockade of let-7 expression.
With all the efforts and advances that continue to be applied to the development of miRNA-mediated therapies, obstacles still remain. One obstacle that should be emphasized is maintaining specificity for the target. miRNA targeting is known to be sequence specific rather than gene specific, and the let-7 family has been shown to target multiple genes. It should be noted that silencing genes by miRNAs require partial complementary binding between miRNAs and protein-coding transcripts (44). Therefore, it is important to evaluate effects of specific miRNA-mediated therapy on a proteome-wide scale to prevent unexpected gene alteration in nontumor cells.
Conclusion and Prospects
In summary, let-7 controls multiple targets to regulate stemness and differentiation as required for proper development and tumor suppression. The identity of these targets, and their physiological relevance, has primarily emerged. Patents for novel cancer therapeutics, using let-7 based therapies have been filed in Australia (2007/333109 A1), US (20090163430), and China (CN102038959).
We are now starting to learn how to regulate the expression of let-7 and to find out which tumor-specific genes are targeted by let-7 for future let-7 family-based therapies. Such investigations will be valuable in helping us to understand the regulation of cell fates, in particular stem/progenitor cell fates. Therefore, it will be necessary to define the transcriptional regulatory networks and cell signaling pathways that distinguish normal stem/progenitor cells from transformed cells. Although studies show a promising therapeutic approach for treating various kinds of cancer by let-7, it should be noted that these studies looked at the impairment of tumor initiation, not remission of preexisting tumors, the latter being much associated with the clinically relevant situation. Other targets such as HMGA2 or lin28 might provide the profound regulation of stemness and differentiation as substitutes for let-7 based cancer therapy.
In the present day, many open questions about let-7 function in normal stemness, development and differentiation of stem/progenitor cells are still unsolved. The mechanisms of let-7 resistance might emerge during the extended treatment of exogenous let-7. Mostly, additional pluripotent factors or inhibitors for substituting the let-7 family are continuously being addressed in cancer therapies.
Footnotes
Acknowledgments
This study was supported by grants from National Science Council (NSC 98-2314-B-039-008-MY2, NSC 102-2314-B-039-022, and NSC 102-2314-B-039-021-MY3) and China Medical University and Hospital [CMU99-N2-01-(1&2), DMR-101-116, and DMR-102-053] awarded to Dr. Shao-Chih Chiu. The authors declare no conflicts of interest.
