Abstract
Endothelial progenitor cell (EPC) transplantation has beneficial effects for therapeutic neovascularization. We therefore assessed the effect of a therapeutic strategy based on EPC administation in the healing of radiation-induced damage. To improve cell therapy for clinical use, we used pretreatment with ephrin B2-Fc (Eph-B2-Fc) and/or coadministration with smooth muscle progenitor cells. At day 3, EPCs promoted dermal wound healing in both nonirradiated and irradiated mice by 1.2- and 1.15-fold, respectively, compared with animals injected with phosphate-buffered saline. In addition, EPCs also improved skin-blood perfusion and capillary density in both irradiated and nonirradiated mice compared with PBS-injected animals. We also demonstrated that activation with Eph-B2-Fc increased wound closure by 1.6-fold compared with unstimulated EPCs in nonirradiated mice. Interestingly, the beneficial effect of Eph-B2-Fc was abolished in irradiated animals. In addition, we found that Eph-B2-Fc stimulation did not improve EPC-induced vascular permeability or adhesiveness compared to unstimulated EPCs. We hypothesized that this effect was due to high oxidative stress during irradiation, leading to inhibition of EPCs' beneficial effect on vascular function. In this line, we demonstated that, in irradiated conditions, N-acetyl-l-cysteine treatment restored the beneficial effect of EPC stimulation with Eph-B2-Fc in the wound healing process. In conclusion, stimulation by Eph-B2-Fc improved the beneficial effect of EPCs in physiological conditions and irradiated conditions only in association with antioxidant treatment. Additionally, cotherapy was beneficial in pathological conditions.
Keywords
Introduction
Cutaneous wound healing is a complex process involving inflammatory reactions, cell migration and proliferation, and tissue remodeling. In particular, formation of new blood vessels is required to sustain the newly formed granulation tissue (3). However, high-dose ionizing irradiation, such as occurs in nuclear or radiotherapy accidents, leads to deficient wound healing because of inadequate angiogenesis (4, 31). The development of strategies designed to promote wound healing in the setting of irradiation is a major therapeutic challenge.
Recent studies have illustrated that bone marrow-derived stem cells and adipose tissue-derived stromal cells participate in cutaneous wound healing and skin regeneration in physiological and irradiated conditions (9). In addition, human peripheral blood-derived endothelial progenitor cells (EPCs) restore neovascularization in models of tissue ischemia, at least in part by their capacity to promote the recruitment of monocytes/macrophages into the wound area (15, 16).
In addition, EPCs have been shown to incorporate into the injured vasculature, promoting neovascularization and subsequent functional recovery of the surrounding tissues. EPCs promote neovascularization by differentiating in situ into endothelial cells and by secreting growth factors, cytokines, and proteases that support angiogenic and vasculogenic processes (14, 17, 18). Unfortunately, transplantation of autologous EPCs has several limitations, including the limited supply of expanded progenitors, and the yield is currently so low that very large blood volumes have to be processed to obtain sufficient EPCs for therapeutic use. In addition, strategies to improve the therapeutic potential of cell therapy need to be developed to counteract progenitor cell dysfunction in patients with cardiovascular risk factors (23, 25, 26, 28).
Recent data indicate that the required number of efficient EPCs can be reduced by potentiating EPCs and their proangiogenic effect using concomitant infusion of proangiogenic proteins (32) or by activating EPCs before their injection (10, 28).
One important family of surface receptors is the ephrin receptors. Erythropoietin-producing human hepatocellular carcinoma (ephrin) receptors belong to the largest family of tyrosine kinase receptors. Originally identified as neuronal guidance molecules, they are also expressed by nonneural cells, including endothelial cells, hematopoietic cells, and tumor cells (7). Studies in mouse embryos showed that the two ligand-receptor proteins, ephrin B2/ephrin receptor B4 (Eph B4), are essential for embryonic heart development and angiogenesis (29). The interaction between ephrin B2/Eph B4 is thought to play an important role not only in cell–cell and cell–extracellular matrix interactions but also in cell migration, adhesion, and proliferation (6). In recent studies, activation of Eph B4 with an ephrin B2 fragment, crystallizable (Fc) chimeric protein enhanced the proangiogenic capacity of EPCs in a mouse hindlimb model (10) and stimulated postnatal neovascularization in the murine cornea (12).
In addition, neovascularization requires that both endothelial cells and mural cells proliferate, migrate to the proper location, and assemble into vascular structures. Among mural cells, pericytes are emerging as critical regulators of vascular function because they are thought to stabilize the vessel wall and to regulate endothelial cell survival, growth maturation, and permeability (1). Pericytes and smooth muscle cells can be obtained from smooth muscle progenitor cells (SMPCs), and of interest, coadministration of EPCs and SMPCs has been shown to potentiate EPC-related actions on postischemic vessel growth (10).
In this work, we sought to determine whether EPCs accelerate wound healing in physiological and pathological conditions in vivo and in vitro. In addition, we proposed to improve cell-based therapeutics in order to circumvent the problems of insufficient cell number and low efficiency of incorporation, by combining two distinct strategies: the use of activated EPCs with Eph-B2-Fc or coadministration with SMPCs.
Materials and Methods
Cell Culture
EPCs and SMPCs were isolated and pooled from three human female umbilical cord blood samples (considered biological waste under French law) and were differentiated ex vivo as previously described (19). They were grown in rat tail type I collagen-coated flasks (Sigma-Aldrich Chimie, Saint Quentin Fallavier, France) in M199 medium (Invitrogen, Cergy Pontoise, France) supplemented with 20% fetal calf serum (FCS; Dominique Dutscher, Brumath, France), 25 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (Invitrogen), antibiotic and antimycotic solution (Invitrogen), and 10 ng/ml recombinant human vascular endothelial growth factor (VEGF; R&D Systems, Oxford, UK) and human platelet-derived growth factor (R&D Systems). All experiments with EPCs and SMPCs were carried out between passages 9 and 15.
Phenotypic characterization of EPCs and SMPCs was then done using specific markers as previously described (19).
Mouse Model of Skin Wound Injury and Experimental Procedures
All animal procedures were carried out in accordance with French government regulations (Services veterinaires de la santé et de la production animale and with the approval of the IRSN Ethics Committee: P09-14). Ten-week-old male athymic nude mice (Harlan, Gannat, France) were anesthetized by intraperitoneal injection of ketamine and xylazine (Virbac France, Carros, France and Bayer Healthcare, Puteaux, France), n = 10 per group. The backs of the mice were shaved and sterilized with alcohol. A sterile 8-mm dermal punch biopsy tool (Centravet, Plancoet, France) was used to outline a pattern for the wound on the dorsum of the mice. An 8-mm wound was chosen due to their large dorsal surface. The mice were then treated with 100 μl of phosphate-buffered saline (PBS; Life Technologies, Saint Aubin, France) containing 1 × 106 EPCs. The EPCs were pretreated with or without 3 μg/ml of either Eph-B2-Fc (R&D Systems, 496 EB 200) or Eph B4-Fc (R&D Systems, 496 EB) for 6 h at 37°C and washed twice with PBS to remove unbound recombinant proteins. Local irradiation (20 Gy 1.51 Gy/min) of the flank skin was also performed before punch application in an additional group of mice. These animals also received antioxidant N-acetyl-l-cysteine (NAC; 200 mg/kg/day, IPl; Sigma-Aldrich Chimie).
In the second set of experiments, 6 h after injury, mice that received 100 μl of PBS containing EPCs (0.5 × 106), SMPCs (0.5 × 106), or EPCs plus SMPCs (0.5 × 106 + 0.5 × 106) from a pool of three donors were intramuscularly injected in the skin wound model in nonirradiated and irradiated conditions (10 mice per group).
Wound Closure
Wound areas were measured at days 3, 7, 10, and 14 after punch biopsy. Image analysis software (ImageJ; NIH, Bethesda, MD, USA) was used to determine the area of wound healing. One hundred percent indicated the maximum size of the wound area at day 0. Standardized digital photographs of the wounds were taken, with the same distance between the camera and preanesthetized animals for each animal. The open wound areas were determined with an image analyzer, and the total pixels that cover the unhealed areas were drawn onto the digital photographs using a pattern overlay in ImageJ. The number of pixels covering an open wound area on a given day was divided by the number of pixels spreading over the open wound on day 0 to obtain the percentage of closure. Mice were randomized before punch application and were then distributed into all the different experimental groups. All the groups were used in the same set of experiments. Results were obtained after blind evaluation.
Immunohistochemistry
Skin sections were collected and progressively frozen in isopentane solution (VWR International, Fontenay sous Bois, France) cooled in liquid nitrogen. Sections (5 μm) were fixed in 100% cold acetone (VWR International), blocked with 20% goat serum (Sigma-Aldrich Chimie) for 20 min, and incubated with the appropriate primary rat anti-mouse cluster of differentiation 31 (CD31; CBL1337, 1/100; Millipore, Fontenay sous Bois, France), rat anti-mouse E-selectin (ref 550290, 1/20; BD Pharmingen, Le Pont de Claix, France), or rat anti-mouse P-selectin (ref 550289, 1/25; BD Pharmingen) antibodies, for 1 h at room temperature. Sections were washed three times with PBS and incubated with goat anti-rat secondary antibodies coupled to fluorescein isothiocyanate (FITC) for CD31 analysis (Alexa 488, A11006, 1/200; Molecular Probes, Saint Aubin, France) or to Alexa Fluor 568 for E-selectin and P-selectin analysis (Alexa 568, A 11077, 1/200; Molecular Probes).
Oxidative Fluorescent Microtopography
The oxidative fluorescent dye dihydroethidium (DHE; Invitrogen, Molecular Probes) was used to evaluate in situ production of superoxide. DHE is freely permeable to cells and in the presence of O2-. is oxidized to eithidum bromide (EtBr), where it is trapped by intercalation with DNA. EtBr was excited at 488 nm, and the emission spectrum was recorded at 610 nm. In cell-free assays, addition of hydrogen peroxide to DHE does not significantly increase EtBr fluorescence (20). Unfixed frozen skin tissue from irradiated mice treated with or without NAC (200 mg/kg/day, IP) was cut into 7-mm-thick sections and placed on a glass slide. DHE (2 μM) was topically applied to each tissue section and coverslipped. Slides were incubated in a light-protected humidified chamber at 37°C for 30 min. Images were obtained with a laser scanning confocal microscope equipped with a krypton/argon laser (Zeiss, Le Pecq, France).
Laser Doppler Perfusion Imaging System
To provide functional evidence for wound trauma-induced changes in neovascularization, laser Doppler perfusion imaging experiments were performed in mice. A full-field laser perfusion imager (MoorFLPI; Moor Instruments, Ltd., Devon, UK) was used to quantitatively measure circulatory blood flow intensity. This imager uses laser speckle contrast imaging, which exploits the random speckle pattern that is generated when tissue is illuminated by laser light. The random speckle pattern changes when blood cells move within the region of interest (ROI). The circulatory blood flow intensity of each ROI will be recorded as flux with perfusion unit, which is related to the product of average speed and concentration of moving red blood cells in the tissue sample volume. The images will be recorded and analyzed in real time by the Moor FLPI software version 3.0 (Moor Instruments, Ltd.). A side-stream dark-field (MicroScan, Microvision Medical, Amsterdam, the Netherlands) video microscope will be used to visualize the small vessel (less than 20 μm) blood flow classification of each small vessel, perfused small vessel density, microvascular flow index, and heterogeneity index. The analyses were done by a single investigator who was blinded to grouping.
Capillary Density
Capillary density was measured by immunohistochemistry using rat anti-mouse CD31 antibodies (1/100, CBL 1337; Millipore, Saint Quentin en Yvelines, France) in all experimental groups, at day 3 after injury.
Plasma Levels of Vascular Endothelial Growth Factor and Stromal-Derived Factor.
Plasma VEGF and stromalderived factor 1 (SDF-1) levels were measured with a specific ELISA (R&D Systems Europe, Lille, France), following the manufacturer's protocol. Peripheral blood was taken 7 and 14 days after skin injury by retro-orbital puncture.
HUVEC and EPC Tube Formation on Matrigel
Five hundred microliters of Matrigel (BD Biosciences, Le Pont de Claix, France) was added to a 12-well plate (Dominique Dutscher, Brumath, France).
Female human umbilical vein endothelial cells (HUVECs; CC-2519, n = 3; Lonza, Verviers, Belgium) were uniformly irradiated [in a γ radiation field with a single dose of 20 Gy delivered from a 137Cs source (IBL 637, CISBio International, Saclay, France), dose rate approximately 0.7 Gy/min] or were not irradiated. They were labeled with CM-DiI red dye (1 g/ml; Invitrogen, Life Technologies), and EPCs were labeled with SP-Dioc18 green dye (2 g/ml; Invitrogen) to induce tube formation. HUVECs were added to the Matrigel and then incubated overnight in medium M199 containing 10% FCS or in conditioned medium of irradiated HUVECs. The conditioned medium was obtained from the supernatant of HUVECs uniformly irradiated with a single dose of 20 Gy, dose rate approximately 0.7 Gy/min, and incubated for 4 days. EPCs or EPCs activated with B2 were then added to the endothelial network (12 × 105 EPCs per well) for 8 h. For quantitative analysis, Matrigel wells were observed under an Axiovert 25 microscope (Zeiss), and Histolab software (Microvision Instruments, Evry, France) was used to count the number of sprouts in 10 fields of each well. Cells were visualized by fluorescence using an inverted-phase microscope (Zeiss).
Incorporation of EPCs Into the HUVEC Network
EPCs (3 × 104 per well) labeled with CM-DiI red dye (Invitrogen) were mixed with irradiated (20 Gy) or nonirradiated HUVECs (12 × 105 per well) labeled with CM-DiI green dye (Invitrogen). Cells were visualized by fluorescence using an inverted phase microscope. For quantitative analysis, the number of EPCs incorporated into the HUVEC network was determined as a ratio of the number of EPCs to the total area of the HUVEC network using Histolab software (Microvision Instruments).
RT PCR RNA Isolation and RT Real-Time Quantitative PCR
Total RNAs were isolated from irradiated HUVEC culture (20 Gy) using RNeasy Mini Kit (Qiagen, Les Ulis Courtaboeuf, France), according to the manufacturer's recommendations. TaqMan Gene Expression Assays were used with P- and E-selectin (respectively, Hs00927900_m1 and Hs00950401_m1; Applied Biosystems, Foster City, CA, USA). Fluorescence data were analyzed after PCR completion, with an ABI PRISM 7700 Sequence Detection System instrument (Applied Biosystems). Significant PCR fluorescent signals were normalized to those of the housekeeping gene: glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Predeveloped Taqman Assay; Applied Biosystems). Relative mRNA was calculated using the comparative ΔCt method.
Determination of Vascular Permeability
Vascular permeability was studied in irradiated or nonirradiated animals treated or not with activated EPCs, 3 days after injury. Twenty-five milligrams per kilogram FITC-labeled 77-kDa dextran (Sigma-Aldrich Chimie) was administrated by intraocular perfusion. Blood vessels were visualized with a fluorescence-inverted microscope (Nikon, Champigny sur Marne, France). Fluorescence intensity was recorded 30 s, 10 min, 20 min, and 30 min after FITC-dextran injection with an Exwave HAD digital color camera (Sony, Zaventem, Belgium). Fluorescence intensity was quantified using the Histolab 4.3.6 software and was measured within the vessel under study (I int) and in a contiguous area of perivascular interstitium (I ext). Background fluorescence intensity (Bg) was subtracted from each value. An index of vascular leakage (IP) was calculated by dividing (I ext − Bg) by (I int − Bg). Experiments were performed, and 30 vessels/animals were analyzed for the calculation of the permeability index. Sampling area was proximal to the punch lesion.
Adhesion Assay
EPC Adhesion to Recombinant E-Selectin and P-Selectin–Fc Fusion Proteins
The 96-well tissue culture plates (Dominique Dutscher) were coated with 100 μl/well of a 25 μg/ml human immunoglobulin G fragment, crystallizable γ (IgG Fcγ; R&D Systems) solution in PBS overnight at 4°C. Unbound antibody was discarded, and wells were blocked with 1% bovine serum albumin (BSA; Sigma-Aldrich Chimie) for 1 h at room temperature. After washing with PBS, 100 μl of human recombinant E-selectin or P-selectin fusion proteins (5 μg/ml or 10 μg/ml in 0.01% BSA, respectively; R&D Systems) was added, and the plates were incubated for 1 h at 37°C. Then, 100 μl of a 6 × 105 cell suspension pretreated with the fusion proteins was added, and plates were incubated for 1 h at 37°C to allow cell adhesion. Cells were washed twice with M199 medium, fixed in 1% glutaraldehyde (Sigma-Aldrich Chimie), and then stained with crystal violet (Sigma-Aldrich Chimie). Cell adhesion was quantified by measuring optical density at 570 nm.
Endothelial–EPC Interactions in the Parallel-Plate Flow Chamber
Primary female human microvascular endothelial cells of dermal origin (HMVECs-D; CC-2543, n = 3; Lonza) were routinely cultured at 37°C in 5% CO2 according to the manufacturer's recommendations in the supplier' sendothelial cell growth medium (endothelial basal medium, EBM-2; CC-3156; Lonza). HMVECs-D were used between the fifth and eighth passages. For the flow experiments, 7 × 104 endothelial cells were seeded onto 0.17 × 24 × 60-mm glass slides (Diamant Star; CML, Nemours, France). Cells were allowed to reach confluence before irradiation. The endothelial cells were uniformly irradiated in a γ radiation field with a single dose of 20 Gy delivered from a 137Cs source (IBL 637, dose rate approximately 0.7 Gy/min). Sham-irradiated control endothelial cells were treated under the same conditions. Irradiated EPCs and EPCs activated with Eph-B2-Fc were then treated with or without the antioxidant NAC (5 mM) just before irradiation. The flow experiments were performed 4 days later.
The glass slide coated with a monolayer of HMVECs was placed in the parallel-plate laminar flow chamber as previously described (27).
Patients, Immunohistology
Six patients treated for mammalian adenocarcinoma (women from 45 to 68 years old) who underwent radiotherapy (45 Gy, 2 Gy by fraction) were included in this study. Formalin-fixed paraffin-embedded tissue samples were obtained following institutional ethical guidelines (Institut Montsouris) and French Medical Research Council Guidelines. For each patient, specimens of normal tissue were taken in the irradiated field adjacent to the tumor and from normal tissue at a large distance from the tumor. No consent was required, and sections from patients were obtained retrospectively (several years after surgery), and data were analyzed anonymously. For immunohistochemistry, 5-μm sections were used to immunolocalize P- and E-selectin (respectively, ab6632: 1/500 and ab49506: 1/20; Abcam, Paris, France) and counterstained with 4′,6-diamidino-2-phenylindole (DAPI, Vectashield; Clinisciences, Nanterre, France).
Statistical Analysis
A one-way ANOVA was used to compare variables (Jandel Scientific, Systat software, San Jose, CA, USA). Post hoc Tukey's t-test comparisons were then performed to identify which group differences account for the significant overall ANOVA. A value of p < 0.05 was considered significant.
Results
Eph-B2-Fc Pretreatment Accelerated Wound Healing in Physiological Conditions
In order to determine the potential effect of ephrin B2/Eph B4 activation, we stimulated EPCs with Eph-B2-Fc and Eph B4-Fc before local injection in skin-injured mice.
Eph-B2-Fc Pretreatment on EPC Proangiogenic Effect
Wound Closure
Day 3 after punch biopsy, transplantation of EPCs accelerated wound healing by 1.2-fold compared with that of control animals injected with PBS (Fig. 1A). The wound-healing area of Eph-B2-Fc-stimulated EPCs was 1.27- and 1.6-fold higher than that of mice treated with untreated EPCs (p < 0.05) or PBS (p < 0.01), respectively (Fig. 1A). No such improvement was detected when cells were stimulated with Eph-B4-Fc. These results demonstrate that activation of Eph-B4 by Eph-B2-Fc enhanced the therapeutic potential of EPCs in wound healing in physiological conditions.
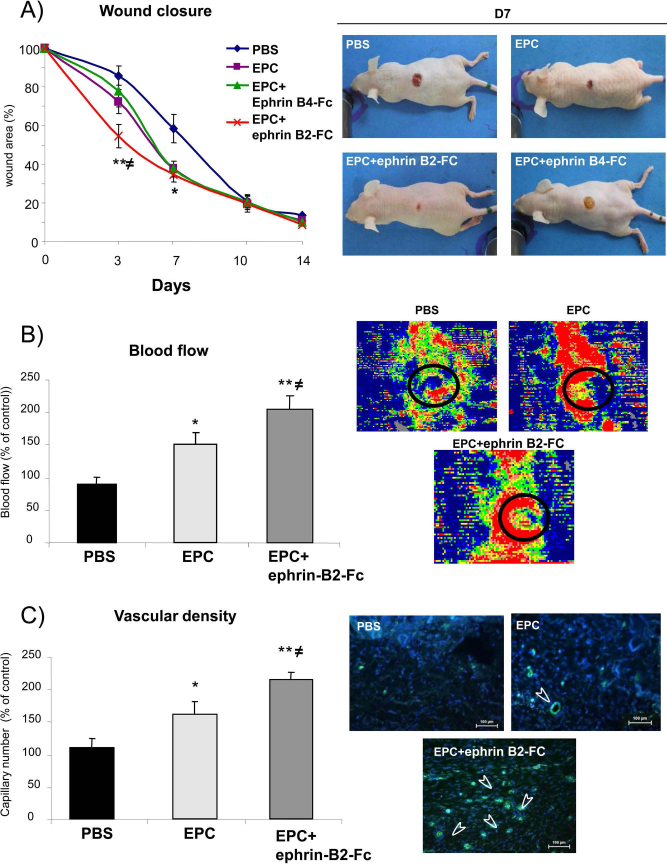
Eph-B2-Fc increases EPC proangiogenic potential in wound healing 3 days after wound injury. (A) Kinetics of wound healing of (left) and representative photomicrographs (right) of dorsal wound healing of unstimulated EPCs and Eph B4- or Eph-B2-Fc-stimulated EPCs injected into animals compared to PBS injection. (B) Quantification (left) and representative photomicrographs (right) of cutaneous blood flow perfusion of unstimulated EPCs and stimulated EPCs injected into animals 3 days after skin injury. (C) Quantification (left) of vascular number in animals injected with PBS, unstimulated EPCs, or stimulated EPCs. Representative photomicrographs (right) of skin vascular density after skin injury. Values are expressed as mean ± SEM; n = 10 per group. *p < 0.05 and **p < 0.01 versus PBS-injected mice and ≠p < 0.05 versus mice injected with stimulated EPCs.
Cutaneous Blood Perfusion
Day 3 after punch biopsy, local injection of EPCs increased blood perfusion by 1.6-fold with reference to control animals receiving PBS (p < 0.05), whereas Eph-B2-Fc cell stimulation resulted in 1.33- and 2.25-fold greater recovery of skin flow compared with that in untreated EPCs (p < 0.05) or PBS-injected animals (p < 0.01) (Fig. 1B).
Vascular Density
Changes in tissue perfusion were associated with vascular network modification. Day 3 after punch biopsy, vessel density was 1.47- and twofold higher in mice injected with Eph-B2-Fc-stimulated cells compared to unstimulated EPCs (p < 0.05) and PBS-injected animals, respectively (p < 0.01) (Fig. 1C).
Incorporation of EPCs Into the Skin Wound Area
We used fluorescence labeling to detect injected EPCs stained with an anti-human CD 31 antibody (green labeling). Our results indicated that transplanted EPCs were incorporated into the mouse vasculature of the skin wound and specifically in the dermis (Fig. 2A).

EPC localization in the wound bed associated with SDF-1 secretion. (A) Transplanted EPCs home to ischemic tissue. Representative photomicrographs of incorporated EPCs identified by double fluorescence labeling in skin wound. Transplanted human EPCs were stained with red DiI before injection and costained with biotinylated anti-human cluster of differentiation 31 (CD 31) antibody (green). In histological sections retrieved from skin tissue 7 days after injection, nuclei were stained with DAPI (blue labeling). Arrowheads indicate labeled EPCs. (B) Quantitative evaluation of murine VEGF and stromal cell-derived factor-1α (SDF-1α) protein levels in PBS- and EPC-injected mice 7 and 14 days after wound injury. Values are expressed as mean ± SEM; n = 10 per group. **p < 0.01 versus PBS-injected mice.
Effect of EPCs on Human SDF and VEGF Modulation Plasma Levels
VEGF and SDF-1 participate in progenitor cell mobilization from the bone marrow to the circulation. Seven days after skin injury, we observed that plasma SDF-1 level increased 1.6-fold in EPC-injected animals compared with untreated mice (p < 0.01) (Fig. 2B). No changes were observed in plasma VEGF level in our condition (Fig. 2B).
EPCs Stimulate Tube Formation in Matrigel
We analyzed the ability of EPCs to promote endothelial cell network formation using a Matrigel culture assay. EPCs triggered the formation of endothelial cell networks and increased the number of sprouts 1.6-fold compared with endothelial cells alone (p < 0.05) (Fig. 3A). Interestingly, Eph-B2-Fc-activated EPCs stimulated a higher endothelial cell network formation (p < 0.01).

Eph-B2-Fc increases EPC vascular network formation. (A, left) Quantitative analysis of capillary tube formation on Matrigel of HUVECs evaluated by the number of sproutings in 10 fields of each well. EPCs or EPCs activated with B2 were added to the endothelial network for 8 h. (A, right) Representative photomicrographs of vascular network. (B) Representative photomicrographs of EPCs (green DiI) and HUVECs (red DiI) in Matrigel coculture assay. Arrows indicate incorporated EPCs (green DiI) on HUVECs. Values are expressed as mean ± SEM; n = 3. *p < 0.05 and **p < 0.01 versus HUVECs alone and ≠p < 0.05 versus HUVECs plus EPCs.
We next defined the mechanism involved in the beneficial effect of EPCs on endothelial tube formation. As shown in Figure 3B, the EPCs were colocalized with endothelial cells, suggesting that the beneficial effect on endothelial cell network stabilization and sprouting is due to a direct effect of EPCs on endothelial cells.
EPCs Pretreated Eph-B2-Fc Did Not Accelerate Wound Healing in Irradiated Conditions
Angiogenic Effect
The wound-healing process was significantly altered in irradiated mice compared with control animals, as previously shown (9). Day 7 after skin injury, EPC injection accelerated the rate of wound healing by twofold compared with PBS-injected animals (p < 0.01) (Fig. 4A). These observations were associated with upregulation of skin blood perfusion and vascular density. Interestingly, in irradiated conditions, EPCs stimulated by Eph-B2-Fc have the same effect as nontreated cells on wound healing (Fig. 4A) and cutaneous blood perfusion (Fig. 4B), but not vascular density (Fig. 4C). Our data demonstrated that activation of Eph-B4 by Eph-B2-Fc did not potentiate the beneficial effect of EPCs on wound healing in irradiated conditions.

Eph-B2-Fc did not increase EPC proangiogenic potential in wound healing in irradiated condition. (A) Kinetics of skin wound healing (left) and representative photomicrographs (right) of dorsal wound healing, 7 days after skin injury in irradiated mice injected with PBS, unstimulated EPCs, and stimulated EPCs. (B) Quantification (left) and representative photomicrographs (right) at day 7 after wound injury of cutaneous blood flow perfusion of animals injected with PBS, unstimulated EPCs, or stimulated EPCs. (C, left) Quantification at day 7 after wound injury of vascular number in irradiated animals injected with PBS-stimulated EPCs, unstimulated EPCs, and stimulated EPCs. (C, right) Representative photomicrographs of skin vascular density after skin injury. Values are expressed as mean ± SEM; n = 10 per group. *p < 0.05 and **p < 0.01 versus PBS-injected irradiated mice.
These results were confirmed by in vitro Matrigel assays. Irradiation hampered the formation of endothelial cell networks and reduced the number of sprouts compared with nonirradiated cells (Fig. 5A). Interestingly, addition of EPCs enhanced this effect and restored endothelial cell network stabilization and sprouting (Fig. 5B). However, Eph-B2-Fc pretreatment did not enhance EPC-related effects compared to PBS-injected animals, suggesting that the pretreatment has lost the beneficial effect obtained in nonirradiated condition.

Eph-B2-Fc did not induce EPC vascular network formation in irradiated condition. (A, left) Quantitative analyses of capillary tube formation on Matrigel of irradiated HUVECs incubated overnight with 10% FCS medium. EPCs or EPCs activated with ephrin B2 were added to the endothelial network for 8 h. (A, right) Representative photomicrographs of vascular network. (B) Representative photomicrographs of EPC (green DiI) and HUVECs (red DiI) in Matrigel coculture assay. Arrows indicate adhesion of EPCs (green DiI) to irradiated HUVECs. n = 3. **p < 0.01 versus irradiated HUVECs alone.
Eph-B2-Fc Stimulation Did Not Enhance Vascular Permeability in Irradiated Condition
Endothelial cells have tight junctions and adherent junctions that regulate vascular permeability. EPCs were injected 4 h after skin wound injury, followed by intravenous injections of dextran-FITC to analyze vascular permeability. Ten and 20 min after dextran-FITC injection, EPC administration increased vascular permeability by 3.18-fold compared with PBS-injected animals (p < 0.01) (Fig. 6A). Interestingly, Eph-B2-Fc-stimulated EPCs did not modulate vascular permeability.
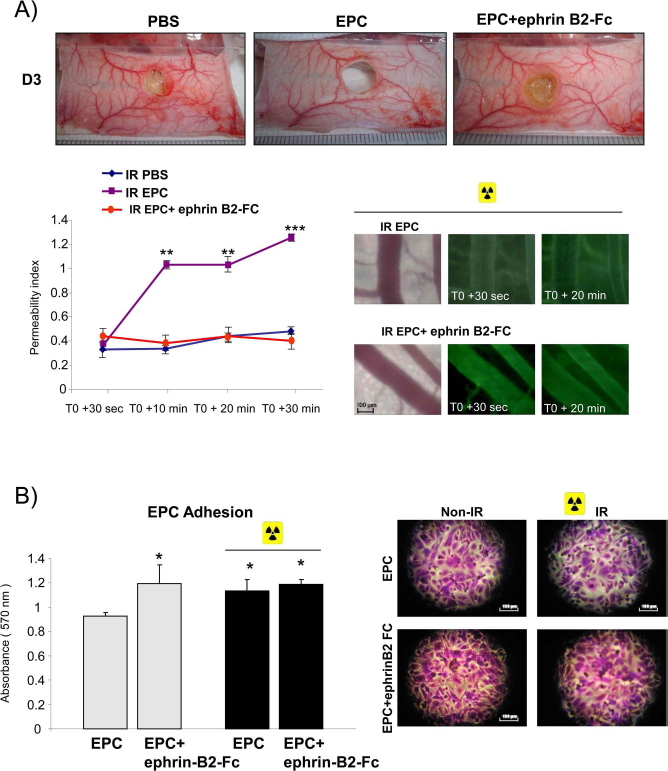
Kinetics of skin edema and adhesion. (A, top) Changes in vascular permeability were evidenced by diffusion of dextran-FITC 30 s, 10 min, 20 min, and 30 min after injection in the vessel wall. (A, bottom left) Quantitative evaluation of vessel permeability was performed 3 days after injury, n = 3. **p < 0.01, ***p < 0.001 versus irradiated PBS-injected animals. (A, bottom right) Representative photomicrographs of dextran-FITC diffusion 30 s and 20 min after injection in vessel wall. (B, left) Eph-B2-Fc induced EPC adhesion to HUVECs in nonirradiated conditions, whereas no changes were observed in EPC adhesion in irradiated conditions. Data are expressed as means ± SEM; n = 3 *p < 0.05 versus unstimulated EPCs in nonirradiated conditions. (B, right) Representative photomicrographs of EPC adhesion to HUVECs in nonirradiated or irradiated condition.
Eph-B2-Fc Stimulation Did Not Stimulate Adhesiveness to an Endothelial Monolayer in Irradiated Condition
Our results have shown that in nonirradiated conditions, the number of adhering cells was 1.2-fold higher for EPCs treated with Eph-B2-Fc than for untreated EPCs (p < 0.05) (Fig. 6B). In addition, we observed that irradiation increased EPC adhesion compared to nonirradiated cells (p < 0.05), whereas Eph-B2-Fc-stimulated EPCs did not further enhance adhesion of EPCs to HUVECs.
Effect of Irradiation on P- and E-Selectin mRNA In Vitro, In Vivo, and in Irradiated Patients
Inflammatory reaction has been shown to enhance E- and P-selectin-dependent adhesion of EPCs. We therefore analyzed E-and P-selectin expression in our experimental conditions.
E- and P-selectins are expressed by activated endothelium. E-selectin was induced in HMVECs in a dose-dependent manner, 3 h after irradiation (Fig. 7A). E-selectin expression decreased 16 and 24 h after irradiation. No difference in P-selectin expression was observed after irradiation. Interestingly, P-selectin RNA expression was upregulated at 3 days compared to 1 day after skin injury in both conditions, nonirradiated and irradiated animals, whereas no significant changes were observed in E-selectin RNA expression in vivo in each condition at day 1 and day 3 after skin injury (Fig. 7B, left and right panels). Finally, E-selectin was overexpressed in homogenous areas of the dermis of irradiated tissue. Conversely, P-selectin was expressed and localized in the dermis of both nonirradiated and irradiated patients (Fig. 7C). Altogether, these results suggest that the changes observed in EPC adhesiveness in the irradiated condition were unlikely related to modification in E- and P-selectin levels.

Time-dependent expression of selectins in the endothelium after irradiation. (A) RT-PCR analysis of P- and E-selectin mRNA expression in HMVECs-D 3, 16, and 24 h after irradiation (20 Gy). Data were normalized for loading with GAPDH; n = 3. (B) RT-PCR analysis of P- and E-selectin mRNA expression in irradiated and nonirradiated mice, 1 and 3 days after skin injury. Values are expressed as mean ± SEM; n = 3 per group. *p < 0.05 versus nonirradiated mice day 1 and ≠p < 0.05 versus irradiated mice day 1. (C) Immunohistochemical staining of P- and E-selectin in humans was performed in tissues from six patients treated for radiotherapy for mammalian tumors.
Deleterious Effect of Oxidative Stress on Eph-B2-Fc Stimulated EPC Potential
Antioxidant Treatment Restored Beneficial Effect of Eph-B2-Fc Stimulated EPCs on Wound Healing
The generation of reactive oxygen species (ROS) is the primary event of tissue radiation interactions leading to radiation injury and rapid progression of oxidative stress (11). An increase in ROS generation may lead to pathological tissue stress responses (8). Our hypothesis is that ROS generation induced by radiation may block Eph-B2-Fc-stimulated EPC adhesion to the endothelium.
We assessed the role of ROS overproduction in wound healing 7 days after injury. We found that the scavenging of ROS by NAC restored the wound closure in irradiated mice injected with Eph-B2-Fc-stimulated EPCs compared with untreated EPCs (Fig. 8A).

Oxidative stress inhibitor beneficial effect in irradiated condition. (A) Skin wound healing process 7 days after injury in mice treated with PBS, Eph-B2-Fc-stimulated EPCs, and EPCs treated with NAC in irradiated conditions. Values are expressed as mean ± SEM; n = 10 per group. **p < 0.01 and ***p < 0.001 versus PBS-injected mice and ≠p < 0.05 versus mice treated with unstimulated EPCs. (B) Representative immunohistochemical image of ROS production in irradiated mice (DHE red staining). (C) Total numbers of adherent progenitors of EPCs at wall shear rates of 1,000 s−1, 4 h after 10 Gy irradiation of EPC and NAC treatment. Each histogram shows the mean ± SEM of data from 40 fields randomly chosen in two separate experiments. Representative photomicrographs of EPCs and irradiated endothelial cells.
Changes in ROS Levels in Skin Tissue
Vascular ROS formation assessed by DHE fluorescence measurement was detectable in irradiated tissue in mice. As expected, superoxide production was downregulated in the irradiated tissue of mice treated with NAC compared with untreated irradiated animals (Fig. 8B).
Decrease in ROS Contents Enhanced the Effect of EPC Adhesion in Irradiated Conditions; no Effect on Eph-B2-Fc-Stimulated EPCs Adhesion Capacity
In a second set of experiments, we evaluated the resistance to detachment by high shear stress of EPCs stimulated or not. We used confocal microscopy to visualize focal adhesions in real time. Thus, we investigated the effect of antioxidant treatment on Eph-B2-Fc-stimulated EPCs on the EPC adhesion to the endothelium.
Three hours after HMVEC irradiation, we observed that the number of adherent EPCs was decreased at 1,000 s−1 wall shear rates compared with no shear stress (Fig. 8C).
Interestingly, after NAC treatment, the number of adherent EPCs was significantly increased 1.3-fold at 1,000 s−1 wall shear rates, compared with untreated EPCs (p < 0.01), whereas no significant effect was observed with Eph-B2-Fc-stimulated EPCs (Fig. 8C). These results suggest that in an antioxidant environment, EPCs enhanced their capacity to improve adhesion. However, this pretreatment was still inefficient to restore the Eph-B2-Fc-stimulated EPC adhesion capacity.
EPC Potentiation by Coadministration with SMPCs
Beneficial Effect of EPC and SMPC Biotherapy on Wound Healing in Irradiated Conditions
Since irradiation-induced ROS accumulation blunted the beneficial effects of Eph-B2-Fc stimulation, we sought to define an alternative strategy to enhance EPC-mediated therapeutic effects. We therefore examined the potential therapeutic effect of EPC and SMPC coadministration on wound healing in irradiated and physiological conditions. Indeed, in several studies, the interaction between endothelial cells and mural cells has been described as a key feature in the regulation of vascular formation and stabilization (1). Our results show that 7 days after injury, SMPC injection alone significantly accelerated wound healing compared to PBS injection (p < 0.01) (Fig. 9A). Moreover, administration of EPCs increased 1.2-fold wound closure compared with PBS-treated animals (p < 0.05). Finally, coadministration of SMPCs further improved the EPC-related effect on wound healing and tissue perfusion compared to EPCs and SMPCs injected alone (Fig. 9A).

Coadministration of EPCs and SMPCs has beneficial effect on wound healing process in irradiated condition. (A, top) Morphology of EPCs (left) and of SMPCs (right) cultured for 2 weeks. (A, bottom) Skin wound healing (left) and cutaneous blood flow perfusion (right) of mice treated with PBS, EPCs, and SMPCs alone and EPCs + SMPCs, 7 days after injury, in irradiated conditions. **p < 0.01 and ***p < 0.001 versus PBS injected animals; ≠p < 0.05 versus EPC injected animals. (B) Homing and incorporation of EPCs into skin wound, 7 days after injury, shown as representative photomicrographs of incorporated EPCs (red labeling; DiI) in histological sections from the wound.
Incorporation of EPCs Into the Wound
We also evaluated incorporation of EPCs into mouse microvasculature by red labeling with CM-DiI red dye. Histological analysis showed that we can localize EPCs in the skin tissue, and coadministration enhanced the number of EPCs in the skin lesion (Fig. 9B). Moreover, we were unable to detect SMPCs in the tissue in our experimental conditions.
Discussion
We report that activation of Eph-B4 with an Eph-B2-Fc chimeric protein improved the effect of EPCs on wound closure in physiological conditions through stimulation of angiogenesis and adhesion to E-selectin and P-selectin, whereas no effect was seen in irradiated conditions. Our results show that inhibitors of oxidative stress restore the beneficial effect of stimulated EPCs in irradiated conditions by stimulating wound closure. Finally, coadministration of EPCs and SMPCs enhanced the efficiency of proangiogenic therapy in this deleterious condition.
In support of these findings, several studies have reported the important roles of Eph receptors and ephrins in tumor angiogenesis (21). Furthermore, in a recent study, ephrin B2 activation induced dynamic changes in cell morphology and invasive behavior of endothelial cells (2). More recently, Wang et al. suggested that ephrin B2 controls VEGF-induced angiogenesis and lymphangiogenesis in a mouse and zebra fish model (30). In our study, we provide evidence that Eph-B2-Fc chimeric protein activation of EPCs accelerates wound healing compared with untreated EPCs by stimulating cutaneous blood perfusion and vascular density. We also found that Eph-B2-Fc-stimulated EPCs promoted capillary tube formation and sprouting more than untreated EPCs. These findings are in agreement with previous studies in a mouse model indicating that Eph-B2-Fc activation enhances postischemic angiogenesis (10). Adhesion molecules (P- and E-selectins) previously known to be involved in the phase of rolling and firm adhesion of leucocytes were thus identified as key regulators of EPC homing. In this study, the authors have demonstrated that activation of EPCs by ephrin B2 leads to higher expression of P-selectin glycoprotein ligand-1 (PSGL-1), a selectin ligand, and subsequently increased adhesion to P- and E-selectin. Other studies have shown that E-selectin also potentiates angiogenesis in ischemic hindlimbs in part by mediating EPC–endothelial cell interaction (22). β2 integrins expressed on the cell surface of EPCs mediate the adhesion and transmigration of EPCs to the damaged endothelial monolayer.
Transplantation of EPCs improves neovascularization of ischemic hindlimb and ischemic heart by integrating new blood vessels and/or secreting proangiogenic factors (15, 16). We have found that human EPCs were incorporated into blood vessels and increased their density. In response to vascular injury, stem cells have to be rapidly mobilized and recruited to the damaged area. The chemokine SDF-1 and its receptor CXCR-4 play a major role in the recruitment of stem cells to ischemic areas (33). In our experiments, we have found that EPCs upregulated SDF-1α plasma levels after skin injury, whereas VEGF secretion is not modulated. Another study recently highlighted the importance of multiple cytokines such as VEGF, angiopoietin-1, SDF-1, and placental growth factor secreted by resident cells in mobilizing bone marrow cells (BMCs) after transplantation of human EPCs into the myocardium of immunodeficient mice (5). We demonstrated that the proangiogenic effect of Eph-B2-Fc stimulation is not mediated by upregulation of these angiogenic factors.
A major complication of ionizing radiation is deficient wound repair associated with changes in the vasculature, leading to progressive loss of vessels (9). The present study clearly demonstrates that the topical administration of EPCs into full-thickness wounds normalizes wound healing in irradiated mice. However, Eph-B2-Fc activation did not improve the proangiogenic capacity of EPCs to accelerate wound healing in irradiated conditions. We also demonstrated that in Matrigel, the endothelial capillary network was ameliorated in the presence of EPCs compared with endothelial cells alone, but there was no stimulation of tube formation by Eph-B2-Fc activation. These results suggest that a deleterious environment in irradiated conditions altered the beneficial effect of Eph-B2-Fc.
As demonstrated previously, Eph-B2-Fc activation enhances the proangiogenic capacity of EPCs by inducing PSGL-1 expression and adhesion to P- and E-selectin (10). We have been therefore tempted to conclude that the inefficiency of Eph-B2-Fc activation in irradiated conditions may be due to downregulation of P- and E-selectin expression after irradiation. Conversely, our results demonstrate that irradiation upregulated expression of endothelial adhesion molecules such as E-selectin and P-selectin in vitro, in vivo, and in patients. Finally, to elucidate the cellular and molecular mechanisms associated with the inactivation of EPCs, stimulated or not, we first assessed vascular permeability. BMC-released nitric oxide (NO) induces vasodilatation of preexisting vascular networks and increases vascular permeability, effects that are crucial to BMC-stimulated neovascularization (24). Our results have shown that EPC injection increases vascular permeability in accordance with the findings of You et al., which suggested that progenitor cell-induced NO-dependent vasodilatation and hyperpermeability were required for their proangiogenic effect (32). It is likely that the effect of EPCs on vascular permeability may enhance their infiltration into the injury area. Interestingly, no effect of Eph-B2-Fc activation on vascular permeability was observed. These results suggest that the inactivation of Eph-B2-Fc treatment in irradiated conditions may be due to its inability to induce vascular permeability.
It has been reported that the generation of ROS is the primary event of tissue–radiation interactions leading to radiation injury and rapid progression of oxidative stress (11). Furthermore, increased ROS production may contribute to vascular alteration and endothelial dysfunction (13). We showed that NAC administration restored Eph-B2-Fc activation in EPC-accelerated wound healing in irradiated conditions. Furthermore, we demonstrated a consistent effect of increasing shear rate, resulting in decreased cell adhesion. In irradiation conditions, NAC administration increased EPC adhesion and reduced EPC detachment after enhancement of shear stress rate, whereas no beneficial effect was observed on Eph-B2-Fc-stimulated EPCs.
A considerable body of evidence suggests that oxidative stress results in disruption of noncovalent bonds between adhesion molecules and their ligands, which explains the inactivation of stimulated EPCs in irradiated conditions. In our condition, we can suppose that the beneficial effect of oxidative stress inhibitors may affect the vascular permeability rather than adhesion capacity.
Finally, we provide evidence that under irradiated conditions, cotransplantation of EPCs and SMPCs derived from umbilical cord blood could be more appropriate in tightly orchestrating the complex process of neovascularization. Using a dual labeling strategy, we were able to track the homing of injected EPCs in skin wound injury. No SMPC engraftment was detectable in the wound, suggesting that the increased efficacy of cotherapy results from paracrine secretion by SMPC- and EPC-related functions. To determine the potential mechanism of the SMPC effect, Foubert et al. showed that knockdown of angiopoietin-1 (Ang-1) expression in SMPCs or Tunica interna endothelial cell kinase 2 (Tie2; Ang-1 receptor) expression in EPCs by siRNA diminished the therapeutic effect of the cell therapy. They concluded that production of Ang-1 by SMPC activates Tie-expressing EPCs, resulting in an increase in EPC survival and formation of a stable vascular network.
In conclusion, our study demonstrated that EPCs participate in dermal wound healing in physiological and pathological conditions. Stimulation by Eph-B2-Fc improved the beneficial effect of EPCs in physiological conditions and irradiated conditions with antioxidant treatment. Moreover, coadministration of EPCs and SMPCs may improve the therapeutic effect of cell therapy. In consequence, the development of a new therapeutic approach using adult stem cells to control inflammation, accelerate reepithelialization, and vessel growth constitute a major challenge for tissue regeneration or repair after irradiation. Finally, this study is important because it suggests potential strategies to overcome the limited therapeutic successes seen in early clinical trials of human progenitor cell therapy. Use of multiple cell types, or genetically engineered cells, or combinations of progenitor cells with cytokines, growth factors, or transcription factors is likely to be more beneficial than EPCs alone.
Footnotes
Acknowledgments
We thank Dr. Teni Ebrahimian for her contribution to the in vivo experiments and cell culture. The authors declare no conflicts of interest.
