Abstract
Mesenchymal stromal cells (MSCs) play an important role in tissue regeneration mainly through the secretion of trophic factors that enhance the repair of damaged tissues. The main goal of this work was to study the paracrine mechanisms by which an umbilical cord tissue-derived MSC population (UCX®) promotes the migration capacity of human dermal fibroblasts and keratinocytes, which is highly relevant for skin regeneration. Furthermore, the differences between paracrine activities of MSCs from the umbilical cord tissue and the bone marrow (BM-MSCs) were also evaluated. In vitro scratch assays revealed that conditioned media (CM) obtained from both growing and stationary-phase UCX® cultures induced human dermal fibroblast (HDF) and keratinocyte (HaCaT) migration. These assays showed that the motogenic activity of UCX® CM to HaCaTs was significantly higher than to HDFs, in opposition to the effect seen with CM produced by BM-MSCs that preferentially induced HDF migration. Accordingly, a comparative quantification of key factors with vital importance in the consecutive stages of wound healing revealed very different secretome profiles between UCX® and BM-MSCs. The relatively higher UCX® expression of EGF, FGF-2, and KGF strongly supports early induction of keratinocyte migration and function, whereas the UCX®-specific expression of G-CSF suggested additional roles in mobilization of healing-related cells including CD34-/CD45- precursors (MSCs) known to be involved in tissue regeneration. Accordingly, in vitro chemotaxis assays and an in vivo transplantation model for chemoattraction confirmed that UCX® are chemotactic to CD34-/CD45- BM-MSCs via a cell-specific mobilization mechanism mediated by G-CSF. Overall, the results strongly suggest different paracrine activities between MSCs derived from different tissue sources, revealing the potential of UCX® to extend the regenerative capacity of the organism by complementing the role of endogenous BM-MSCs.
Keywords
Introduction
The healing of cutaneous wounds, like any other type of injury, represents a complex process, integrating a number of sequential molecular events involving clotting, inflammation, cellular migration, proliferation, extracellular matrix (ECM) deposition, angiogenesis, vasculogenesis, and remodeling of the mature scar tissue (29,36,50,58). In a number of medical conditions, such as diabetes, chronic renal failure, or arterial or venous insufficiency, the success of this process is jeopardized, resulting in numerous complications and increased morbidity. Hence, a number of novel strategies have been attempted, such as the local application of growth factors and stem cells, to improve the regeneration process (6,35).
Mesenchymal stromal cells (MSCs) are a subset of stem cells that can be isolated from a number of adult and neonatal tissues (13,16,56). MSCs have been shown to possess modulatory properties of both immune and inflammatory reactions, although the specific mechanism by which MSCs display such behaviors is still under investigation (18,26,57).
In situ engraftment of MSCs to sites of tissue damage after systemic infusion or localized transplantation has been demonstrated, yet increasing evidence also shows that MSCs have the ability to induce tissue regeneration without local engraftment and differentiation. Indeed, several authors have hypothesized that the MSC-secreted factors enhance and assist in the repair and regeneration of damaged tissues via paracrine activation of surrounding cells (7,8,12,24,38). Secreted bioactive factors may induce cell migration, proliferation, and even differentiation, resulting in, for example, angiogenesis and vasculogenesis (6,38,44). The paracrine secretion of trophic factors has the potential to induce profound effects on the MSC local environment, as suggested by the cytokine-mediated control of hematopoiesis, which is thought to be regulated by MSCs in the vascular niche of the bone marrow (8,44,59). The paracrine action of MSCs has been implicated in cutaneous wound healing, where it is suggested that these cells secrete a number of promitogenic and motogenic growth factors and cytokines, thus leading to enhanced tissue regeneration (10,59,60).
Most of the data so far have been generated with the prospective autologous application, using bone marrow-derived MSCs (BM-MSCs) (9,60). BM-MSCs are known to be endogenously mobilized and to coexist within sites of injury, actively participating in the healing process (9,10,60). However, increasing evidence has been supporting neonatal tissues, such as the umbilical cord tissue and placenta, as better sources of MSCs due to several reasons. Neonatal tissues confer ease of isolation from noninvasive and noncontroversial human sources. Furthermore, a relatively higher cell yield is obtained, and the resulting MSCs have higher expansion capacity and often higher regenerative potency (2,28,60,61). UCX® in particular are MSCs obtained from the umbilical cord stromal tissue using a proprietary method that has recently been adapted according to advanced therapy medicinal product (ATMP) certification requirements [EMEA/CAT/486831/ 2008/corr (17)] (36a). UCX® comply with the MSC definition provided by the International Society for Cellular Therapy (ISCT) (15). Namely, UCX® present fibroblast-like morphology while adherent to plastic; more than 98% of UCX® within the population express the markers cluster of differentiation 105 (CD105), CD73, CD90, and CD44, whereas less than 2% express CD45, CD34, CD31, CD14, CD19, and human leukocyte antigen (HLA)-DR. Finally, UCX® can be differentiated into osteoblast, chondrocyte, and adipocyte-like cells (46). UCX® have proven to be nonimmunogenic in vitro and in vivo and to promote angiogenesis in the scope of an acute myocardial infarction model (38a). Furthermore, UCX® have recently been demonstrated to have the capacity of repressing T-cell activation and promoting the expansion of regulatory T-cells (Tregs) in vitro. Most importantly, the UCX® capacity for modulating the inflammatory reaction and for suppressing the immune system was shown to be higher than that of BM-MSCs (46). These observations have led us to propose that MSCs from different tissue sources should not be treated as equal in terms of their regenerative properties and should therefore be discussed and evaluated as separate therapeutic entities (46).
In this work, the motogenic effect of UCX® to human dermal fibroblasts (HDFs) and human keratinocytes (HaCaTs) was demonstrated and shown to be very different from that caused by BM-MSCs, further supporting a distinct, but nevertheless complementary, regenerative role for UCX® (55). Accordingly, a comparative quantification of a selection of paracrine factors with vital importance during the consecutive phases of wound healing revealed very different secretome profiles between the two MSC types. While the UCX® trophic factor profile suggested a direct impact on the earlier homeostasis and inflammation stages of wound healing, the BM-MSC trophic profile was better suited to promote later proliferative and final remodeling activities. Additionally, the UCX®-specific expression of granulocyte-colony stimulating factor (G-CSF) was shown to attribute UCX® with a chemotactic recruitment activity to other circulating or resident BM-MSCs. Such capacity was confirmed in vivo through transplantation of UCX® and BM-MSC-seeded scaffolds in the inner wall of the peritoneal cavity of severe combined immunodeficient (SCID) mice and by monitoring interscaffold-specific MSC migration.
Overall, our results are consistent with different regenerative activities between MSCs from different tissue sources and confirm the potential of UCX® as an active substance for developing ATMPs.
Materials and Methods
Ethical and Regulatory Aspects
This study was approved by the Ethics Committee of the Cascais Hospital Dr. José de Almeida (Portugal), in the scope of a research protocol with ECBio, S.A. (Amadora, Portugal). Umbilical cord procurement involving donations, with written informed consent, was made according to Directive 2004/23/EC. All the experimental procedures involving animal models were carried out with the permission of the local laboratory animal committees and in accordance with internationally accepted guidelines.
UCX® Isolation and Culture
UCX® used in this work were derived from a total of seven umbilical cords (three females and four males) from healthy donors, isolated according to Santos et al. (47) and, when necessary, thawed from a low passage (P3–P5) working cell bank (WCB). In brief, UCX® were isolated from human umbilical cord tissue sections that were digested using a precise ratio between tissue mass, tissue digestion enzyme activity units, digestion solution volume, and void volume using collagenase. Cells dissociated from the tissue were recovered by adherence to the surface of a culture flask (Nunc, Schnelldorf, Germany) during a static horizontal incubation period and cultured with α-modified minimum essential medium (α-MEM; Sigma-Aldrich, Madrid, Spain) supplemented with 20% fetal bovine serum (FBS; Gibco, Life Technologies, Madrid, Spain), herein designated as MSC complete medium, at 37°C humidified atmosphere with 7% CO2 in air and cryopreserved (47). The WCB was generated by expanding UCX® in standard MSC culture conditions (46). Cells were characterized by their capacity to adhere to plastic in standard MSC culture conditions, specific surface marker expression, and trilineage differentiation capacity (46). All cell culture media and supplements were purchased from Sigma-Aldrich, unless otherwise indicated.
Cell Characterization by Flow Cytometry
For surface marker expression analyses (
Cell Characterization by Trilineage Differentiation
To induce adipogenic differentiation (
Production of UCX® and BM-MSC Conditioned Media (CM)
For conditioned media (CM) production, UCX® and BM-MSCs (Cat. P10576, batch 2095; Innoprot, Bizkaia, Spain) were first sequentially adapted to low serum concentrations by a reduction of the amount of FBS in the cell culture medium from 20% to 2% (Fig. 1a). When at 2% FBS, cultures were maintained until 80% confluence after which (i) either UCX® or BM-MSCs were trypsinized (Gibco, Life Technologies, Madrid, Spain) and seeded at a 1 × 104 cells/cm2 density in MSC medium supplemented with 2% FBS and cultured for a conditioning period of 7 and 4 days, respectively (growing UCX®-CM and growing BM-MSC-CM), or (ii) UCX® medium was simply replaced with MSC medium supplemented with 2% FBS and maintained in culture for a conditioning period of 3 days (confluent UCX®-CM). The control sample consisted of MSC complete medium containing 2% FBS that was never in contact with cells. CM and control were concentrated with 5 kDa cutoff spin concentrators (Agilent Technologies, Santa Clara, CA, USA) as per manufacturer's recommendations. CM used for comparative studies based on scratch assays were normalized for total protein concentration. Protein content was quantified using the bicinchoninic acid protein assay kit (Thermo Scientific, Waltham, MA, USA) according to the manufacturer's instructions.

Motogenic effect of UCX®-CM and BM-MSC-CM to HDF and HaCaT cells. (a) Scheme of CM production as described in Materials and Methods. (b) Images of the scratch area on HDF and HaCaT cell cultures at time 0 h and 20 h or 40 h, respectively, after CM treatment. (c) Graphical representation of the integrated area migrated by HDF and HaCaT cells after contact with different CMs.
In Vitro Scratch Assays
The in vitro scratch assays were performed on primary HDFs (from a 38-year-old Caucasian female, Cat. DF-F; Zen-Bio, Research Triangle Park, NC, USA) and on HaCaTs (from a 62-year-old Caucasian male, Cat. 300-493; CLS Cell Lines Service GmbH, Eppelheim, Germany) essentially as described by Liang et al. (33). Primary HDF and HaCaT cells were seeded into 24-well plates (Nunc, Schnelldorf, Germany) at a density of 2.5 × 104 cells/cm2 and 4.0 × 104 cells/cm2, respectively, with DMEM-High Glucose (DMEM basal medium) supplemented with 10% FBS, herein designated as DMEM complete medium. Once cultures reached 80% confluence, culture medium was replaced with DMEM basal medium without FBS for 24 h to guarantee the inhibition of cell proliferation, while enabling cell migration. Scratches of approximately 0.5 mm in width were then performed on the monolayer with a sterile P200 pipette tip (Orange, Braine-L'Alleud, Belgium). After performing the scratch, the culture medium was replaced with 200 μl of DMEM basal medium supplemented with the different groups of CM and control diluted to a final concentration of 500 μg/ml total protein. Additional controls consisting of CM generated from HDF or HaCaT cultures were also performed in order to monitor for self-chemotactic effects (data not shown). The area of the scratch was measured at 0 h for both cell types and at 20 h and 40 h postscratch for HDF and HaCaT cells, respectively. UCX®-CMs were derived from a total of
Trophic Factor Quantification
A commercially available kit (FlowCytomix™; eBioscience) was used to determine the concentrations of epidermal growth factor (EGF), fibroblast growth factor-2 (FGF-2), G-CSF, hepatocyte growth factor (HGF), interleukin-6 (IL-6), TGF-β1, and vascular endothelial growth factor-A (VEGF-A) in CM from growing UCX®, growing BM-MSCs, and control samples. Samples were acquired on a Gallios imaging flow cytometer, and the results were obtained using FlowCytomix Pro 3.0 Software. Quantification of keratinocyte growth factor (KGF) in these samples was performed using an enzyme-linked immunosorbent assay kit (Quantikine; R&D Systems, Minneapolis, MN, USA). The quantification of all growth factors was obtained from a total of
In Vitro Chemotaxis Assay
BM-MSCs were labeled with octadecyl indocarbocyanine (DiD, 1 μM; Life Technologies, Madrid, Spain) for 30 min at 37°C. Fifty thousand labeled BM-MSCs were seeded in serum-free media in the upper compartment of a Boyden chamber (Corning, Amsterdam, Netherlands) and were allowed to migrate through the porous membrane (8 μm) for 48 h in response to the following stimuli from the lower compartment: (i) serum-free media (negative control), (ii) serum-free media supplemented with the universal chemoattractant EGF at 10 ng/ml (positive control), (iii) 200,000 nonlabeled BM-MSCs in serum-free media seeded on the bottom of the well (control for self-mediated chemotaxis), (iv) 200,000 nonlabeled UCX® in serum-free media seeded on the bottom of the well, and (v) 200,000 UCX® in serum-free media supplemented with anti-G-CSF as an inhibitor (hG-CSF, Anti-G-CSF Human, 500-P43-B; Tebu-Bio). Upon incubation, migratory BM-MSCs were removed from the bottom of the membrane using trypsin and quantified using a FLUOstar OPTIMA fluorimeter (BMG Labtech, Ortengerg, Germany) operated at 549 nm excitation/565 nm emission. Results from three independent experiments are represented as ratios compared to negative controls.
In Vivo Scaffold-Based Transplantation Model for Chemoattraction
Either BM-MSCs or UCX® labeled with 1 μM DiD was suspended in culture medium at an appropriate concentration (50,000 cells/20 μl) and seeded into 3D Insert-polystyrene/polycaprolactone (PS/PCL) with nanomesh scaffolds (3D-Biotek, Hillsborough, NJ, USA) 24 h before implantation in the inner wall of the peritoneal cavity of 8- to 9-week SCID mice (female SCID beige mice; Charles River, Barcelona, Spain). For this, mice were anesthetized with Ketolar® (75 mg/kg; Parke-Davis, Pfizer, Madrid, Spain) and metomidine (1 mg/kg; Orion Pharma, Barcelona, Spain) and an incision performed in the peritoneum to introduce and fix the scaffolds with BM-MSCs and UCX® in the left upper part and lower part of the peritoneal cavity at 1 cm of distance approximately, with the help of surgical glue (Vetbond®; 3M, St. Paul, MN, USA). Atipamezol (1 mg/kg; Orion Pharma), a metomidine antagonist, was supplied after operation. One week after implantation, mice were sacrificed, and MSC migration was evaluated using an in vivo imaging system (IVIS; Perkin-Elmer, Santa Clara, CA, USA) operated at 644 nm excitation/665 nm emission. Figures are representative images of two independent experiments using a minimum of four animals per group.
Statistical Analysis
All statistical analyses were done with SPSS 21.0 (Chicago, IL, USA). Results are presented as mean and with standard errors of the mean (SEM) where possible. To estimate the significance of the differences between groups, the parametric Student's
Results
In this work, UCX® were freshly isolated from a total of seven umbilical cord samples, and when necessary for experimentation, cells were thawed from a low passage WCB (P3–P5). UCX® and BM-MSCs were expanded in all cases and consistently presented fibroblast-like morphology while adherent to plastic; more than 98% of the cells within the corresponding populations consistently expressed CD105, CD73, CD90, and CD44, whereas less than 2% expressed CD45, CD34, CD31, CD14, CD19, and HLA-DR. Finally, both types of MSCs could consistently be differentiated into osteoblast, chondrocyte, and adipocyte-like cells following the MSC criteria as defined by the ISCT (15) (results not shown).
In Vitro Motogenic Effects of UCX® and BM-MSC to HDFs and HaCaTs
In order to evaluate the motogenic potential of UCX® to HDFs and HaCaTs, CM was derived from UCX® and its activity compared to that derived from BM-MSCs (Fig. 1a). Both CMs were supplied to HDF and HaCaT cell cultures and specific cell migration evaluated using in vitro scratch assays. Prior to CM production, both types of MSCs were progressively adapted to growth conditions containing sequentially lower FBS concentrations in order to reduce the amount of serum components in the CM that could potentially mask their trophic activity (Fig. 1a). CM was finally produced by cultures growing in 2% FBS, the lowest concentration that still allowed for MSC growth and expansion. Also, in an attempt to elucidate for the importance of metabolic rate in the quality of the secreted trophic factors, CM were produced by UCX® cultures in exponential phase (growing UCX®-CM) and stationary phase (confluent UCX®-CM). Both HDF and HaCaT cells in contact with either growing UCX®-CM or confluent UCX®-CM exhibited a significantly increased migration capacity, with a slightly more prominent effect being observed for CM produced by more metabolically active, exponentially growing UCX® (Fig. 1b, c). Interestingly, the motogenic effect of growing UCX®-CM was considerably higher with keratinocytes (more than threefold higher than control) than fibroblasts (1.8-fold higher than control), contrarily to the cell migration promoted by CM produced by exponential phase BM-MSC cultures (growing BM-MSC-CM) that showed a clear selectivity for fibroblasts (twofold higher than control) (Fig. 1c). The results demonstrated that the two different types of MSCs have diverse paracrine activities, resulting in differential motogenic effects to HDFs and HaCaTs.
Trophic Factor Quantification in UCX® and BM-MSC Conditioned Media
In order to establish a correlation between the differential motogenic effects caused by the paracrine activity of UCX® and BM-MSCs to HDFs and HaCaTs, respectively, a number of essential trophic factors known to be representative of the several phases of wound healing were quantified. Thus, the concentrations of HGF, G-CSF, TGF-β1, KGF, EGF, FGF-2, VEGF-A, and IL-6 were determined in growing UCX®-CM, growing BM-MSC-CM, and CM control samples (Fig. 2a). The results clearly show different secretome profiles between UCX® and BM-MSCs under our growth conditions (Fig. 2a). In turn, a pie chart representation of trophic factor expression distribution between growing UCX®-CM and growing BM-MSC-CM revealed that i) factors secreted preferentially by UCX® (such as G-CSF, EGF, FGF-2, and KGF) have been commonly implicated in earlier stages of wound healing, namely, homeostasis and inflammation (1,5,27,30,40–42,48,53,62), while ii) factors secreted preferentially by BM-MSCs (such as TGF-β1, IL-6, and VEGF-A) have been more closely associated with the later proliferation and remodeling phases of wound resolution (4,19,20,22,32,34,39) (Fig. 2b). Moreover, given what is known about G-CSF-mediated mobilization of circulating BM-MSCs to sites of injury (14,21,43,45,54), a significant UCX®-specific expression of G-CSF led us to propose a mechanism where UCX® could further promote chemotactic recruitment of BM-MSCs (Fig. 2b).
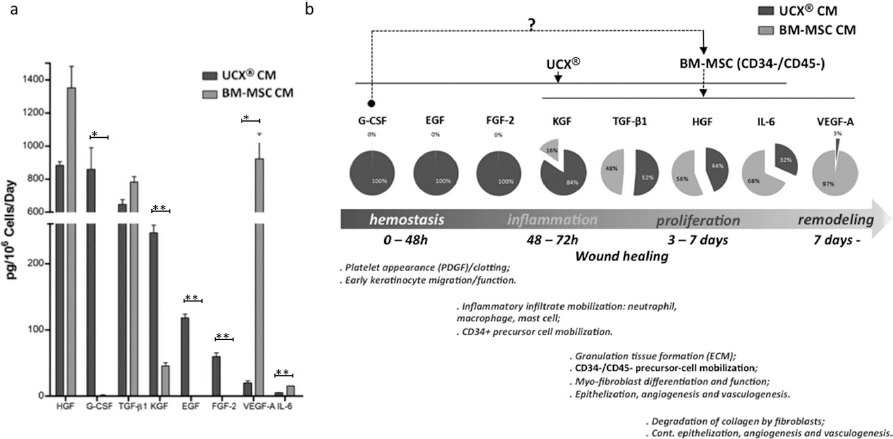
Trophic factor quantification in growing UCX®-CM and growing BM-MSC-CM. (a) Quantification of HGF, G-CSF, TGF-β1, KGF, EGF, FGF-2, VEGF-A, and IL-6 in growing UCX®-CM and growing BM-MSC-CM normalized against control background and expressed in pg of growth factor/cell number in culture/number of conditioning days (pg/106 cells/day).
In Vitro Evaluation of G-CSF-Mediated UCX® Chemotactic Mobilization of BM-MSCs
The hypothesis that UCX® could promote chemotactic mobilization of BM-MSCs was first evaluated in vitro. For this, DiD-labeled BM-MSCs were placed in the upper compartment of Boyden chambers and allowed to migrate toward the lower compartment in response to different chemotactic stimuli. As shown in Figure 3, BM-MSCs migrate in the presence of EGF (universal chemoattractant positive control) or BM-MSCs (self-mediated chemotaxis positive control). In turn, UCX® seeded on the bottom of the well were clearly chemotactic to BM-MSCs, resulting in a 1.7-fold migration increase when compared to self-mediated chemotaxis. Moreover, when the chemotactic assay was performed in the presence of hG-CSF neutralizing antibodies (α–G-CSF), the UCX®-mediated recruitment was significantly hindered (Fig. 3). These results indicate that (i) UCX® are chemotactic to BM-MSCs, and (ii) cell-specific expression of G-CSF by UCX® is an important factor determining the paracrine chemotactic activity to BM-MSCs in vitro.

G-CSF-mediated chemotactic activity of UCX® cells to BM-MSCs in vitro. Graphical representation of the relative BM-MSC migration through the Boyden chamber pores (8 μm) and schematic representation of experimental setup. UCX® seeded on the bottom of the well were chemotactic to BM-MSCs. Moreover, when the chemotactic assay was performed in the presence of α-G-CSF neutralizing antibodies, the UCX®-mediated recruitment was significantly hindered. Relative migration was determined with respect to the negative control. *
In Vivo Evaluation of UCX® Potential for Chemotactic Mobilization of BM-MSCs
In order to confirm whether the UCX® chemotactic effect to BM-MSCs was kept intact in vivo, two 3D-Biotek Insert-PS/PCL scaffolds were transplanted into the peritoneum of SCID mice, one seeded with BM-MSCs and the other with UCX®. MSC-specific chemotactic mobilization was assessed by coupling either fluorescently labeled UCX® or BM-MSCs with their nonlabeled counterparts (Fig. 4). The left panel on Figure 4 shows fluorescence emission by MSCs at time 0, right after scaffold implantation. In turn, the center panels show the location of both scaffolds in the internal wall of the peritoneum 1 week past cell transplantation, at sacrifice day 7. Panels on the right show the corresponding fluorescent image at day 7, and the smaller insets, on the bottom right-hand corner of these panels, are fluorescent images of the corresponding scaffolds ex vivo. The experiment with DiD-labeled BM-MSCs can be followed in the upper panels of Figure 4 (A, B). DiD-labeled BM-MSCs seeded in the upper scaffold migrated toward the lower scaffold containing unlabeled UCX® as demonstrated by a strong fluorescent signal emanating from the bottom scaffold, as well from a migration path connecting both scaffolds (Fig. 4A, B). In turn, when labeled UCX® were located in the upper compartment, a mobilization of much lower significance was detected toward the lower scaffold seeded with unlabeled BM-MSCs (Fig. 4C, D). The column histogram in Figure 4 shows the significant difference between the quantified fluorescence intensity, in arbitrary units, in the UCX® scaffold, resulting from the mobilization of DiD-labeled BM-MSCs (Fig. 4B), compared to fluorescence intensity in the BM-MSC scaffold, resulting from a much lower mobilization of DiD-labeled UCX® (

Chemotactic activity of UCX® cells to BM-MSCs in vivo using a SCID mouse intraperitoneal, two-scaffold, implant model. (Left panels): Photos of mice right after small-incision scaffold implantation, at day 0. (Right panels) Postmortem images of mice at day 7. (A, C, E) Photos showing scaffold location. (B, D, F) Images of mice with superimposed in vivo imaging system (IVIS) detection, representing the density (color gradient) of octadecyl indocarbocyanine (DiD)-labeled cells. The bottom right-hand panels are ex vivo IVIS images of scaffolds, after excision from animals. The column histogram consists of quantified fluorescence intensity, in arbitrary units, in the UCX® scaffold, resulting from the mobilization of DiD-labeled BM-MSCs (B), compared to fluorescence intensity in the BM-MSC scaffold, resulting from a much lower mobilization of DiD-labeled UCX® (D)
Discussion
In a still widely accepted classical model of wound healing, four consecutive and overlapping phases can be considered: homeostasis, inflammation, proliferation, and remodeling, although many authors congregate homeostasis and inflammation in a single inflammatory stage (37). Figure 2b depicts the wound healing process attempting to divide the four healing phases within a temporal sequence of events. Nevertheless, in vivo, there are overlapping sequences occurring simultaneously within the wound site.
Accumulated evidence in this field has drawn up an extensive list of growth factors and cytokines secreted by MSCs that are involved in the various processes leading to tissue regeneration (4,20,32,58). Within the cutaneous wound milieu, inflammation, cellular migration, proliferation, differentiation, ECM deposition, angiogenesis, vasculogenesis, maturation, and remodeling are all necessary responses where MSC-secreted trophic factors may play a role (9,10,24,25,38,49–51,55,59).
In line with the previously demonstrated differential capacity between UCX® and BM-MSCs for immune suppression (46), we herein report a clear, distinct, but nonetheless complementary effect of their paracrine motogenic activities to HDFs and HaCaTs. In agreement with what had been previously reported for BM-MSCs (55), we demonstrate a preferential trophism of BM-MSCs to HDFs when compared to HaCaTs. Conversely, our results showed that the effect of UCX® on inducing HaCaT migration was significantly higher when compared to the corresponding effect on HDFs. Given the relatively higher importance of keratinocyte migration in the early stages of wound healing, we hypothesized that the paracrine healing potential of UCX® could therefore be more relevant during the initial stages leading to skin regeneration, where early keratinocyte migration plays a major role (9,55).
Accordingly, UCX® and BM-MSCs presented clearly distinct secretome profiles. Furthermore, within our specific pool, those factors either exclusively or majorly expressed by UCX® could in fact be directly correlated to events occurring during the earlier phases of the healing process. The relatively secluded expression of EGF, FGF-2, and KGF by UCX®, contrasting with the relatively cloistered expression of VEGF-A (and to some extent IL-6) by BM-MSCs, could alone explain the differential effects observed on HDF and HaCaT migration. EGF, for example, has been shown not only to stimulate HaCaT migration, but also to induce the autocrine secretion of all members of the EGF family. Some of these EGF family members possess even higher promitogenic and motogenic-inducing capacities upon HaCaTs so important for hemostasis. In addition, increasing evidence suggests that the EGF receptor pathway has a major impact on the early inflammatory/immune reactions necessary for proper skin regeneration early after physical injury (31,40,48,53). In turn, FGF-2 is essential for the organization and morphogenesis of suprabasal HaCaTs acting through a chemotactic mechanism (5,27,42). Additionally, FGF-2 has been shown to induce leukocyte recruitment on endothelium, implicating this growth factor on early inflammatory stages of the healing process (62). Finally, although KGF has preferentially been correlated with epithelialization events (1), its early action on wound healing has been recently shown in vivo in a mouse model (41). Experiments demonstrated that KGF is essential to the proliferation of keratinocytes and that the early activity of KGF on keratinocyte function may also play a role in the regulation of VEGF gene expression and consequently on angiogenesis during following stages of the healing process (41). On the other hand, in our culture conditions, factors such as the dominant VEGF isoform VEGF-A (VEGF165) and IL-6 were secreted preferentially by BM-MSCs. VEGF-A, in particular, has been shown to be expressed in a specific temporal pattern in wounds, namely, in the later proliferation phase occurring 3 to 7 days postwound, when capillary growth and differentiation are at a maximum (Fig. 2b) (3). Similarly, VEGF-A-inducible matrix metalloproteinase 1 (MMP-1), MMP-2, and tissue inhibitor of metalloproteinase 1, all peaking at 2 to 5 days after excisional wounding (22), support a role of VEGF-A during fibroblast proliferation and migration, ECM synthesis leading to granulation tissue formation, angiogenesis, and vasculogenesis (20). In accordance with our results, VEGF-A has been targeted primarily at fibroblasts (and macrophages) in deeper wounds (39), thus correlating the BM-MSC-CM fibroblast-specific motogenic activity seen by us with its VEGF-A contents. Like VEGF-A, IL-6 was also mainly expressed by BM-MSCs in our assays. Pursuant to what is known about VEGF-A, IL-6 has also been shown to be crucial to epithelialization and to influence granulation tissue formation during the later stages of tissue regeneration (34).
In summary, the functional analysis of the factors included in our test pool does substantiate our hypothesis that, contrasting to BM-MSCs, the direct impact of UCX® paracrine activity could be centered on the earlier phases of the regenerative process. Nevertheless, equivalent expression of other important factors, such as HGF and TGF-β1, further suggest overlapping roles between UCX® and BM-MSCs. HGF is a cytokine known to play multiple roles during the various stages of wound healing, from neutrophil, monocyte, and mast cell recruitment to the secretion of matrix metalloproteinases, ECM proteins, integrins, and collagenases necessary for keratinocyte migration across the fibronectin-rich provisional wound matrix (4,19,20,23,32,58). In turn, TGF-β1 is highly expressed by keratinocytes and has been shown, along with elements from the VEGF family, to be a key factor for the formation of granulation tissue due to its promitogenic effect upon dermal fibroblasts, while also inducing the expression of ECM proteins and matrix deposition by these same cells (32). Furthermore, TGF-β1 aids in the angiogenic process and has been reported as necessary for fibroblast differentiation into myofibroblasts that are responsible for wound contraction (4,20,32).
Finally, our secretome analysis has revealed a quite remarkable UCX®-specific expression of G-CSF in our growth conditions. In the context of tissue regeneration, it has come to our understanding that circulating factors and systemic-derived cells play an important role in numerous aspects of homeostasis and repair. The capacity of G-CSF to mobilize platelets, infiltrating inflammatory cells and circulating hematopoietic stem cells has been well documented and currently widely used for clinical transplantation (11). However, a putative circulating population of CD45-/ CD34- nucleated cells has also been found in the peripheral blood of human adults that could be mobilized to the injured tissue through a mechanism involving G-CSF (14,21, 43,45,54). Studies using parabiotic models have demonstrated the participation of circulating MSCs in wound repair (52). Autologous BM-MSCs multiply in the bone marrow and migrate to the injured sites immediately after burn injuries, contributing to the inflammatory response, autolysis, and neovascularization along with hematopoietic and endothelial progenitor cells (6). More recently, the relative contribution of grafted human MSCs and host stem/progenitor cells in promoting wound healing has been evaluated by using a novel asymmetric wound model in normal and impaired healing diabetic (db/db) mice (49). MSCs significantly improved healing in both normal and impaired animals by inducing wingless-related mouse mammary tumor virus integration site 3a (Wnt3a), VEGF, and platelet-derived growth factor receptor-a and increasing the number of preexisting host MSCs recruited to the wound bed. Improvement was seen in both the grafted and nongrafted sides, suggesting a systemic response to MSC engraftment. Most importantly, healing was enhanced despite the rapid loss of grafted human MSCs, suggesting that mobilizing the host response was the major outcome of grafting MSCs to tissue repair (49).
As demonstrated in vitro, a G-CSF-dependent pathway is possibly implicated in the motogenic potential of UCX® in vivo. Such a discovery, together with distinct motogenic activities to skin cells, could constitute important distinctive factors between UCX® and BM-MSCs in terms of their future applications. During the early phases of healing, the paracrine activity of UCX® can directly benefit the process in two ways: (i) by mitogenic and motogenic activity to keratinocytes and (ii) by promoting G-CSF-mediated direct recruitment of platelets, infiltrate cells, such as neutrophils, macrophages, mast cells, and CD34+ hematopoietic precursor cells (Fig. 5). In turn, the relatively lower trophism of UCX® to fibroblasts, so important during the later phases of healing, would be compensated by inducing the chemotactic mobilization of either resident or circulating BM-MSCs with potential to promote the formation of granulation tissue, contraction by myofibroblasts, angiogenesis, vasculogenesis, and epithelialization (Fig. 5).

UCX®-mediated therapeutic pathways. Representation of the different pathways by which UCX® can actively impact the wound healing process. Gray arrows highlight G-CSF-mediated mechanisms, namely, early impact recruitment of platelets, immune infiltrate cells, and CD34+ hematopoietic precursors, and later-impact recruitment of CD34-/CD45- BM-MSCs. ECM, extracellular matrix.
Overall, the results presented in this work validate the motogenic potential of UCX® to dermal fibroblasts and keratinocytes and consolidate distinct paracrine activities of MSCs from different tissue sources. The G-CSF-mediated BM-MSC recruiting capacity of UCX® extends its potential to a full range of events leading to tissue regeneration and homeostasis.
Footnotes
Acknowledgments
