Abstract
Olfactory ensheathing cells (OECs), the only glial cell type that normally penetrates the transition zone between the peripheral and central nervous systems, are one of the most promising candidates for cell transplantation in repairing spinal cord injury (SCI). However, we must manipulate and regulate OECs' behavior to make these cells effective in cell transplantation. In the present study, we assessed the response of rat OECs to different variants of nanofibrous silk fibroin mats with regard to cell morphology, adhesion, proliferation, and migration and the related gene and protein expression. Results showed that OECs adhere and spread more easily on Tussah silk fibroin (TSF) fibers than Bombyx mori silk fibroin fibers, resulting in a higher rate of cell proliferation and gene and protein expression, examined by RT-PCR and ELISA. In addition, the morphology of OECs on microfibers is mainly polygonal with short protrusions, whereas the OECs on nanofibers exhibit a bipolar shape with long protrusions that align along the fibers, especially when aligned fibers are employed. Moreover, OECs on silk fibroin nanofibers migrate more efficiently than those on poly-l-lysine (PLL). Based on the experimental results, the morphology, adhesion, spread, gene and protein expression, and migration of OECs could be modulated and regulated by adjusting the contents and structure of silk fibroin nanofibers, shedding light on the control of OECs' behavior in nerve tissue engineering and thus the future therapeutic intervention for nerve repair after injury. This manuscript is published as part of the International Association of Neurorestoratology (IANR) supplement issue of Cell Transplantation.
Keywords
Introduction
More than 11,000 traumatic spinal cord injuries (SCI) occur in the US each year (11). Unfortunately, the repair of SCI continues to be a major challenge, and no effective therapies are currently available (19,23). Cell transplantation is one of the most promising therapeutic approaches to promote axon regeneration (25). In particular, olfactory ensheathing cells (OECs) are excellent candidates for this therapeutic approach as they are able to remyelinate the demyelinated axons and support regrowth of transected axons after transplantation to repair the adult SCI (12). However, it is necessary to be able to manipulate and regulate the OECs' behavior to make them effective in cell transplantation.
The interaction of mammalian cells with nanoscale topography has been proven to be an important signaling modality in controlling cell shape and function (3). The naturally derived component of the extracellular matrix (ECM) provides the necessary biological properties in modulate biomechanical cues and cell–matrix interactions and thereby direct homogenous differentiation into targeted tissues (7,14,24). Studies show that ECM scaffolds promoted OEC adhesion, proliferation, and process extension (18,22).
Electrospinning is a versatile technique that is used to produce nanofiber-based biomaterial scaffolds. The scaffolds have promising applications in tissue engineering and regenerative medicine since they mimic the ECM topography in vivo (13). Many researchers have reported the biocompatibility of cells and electrospun silk fibroin fibers and concluded that silk fibroin nanofiber has positive effects on cell adhesion, morphology, viability, proliferation, and differentiated functions (32). Previously, we found that OECs remained viable with original morphology and phenotype on Bombyx mori silk fibroin (BSF) fibers (22). In recent years, several studies have indicated that nanofiber diameter and alignment, chemical compositions, and structures of the biomaterial scaffolds had a significant effect on cell behaviors, including cell proliferation and differentiation (4,16,27). However, there is scarcely any data about the interaction between OECs and the biomaterial scaffolds, even though silk fibroin is a widely used biomaterial in tissue engineering. The aim of this work was to map OEC morphologies and alignment, to determine the effect of composition and topography of silk fibroin nanofiber on OECs' behavior, and then to develop an implantable silk fibroin scaffold seeded with OECs to foster an environment for spinal cord regeneration.
Materials and Methods
Fabrication and Processing of BSF and TSF Fibers
Bombyx mori silk fibroin (BSF; Huajia Group, Suzhou, China) fibers and Tussah silk fibroin (TSF; Huajia Group) fibers, aligned and randomized with varying diameter, were fabricated via electrospinning with a homemade stainless steel cylindrical collector, as in our previous reports (28,30). The resulting silk fibroin fibers were collected on 24 × 24 mm coverslips (Hengtai Glass Instrument Factory, Yancheng, China) used for cell culture study after sterilization by γ-ray irradiation (Soochow University, Suzhou Jiangsu, China). For posttreatment, the electrospun silk fibroin fibers were immersed in 75% (v/v) ethanol for 1 h and then dried in air.
Morphology and Structure of Silk Fibroin Fibers
The morphology of electrospun BSF and TSF fibers was observed using a scanning electron microscope (SEM; Hitachi S-520, Tokyo, Japan) at 20°C, 60 RH. Samples were mounted on a copper plate and sputter-coated with a gold layer 20–30 nm thick prior to imaging. To assess the average diameter, over 100 individual fibers were measured with NIH Image J software (NIH, Bethesda, MD, USA) using SEM images from at least five different sections of each sample.
Fourier transform infrared (FTIR) spectra were obtained using a Magna spectrometer (NicoLET5700, Thermonicolet, Madison, WI, USA) in the spectral region of 400–4,000 cm−1. The powdered silk fibroin fibers were pressed into potassium bromide (KBr, Sanxing Chemical Co., Ltd., Shandong, China) pellets prior to data collection.
Cell Seeding and Culture
Primary cultures of OECs were prepared from 1-monthold male Sprague–Dawley rats (Experimental Animal Center of Soochow University) as reported previously (22). All experiment procedures were carried out in accordance with the regulations of and were approved by the administration of affairs concerning experimental animals of Soochow University. The complete culture medium for OECs consists of Dulbecco's modified Eagle's medium (DMEM)/F-12 (Gibco, Grand Island, NY, USA), with 10% fetal bovine serum (FBS; Gibco), 1% glutamine (Sigma, St. Louis, MA, USA), and 2% penicillin–streptomycin (Hayao, Haerbin, China). After 10 days in culture, OECs were used for biocompatibility evaluation with SF nanofibers. The OECs were trypsinized by 0.125% trypsin (Sigma), centrifuged, and resuspended. Then the OECs were seeded at a density of 1.0 × 105 cells on the culture medium of prewetted BSF and TSF fibers or PLL (Sigma)-coated coverslips in 35-mm Petri dishes (Corning Inc., Corning, NY, USA). The fibers were each incubated in 1 ml of complete culture medium at 37°C, 30 min before cell seeding. One milliliter of cell suspension was placed in each dish (only on the fibers), and 1 ml culture medium was added after 4 h. The culture medium was changed every 3 days. Cells were observed every day using inverted phase-contrast microscope.
Cell Proliferation Assay
The OECs were trypsinized by 0.125% trypsin, centrifuged, resuspended, and then seeded at 1 × 104 cells/well on 96-well microtiter plates (Corning Inc.) to measure proliferation. The proliferation of OECs on BSF, TSF fibers, and PLL was examined at days 1, 3, 5, and 7. At each time point, 20 μl 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma) dissolved in PBS (Sigma) at 5 mg/ml was added to each well to continue incubation at 37°C for 4 h. Then 200 μl dimethyl sulfoxide (DMSO; Sigma) was added to each well to dissolve the dark blue crystals. The absorbance was measured at 490 nm with a reference wavelength of 570 nm in a microplate reader (Bio-Rad, Hercules, CA, USA). Cell number was correlated to optical density (OD).
Cell Immunostaining
The OECs were seeded at a density of 1.0 × 105 cells on the BSF and TSF fibers or PLL-coated coverslips in 35-mm Petri dishes. After 5 days in culture, the OECs on BSF and TSF fibers and PLL were confirmed by immunostaining. Cells on fibers and PLL were fixed for 30 min at room temperature with 4% paraformaldehyde (Zhongde, Tianjin, China) and blocked for 30 min in PBS containing 3% bovine serum albumin (BSA; Sigma), 0.2% Triton X-100 (Sigma), and 0.02% NaN3 (v/v) (Sigma). Cells were incubated with rabbit polyclonal antibody nerve growth factor receptor (NGFR) p75 (Wuhan Boster, Wuhan, China) diluted 1:200 in PBS at 4°C overnight. After three washes with PBS, cells were incubated with fluorescein isothiocyanate FITC-conjugated goat anti-rabbit IgG (Boster) secondary antibodies diluted 1:70 in PBS for 1 h at room temperature. Finally, cells were washed three times with PBS, and then cell nuclei were stained with Hoechst 33258 (Sigma) diluted 1:100 in PBS. Coverslips were washed and mounted with 50% glycerine (Fuyi, Zhengzhou, China) in PBS before imaging (Fluorescence microscope AF6000, Leica, Wetzlar, Germany).
Cell Viability and Morphology
In order to quantitatively assess cell viability, a live/dead viability/cytotoxicity kit (Invitrogen, Carlsbad, CA, USA), consisting of calcein AM and ethidium homodimer-1 (Ethd-1), was used at days 1, 3, and 5 on BSF and TSF fibers and PLL in 35-mm Petri dishes. Calcein AM was converted to calcein by the intracellular esterase present in live cells, resulting in a bright green fluorescence. EthD-1 could penetrate damaged membranes and bind to nucleic acids of dead cells, causing intense red fluorescence. The medium was removed, and the cells were washed with PBS after incubating for 30 min with 1 ml DMEM containing 0.5 mM calcein AM and 2 mM EthD-1 for each well, prior to analysis via fluorescent microscopy. About 3,000 total cells per dish were counted from randomly selected fields.
RT-PCR Analysis
Total RNA was isolated from cells using a total RNA kit (Omega Bio-Tek, Norcross, GA, USA) following the manufacturer's instructions. Briefly, OECs cultured on silk fibroin fibers and PLL in 35-mm Petri dishes for 4 days were washed twice by PBS, harvested by trypsinization, and then transferred into 2-ml Eppendorf tubes (Eppendorf, Hamburg, Germany). The tubes were centrifuged at 15,000 × g for 5 min. The supernatants were removed before the pellets were lysed in 350 μl TRK Lysis Buffer (Omega Bio-Tek) with 2% β-mercaptoethanol (Omega Bio-Tek). The supernatants of lysates after centrifugation were collected with 2-ml Eppendorf tubes, and 350 μl of 70% ethanol (v/v) was added to the supernatants. Then, a Hibind RNA mini column was applied to the centrifuge to digest (DNase I), wash (RNA Wash Buffers I and II, respectively), and elute RNA (All Omega Bio-Tek). cDNA synthesis and PCR amplification were performed as follows. For reverse transcription, 4 μl of total cellular RNA was used as templates in a 10-μl reaction containing 0.5 μl dNTP (10 mM), 1 μl Oligo dT (50 μM), and 0.25 μl Moloney Murine Leukemia Virus Reverse Transcriptase (RTase M-MLV; 200 U/μl) (TaKaRa, Dalian, China). The reaction was performed at 42°C for 1 h and 72°C for 15 min. For PCR amplification, 2 μl cDNA was used as a template with the appropriate primers, and PCR conditions were as follows: 94°C for 5 min, 30–35 cycles of 94°C for 30 s, 55–59°C (depending on primers used) for 30 s, 72°C for 60 s, and 72°C for 10 min. β-Actin mRNA served as an internal control. PCR products were visualized on 1% agarose gels (Invitrogen) stained with ethidium bromide (Invitrogen). The primers were synthesized at Sangon Company (Shanghai, China) (Table 1).
The Sequences of Rat Primers Designed and Used for RT-PCR Experiments

MBP, myelin basic protein; NGF, nerve growth factor; GDNF, glial-derived growth factor; BDNF, brain-derived neurotrophic factor; NCAM, neural cell adhesion molecule; VEGF, vascular endothelial growth factor.
ELISA Analysis
OEC culture media were collected for analysis of neurotrophic factors. Myelin basic protein (MBP), nerve growth factor (NGF), glial-derived growth factor (GDNF), vascular endothelial growth factor (VEGF), neural cell adhesion molecule (NCAM), and brain-derived neurotrophic factor (BDNF) levels in culture media secreted by OECs on silk fibroin fibers and PLL were determined using ELISA Kits (Boster). The assays were performed following the manufacturer's instructions. Briefly, standard and test samples (4-day culture medium) were incubated for 90 min at 37°C in 96-well plates. The chromogenic substrate solutions of tetramethyl benzidine (TMB chromogenic buffer) were added to the plates, and the reaction stopped by adding the TMB stop buffer. The colors were measured at 450 nm in a microplate reader (Bio-Rad). Standard curves were generated using known amounts of the growth factors, and samples diluents were used as controls. The standard curves for NGF, GDNF, and BDNF provided linear plots of absorbance versus concentration. The linear plots were used to determine the concentration of NGF, GDNF, and BDNF in the test samples. All experiments were performed in triplicate.
Cell Migration Analysis
The migration behavior of OECs on TSF fibers and PLL in 35-mm Petri dishes was directly observed using the Leica AF6000 fluorescent microscope. The method was followed as previously described (31). In this work, we applied a systematic sampling, and all cells within the migration region of the chamber (Weber Scientific International, Ltd., Teddington, UK) were recorded and analyzed without the addition of growth factors. Data were recorded every 5 min using a 10x objective. Then the cell migration behaviors, including migration tracks, turning behavior, and migration distances, were analyzed.
Scanning Electron Microscopy (SEM)
The SEM was employed to study the morphology of cells grown on the fibers. Samples were rinsed with PBS three times, fixed in 4% glutaraldehyde (Youran, Zhengzhou, China) for 90 min, and postfixed with 1% osmic acid (Hengyuan, Shanghai, China) for 1 h. Then successive dehydration was performed in a graded concentration (50–100% v/v) of ethanol. Subsequently, the samples were dried in a critical point drier (Hitachi, Tokyo, Japan) and coated with gold for the observation of cell morphology.
Statistical Analysis
Data were expressed as mean ± standard deviation (SD). One-way ANOVA followed by Student–Newman–Keuls test was used to analyze the data. A value of p < 0.05 was considered as significant difference.
Results
Morphology and Structure of Silk Fibroin Fibers
SEM micrographs of electrospun BSF and TSF fibers revealed porous, nanoscaled fibrous structures formed during electrospinning. BSF and TSF nanofiber showed a consistent thickness with an average diameter of about 392.5 and 386.7 nm, respectively, as shown in Figure 1A. FTIR is a powerful and common tool for secondary structure analysis and is frequently used to investigate the conformation changes of silk fibroin. Secondary structure in BSF and TSF fibers after ethanol treatment was determined by FTIR, as shown in Figure 1B. The absorption bands of BSF (sample a) and TSF (sample b) at wave numbers of 1,628 cm−1 (amide I), 1,520 cm−1 (amide II), 1,265 cm−1 (amide III), and 700 cm−1 (amide V) were attributed to the β-sheet structure (30). The absorption peak at 895 cm−1 (amide IV) was characteristic of TSF β-sheet structure (30). These FTIR results suggest the prevailing β-sheet structure of electrospun BSF and TSF nanofiber after ethanol treatment. Silk fibroin, like many other fibrous proteins, is composed of antiparallel β-sheet responsible for its water insolubility. During the ethanol treatment process, silk fibroin undergoes a conformational transition from α-helices or random coils into β-sheet structure.

The structure of the Bombyx mori silk fibroin (BSF) and Tussah silk fibroin (TSF) fibers. Scanning electron microscope (SEM) images (A) and Fourier transform infrared (FTIR) spectra (B) of BSF (a) and TSF (b) fibers. The average diameter of the fibers is shown below part A.
Characterization of OECs on BSF and TSF Fibers
Inverted light microscopy was employed to observe cell attachment and spreading on BSF and TSF fibers (Fig. 2), in which the speeds were found to be different for each fiber type. The percentage of OECs that acquired spread morphology on TSF fibers after 2 h, denoting full attachment, was higher on the PLL and BSF fibers. Thus, TSF fibers maintained or, in some cases, even enhanced cell interactions in a short time. The morphological characteristics of cells on BSF and TSF fibers were typical for OECs (Fig. 3) (for NGFR p75 staining), suggesting that silk fibroin fibers supported the survival and phenotype maintenance of OECs as we previously reported (22).
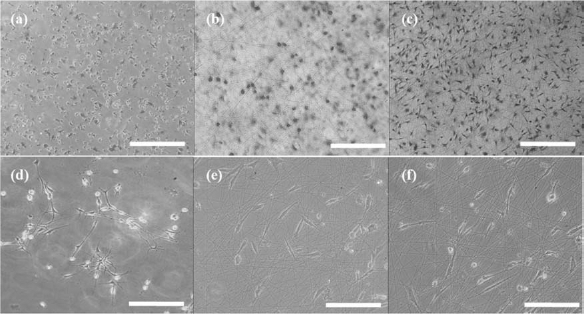
Growth of olfactory ensheathing cells (OECs) on vehicle or fibers. OECs growing on poly-l-lysine (PLL) (a and d), BSF fibers (b and e), and TSF fibers (c and f) for 2 h (a, b, and c) and 1 day (d, e, and f). Scale bars: 500 μm (a, b, c) and 250 μm (d, e, f).

NGFR Immunostaining of OECs. Immunostaining performed on OECs after 5 days of culture on PLL and silk fibroin fibers for OEC marker nerve growth factor receptor (NGFR) p75 (red) and Hoechst 33258 (blue). The majority of cells are NGFR p75 positive on PLL (A), BSF fibers (B), and TSF fibers (C). Scale bars: 50 μm.
In order to evaluate the cell viability and proliferation, OECs were cultured on the aforementioned BSF and TSF fibers, and then the live/dead assay and MTT were performed at days 1, 3, and 5. Cells readily survived with no significant differences in viability on all of the fibers tested (Fig. 4). The statistical analysis results showed that the percentage of dead cells on PLL, BSF, and TSF fibers was 5.3%, 6.2%, and 4.9%, respectively. A significant increase in cell numbers was directly observed in Figure 4. The growth of cells on TSF fibers was significantly greater than for other scaffolds. This effect extended to the rest of the culture time, resulting in a higher cell proliferation on TSF fibers than on PLL and BSF fibers (Fig. 5).
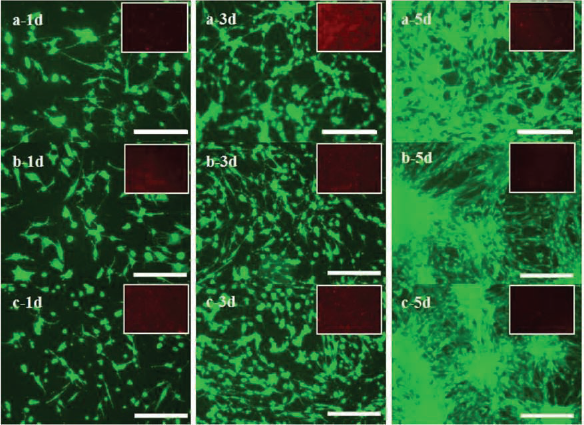
OEC viability on silk fibroin fibers. Cell viability of OECs cultured on silk fibroin fibers at days 1, 3, and 5 as determined by live/dead staining. Scale bars: 250 μm. (a) PLL, (b) BSF fibers, and (c) TSF fibers.

Results of cell proliferation by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. The OD value of OECs on TSF fibers was higher than that on PLL and BSF fibers at days 3, 5, and 7; **p < 0.05.
The gene expression patterns of OECs included MBP, BDNF, NGF, GDNF, NCAM, and VEGF. The gene expression level of OECs was examined by RT-PCR. As shown in Figure 6A, the level of NGF expression was detected strongly in the three experimental groups, while the remaining genes were weakly detected with the obvious exception of the high levels of BDNF on TSF fibers. Statistics indicated that the expression of NGF, BDNF, and VEGF was significantly higher in OECs cultured on TSF fibers than on BSF and PLL. To confirm the PCR results and determine the level of protein expression, an ELISA was applied at corresponding time points. As Figure 7 shows, NGF, BDNF, and NCAM expression levels in OECs on TSF fibers were significantly higher than that on BSF fibers and PLL, thus similar to the PCR results (except for a significant increase in NCAM rather than VEGF).

Gene expression in OECs cultured on TSF fibers, PLL, and (B) SF fibers for 5 days analyzed by semiquantitative RT-PCR. (A) RT-PCR gel images. (B) Band density measurement results of the gene and β-actin gene used for the control; **p < 0.05. Cells grown on TSF fibers expressed higher levels of nerve growth factor (NGF), brain-derived trophic factor (BDNF), and vascular endothelial growth factor (VEGF), while there was a trend toward increased BDNF and VEGF for (B) SF fibers. MBP, myelin basic protein; GDNF, glial-derived neurotrophic factor; NCAM, neural cell adhesion molecule.

Quantification of neurotrophic factors using ELISA analysis. Histograms of MBP, BDNF, NGF, GDNF, NCAM, VEGF expression levels in culture medium secreted by OECs after 5 days in culture. NGF, BDNF, and NCAM expression levels in OECs on TSF fibers were significantly higher than those in SF fibers and PLL, **p < 0.05.
TSF Fibers Regulate the Morphology and Alignment of OECs
To further analyze the topographic effect on the cell behavior of OECs, TSF fibers with different topographies including changes in fiber diameter and alignment were fabricated. BSF fibers supported OEC adhesion and spreading as previously described (22). However, the morphology of OECs was significantly affected by fiber diameters, as shown in Figure 8. Generally, most OECs present in a polygonal orientation, which seem to be trapped in the pores of the electrospun TSF fibers with an average diameter of 1,200 nm (Fig. 8A, B, and C). In contrast, a completely different cell morphology was observed on 400-nm TSF fibers, on which OECs acquired a spindle-like and bipolar shape (Fig. 8D, and E). Further more, the OECs' arrangement could be controlled by aligning 400-nm fibers parallel to the fiber direction. To our surprise, significant changes in the cell nucleus were observed on aligned 400 TSF fibers, consistent with previous research (5). The shape of the OECs' nucleus was mainly irregular but spherical on random 400- and 1200-nm TSF fibers but changed to a spindle pattern with a large length/diameter (L/D) ratio of 2.7 ± 0.2 on aligned 400 TSF fibers.
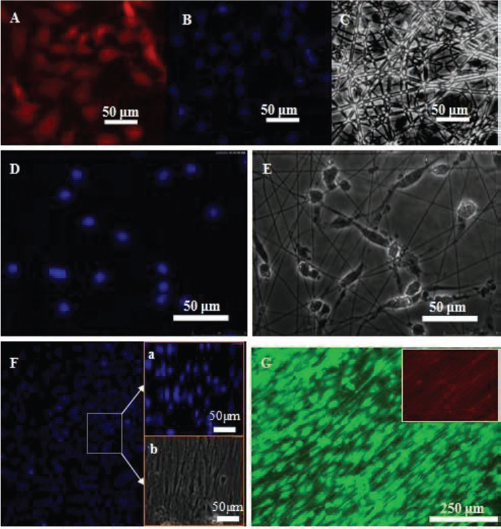
The morphology of OECs on TSF fibers was observed through P75, DAPI, and live/dead staining. Cells on 1200-nm TSF fibers were mainly polygonal in shape and retained a cobblestone appearance (A, B, and C); cells on 400-nm TSF fibers showed mainly bipolar and spindle shape (D and E); cells on aligned 400-nm TSF fibers demonstrated the direction of OEC elongation exactly parallel to the direction of fiber alignment. In addition, the cell nucleus changed their original round shape to elongated and spindle-like shape along the fiber direction (F and G). Immunostaining performed on OECs after 5 days of culture on PLL and TSF fibers. NGFR p75 staining (A), Hoechst 33258 staining (B, D, F, F, a), phase image of cells (C, E, and F, b), and live/dead staining (G).
Cells on TSF fibers were processed for SEM imaging in order to more clearly visualize cell shape and cell–substrate interaction. The results indicated that OECs were securely attached on both random and aligned TSF fibers with elongated processes and a high number of cell-to-cell contacts but exhibited different morphologies: OECs on random TSF fibers possessed bipolar and tripolar extensions with a more spread morphology (Fig. 9A), while OECs on aligned TSF fibers exhibited a stretched and bipolar spindle-shaped morphology (Fig. 9B), which provided morphological evidence of the possibility of the regulation of the OECs' shape and arrangement by random and aligned TSF fibers.

Scanning electron microscope (SEM) images of OECs cultured on random (A) and aligned (B) 400-nm TSF fibers for 5 days. Scale bars: 100 μm.
Migration of OECs on TSF Fibers
Migration of OECs after implantation is essential for the repair of SCI. To determine the effect of TSF fibers on OEC migration, we investigated the migratory behavior of OECs on PLL and TSF fibers by fluorescent microscopy (Leica AF600). OECs cultured on PLL migrated in a random fashion (Fig. 10A), whereas those cultured on TSF fibers demonstrated a fiber-dependent migration (Fig. 10B), suggesting an effective way to guide cell migration.

Migration tracks of OECs on PLL and TSF fibers. Phase-contrast photos showing migratory behavior of two representative cells (in circles) on PLL (A) and TSF fibers (B). Arrows indicates the migration direction of OECs on PLL and TSF fibers. Scale bars: 100 μm.
Discussion
The capacity of axonal regeneration in SCI is extremely limited due to the active inhibitory molecules and the lack of a permissive environment (19) at sites of injury that inhibit axon regeneration. Therefore, ways to supplement cells to improve inhibitory environment and activate endogenous cells are key challenges (26). Implantation of tissue-engineered scaffolds is one of the most promising therapeutic strategies for inducing nerve regenerations following spinal cord injuries (18,23). Understanding the behavior of OECs and identification of elements of biomaterial scaffolds that control the behavior of OECs may eventually enable us to manipulate nerve cell growth and targeting. In this study, we found that the electrospun BSF and TSF fibers are promising growth matrices for OECs. Consistent with earlier studies indicating that substrates made up of BSF and TSF supported the survival and growth of neurons and Schwann cells (28,29), our results further demonstrated that cell behaviors, including cell adhesion, growth, and migration, could be regulated by electrospun silk fibroin fibers, suggesting a potential use of silk fibroin for preparing the tissue-engineered scaffolds to treat SCI.
In the present study, we obtained BSF and TSF nanofiber by electrospinning and analyzed the OECs' behavior on these fibers. Results from our current study demonstrated that the number of OECs attaching on the TSF fibers seemed higher than on the BSF fibers and PLL (Fig. 2a, b, and c), which might be perpetuated by the presence of the Arg-Gly-Asp (RGD) sequence and the hydrophilicity. It is well known that TSF contains RGD sequences that are recognized by cell surface receptors, such as integrins, which mediate cell adhesion (9,17,20). The conjugation of RGD molecules to a scaffold surface has been proven to improve cell adhesion and spread (2,33). The biomaterial scaffold hydrophilicity also has a profound impact on cell–matrix interactions and cellular behaviors such as adhesion, shape, motility, cytoskeletal organization, and differentiation, which correlate well with protein absorption at the scaffold interfaces (1). It is reported that cell adhesion is more enhanced on hydrophilic hydrogel surfaces than that on hydrophobic blocks. Compared with BSF, the hydrophilicity of TSF is higher due to more polar amino acids (30). In the current study, after cell adhesion, the OECs showed polygonal morphology on PLL (Fig. 2d) and mainly a bipolar shape on silk fibroin fibers with protrusions along the fibers (Fig. 2e and f) due to the effect of fiber guidance. Fluorescence microscopy showed that OECs was stained positively for NGFR p75, suggesting that silk fibroin fibers could keep the OECs' phenotype. The improved adhesion and spreading of OECs on TSF fibers had an effect on the cell proliferation and protein expression (Figs. 5, 6, and 7). A higher cell proliferation was observed when OECs were cultured on TSF fibers than that on PLL and BSF fibers. Plus, the levels of some gene and protein expressions were higher on TSF fibers than on BSF fibers and PLL. The mechanism of OECs stimulating SCI regeneration is still unclear, though OEC transplantation was able to promote the growth of axons, producing new myelin sheaths around demyelinated or amyelinated axons and, in some cases, functional recovery (8,10). The growth factors produced by OECs were thought to play important roles in SCI repair (19). Taken together, these results imply that the TSF nanofibrous scaffold is a promising growth matrix for OECs.
Further, the effects of fiber diameter and alignment of TSF fibers on OECs' shape and migration were investigated. OECs exhibited different shapes on TSF micro-and nanofibers with an average diameter of 1.2 μm and 400 nm (Fig. 8). It seemed that OECs were trapped in the pores of TSF microfibers and showed polygons, indicating the confining extension of OEC protrusions (Fig. 8A, B, and C); however, OECs cultured on TSF fibers demonstrated a spindle-like and bipolar shape (Fig. 8D and E). In addition, OECs cultured on the aligned fibers exhibited classical contact guidance by growing parallel to the TSF fibers; even the cell nucleus changed their original round shape to an elongated one along the parallel fibers (Figs. 8F, G and 9). Combining the technologies of geometric confinement with the modern toolbox of cell biology is feasible to control and understand the diversity of cell shapes and function (21). The different cell shapes on TSF micro- and nanofibers demonstrated the significant effect of fiber diameter on cell shape, which plays an important role in keeping cell function (15,21). The aligned OECs suggest that the favorite growing direction of OECs is parallel to the TSF nanofiber, and the process is dynamically directed over time. In some nonmammalian vertebrates, successful central nervous system regeneration is attributed to the alignment of reactive glial, which guides axons across the lesion site (6).
The lack of the aligned neuroglia pathway guiding cell growth and migration is one of the key factors in the failure of axonal regeneration. Migration tracks of OECs on PLL and TSF fibers revealed an intriguing migration behavior: OECs cultured on PLL made random turns during migration (Fig. 10A), while those cultured on TSF fibers exhibited a linear and fiber-dependent migratory behavior (Fig. 10B), suggesting a way to regulate cell migration behavior by the fibers guiding the cells through contact.
Conclusions
To develop OEC-based scaffolds for SCI repair, BSF and TSF fibers were prepared by electrospinning, and their effects on OECs behavior were investigated. It was found that TSF fibers exhibited a much higher rate of cell attachment, proliferation, and protein expression of cultured OECs compared with BSF fibers and PLL. Moreover, the diameters and the alignment of TSF fibers significantly affected the OECs' shape: OECs elongated with the fiber direction in the aligned scaffolds, and the cell nuclei were also observed to be parallel to the aligned fiber. Furthermore, OECs exhibited a TSF fiber-dependent linear migratory behavior, suggesting that the TSF nanofiber alignment represents an attractive guidance cue for the migration of OECs. A combination of OECs and optimized TSF fibers may provide a new platform for possible SCI repair.
Footnotes
Acknowledgments
This work was supported by the health department scientific research projects of Jiangsu Province (H200920), the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), the National Natural Science Foundation of China (Nos. 30870642, 31071220, and 81271723), the Natural Science Foundation of Jiangsu Province (BK2009119), China Project 211 (reconstruction of spinal functions and bone tissue engineering after minimal invasive surgery), and the Applied Basic Research Project of Suzhou. The authors declare no conflict of interest.
