Abstract
Cellular therapy for myocardial repair has been one of the most intensely investigated interventional strategies for acute myocardial infarction. Although the therapeutic potential of stem cells has been demonstrated in various studies, the underlying mechanisms for such improvements are poorly understood. In the present study, we investigated the long-term effects of stem cell therapy on both myocardial fiber organization and regional contractile function using a rat model of postinfarct remodeling. Human nonhematopoietic umbilical cord blood stem cells (nh-UCBSCs) were administered via tail vein to rats 2 days after infarct surgery. Animals were maintained without immunosuppressive therapy. In vivo and ex vivo MR imaging was performed on infarct hearts 10 months after cell transplantation. Compared to the age-matched rats exposed to the identical surgery, both global and regional cardiac functions of the nh-UCBSC-treated hearts, such as ejection fraction, ventricular strain, and torsion, were significantly improved. More importantly, the treated hearts exhibited preserved fiber orientation and water diffusivities that were similar to those in sham-operated control hearts. These data provide the first evidence that nh-UCBSC treatment may prevent/delay untoward structural remodeling in postinfarct hearts, which supports the improved LV function observed in vivo in the absence of immunosuppression, suggesting a beneficial paracrine effect occurred with the cellular therapy.
Keywords
Introduction
Myocardial infarction (MI), induced by coronary artery disease, is one of the major causes of heart failure in the world. Owing to the loss of viable myocardium after MI, the heart works continuously under elevated wall stress, which results in progressive myocardial hypertrophy and left ventricular (LV) dilatation that eventually leads to heart failure. In the past 10 years, implantation of progenitor cells for myocardial repair in postinfarct hearts has been extensively investigated both experimentally and clinically (16,17,27). Although different levels of functional benefit have been reported from studies using different animal models, delivery modes, and cell types and doses (1,12), the underlying mechanisms of the functional improvement remain poorly understood. Overall, the level of muscle cell regeneration is low (21,41). If we assume that a large infarct irreversibly causes a 30% loss of LV mass and stem cell-induced muscle regeneration is 10% of the damaged tissue, this would lead to only 3% of the total LV myocytes from stem cell differentiation. On the other hand, it has been reported that a paracrine effect plays an important role in observed beneficial effects of cellular therapy for myocardial repair (11). Further, a recent mouse study that compared marrow-derived progenitor cells for myocardial repair in immune-deficient and immune-competent mice suggests that inflammatory response may also play an important role in the paracrine effect benefiting the recipient myocardium (25). Regardless of whether the beneficial effects are due to the paracrine effect or from engrafted stem cells, the observed functional improvement must be a result of the functional changes of the native ventricular muscle structure, coronary circulation, and the pacemaker and conduction system or the integration of exogenous stem cells into the existing ventricular structure.
In the normal heart, the orientation of LV fibers changes smoothly from left-handed fibers in the subepicardium to circumferential fibers in the midwall and to right-handed fibers in the subendocardium (28,29). This unique three-dimensional structure of myocardial fibers plays an important role in determining ventricular function and the electromechanical properties of the heart (2,32). Early descriptions of myocardial fiber structure are based on histological measurements, which are destructive and time consuming (22,29). Recently, diffusion tensor magnetic resonance imaging (DTMRI) has emerged as a promising method for nondestructive reconstruction of the three-dimensional fiber structure throughout the whole ventricle at high spatial resolution (4,5). By comparing DTMRI and conventional histological analysis, several studies have demonstrated a direct correlation between the fiber angle measurements, validating the utility of DTMRI as a fast and nondestructive technique to characterize fiber architecture in the myocardium (13,14,26).
Alterations of cardiac fiber architecture after MI have been previously investigated using DTMRI (7,30,33,35). Chen et al. observed a significant increase in water diffusivity and a significant decrease in relative diffusion anisotropy in remodeled rat myocardium 1 month after infarction (7). The changes in diffusion anisotropy were found to correlate with the degree of microscopic fiber disarray. Recent studies in both humans and porcine models further confirmed these findings. In addition, a redistribution of fiber structure was observed with more left-handed fibers in infarct myocardium (33,35). Furthermore, a recent study on cardiomyopathic Syrian hamsters showed that changes in transverse diffusivity may reflect calcium deposition in the lesion areas (19). These studies demonstrate the capability of multiparametric DTMRI in delineating various aspects of structural remodeling in diseased hearts.
In the present study, we investigated the long-term effects of stem cell therapy on myofiber orientation and cardiac function in postinfarct rat hearts. Infarcted rats were treated with human nonhematopoietic umbilical cord blood stem cells (nh-UCBSCs) in the absence of immunosuppressive therapy. These nh-UCBSCs were previously shown to exhibit neurorestorative effects in a rodent model of ischemic brain injury (37). Global and regional LV function and myocardial fiber structure were characterized using cardiac functional MRI and DTMRI, respectively. Compared to the age-matched untreated rat hearts with MI, the regional myocardial function of nh-UCBSC-treated hearts was significantly improved, in conjunction with preserved myocardial fiber structure that may serve as an underlying mechanism for the observed functional improvement.
Materials and Methods
All animal protocols used in this study were approved by the Institutional Animal Care and Use Committees of both the University of Minnesota and Case Western Reserve University.
nh-UCBSCs were collected from a male donor. Informed consent for donation was obtained before collection. The use of human-derived cells was approved by the University of Minnesota.
MI Surgery and Treatment with nh-UCBSCs
Twelve male Lewis rats (250—275 g, 2—3 months old; Harlan Laboratories, Indianapolis, IN, USA) were used in the study. MI was induced in seven rats through the occlusion of the left anterior descending artery (LAD). Briefly, the animal was anesthetized in an isoflurane (Butler Schein Animal Health, Dublin, OH, USA) induction chamber (3%), intubated, and then placed on a ventilator (Harvard Apparatus, Southnatick, MA, USA). A thoracotomy was performed to expose the heart. The LAD was ligated using polypropylene suture (6-0 Prolene; Ethicon, San Angelo, TX, USA). The chest was closed, and animals were allowed to recover with postsurgical monitoring for 2 h. The remaining five sham-operated animals were used as the controls (CONT, n = 5).
The seven rats that underwent MI surgery were randomly allocated into two groups: 1) LAD ligation without any further therapy (MI, n = 3) and 2) LAD ligation with nh-UCBSC injection (MI + Cell, n = 4). The procedure to isolate nh-UCBSCs was described in detail previously (37). The isolated cells were cultured in a T25 flask (Fisher Scientific, Hampton, NH, USA) in 7 ml Dulbecco's modified Eagle's medium/nutrient mixture F-12 (Ham) 1:1 (DMEM/F12; Invitrogen, Carlsbad, CA, USA) supplemented with 1 × N2 supplement (Invitrogen), 10% fetal bovine serum (FBS; Invitrogen), 10 ng/ml recombinant human basic fibroblast growth factor (bFGF; R&D Systems, Minneapolis, MN, USA), 10 ng/ml recombinant human epidermal growth factor (EGF; R&D Systems), 100 U penicillin (Invitrogen), and 1,000 U streptomycin (Invitrogen). When cells were 70—80% confluent, they were detached with 0.25% trypsin-EDTA (Invitrogen) and split at 1:4 for future usage. Two days after surgery, nh-UCBSCs were administered via tail vein injection (106/500 μl PBS). For the rats in the MI group, the same volume of PBS (Sigma-Aldrich, St. Louis, MO, USA) without any cells was injected. None of the animals were given immunosuppressive therapy.
In Vivo MR Imaging
Ten months after the surgery, in vivo functional MRI studies were performed on a 9.4T Bruker Biospec (Billerica, MA, USA) horizontal bore scanner with a 10-cm volume coil (Bruker Biospin). Anesthesia was induced by 2% isoflurane in O2 and was maintained at 1.5% isoflurane in O2 by a nose cone built in-house during imaging. An MR-compatible small animal gating and monitoring system (SA Instruments, Stony Brook, NY, USA) was used for electrocardiography (ECG) gating, respiration gating, and temperature control. The ECG signal was obtained from two ECG electrodes that were attached to the left paw and the right leg of the rat. The respiration patterns were acquired from a respiratory pad placed under the chest wall of the animal in the prone position. A rectal temperature probe was inserted to measure the body temperature. The body temperature was maintained at 34.2 ± 1.2°C by blowing hot air into the magnet through a feedback control system. A series of scout images were acquired to obtain the LV long-axis plane. After acquiring a horizontal long-axis image (four-chamber view), the LV short-axis planes at the base, midventricle, and apex were prescribed as perpendicular to the LV long-axis with an interslice distance of 3 to 3.5 mm.
Displacement encoding with stimulated echo (DENSE) MRI was used to quantify the myocardial wall motion at high spatial resolution as previously described (43). Multiphase DENSE images were acquired at the base, midventricle, and apex for each rat. The DENSE imaging sequence used a spatial modulation of the magnetization 11 (SPAMM11) tagging preparation module with fast low-angle single-shot (FLASH) readout acquisition. To measure displacement in one direction, displacement encoding/unencoding gradients with the same magnitude, but opposite polarities, were used in two DENSE acquisitions to correct for baseline phase errors and to increase the dynamic range of the displacement encoding. A total of four cine data sets were acquired to quantify myocardial motion in two orthogonal directions. The displacement encoding gradient was chosen to yield an encoding frequency (kx) of 0.77 cycles/mm. Additional imaging parameters included the following: flip angle, 20°; echo time (TE), 3.0 ms; field of view, 5 × 5 cm2; matrix size, 128 × 128; slice thickness, 1.5 mm; number of averages, 6. Relaxation time (TR) was adjusted according to the heart rate (HR). A total of 17 frames were acquired in one cardiac cycle to yield a temporal resolution of 10~13 ms. Cine FLASH images with the same geometric parameters were also acquired at each imaging level to calculate the LV volumes and the ejection fraction (EF). The imaging parameters were flip angle, 20°; TE, 2.1 ms; number of averages, 4.
Hemodynamic Measurements with the Pressure—Volume Conductance Catheter
Hemodynamic studies were performed 1 week after the in vivo MRI scan. During the terminal surgery, a 2.0-Fr pressure—volume (PV) catheter (SPR-838; Millar Instruments, Houston, TX, USA) was inserted into the right carotid artery and advanced into the LV (23,24), and the position was optimized by aiming for maximal stroke volume (SV). A 14-gauge angiocath (BD Biosciences, Franklin Lakes, NJ, USA) was inserted into the right jugular vein for the infusion of saline boluses for calibration. After 15 min of stabilization, steady-state parameters, including HR, maximal LV systolic pressure, and LV end-diastolic pressure were recorded using MPVS-400 signal conditioning hardware with integrated AD Instruments PowerLab DAQ Technology (AD Instruments, Mountain View, CA, USA). Maximal slopes of systolic and diastolic pressure (+dP/dt and −dP/dt) were calculated using PVAN 3.2 software (Millar Instruments) (23,24).
Ex Vivo Diffusion Tensor Imaging
After hemodynamic measurements, the heart was excised and fixed for DTMRI study as previously described (7,19). Briefly, the heart was first perfused with Krebs buffer (Sigma-Aldrich) and then arrested in diastole by perfusion with a 50-ml solution of KCl (20 mM; Sigma-Aldrich) and NaCl (120 mM; Sigma-Aldrich). The perfusate was then switched to 10% formalin (Fisher Scientific) for rapid fixation. The procedures were all performed at room temperature (20°C). The fixed heart was then taken off the perfusion column, immersed in formalin, and stored in the refrigerator at 4°C for further fixation. One day before imaging, the heart was taken out from the formalin solution, rinsed, and suspended in 1 × PBS.
Diffusion-weighted, short-axis images of the fixed hearts were acquired in a 9.4T vertical bore magnet (Bruker Biospin) with a 2-cm birdcage coil (M2M Imaging, Cleveland, OH, USA). The acquisition utilized a standard Stejskal—Tanner spin-echo sequence. Seven to nine short-axis images were acquired to cover the whole LV. Imaging parameters included the following: TR, 2.5 s; TE, 18 ms; diffusion time (D), 20 ms; diffusion gradient on time (d), 10 ms; number of diffusion-encoding directions, 6; b, 800 s/mm2; field of view, 2 × 2 cm2; matrix size, 128 × 128; slice thickness, 1 mm; number of averages, 8. The total acquisition time for each heart was about 5 h.
Image Reconstruction and Data Analysis
In vivo MR images were reconstructed and analyzed offline using an in-house developed, MATLAB-based image analysis tool package with a custom-designed graphic user interface. LV contours were traced using cine FLASH images to calculate LV diameter, wall thickness, and LV volume. The LV EF was then calculated from LV end-diastolic volume (EDV) and end-systolic volume (ESV) as previously described (42).
The image processing and displacement calculation from DENSE images were described in detail previously (43). Briefly, a k-space filter with a cutoff frequency of 0.48 cycles/mm was used to eliminate the T1-relaxation echo and the residual complex conjugate echo. The raw data was zero filled, and a 2D inverse Fourier transform was applied to generate the complex images. The two phase images obtained from the same direction, but opposite-encoding gradient polarities, were subtracted to eliminate baseline phase errors. Subsequently, displacement-encoded phase images were unwrapped using an algorithm similar to the 2D quality-guided path-following method. The 2D displacement map was calculated, and the Lagrangian strain tensor, E, was computed within each four adjacent-pixel element from the 2D displacement as previously described (43). LV twist angles and torsion were then quantified from the displacement. The twist angle was defined as the rotation of each voxel compared to its original position at end-diastole. Torsion was calculated as the net twist between apical and basal slices normalized by the interslice distance (20).
The ex vivo diffusion-weighted images were transferred to a PC for analysis with an in-house developed MATLAB software (7). The three eigenvalues of the diffusion tensor, λ1, λ2, λ3, and their corresponding eigenvectors were calculated on a pixel-by-pixel basis for the entire LV. All the eigenvalues were then normalized to the mean diffusivity of the surrounding PBS solution. The mean diffusivity (λ) was computed as the average of the three normalized eigenvalues. The fractional anisotropy (FA) was used to quantify the diffusion anisotropy. The myofiber helix angle was further determined from the primary diffusion eigenvector to delineate myocardial fiber orientation as previously described (7). Angular dispersion, which is defined as the standard deviation of the helix angles, was used to evaluate the coherence of fiber orientation at each transmural depth. For the MI group, four or five slices that cover the infarct region in the apex and midventricle were selected. The results were compared to the data obtained from similar regions of the LV in both CONT and MI + Cell groups.
The myocardium in diffusion-weighted images was divided into three segments, that is, infarct, border, and remote zones (Fig. 1) (33). The infarcted myocardium was identified as the region with reduced wall thickness in the MI group (Fig. 1a). In the MI + Cell rats, no obvious wall thinning was observed, and a segment of anterior myocardium with the same size as the average infarct size in MI rats was selected as the infarct segment. The rest of the myocardium was divided into six equiangular segments. The two segments that were adjacent to the infarct region were classified as the border zones, and the four remaining sections were defined as the remote zones. In the CONT rats, sham infarct, border, and remote zones were defined similarly as the MI + Cell rats (Fig. 1c). The ventricular geometry in DENSE images was correlated to DTMRI images based on the position of the right ventricle and papillary muscles (Fig. 1b, d).

Segmentation of LV into three segments. Segmentation of the LV into the infarct, border, and remote zones in a fixed (a) and in vivo (b) heart from the MI group and a fixed (c) and in vivo (d) heart from the control group.
Immunohistochemistry Analysis
The hearts were fixed in 10% formalin, processed, and paraffin embedded. Paraffin sections of 5-μm thickness were used for immunohistochemistry studies. To visualize the scar tissue, tissue sections underwent trichrome staining (Sigma-Aldrich) as per instructions. To quantify the infarct zone, trichrome-stained heart sections were scanned. The percentage of infarction was calculated as the ratio of the infarct area to LV free wall area (not including LV septal wall). The percentage of wall thickness in the infarct zone was calculated as the ratio of the wall thickness in the infarct region to the average LV free wall thickness of normal control rats (n = 3 or 4 animals in each group). The percentage of fibrosis in the infarct zone was calculated as the ratio of the numbers of pixels representing fibrotic and total myocardial tissue, respectively.
V-Kit Hardy—Zuckerman 4 feline sarcoma viral oncogene homolog positive (c-Kit+) cardiac progenitor cell (CPC) density was calculated as the number of total c-Kit+ CPCs divided by LV free wall area (cm2). CPCs were quantified at 400× magnification in eight microscopic fields/heart (n = 3 or 4 animals in each group) after double fluorescent immunostaining for c-Kit (1:50 goat anti-c-Kit IgG primary antibody; R&D Systems) and α-sarcomere actin (a-SA, 1:100 mouse anti α-SA IgM primary antibody; Sigma-Aldrich) expressions followed by 1-h incubation with donkey anti-goat IgG conjugated with fluorescein isothiocyanate (FITC; 1:100) and donkey anti-mouse IgM conjugated with tetra rhodamine isothiocyanate (TRITC; 1:200) (Jackson ImmunoResearch, West Grove, PA, USA). All tissue sections were counterstained with 0.2 μg/ml 4, 6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich) to visualize the nucleus.
The blood vessel density at the infarct and border zones was measured at 400× magnification in eight microscopic fields/heart (n = 3 or 4 animals in each group) after double fluorescent immunostaining for von Willebrand factor—coagulation factor 8 (1:100 rabbit anti-vWF-VIII IgG primary antibody; Dako, Glostrup, Denmark) and smooth muscle actin (SMA, 1:500 mouse anti-SMA IgG conjugated with TRITC; Sigma-Aldrich) expressions followed by 1-h incubation with 1:100 donkey anti-rabbit IgG conjugated with FITC (Jackson ImmunoResearch, West Grove, PA, USA).
Statistical Analysis
All the results are presented as mean ± SD. Comparisons for multiple groups were performed with one-way ANOVA followed by Tukey's test. Significance of differences was assigned at p < 0.05.
Results
Animal Characteristics and Global Cardiac Function
The morphological and physiological data of the animals are presented in Table 1. The body weight and the LV length were similar among all three groups. Significant increases in LV diameter and EDV were observed in both MI and MI + Cell groups, which suggests LV dilation. Compared to the MI group, the ESV in the MI + Cell group decreased by 21%. As a result, LV EF in nh-UCBSC-treated rats was significantly improved (p < 0.05) and was similar to that of the CONT rats (p = N.S.).
Body Weights and Cine Magnetic Resonance Imaging (MRI) Data

MI + Cell, myocardial infraction + cell treatment; EDV, end-diastolic volume; ESV, end-systolic volume; EF, ejection fraction; LV, left ventricle; bpm: beats per minute.
p< 0.05 compared to control (CONT) group.
p < 0.05 compared to myocardial infarction only (MI) group.
End-diastolic wall thickness at the apex, midventricle, and base was measured from in vivo cine images (Table 2). In the MI group, wall thickness in the infarct zone was reduced by 50% compared to the CONT rats. In addition, wall thickness at the border zone was also significantly decreased compared to the CONT rats. In the MI + Cell group, no statistical difference in wall thickness was observed compared to the controls.
End-Diastolic Wall Thickness (mm) Assessed From In Vivo Cine MRI

p < 0.05 compared to CONT group.
p < 0.05 compared to MI group.
Hemodynamics Under Steady-State Conditions
Steady-state in vivo hemodynamic properties assessed using a PV conductance catheter are reported in Table 3. Peak LV +dP/dt and −dP/dt decreased significantly in the MI group compared to the CONT group. However, ES pressure and ED pressure were not different. EF was also significantly reduced in the MI group. In contrast, no hemodynamic changes were observed in the MI + Cell group. EF in MI + Cell rats was also similar to that of the CONT rats, which is consistent with the in vivo MRI measurements (Table 1).
Steady-State Hemodynamic Properties Assessed by In Vivo PV Conductance Catheter

PV, pressure—volume; ES, end-systolic; ED, end-diastolic; EF, ejection fraction.
p < 0.05 compared to CONT group.
p < 0.05 compared to MI group.
Myocardial Strain and Torsion
Figure 2a and b shows the radial (Er) and circumferential (Ec) strains in the infarct, border, and remote zones at apex. Owing to the significant wall thinning in MI rats, myocardial strains in the infarct zones cannot be reliably quantified. Compared to the CONT group, a significant decrease in both radial and circumferential strains was observed in the border zone of the MI group. However, in nh-UCBSC-treated hearts, wall strains were similar to the controls in the infarct, border, and remote zones. At the midventricular and basal levels, no significant differences in myocardial wall strains were observed among all three groups (data not shown).

Characteristics of in vivo cardiac function. Radial (a) and circumferential (b) strains at apex and ventricular torsion (c) in control (CONT), MI only (MI), and MI + Cell groups. †Myocardial strains in the infarct zone of MI rats were not quantified due to significant wall thinning. *p < 0.05 CONT versus MI. #p < 0.05 MI versus MI + Cell.
Improved ventricular function in nh-UCBSC-treated rats was further demonstrated by the measurement of ventricular torsion (Fig. 2c). At peak systole, LV torsion was 23.6 ± 2.6°/cm for the CONT group. It decreased significantly to 14.3 ± 1.8°/cm in the MI group (p < 0.05). However, torsion in the MI + Cell hearts (21.2 ± 4.9°/cm) was similar to that of the controls, a 48% increase compared to the MI group.
Myocardial Fiber Structure
The histograms of the helix angle distributions are presented in Figure 3. Compared to the control rats, there was a significant reduction in the percentage of right-handed fibers (30° to 90°) in all three zones of the untreated hearts, suggesting a loss of endocardial fibers. As a result, the percentage of circumferential fibers (-30° to 30°) increased by 18.0%, 22.8%, and 26.9% in the infarct, border, and remote zones, respectively. The percentage of left-handed fibers (-90° to −30°) in the remote zone also decreased significantly in MI rats. The nh-UCBSC treatment prevented such a shift toward more circumferential orientation in all three zones. No difference was observed in the percentage of the three types of fibers between the control and the therapy group.

Fiber helix angle distribution in fixed hearts. Histograms of fiber helix angles in the infarct (a), border (b), and remote (c) zones for CONT, MI, and MI + Cell groups. *p < 0.05 CONT versus MI. #p < 0.05 MI versus MI + Cell.
Increased angular dispersion of the helix angle was observed in the infarct zone for both MI and MI + Cell rats. Angular dispersion ranged from 11.2° to 17.0° in the sham “infarct” zone for the CONT group. In the infarct zone, the range of angular dispersion increased to 22.8°~ 28.7° for the MI group and 16.1°~21.7° for the MI + Cell group. However, in the remote zone, the range of angular dispersion was similar among all three groups (CONT, 10.9°~22.8°; MI, 13.0°~17.8°; MI + Cell, 12.4°~23.3°).
In addition to the changes in fiber helix angles, water diffusivity was also significantly reduced in the nh-UCBSC-treated hearts compared to the MI group (Fig. 4a). No significant difference in mean diffusivity was observed between the CONT and the MI + Cell group (p = N.S.). Further analysis showed that all three eigenvalues in the infarct zone of the MI + Cell group were significantly reduced compared to the MI group, which contributed to the overall decrease of the mean diffusivity (Table 4). In addition, the primary eigenvalue (λ1) in both the border zone and remote zone were also significantly decreased with nh-UCBSC treatment compared to untreated MI rats (Table 4).

Diffusivity and FA in fixed hearts. Mean water diffusivity (a) and fractional diffusion anisotropy (FA) (b) in the infarct, border, and remote zones for the CONT, MI, and MI + Cell groups. *p < 0.05 CONT versus MI. #p < 0.05 MI versus MI + Cell.
Three Normalized Eigenvalues in the Infarct, Border, and Remote Zones for Control, Untreated, and Therapy Groups

p < 0.05 compared to CONT group.
p < 0.05 compared to MI group.
Figure 4b shows the fractional diffusion anisotropy (FA) in the infarct, border, and remote zones in all three groups. Compared to the control rats, a trend toward decreased FA was observed in the MI group in the infarct and border zones by 21.1% and 18.9%, respectively. A similar trend of reduction was also observed in nh-UCBSC-treated hearts. No evident difference in FA was observed in the remote region among all three groups.
Histological Analysis
Masson's trichrome staining showed reduced fibrosis in rat hearts treated with nh-UCBSCs (Fig. 5a—f). Although the area of scar zone was similar between the two groups (Fig. 5g), significantly increased wall thickness was found in the MI + Cell group (Fig. 5h). The MI + Cell group also showed significantly reduced fibrosis in the infarct zone (Fig. 5i). There is no difference in c-Kit+ CPCs between the MI and MI + Cell groups (Fig. 6). Compared to the MI group, a trend of an increase in the number of blood vessels was observed (Fig. 7).
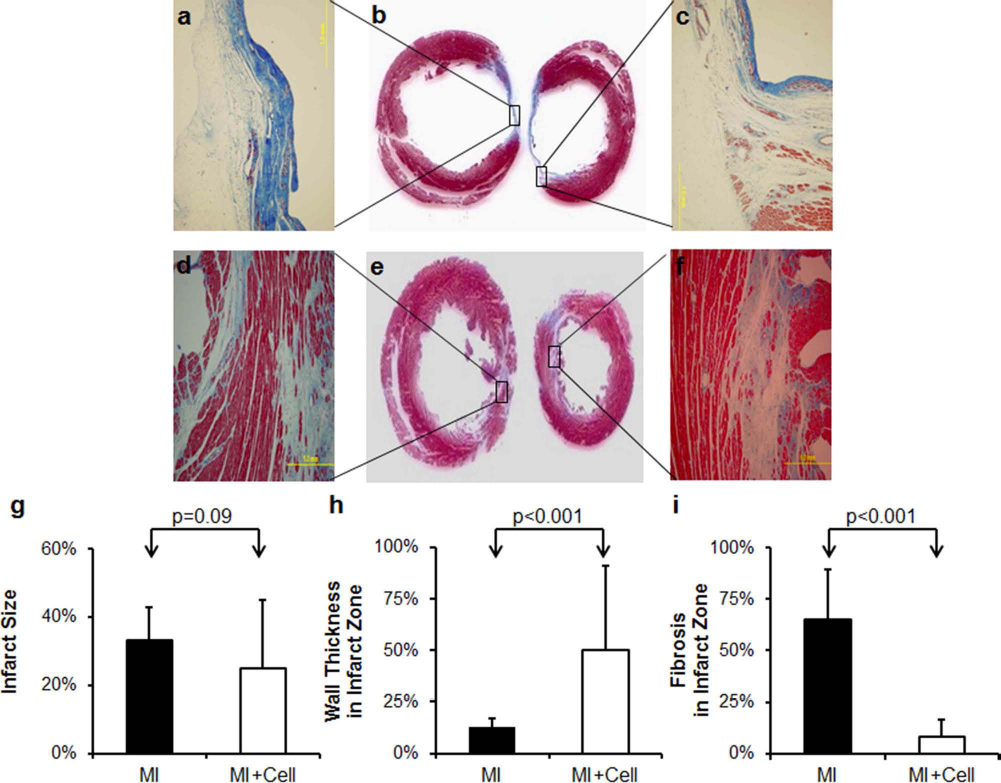
Masson's trichrome staining of fixed hearts. (a, c) Magnified pictures of scar zone of rat hearts from the MI group (b). (d, f) Magnified pictures of scar zone of rat hearts from the MI + Cell group (e). Color representation: nuclei, black; cytoplasm, red; muscle fiber, red; collagen, blue (scale bar: 1 mm). (g) Percentage of infarct area. (h) Percentage of wall thickness in infarct zone. (i) Percentage of fibrosis in infarct zone.

Quantification of c-Kit+ CPCs. (a) Representative picture from a heart in the MI group. (b) Representative picture from a heart in the MI + Cell group. (c) Quantification of V-Kit Hardy—Zuckerman 4 feline sarcoma viral oncogene homolog-positive (c-Kit+) CPCs (scale bar: 100 μm). α-SA, α-sarcomere actin; DAPI, 4′,6-diamidino-2-phenylindole.
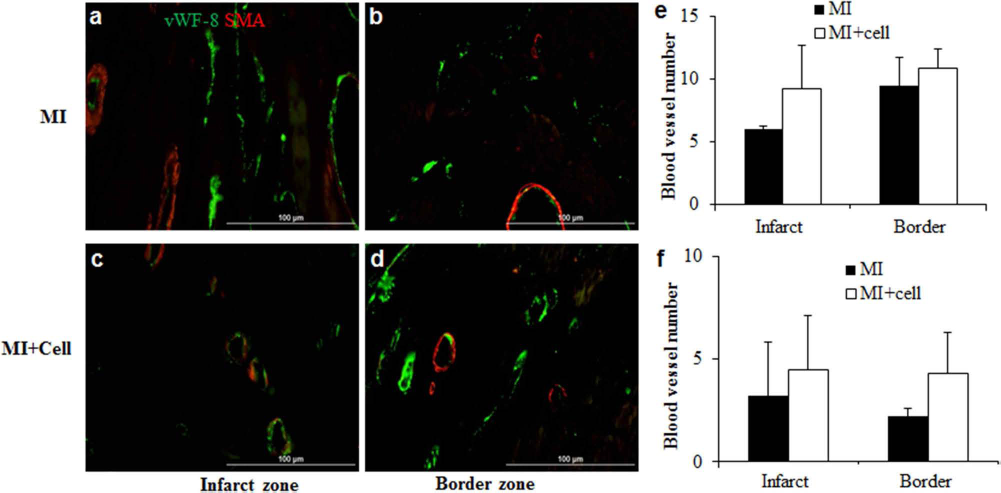
Dual fluorescent immunostaining for blood vessel (arterioles) density quantification. Representative pictures of blood vessel density in infarct zone (a) and border zone (b) of the MI group. Representative pictures of blood vessel density in infarct zone (c) and border zone (d) of the MI + Cell group. Quantification of blood vessel density (number/400× magnification field) was based on immune-staining images for von Willebrand Factor—coagulation factor 8 (vWF-8; e) and smooth muscle actin (SMA; f). Scale bar: 100 μm.
Discussion
Stem cell therapy for myocardial repair has been investigated extensively for the past 10 years. Although different levels of improvement in LV function have been reported in response to different cell types, LV injury model, or cell delivery modes, the underlying mechanisms of the beneficial effects are not fully defined. In the current study, the long-term effects of stem cell therapy on both ventricular function and myocardial fiber structure were investigated in postinfarct hearts. In addition to improved contractile performance, nh-UCBSC-treated hearts demonstrated preserved right-handed fibers that were similar to that in control hearts. While previous study reported preserved global ventricular structure by stem cell treatment (10), our results demonstrate for the first time that improvement in LV contractile function is associated with preserved myofiber orientation. The preserved structure observed with nh-UCBSC treatment may provide the structural basis for improved cardiac function in postinfarct hearts.
Global and regional LV function was significantly improved with nh-UCBSC treatment. This functional improvement has been reported in many studies using various imaging methods, including MRI (1,12). However, most of these studies focused on the short-term effects of stem cell treatment on myocardial function, while the prolonged effect remains to be investigated. In the current study, our results demonstrate the long-term effects of improved cardiac function in postinfarct myocardium with nh-UCBSC treatment. Both MI and MI + Cell groups showed significantly increased EDV (Table 1), which may lead to improved EF by increasing diastolic filling. However, the nh-UCBSC-treated hearts also showed decreased ESV compared to untreated hearts, suggesting that improved systolic contractility also contributed to the observed increase in EF. Further, nh-UCBSC-treated hearts showed increased peak LV +dP/dt and −dP/dt compared to the MI group (Table 3), providing further evidence of improved systolic and diastolic function.
During the MRI experiments, we typically observed a 1~2°C fluctuation in body temperature measured by a rectal probe. To avoid overheating the animals, the temperature control system was set to 35°C during MRI scanning, leading to an average body temperature of 34.2 ± 1.2°C. A previous study in rats reported that body temperature measured by rectal probe was typically lower (~0.6°C) than body temperatures recorded by chronically implanted electrodes, with the difference being smaller at low and greater at high basal temperatures (3). Further, the observed torsion and strain values in the current study were similar to that from a previous study when the temperature was maintained at 35.8 ± 0.8°C (8).
The unique three-dimensional architecture of myocardial fibers renders the heart an efficient pump. Previous studies employing mathematical modeling demonstrated that a 15% shortening of circumferentially oriented fibers can only induce ~30% reduction in ventricular volume from end-diastole to end-systole; however, obliquely oriented fibers can lead to more than 60% volume reduction (15). In addition, the twisting motion and torsion of the LV is a consequence of the obliquely oriented fiber structure. Therefore, the efficacy of cellular therapy relies on the preservation of the structure of ventricular myocardium. In the current study, examination of myofiber structure with DTMRI revealed that nh-UCBSC treatment can preserve fiber orientation within the myocardium, as demonstrated by the similar percentage of left-handed, circumferential, and right-handed fibers compared to the control hearts. This preserved structure contributed to decreased ESV (Table 1) and elevated ventricular torsion (Fig. 2c) in nh-UCBSC-treated hearts, leading to normalized EF.
Postinfarct myocardial remodeling is a prolonged process that involves changes in both cell types and structure. In the infarct zone, different cell types exist at different stages of the remodeling process. In the early stage, macrophages differentiated from monocytes in the blood are first recruited to the injury site to remove necrotic cardiac myocytes and apoptotic neutrophils from the myocardium. Later, more fibroblasts and endothelial cells will emerge during the wound healing process. Once the scar is formed, it will gradually expand to the border zone and induce global remodeling of the whole heart (18,39). These cellular remodeling processes can impact DTMRI measurements of myocardial structure.
Previously, DTMRI has been applied to study myofiber structure in both humans and animals (6,7,9,13,14,19, 30,33,35). Several studies also examined the fiber structure in postinfarct myocardium (7,30,33,35). Consistent with previous findings in both human and rodent studies, we observed reduced right-handed fibers and increased angular dispersion in untreated infarct hearts (7,34,35). These changes are likely due to the loss of viable myocytes in the subendocardial area after the infarction. As a result, the percentage of circumferential fibers was increased. Interestingly, this redistribution of myocardial fibers occurred not only in the infarct zone, but also in the border and remote zones, suggesting that the whole myocardium underwent structural remodeling as a result of long-term postinfarct remodeling. While the fiber redistribution in all three zones was consistent with a previous ex vivo study in a porcine model (33,36), our current finding of decreased left-handed fibers in the remote zone has not yet been reported. Further investigations are needed to elucidate whether this reduction is due to the long-term remodeling after infarction.
In addition to fiber orientation, we also found preserved water diffusivity in nh-UCBSC-treated rats compared to the controls, while untreated hearts showed significantly increased water diffusivity in the infarct and border zones. The exact mechanisms for the preserved diffusivity in nh-UCBSC-treated hearts are not completely understood. We have previously shown that the increased water diffusivity was associated with enlarged extracellular space following cell death (7). It is possible that nh-UCBSC treatment and the subsequent paracrine effect caused less cell death and limited the expansion of extracellular space. However, this hypothesis needs to be examined at an earlier time point, as myocardial apoptosis is expected to be very low 10 months after acute infarction. Another possibility is that the integration of nh-UCBSCs in the infarct myocardium replenished the extracellular space after initial cell death. However, the sparseness of nh-UCBSCs surrounding the regions of ischemic injury makes this an unlikely possibility (Fig. 6b).
In spite of the preserved fiber orientation and water diffusivity, similar decreases in diffusion anisotropy were observed in the infarct zone in both untreated and treated hearts. In addition, nh-UCBSC-treated hearts also showed increased angular dispersion compared to the controls. Changes in cell morphology, extracellular space, and fiber orientation can all lead to alterations in the diffusion anisotropy. Our previous study demonstrated that decreased anisotropy and increased angular dispersion in infarcted myocardium were associated with microscopic fiber disarray and extracellular space expansion (7). Given that the water diffusivity in nh-UCBSC-treated hearts was similar to that of the controls, the reduced diffusion anisotropy was unlikely due to an enlarged extracellular space. Hence, the decreased diffusion anisotropy and increased angular dispersion suggest fiber disarray in nh-UCBSC-treated hearts.
In the current study, human nh-UCBSCs were used in immune-competent rats without immunosuppressive agents. Previously, we have used human bone marrow-derived stem cells on both immune-deficient [nonobese diabetic/severe combined immunodeficient (NOD/SCID)] and immune-competent mice with acute myocardial infarction. Our results show that although the engraftment rate is significantly lower in immune-competent mice, the beneficial effects on LV function are significantly better for the immune-competent mice than the NOD/SCID mice, suggesting a cytokine effect (31). In another study using a swine model of postinfarct LV remodeling (40), we compared animals with or without immunosuppression cyclosporine A (CsA, 15 mg/kg/day). The results of that study demonstrate that mesenchymal stem cells have an immune-privilege window of at least 4 weeks and that cellular therapy-induced beneficial effects on LV function were similar between groups with or without immunosuppression.
No significant increase in the number of c-Kit+ cells was observed in nh-UCBSC-treated hearts (Fig. 6). Although it is possible that the c-Kit marker of the engrafted nh-UCBSCs was silenced during differentiation over a period of 10 months, the observed functional benefits are more likely related to the mobilization of the endogenous c-Kit+ cells induced by cellular therapy, which was observed in our recent study using a swine model treated with human embryonic stem cell-derived vascular cells (38).
One limitation of the current study is the low heart rate in both MRI and hemodynamic studies (Tables 1, 3). As myocardial contractility is impacted by HR, the measurements acquired at lower HR may deviate from the normal baseline. Another limitation of this study is the small number of animals in each group. Owing to the high mortality in older rats and the cost of maintaining the animals for a long period of time, only three to five animals were examined in each group. The small sample size may reduce the power of statistical analysis to detect a difference. Such may be the case in anisotropy measurement. While our current study only showed a trend toward decreased diffusion anisotropy in the infarcted hearts, the observed values in both the control and the untreated groups were similar to those reported in the literature where statistical difference was detected (33,36). Furthermore, even with the small sample size, significant changes in both cardiac function and myofiber orientation were still observed with nh-UCBSC treatment.
Conclusions
In conclusion, we investigated the long-term effect of stem cell therapy on myocardial function and myofiber orientation in the postinfarct rat heart. While the functional data suggested significantly improved global and regional functions in nh-UCBSC-treated hearts, DTMRI studies showed that nh-UCBSC treatment may have the additional benefit of preventing/delaying untoward structural remodeling in postinfarcted hearts. These data demonstrate a novel finding that nh-UCBSC treatment preserves myocardial fiber structure that supports the improved LV regional and chamber function.
Footnotes
Acknowledgments
This work was supported by National Heart, Lung, and Blood Institute Grants R01 HL73315 and HL86935 (to X. Yu), HL50470, HL 67828, HL 95077, HL100407 (to J. Zhang), American Heart Association postdoctoral fellowship 09POST2080107 (to Y. Chen), and a Graduate School Grant-in-Aid from the University of Minnesota (to W. C. Low). The authors declare no conflicts of interest.
