Abstract
The present study aimed to evaluate the effect of bone morphogenetic protein 9 (BMP9) and BMP13 on cardiac differentiation of C3H10T1/2 cells in vitro and to characterize the differentiated cells on their ultrastructure and transmembrane electrophysiological features. C3H10T1/2 cells were transfected with the vectors for BMP9 or BMP13 and differentiated into cardiomyocytes in vitro for up to 28 days. The expression of cardiac-specific genes Gata4 and Mef2c and proteins troponin T (cTnT) and connexin 43 (Cx43) was significantly increased in the cells transfected with BMP9 or BMP13 after differentiation over the controls as evaluated using quantitative RT-PCR, Western blotting, and immunofluorescence staining. Transmission electron microscopy and Masson trichrome staining showed that the specific myocardial leap dish and myofilament-like structure were present in the cells overexpressing BMP9 or BMP13, not in the control cells. Whole-cell patch-clamping study demonstrated the presence of delayed rectifier potassium current, inward rectifier potassium current, and T-type calcium current in the cells overexpressing BMP9 or BMP13. Sodium current was detected in a small number of cells overexpressing BMP9, not in the BMP13-transfected cells or the control cells. The expression of Mef2c gene and Cx43 and cTnT proteins was also significantly higher in the cells overexpressing BMP9 than those overexpressing BMP13. Our data indicate that BMP9 and BMP13 (BMP9 might be more effective) promoted the differentiation of C3H10T1/2 cells into cardiomyocyte-like cells with cellular ultrastructures and ion channel currents similar to mature cardiomyocytes in vitro.
Keywords
Introduction
Myocardial injury often leads to impaired cardiac function due to decreased cardiomyocyte number and/or function. Cell transplantation has become an attractive option for cardiac repair and regeneration after injuries. Bone marrow stem cells have been considered as a major source for cell-based therapy (11). C3H10T1/2 cells were originally isolated from C3H mouse embryos and are functionally similar to bone marrow stem cells (47) and exhibit multilineage differentiation capacities both in vitro and in vivo without spontaneous transformation or tumorigenicity (35). C3H10T1/2 cells are able to be induced to differentiate into osteoblasts, chondrocytes, adipocytes, and striated muscle cells (3,40).
Bone morphogenetic proteins (BMPs) are a transforming growth factor-β (TGF-β) superfamily and are critically involved in embryogenesis and morphogenesis of a variety of tissues and organ systems including cardiovascular development (15,27,44,45). It is well known that BMPs, especially BMP2 and BMP4, play an essential role in the differentiation of stem cells into myocardial cells (42,43). BMP9 and BMP13 are part of the TGF-β superfamily and are shown to be extensively involved in the embryonic development and formation of vascular endothelial cells, bone cells, and the basal forebrain cholinergic neurons (2,4,25). However, it is not clear whether BMP9 and BMP13 have the capability of inducing cardiomyocyte differentiation of stem cells.
The present study was designed to evaluate the effect of BMP9 and BMP13 on the differentiation of C3H10T1/ 2 cells into cardiomyocytes in vitro. Special efforts were made to characterize the differentiated cardiomyocytes on their ultrastructure using a transmission electron microscope and membrane electrophysiological features using a whole-cell patch-clamping technique in addition to evaluating the expression of cardiac-specific proteins and genes.
Materials and Methods
Culture and Treatment of C3H10T1/2 Cells
The mouse C3H10T1/2 cells (a gift from the Molecular Oncology Laboratory at the University of Chicago Medical Center, Chicago, IL, USA) were cultured in Dulbecco's modified Eagle medium (DMEM)/high glucose (Thermo, Logan, UT, USA) containing 10% fetal bovine serum (FBS; Thermo) at 37°C in a humidified incubator with 5% CO2. At 70–80% confluence, the cells were subcultured for four passages and then randomly divided into four groups for experiments: BMP9 group, BMP13 group, green fluorescent protein (GFP) group, and control group (without any treatment). Recombinant adenoviruses expressing BMP9, BMP13, and GFP were generated as previously described (a gift from the Molecular Oncology Laboratory at the University of Chicago Medical Center, IL, USA) (14,16). The transfection efficiency of pAdEasy–BMP9, pAdEasy–BMP13, and pAdEasy–GFP in the cells was evaluated with flow cytometry. The cells were then collected for Western blotting, quantitative RT-PCR, immunofluorescence staining, transmission electron microscopic examination, and membrane electrophysiological studies as detailed blow at days 7, 14, 21, and 28 of transfection and differentiation.
Flow Cytometry
After different treatments and culture, the C3H10T1/2 cells were collected in the 2-ml EP tube (Axygen, Union City, CA, USA) after washing with phosphate-buffered saline (PBS; Sigma, Carlsbad, CA, USA). The transfection rates (the rates of cells with GFP, which expresses in pAdEasy–BMP9, pAdEasy–BMP13, and pAdEasy–GFP as a tracer protein) were analyzed using flow cytometry (FACSCanto; BD Biosciences, San Jose, CA, USA) at the Key Laboratory of Pediatrics in Chongqing, Children's Hospital of Chongqing Medical University (Chongqing, China) as previously described (17,23).
Culture of Neonatal Mouse Cardiac Myocytes
C3H mice (35) (purchased from the Experimental Animal Center of Chongqing Medical University, China) were used as the source of neonatal mouse cardiac myocytes in the present study. The animal protocol was approved by the Animal Care and Use Committee at the Chongqing Medical University (Chongqing, China). Primary cultures of the cardiac myocytes were prepared from the hearts of C3H10T1/2 mice (day 1–2) as described previously (39). The cells were then cultured in DMEM (Thermo) containing 10% FBS, bromodeoxyuridine (final concentration 0.1 mM) (Sigma), and penicillin/streptomycin (final concentration 100 μg/ml) (Invitrogen, Carlsbad, CA, USA) in six-well culture plates (Corning, New York, NY, USA) at a density of 1 × 106 cells/well coated with L-polylysine (Sigma) in a humidified incubator with 5% CO2 at 37°C with medium change after 48 h of culture. After 72 h of culture, the cells were randomly divided into four groups as the positive controls.
RNA Isolation, Reverse Transcription, and Quantitative RT-PCR
Total RNAs were extracted from the cells using RNA extract kit (Bioteke, Beijing, China) and reverse-transcribed (RT) with a kit from Takara (Dalian, China). SYBR Green RealMasterMix kit (Tiangen, Beijing, China) was used for quantitative RT-PCR assay following the manufacturer's instructions to evaluate the transcriptional expression of guanine–adenine–thymine–adenine-binding protein 4 (Gata4); myocyte-specific enhancer factor 2c (Mef2c); potassium voltage-gated channel; shaker-related subfamily, member 5 (Kcna5); potassium inwardly rectifying channel, subfamily J, member 12 (Kcnj12); calcium channel, voltage-dependent, T type, α 1H subunit (Cacna1h); sodium channel, voltage-gated, type V, α subunit (Scn5a) genes with β-actin as the endogenous housekeeping gene to normalize the mRNA levels. The primer sequences were as follows: for Gata4, sense: 5-CCCTCCCGCACGATTTCT-3, antisense: 5-AGAGGCCCAACTCGCTCAA-3; for Mef2c, sense: 5-GCGCAGGGAATGGATACGG-3, antisense: 5-TGCCAGGTGGGATAAGAACG-3; for Bmp9, sense: 5-CAGCCACTGCCAGAAGAC-3, antisense: 5-CATC AGCCAATGGGAAGAAGC-3; for Bmp13, sense: 5-CCAAACTGACTCCCATTAGCATC-3, antisense: 5-CCACCACCATGTCCTCATAC-3; for Kcna5, sense: 5-GGTCCTCACCATTGCTCTG-3, antisense: 5-CTCT GATGCCTTGCTCTTC-3; for Kcnj12, sense: 5-CGAG AAGTCACAACGCTACCT-3, antisense: 5-AACAAC CAGGAGGCAAGAAA-3; for Cacna1h, sense: 5-CAA GGATGGCTGGGTAAACA-3, antisense: 5-CGATGA GCAGGAAGGAGATG-3; for Scn5a, sense: 5-CATCT TCGGCTCCTTCTTCA-3, antisense: 5-TTCTTCTGCT CCTCCGTCAT-3; for β-actin, sense: 5-CACACCCG CCACCAGTTCG-3, antisense: 5-GTCCTTCTGACCC ATTCCCACC-3. The annealing temperature was 60°C for Gata4, Mef2c, Bmp9, Bmp13, and β-actin. Results were expressed as mean of 2-ΔΔCt ± standard deviation (SD) (24).
Immunofluorescence Staining
To evaluate the expression of cardiomyocyte-specific proteins cardiac troponin T2 (cTnT) and connexin 43 [Cx43/gap junction protein α 1 (GJA1)], the cells at different times of differentiation were fixed and prepared for immunofluorescence staining as described (19). The preparations were incubated with the primary antibodies against cTnT (1:200; Abcam, Cambridge, UK) or Cx43/ GJA1 (1:200; Abcam) overnight at 4°C, then exposed to corresponding secondary antibodies as recommended by the manufacturer (1:200; Santa Cruz, Santa Cruz, CA, USA) at 37°C for 30 min. DAPI (4′,6-diamidino-2-phenylindole; Roche, Berlin, Germany) staining for 15 to 20 min was used to visualize the nuclei. The preparations were then examined and analyzed using a fluorescence microscope (Nikon, Tokyo, Japan).
Western Blotting Analysis
After different treatments and culture, the C3H10T1/2 cells and neonatal mouse cardiac myocytes were collected and prepared for Western blotting as previously described (15,44). The protein preparations were loaded on 10% sodium dodecyl sulfate–polyacrylamide gel (Beyotime, Shanghai, China) for electrophoresis and then transferred onto polyvinylidene difluoride membrane (Millipore, Billerica, MA, USA). After blocking with 5% nonfat dried milk (Sangon, Shanghai, China) for 1 h, the membranes were incubated with anti-cardiac troponin T antibody (1:1,000), anti-connexin 43/GJA1 antibody (1:1,000), anti-BMP9/ growth differentiation factor 2 (GDF2) antibody (1:500), anti-BMP13/GDF6 antibody (1:1,000), or rabbit anti-mouse β-actin polyclone antibody (1:2,000) overnight at 4°C as recommended by the manufacturer (all from Abcam). The membrane preparations were then incubated with horseradish peroxidase-conjugated secondary antibody (1:2,000; Santa Cruz Biotechnology) for 1 h at room temperature. The immunoreactive protein bands were identified with an enhanced chemiluminescence luminal reagent (Keygen, Nanjing, China), scanned, and analyzed with Quantity One Version 4.4 software (Bio-Rad, Hercules, CA, USA). All Western blotting experiments were prepared in duplicate for at least three times.
Transmission Electron Microscopic Examination
To evaluate the ultrastructure of the newly differentiated cells after different treatments, the cells were collected and fixed with 2.5% glutaraldehyde (Mengry, Shanghai, China) at 4°C and then further prepared for examination with a transmission electron microscope as previously described (10,49). Briefly, when growing to 70–80% confluence, the cells were collected in the 2-ml EP tube (Axygen) and fixed with 2.5% glutaraldehyde in PBS (Sangon) at 4°C for 2 h. After washing with PBS, the cells were exposed to 2% osmium tetroxide (SPI Supplies, Westchester, PA, USA) and stained with uranyl acetate (SPI Supplies) after dehydration. The preparations were then embedded in epoxy resin (SPI Supplies) and cut into ultrathin sections (60 nm). The ultrastructure of the cells was examined and analyzed in the Electron Microscope Laboratory at Chongqing Medical University (Chongqing, China) with a Hitachi-7500 transmission electron microscope (Hitachi, Tokyo, Japan).
Masson Staining
Cells were cultured in 12-well plates (Corning) with 1 × 1-cm cover glass slide (Menzel-Glaser, Braunschweig, Germany) and prepared for Masson staining with the Trichrome Stain Masson Kit (Sigma) as previously described (21). Briefly, the cell preparations on the small glass slides were rinsed with tap water, then exposed to hematoxylin for 5 min, followed by washing with deionized water for 30 s. The slides were then placed in scarlet–acid fuchsin for 5 min. After washing with deionized water for 30 s, the preparations were exposed to phosphotungstic– phosphomolybdic acid solution for 5 min. The slides were then treated with benzidine blue solution for 5 min and mounted with xylene after dehydration with alcohol. The cell preparations were examined for their ultrastructure using a microscope (Olympus IX70, Tokyo, Japan).
Electrophysiological Characterization of the Cells Using Patch Clamp
To determine if the cells after transfection with BMP9 or BMP13 vector and differentiation exhibited the membrane ion channel activities typically present in mature cardiomyocytes, the cells in different groups were transferred to 24-well plates (Corning) containing 8 × 8-mm slides with three to four cells with GFP on each slide. The cells were then placed in the recording chamber mounted on the stage of an inverted microscope (Olympus IX71) for cellular electrophysiological characterization using a whole-cell membrane patch-clamp technique as previously described (13). Briefly, the cells were superfused with normal Tyrodes solution (mmol/L) (NaCl 136, KCl 5.4, MgCl2 1.0, NaH2PO4 0.33, HEPES 10, glucose 5.5, and CaCl2 1.8, using 2 mmol/L NaOH to adjust pH to 7.4) (Sigma), and their whole-cell membrane currents were recorded using a patch-clamp amplifier (Axon Multiclamp 700B; Molecular Devices, Sunnyvale, CA, USA). For optimal recording, the membrane potential was kept at −80 mV. The membrane currents were recorded under different voltages when a hyperpolarization pulse with the amplitude of −120 ~ +50 mV in 10 mV space and a duration of 300 ms was applied to the cells. The introversion potassium current was recorded under different voltages in the cells with the following ascending voltage stimulus protocol: maintaining the membrane potential at −120 mV and applying the hyperpolarization pulse with the amplitude of −120 ~ 0 mV in 10 mV space for a duration of 300 ms. The currents were recorded at 10 kHz and filtered at 2 kHz. To further evaluate the nature of the individual ion channels, selective ion channel blockers were added in the extracellular fluid: 4–1.2 aminopyridine (4-AP) and BaCl2 for potassium channel, nifedipine for L-type calcium channel, NiCl2 for T-type calcium channel, and tetrodotoxin (TTX) for sodium channel. All the currents were recorded using the patch-clamp amplifier of Axon Multiclamp 700B, processed with a Digidatal 320A converter (AMPI, Sunnyvale, CA, USA) and pclamp9.2 analysis software system (Axon, Molecular Devices), and analyzed with Clampfit9.2 software (Axon, Molecular Devices).
Statistical Analysis
Unless otherwise stated, all experiments were performed at least three times. All data were expressed as mean ± SD. Multiple group comparisons (three or more groups with different treatments) were performed by one-way ANOVA using the SPSS software (version 13.0; SPSS, Inc., Chicago, IL, USA) followed by post hoc conservative Tukey's test to minimize the type I error as appropriate. Differences were considered statistically significant when p < 0.05.
Results
BMP9 and BMP13 Were Successfully Transfected Into and Expressed in the C3H10T1/2 Cells
We first examined the efficacy of vector transfection in the cells using a fluorescence microscope and flow cytometry. As shown in Figure 1A and B, the transfection was successful with over 60% of the cells positive for GFP for all the transfection groups (BMP9, BMP13, and GFP control) 7 days after the transfection. Over 50% of the cells still remained GFP positive 14 days after transfection with the vectors. By 28 days after the transfection, over 40% of the cells were positive for GFP. Morphological evaluation at different time points suggested that the cells were in good condition and remained healthy up to 28 days after the transfection.

Transfection and expression of BMP9 and BMP13 in the C3H10T1/2 cells. The efficacy of vector transfection in the cells was evaluated using fluorescence microscope (×100) and flow cytometry. Over 60% of the cells were positive for GFP for all the transfection groups (BMP9, BMP13, and GFP control) 7 days after the transfection (A, B). By 28 days after the transfection, over 40% of the cells were still positive for GFP. The levels of transcriptional expression (mRNA) of Bmp9 and Bmp13 were examined using quantitative RT-PCR in the cells transfected with vectors for BMP9 and BMP13. The expression of Bmp9 (C) and Bmp13 (D) in the cells transfected with BMP9 and BMP13 vectors was significantly increased over the controls 3 days after the transfection. Blank, blank control; BMP9, cells were transfected with vectors for BMP9; BMP13, cells were transfected with vectors for BMP13; GFP, cells were transfected with vectors for GFP control; Day 7 and 28, 7 days and 28 days after differentiation. *p < 0.05 when compared with blank and GFP controls (n = 5). Scale bar: 200 μm.
The levels of transcriptional expression (mRNA) of BMP9 and BMP13 were examined using quantitative RT-PCR in the cells transfected with vectors for BMP9 and BMP13, respectively. As shown in Figure 1C, the expression of BMP9 in the cells transfected with BMP9 vector was significantly increased by 120–125 times over the controls 3 days after the transfection (p < 0.05). The expression of BMP13 (Fig. 1D) in the cells transfected with BMP13 vector was significantly increased by 125–130 times over the controls (p < 0.05).
Overexpression of BMP9 or BMP13 Enhanced the Expression of Cardiomyocyte-Specific Genes Gata4, Mef2c, and Membrane Ion Channel Genes Kcna5, Kcnj12, Cacna1h, and Scn5a in C3H10T1/2 Cells
The transcriptional expression levels (mRNA) of cardiomyocyte-specific genes Gata4, Mef2c, and membrane ion channel genes Kcna5 (for potassium voltage-gated channel IKur), Kcnj12 (for inward rectifier potassium channel IK1), Cacna1h (for T-type calcium channel ITCa), and Scn5a (for sodium channel INa) (22,32, 34,37) were examined using quantitative RT-PCR in the cells transfected with the vectors for BMP9 and BMP13. As shown in Figure 2A, the expression of Gata4 in the cells transfected with BMP9 or BMP13 vector was significantly increased by 2.5–3.5 times over the controls after 7 days of differentiation (p < 0.05). The expression of Mef2c was also significantly increased by over three times in the cells transfected with BMP9 or BMP13 compared with the controls (Fig. 2B) during the course of cardiac differentiation (p < 0.05). The maximal expression of Mef2c was significantly higher in the cells transfected with BMP9 vector than the ones transfected with BMP13 (p < 0.05) (Fig. 2B). As shown in Figure 3A, the expression of Kcna5 in the cells transfected with BMP9 or BMP13 vector was significantly increased by 3–3.5 times over the controls after 28 days of differentiation (p < 0.05). The expression of Kcnj12 in the cells transfected with BMP9 or BMP13 vector was significantly increased by 1.5–4 times over the controls (Fig. 3B) after 7 days of differentiation (p < 0.05). The expression of Cacna1h was also significantly increased by over two to six times in the cells transfected with BMP9 or BMP13 compared with the controls (Fig. 3C) during the course of cardiac differentiation (p < 0.05), while the expression of Scn5a was significantly increased by over 4.5 times in the cells transfected with BMP9 compared with the controls (p < 0.05) (Fig. 3D), but not in the cells with BMP13 (p > 0.05).
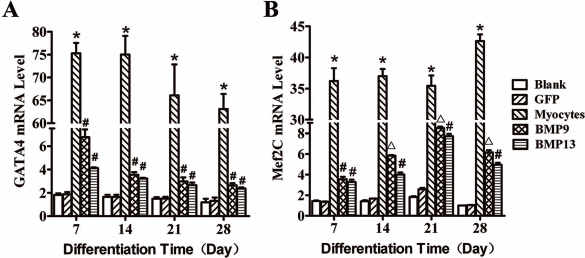
Expression of cardiomyocyte-specific genes Gata4 and Mef2c in C3H10T1/2 cells. The transcriptional expression levels (mRNA) of cardiomyocyte-specific genes guanine–adenine–thymine-adenine-binding protein 4 (Gata4) and myocyte-specific enhancer factor 2c (Mef2c) were examined using quantitative RT-PCR in the cells transfected with the vectors for BMP9 and BMP13. The expression of Gata4 and Mef2c in the cells overexpressing BMP9 or BMP13 was significantly increased over the controls after 7 days of differentiation (A, B). The maximal expression of Mef2c was significantly higher in the cells transfected with BMP9 vector compared to the ones transfected with BMP13 (B). Blank, blank control; BMP9, cells were transfected with vectors for BMP9; BMP13: cells were transfected with vectors for BMP13; GFP, cells were transfected with vectors for GFP control; Myocytes, isolated mouse cardiomyocytes. *p < 0.05 when compared with blank and GFP controls; #p < 0.05 when compared with blank, GFP controls and the mouse cardiomyocytes (positive control); Δp < 0.05 when compared with BMP13 group, blank control, GFP control, and the mouse cardiomyocytes.

Expression of membrane ion channel genes Kcna5, Kcnj12, Cacna1h, and Scn5a during cardiac differentiation of C3H10T1/2 cells. The transcriptional expression levels (mRNA) of membrane ion channel genes Kcna5, Kcnj12, Cacna1h, and Scn5a were examined using quantitative RT-PCR in the cells transfected with the vectors for BMP9 and BMP13. The expression of Kcna5, Kcnj12, and Cacna1h in the cells overexpressing BMP9 or BMP13 was significantly increased over the controls during differentiation (A, B, C). The expression of SCN5A was significantly increased in the cells overexpressing BMP9, but not in cells overexpressing BMP13 (D). Blank, blank control; BMP9, cells were transfected with vectors for BMP9; BMP13, cells were transfected with vectors for BMP13; GFP, cells were transfected with vectors for GFP control; Myocytes, isolated mouse cardiomyocytes. *p < 0.05 when compared with blank and GFP controls; #p < 0.05 when compared with the mouse cardiomyocytes (positive control); Δp < 0.05 when compared with the mouse cardiomyocytes, blank control, GFP control, and BMP13 group.
Expression of Cardiomyocyte-Specific Proteins cTnT and Cx43 Was Increased in the C3H10T1/2 Cells Transfected with BMP9 or BMP13
The expression of cardiomyocyte-specific proteins cTnT and Cx43 was evaluated using immunofluorescence staining and Western blot. No detectable level of cTnT protein or Cx43 protein was present in the control cells. Western blotting analysis demonstrated that the protein contents of cTnT and Cx43 were significantly increased in the cells transfected with BMP9 or BMP13 compared with the controls (p < 0.05) (Fig. 4A–C). It was apparent that the protein levels of Cx43 and cTnT were significantly higher in the cells transfected with BMP9 vector than that in the cells transfected with BMP13 vector (p < 0.05) (Fig. 4B). Similarly, transfection of the C3H10T1/2 cells with BMP9 or BMP13 vector significantly increased the protein levels of cTnT and Cx43 as reflected by the dense intracellular fluorescence (Fig. 4D).

Expression of cardiomyocyte-specific proteins cardiac troponin T2 (cTnT) and connexin 43 (Cx43) during C3H10T1/2 cell differentiation to myocardiocytes. The expression of cardiomyocyte-specific proteins cTnT and Cx43 was evaluated in the cells 28 days after differentiation. Western blotting analysis demonstrated that the protein contents of cTnT and Cx43 were significantly increased in the cells transfected with BMP9 or BMP13 compared with the controls (A, B, C). β-Actin was used as a loading control. It was apparent that the protein levels of Cx43 and cTnT were significantly higher in the cells transfected with BMP9 vector than that in the cells transfected with BMP13 vector (A, B). No detectable level of cTnT protein or Cx43 protein was present in the control cells. Immunofluorescence staining (×400) showed that transfection of the C3H10T1/2 cells with BMP9 or BMP13 vector significantly increased the protein levels of cTnT and Cx43 as reflected by the dense intracellular fluorescence (D). Blank, blank control; BMP9, cells were transfected with vectors for BMP9; BMP13, cells were transfected with vectors for BMP13; GFP, cells were transfected with vectors for GFP control; Myocytes, isolated mouse cardiomyocytes. *p < 0.05 when compared with blank and GFP controls; #p < 0.05 when compared with blank, GFP controls, and the mouse cardiomyocytes (positive control); Δp < 0.05 when compared with BMP13 group, blank control, GFP control, and the mouse cardiomyocytes.
BMP9- or BMP13-Overexpressing C3H10T1/2 Cells Exhibited a Typical Ultrastructure of Cardiomyocytes
Myofilament-like structures and intercalated disc-like structures were observed with transmission electron microscopy in the cells transfected with BMP9 or BMP13 28 days after differentiation that were similar to that in the isolated cardiomyocytes (Fig. 5A–F). As expected, no such structures were present in the control cells. It has been documented that, with Masson staining, collagen fibers turn to green or blue, the nucleus become dark gray or gray blue, and muscle fibers and red blood cells stain red (41). As shown in Figure 5G–J, the cells transfected with BMP9 or BMP13 contained red myofilament-like structure after 28 days of differentiation that were similar to that in the isolated cardiomyocytes, while no such structure was observed in the control cells with Masson staining, suggesting the formation of muscle fibers in the cells expressing BMP9 or BMP13.

Ultrastructures of the differentiated cells. Myofilament-like structures and intercalated disc-like structures were observed with transmission electron microscopy (×10,000) in the cells transfected with BMP9 or BMP13 after 28 days of differentiation that were similar to that in the isolated cardiomyocytes (B, C, D, E, F). No such structures were present in the control cells (A). Masson staining (×400) showed that the cells transfected with BMP9 or BMP13 contained red myofilament-like structure after 28 days of differentiation that were similar to that in the isolated cardiomyocytes (H, I, J). No such structures were present in the control cells (G). The arrows indicate myofilament-like structure, and the circles indicate intercalated disc-like structure. Blank, blank control; BMP9, cells were transfected with vectors for BMP9; BMP13, cells were transfected with vectors for BMP13; Myocytes, isolated mouse cardiomyocytes.
A Variety of Membrane Ion Channel Activities Were Present in the Cells Transfected with BMP9 or BMP13
Using the whole-cell patch-clamp technique, we examined if the membrane ion channel activities typical for mature cardiomyocytes were present in the cells transfected with BMP9 or BMP13 vector after differentiation. We observed that there were two types of K+ currents in the differentiated cells: delayed rectifier potassium current (Ikur) (detected in about 50–60% of cells transfected with BMP9 or BMP13 vectors) and inward rectifier potassium current (IK1) (detected in about 80–90% of cells transfected with BMP9 or BMP13 vectors). T-type calcium current (IT-Ca) was also observed in 30–40% of cells over-expressing BMP9 or BMP13 as shown in Figure 6. Sodium current (INa) was detected in about 10% of cells transfected with BMP9 vector, but not in the cells with BMP13. As expected, all the above currents were detected in cultured neonatal cardiomyocytes. No membrane currents were detected in the control cells.

Transmembrane electric currents in the cells transfected with BMP9 or BMP13. Using a whole-cell patch-clamp technique, the membrane ion channel activities typical for mature cardiomyocytes were evaluated in the cells transfected with BMP9 or BMP13 vector after 28 days of differentiation. Delayed rectifier potassium current (Ikur), and inward rectifier potassium current (IK1), T-type calcium current (IT-Ca) were observed in the cells overexpressing BMP9 or BMP13. Sodium current (INa) was detected in the cells transfected with BMP9 vector, not in the cells with BMP13. All the currents were detected in cultured neonatal cardiomyocytes. No membrane currents were detected in the control cells. The presence of the individual ion channel currents were further verified using the selective ion channel blockers as shown in the individual panels. (A) BMP9/BMP13/Myocytes, Ikur that was blocked by 4-1.2 aminopyridine (4-AP). (B) BMP9/BMP13/Myocytes, IK1 that was inhibited by BaCl2. (C) BMP9/BMP13/Myocytes, IT-Ca that was attenuated with NiCl2. (D) BMP9/BMP13/Myocytes, INa that was diminished with TTX. BMP9, cells were transfected with vectors for BMP9; BMP13, cells were transfected with vectors for BMP13; Myocytes, isolated mouse cardiomyocytes.
The detected individual ion channel currents were further verified using the selective ion channel blockers (Fig. 6). Indeed, Ikur was blocked in the cells when 3 mmol/L 4-AP was added to the extracellular solution (Fig. 6A). The IK1 current was substantially decreased in the cells in the presence of 100 μmol/L BaCl2 in the extracellular solution (Fig. 6B). As expected, IT-Ca current was diminished with addition of 10 mmol/L NiCl2 to the extracellular solution (Fig. 6C), and the INa current was dramatically reduced when the cells were exposed to 10 μmol/L TTX (Fig. 6D).
Discussion
Recent studies demonstrate that BMPs including BMP2, BMP4, BMP6, and BMP7 stimulate myocardial differentiation (18,35). In the present study, we showed that transfection of C3H10T1/2 cells with BMP9 or BMP13 promoted their differentiation into cardiomyocyte-like cells. These differentiated cells expressed cardiac-specific genes Gata4 and Mef2c and proteins cTnT and Cx43 and exhibited the ultrastructures and membrane ion channel activities similar to isolated cardiac myocytes.
Mouse heart development is generally divided into three phases: early stage (0–3 days), middle stage (3–7 days), and late stage (7–14 days) (29). Gata4 and Mef2c are critical genes in cardiac-specific differentiation and expressed in the early phase of cardiac development (7). In the present study, we observed that the expression of Gata4 and Mef2c genes occurred within 7–14 days in the cells after transfection with BMP9 or BMP13. These data were consistent with the observations in the cells after induction with BMP2 (6,20,48), suggesting that both BMP9 and BMP13 were able to promote cardiac differentiation similar to BMP2.
During heart development, cardiac-specific proteins important to cardiac structure and function like cTnT and Cx43 are also expressed. cTnT is an important structure and function protein for the heart (26,30). Cx43 is considered to be the most ubiquitous gap junction protein. It is critically involved in both electrical and chemical signaling between cells and plays an essential role in cell–cell communications in a variety of organ systems including the heart (12,36). The intercalated disk is a cardiac-specific structure that is located in the gap junctions of the myocardial fibers and functions as a bridge connecting the surrounding cardiac myocytes as an electrochemical syncytium through complex chemical information exchanges and electrical impulse conductions among the cells (38). In the present study, we observed that both cTnT and Cx43 were expressed in the cells after transfection with BMP9 or BMP13 with formation of the cardiac-specific intercalated disk in differentiated cells similar to the mature cardiomyocytes on the transmission electron microscopic imaging, suggesting that the differentiated cells after transfection with BMP9 or BMP13 were indeed functional cardiomyocytes.
One of the key features for functioning cardiomyocytes is the cardiac-specific ion channel currents. It has been reported that the cardiomyocytes derived from embryonic stem cells exhibited some characteristic electrophysiological properties for the ion channels (28,46). However, very little is known on the electrophysiological characteristics for the differentiated cardiomyocytes from bone marrow stem cells. In the present study, we observed that the expression of membrane ion channel genes Kcnj12 and Cacna1h occurred within 7 days, and Kcna5 expression within 28 days in the cells after transfection with BMP9 or BMP13, while Scn5a expression occurred within 14 days in the cells after transfection with BMP9. Using the whole-cell patch-clamp technique, we demonstrated that the ultrarapidly activating delayed rectifier outward potassium current and inward rectifier potassium current were present in the cells transfected with either BMP9 or BMP13 after 28 days of differentiation, similar to the ones in the neonatal rat ventricular myocytes (12). It is known that there are two types of calcium channels on the myocardial cell membrane: L-type (IL-Ca) and T-type calcium channel (IT-Ca). IT-Ca is a voltage-gated calcium channel that is mainly distributed in the sinoatrial node cells. In the present study, we detected IT-Ca current in the cells transfected with BMP9 or BMP13 after 28 days of differentiation. In a small portion of the cells transfected with BMP9, sodium current (INa) was detected after 28 days of differentiation. These data suggested that transfection of BMP9 or BMP13 could induce the differentiation of C3H10T1/2 cells into a mixture of different types of cardiomyocytes. Further studies are needed to define the optimal conditions for the differentiation into a homogeneous-specific type(s) of cardiomyocytes with similar electrophysiological properties.
In the present study, we demonstrated that both BMP9 and BMP13 effectively promoted the differentiation of C3H10T1/2 cells into cardiomyocyte-like cells. Interestingly, we also observed that the expression levels of Mef2c gene and Cx43 and cTnT proteins were significantly higher in the cells transfected with BMP9 vector than those transfected with BMP13 vector. The sodium channel genes Scn5a and sodium current was only detected in the cells transfected with BMP9 vector, not in the cells overexpressing BMP13. These data suggested that BMP9 might be a better one to induce cardiac differentiation of C3H10T1/2 cells over BMP13. Previous studies have shown that overexpression of BMP2 could regulate the expression of Gata4, Mef2c, and Nkx2.5 and induce the differentiation of stem cells into cardiomyocytes through p38MAPK, Smads, and PI-3K signaling pathways (5,9,31,33). Expression of BMP2, VEGF, and FGF was increased after myocardial infarction and might contribute to cell differentiation and angiogenesis (1,8). However, the mechanism(s) for BMP9- and BMP13-mediated effect on C3H10T1/2 remains unknown. It is also not clear whether BMP9 and BMP13 have the same or similar effects on bone marrow stem cells in vivo. Further studies are needed to investigate the in vivo cardiac differentiation of C3H10T1/2 cells induced by BMP9 and BMP13 and related mechanism(s).
In summary, in the present study we demonstrated that both BMP9 and BMP13 promoted the differentiation of C3H10T1/2 cells into cardiomyocyte-like cells with expression of cardiac-specific genes and proteins, formation of specific cardiac ultrastructures, and development of ion channel currents similar to mature cardiomyocytes in vitro. Further studies are needed to investigate the underlying mechanisms for BMP9- and BMP13-induced cardiac differentiation of C3H10T1/2 cells and their effects on bone marrow stem cells in vivo.
Footnotes
Acknowledgments
This study was supported by the National Natural Science Foundation of China (Grant No. 30772361). The authors declare no conflicts of interest.
