Abstract
Krabbe's disease (KD) is a demyelinating disorder caused by the deficiency of lysosomal galactocerebrosi-dase (GALC), affecting both the central (CNS) and the peripheral nervous system (PNS). A current therapy, hematopoietic stem cell transplantation (HSCT), is ineffective at correcting the PNS pathology. We have previously shown that systemic delivery of immortalized bone marrow-derived murine mesenchymal stromal cells (BM-MSCs) diminishes the neuropathology of transplanted Twitcher mice, a murine model of KD. In this study, to move one step closer to clinical application, the effectiveness of a systematic delivery of primary BM-MSCs to promote recovery of the Twitcher PNS was assessed. Primary BM-MSCsgrafted to the Twitcher sciatic nerve led to increased GALC activity that was not correlated to decreased psychosine (the toxic GALC substrate) accumulation. Nevertheless, BM-MSC transplantation rescued the axonal phenotype of Twitcher mice in the sciatic nerve, with an increased density of both myelinated and unmyelinated axons in transplanted animals. Whereas no increase in myelination was observed, upon transplantation an increased proliferation of Schwann cell precursors occurred. Supporting these findings, in vitro, BM-MSCs promoted neurite outgrowth of Twitcher sensory neurons and proliferation of Twitcher Schwann cells. Moreover, BM-MSCs expressed nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF) and promoted increased BDNF synthesis by neighboring Schwann cells. Besides their action in neurons and glia, BM-MSCs led to macrophage activation in Twitcher sciatic nerves. In summary, primary BM-MSCs diminish the neuropathology of Twitcher sciatic nerves by coordinately affecting neurons, glia, and macrophages.
Keywords
Introduction
Krabbe's disease (KD) is an inherited demyelinating disorder caused by the autosomal recessive mutation of the lysosomal enzyme galactocerebrosidase (GALC) (49). Due to the incomplete metabolism and accumulation of GALC substrates, especially of the toxic lipid raft-associated sphingolipid psychosine, apoptosis of myelin-forming oligodendrocytes and Schwann cells occurs, affecting both the central (CNS) and the peripheral nervous system (PNS) (15,21,52,56). Another characteristic feature of KD is the strong inflammatory response characterized by the accumulation of macrophages in the PNS or microglia-derived cells (the globoid cells) in the CNS, both building up inclusions of galactocerebroside, the other GALC substrate (1,16).
The only current available therapy for KD patients is hematopoietic stem cell transplantation (HSCT). In this treatment, HSCs allow restoration of enzymatic activity (27), with patients reaching GALC levels close to normal values (9,26). Moreover, it has been shown that HSCT can stabilize or even improve the cognitive functions in KD (9,26). However, even when treated before the onset of symptoms, these patients still present severally impaired peripheral nerve conduction (40), showing that HSCT is not able to successfully correct the PNS pathology. Studies in the Twitcher mouse, the murine model for KD (43), also support these findings, showing that after HSCT animals still present peripheral nerve defects (14,24).
As GALC activity is defective in the Twitcher mouse, neuropathological features are very similar to human KD, and thus this model is a valuable tool for developing effective therapies for this disorder (42,45,46). Besides demyelination, recent reports suggest that axonal degeneration occurs in Twitcher mice (2,12,52). In this respect, it has been shown that in this mouse model the accumulation of psychosine results in the disruption of lipid rafts not only in oligodendrocytes and Schwann cells but also in neurons (52). In a separate study, it was reported that although coinfusion of HSCs and of a GALC-lentiviral vector improved myelination and life span, axonal degeneration was still occurring (12). More recently, Twitcher mice were shown to be affected by an early and progressive axonal dying-back neuropathy, which contributes to the evolution of KD and is contemporaneous to demyeli-nation (2). Together, these reports provide evidence that in KD the so far neglected axonal pathology should be regarded with caution because not only may it be independent of demyelination, but also may contribute more than previously suspected to the observed pathology.
Mesenchymal stromal cell (MSC) delivery has been extensively used as a therapeutic strategy in a number of disorders including diseases affecting the CNS and PNS (4,5,48). This widespread use of MSCs is due to their amenable properties. This cell type is easy to harvest and expand without major ethical problems; has low immuno-genicity, allowing allogeneic transplants (3); has a general lack of tumorigenic properties (6); and has the ability to home to target tissues when injected systemically (55). Lately, the interest in assessing the therapeutic efficacy of bone marrow-derived MSCs (BM-MSCs) in leukodystro-phies including KD has also increased (32,36). Murine BM-MSCs have been injected intracranially into newborn Twitcher mice at postnatal day (PND) 3–4, promoting increased body weight gain, improved motor function, and a longer life span (36). In this report, it was proposed that BM-MSCs act through an immunomodulatory effect, as downregulation of the expression of several proinflam-matory molecules was shown. In a recent study of our group (32), systemic delivery of a telomerase reverse tran-scriptase-immortalized cell line of murine BM-MSCs was shown to diminish the neuropathology of Twitcher mice if delivered systemically at PND 23–25. In transplanted Twitcher mice, immortalized BM-MSCs promoted axonal regeneration and/or survival. Because axonal degeneration might be impeditive for the efficacy of current treatments, as these are mainly focused at improving myelination (12), the addition of BM-MSCs to HSCT might be advantageous in KD. Therefore, the aim of this study was to investigate whether intravenous (IV) delivery of a primary source of BM-MSCs has a similar efficacy as immortalized BM-MSCs, allowing us to move one step forward into the clinical application of this strategy in KD.
Materials and Methods
Animal studies were performed following approval by the Instituto de Biologia Molecular e Celular (IBMC) Animal Ethics Committee and by the Portuguese Veterinarian Board.
BM-MSC Isolation and Cell Culture
As BM-MSC donors, 6- to 8-week-old enhanced green fluorescent protein (EGFP) transgenic mice of both genders in a 129Sv/B6 mixed background expressing EGFP under the control of the β-actin promoter (Jackson Laboratories, Bar Harbor, ME, USA) (33) were used. For isolation of BM-MSCs, tibias and femurs were flushed and mononuclear cells were separated on a Ficoll (Histopaque-1077; Sigma, Sintra, Portugal) gradient and resuspended in Euromed-N (Euroclone, Milan, Italy) containing 10% fetal bovine serum (FBS; Gibco, Life Technologies Europe BV, Porto, Portugal), 20 ng/ml epidermal growth factor (Abcam, Cambridge, UK), 20 ng/ml fibroblast growth fac-tor-2 (Abcam), and 2% B27 (Invitrogen, Madrid, Spain). Forty-eight hours later, nonadherent cells were removed and adherent cells were expanded for approximately 2-3 weeks, with medium changes every 2-3 days, until confluence was reached. BM-MSC-conditioned medium was collected from cultures at 80-90% confluency, growing in medium without supplements for the last 32 h. For adipogenic and osteogenic differentiation, cells were grown until subconfluency after which they were exposed to osteoblastic and adipogenic differentiation media (Cambrex Bio Science, Verviers, Belgium) for 15 days as previously described (23) and subsequently fixed with 4% paraformaldehyde (Sigma). Osteoblasts were visualized by alkaline phosphatase staining using von Kossa (Sigma). Adipocyte lipid granules were assessed through oil red O (Sigma) staining. In the case of cocultures, 100,000/well Twitcher-derived Schwann cells (TwS1; gender unknown) (39) were seeded in 24-well plates (Corning, Corning, NY, USA) and grown for 16 h with Dulbecco's modified Eagle medium (DMEM)/F12 (Invitrogen) with 10% FBS. Subsequently, BM-MSCs (20,000 cells/insert in 1.0-μm-pore Transwells) (BD Falcon, Bedford, MA, USA) were added for 1 day, after which Schwann cell pellets were prepared. Schwann cells grown without BM-MSCs were used as controls.
Reverse Transcription Polymerase Chain Reaction (RT-PCR)
Total ribonucleic acid (RNA) extraction was performed with Trizol (Invitrogen) and reverse transcription with the Superscript II kit (Invitrogen) using oligo-dT BM-MSC expression of selected genes was analyzed with specific primers (Table 1) designed with Primer3 software (37). PCR (95°C for 5 min; 95°C for 1 min, 56°C for 2 min, 72°C for 3 min, 35 cycles; 72°C for 6 min) was performed using 1 μl of complementary deoxyribonucleic acid (cDNA). Quantitative PCR (qPCR) for brain-derived neurotrophic factor (BDNF) was performed with the iQ™ SYBR Green Supermix kit (Biorad, Munich, Germany) and primers designed with Beacon7.5 (Premier Biosoft, Palo Alto, CA, USA; Table 1) at 94°C for 3 min and 94°C for 15 s, 59°C for 20 s, 72°C for 15 s, 40 cycles. β-Actin expression was used for normalization, and quantifications were made using the Livak method (29).
Primers Designed Using the Primer3 Software and Used for RT-PCR to Analyze the Expression of Selected Genes (First Six Genes of the Table) by Primary BM-MSCs and Primers Designed Using the Beacon 7.5 Software Used for the Real-Time qPCR Assessment of BDNF Expression by Schwann Cells Grown in the Presence of BM-MSCs

Expression of β-actin was used as internal control. BM-MSCs, bone marrow derived-mesenchymal stromal cells; NST, nucleostemin; GALC, galacto-cerobrosidase; BDNF, brain-derived neurotrophic factor; NT3, neurotrophin 3; NGF, nerve growth factor.
Twitcher Mice
Twitcher mice and wild-type (WT) littermates of both genders (in a C57BL/6J background) were obtained from heterozygous breeding pairs (Jackson Laboratory) and genotyped as described (38). After weaning at PND 21, mice were provided accessible wet rodent chow. In all procedures, Twitcher mice were euthanized according to the IBMC humane endpoint standard operation procedure established according to FELASA's recommendations (http://www.felasa.eu/) when any of the following features was observed: severely impaired mobility, prolonged dehydration, decreased food intake (for more than 72 h), or severe weight loss (more than 20%). To minimize the putative arbitrariness of some of the end-points, euthanasia was decided with the observer blinded to the animal's experimental group (i.e., transplanted vs. nontransplanted). The life span of transplanted and non-transplanted animals was 38.3 ± 1.3 and 37.7 ± 1.3 days, respectively. Within each group and among different groups, age variations were minimal; thus, the possibility that different stages of disease progression might have contributed to the results observed is reduced.
Transplantation of BM-MSCs
Twenty-two- to 25-day-old Twitcher mice of both genders were subjected to allogeneic transplantation with BM-MSCs; 1.5×106–2.0×106 cells were injected IV into the tail vein. The dose of transplanted cells was chosen using as a reference previous studies with IV delivery of primary BM-MSCs, where the dose of transplanted cells ranged from 1 × 106 to 2 × 106 and transplantation was shown to have a beneficial outcome in the nervous system (31,58). Cell viability was controlled prior to delivery through trypan blue staining (Sigma); only live cells were accounted for transplantation. Control animals were injected with DMEM and are hereafter referred to as non-transplanted. Cyclosporin A (Calbiochem, Nottingham, UK; 10 mg/kg per day) was injected intraperitoneally starting on the day before transplantation and throughout the whole experimental period. Nontransplanted Twitcher mice used as controls were also immunosuppressed.
Immunohistochemistry
Immunohistochemistry (IHC) was performed in 4-μm-thick paraffin longitudinal sections of sciatic nerves and spinal cords. Slides were incubated overnight at 4°C with the following polyclonal antibodies: rabbit anti-mouse glial fibrillary acidic protein (GFAP, 1:500, Dako, Glostrup, Denmark), goat anti-mouse ionized calcium binding adaptor molecule 1 (Iba-1, 1:200, Abcam), rabbit anti-mouse EGFP (1:500; produced at the IBMC), mouse antipro-liferating cell nuclear antigen (PCNA) (1:300, Dako); monoclonal mouse anti-GALC (1:10, generously provided by Dr. Eckman, Mayo Clinic College of Medicine, Jacksonville, FL, USA), and rabbit anti-cleaved caspase-3 (1:300, Cell Signaling, Hitchin, Hertfordshire, UK). The appropriate biotinylated secondary antibodies were used, amplified with the ABC reagent (Vector Laboratories, Peterborough, UK), and developed with 3-amino-9-ethyl carbazole or 3,3′-diaminobenzidine. For EGFP, signal amplification was performed with extravidin-alkaline phosphatase (1:100, Sigma), and Fast Red Naphthol (Sigma) was used for development. Tissue sections without primary antibody were used as negative controls. Nuclei were stained with hematoxylin and eosin (Sigma). For EGFP and GALC colocalization, two consecutive IHCs were performed on the same slide, ensuring no cross-reactivity between the series of antibodies used. Positive cells for EGFP were counted on four longitudinal sections of the sciatic nerve, spaced between each other by 20 μm, in three BM-MSC-transplanted Twitcher mice (age range 35–39 days). The cross-sectional areas of the sciatic nerves were calculated to determine the density of EGFP+ cells grafted. EGFP+ cells were photographed and their coordinates were registered. Subsequently, IHC was performed against GALC. For GFAP and Iba-1, at least four sections from five BM-MSC-transplanted (age range 36–40 days) and three nontransplanted (age range 36–38 days) Twitcher mice were analyzed. For quantification of PCNA+ cells, these were scored in four nonoverlapping micrographs (40× magnification) in three nontransplanted and three transplanted Twitcher mice (age ranges 34–36 and 35–39 days, respectively). Motor neurons positive for anti-cleaved caspase-3 were identified by morphology and scored in a total of 100 nonoverlapping 40× magnified micrographs of the spinal cord in three nontransplanted Twitcher mice (age range 34–36 days) and two BM-MSC transplanted Twitcher mice (age range 35–39 days). The total area scored was calculated; results are presented as the number of cleaved caspase-3-positive motor neurons per square millimeter.
GALC Enzymatic Activity
Sciatic nerves (n = 6 for each group; age range: WT 39–41 days, nontransplanted Twitcher mice 38–42 days, and transplanted Twitcher mice 40–42 days) as well as BM-MSC and TwS1 cell (cocultured for 48 h with or without BM-MSCs) homogenates were prepared in water by sonication. The cell extracts and culture media were assayed for GALC activity using the synthetic substrate 6-hexadecanoylamino-4-methylumbelliferyl-β-D-galac-toside (HMU-βGal; Moscerdam, Oegstgeest, Netherlands), as described (54). Total protein of cell extracts was measured by the Lowry method (30). Fluorescence was measured on a Hitachi F2000 spectrofluorometer (Tokyo, Japan) with excitation and emission wavelengths of 404 and 460 nm, respectively. Specific activity was calculated as nanomoles per hour per milligram protein.
Measurement of Psychosine Levels
Quantification of psychosine was performed by elec-trospray ionization (ESI)-tandem mass spectrometry (MS/ MS) as described (53). Briefly, sciatic nerves (n = 6 for each group; age range: WT 39–41 days, nontransplanted Twitcher mice 38–42 days, and transplanted Twitcher mice 40–42 days) were homogenized in water, and protein quantification was performed by Lowry's method. Methanol and the lactosylsphingosine internal standard (Matreya LLC, Pleasant Gap, PA, USA) were added to the extract. Subsequently, galactosylsphingosine was isolated using a strong cation exchange solid-phase extraction cartridge (Supelclean™; Sigma). The eluate was desalted by C18 solid-phase extraction (Bond Elut®; Agilent Technologies, Lisboa, Portugal), dried, and reconstituted in methanol/ ammonium formate (Sigma). To quantify psychosine by high-performance liquid chromatography (HPLC)-MS/ MS using multiple-reaction monitoring, galactosylsphingosine and lactosylsphingosine were eluted using a linear gradient up to 100% methanol. Quantification of galactosylsphingosine was performed using multiple-reaction monitoring mode. Galactosylsphingosine and the lactosylsphingosine internal standard were identified by precursor ion scanning for mass-to-charge ratio (m/z) 282 and 264 in the positive ion mode. All analyses were performed by ultra performance liquid chromatography-ESI (UPLC-ESI)-MS/MS using a Waters ACQUITY UPLC™ System (Milford, MA, USA) with a Waters Quattro Premier™ XE benchtop tandem quadrupole mass spectrometer.
Morphometric Analysis
Sciatic nerve segments (2 mm) collected distally from the sciatic notch were processed as described (11). One-micrometer-thick transverse sections covering the complete cross-sectional nerve area were stained with p-phenylene-diamine (PPD; Sigma) (13). For each nerve, several 40x magnified photomicrographs covering the entire cross section were taken in a light microscope (Olympus CX31 with DP-25 Camera; Tokyo, Japan); these photos were then loaded in Adobe Photoshop CS5 (San Francisco, CA, USA) to generate a single high-resolution file covering the complete cross section; the total number of myelinated axons in the entire cross-sectional nerve area was counted manually in this composed file. The total cross-sectional area of each nerve was determined from 4x magnified photomicrographs (using the Imaging Software Cell B; Olympus) and the density of myelinated axons was calculated. The g-ratio was calculated by dividing the diameter of each axon by its myelin-including diameter in over 100 axons per animal. Total macrophages, identified by morphology, were counted in one sciatic nerve cross section/animal stained with PPD. To determine the density of unmyelinated axons, ultrathin transverse sciatic nerve sections (50 nm) were cut and stained with uranyl acetate and lead citrate (both Sigma). For each animal, 15 nonoverlapping photomicrographs (6700x magnification) of each section were taken in a transmission electron microscope (JEOL 100CX II, Tokyo, Japan), the number of unmyelinated axons was counted and the total area was calculated. To determine the myelinated axon density, four transplanted (age range 39–44 days), four nontransplanted Twitcher mice (age range 40-44 days), and three WT mice (age range 38–44 days) were used. For g-ratio calculation, n = 6 animals per group were used (age ranges: 39–41 days and 37–40 days for non-transplanted and transplanted Twitcher mice, respectively). To assess the unmyelinated axon density, five transplanted (age range 38–44 days), three nontransplanted Twitcher mice (age range 39–44 days), and four WT mice (age range 38–44 days) were analyzed. To determine macrophage density, six nontransplanted (age range 39–41 days) and six transplanted Twitcher mice (age range 37–40 days) were used. All quantifications were performed with the observer blinded to the experimental condition.
Cocultures of Dorsal Root Ganglia (DRG) Neurons with BM-MSCs
Primary sensory neurons were isolated from 4-week-old Twitcher mice as described (11). Briefly, DRG neurons were treated with 0.125% collagenase IV-S (Sigma) for 3 h at 37°C, and a single-cell suspension was obtained by trituration with a fire-polished Pasteur pipette (VWR International, Carnaxide, Portugal) and centrifuged over a 15% albumin cushion (Sigma) for 10 min at 200 × g. The dissociated neurons were seeded on poly-L-lysine (Sigma)/ laminin (Sigma)-coated coverslides (VWR International) in 24-well plates at 7,500 cells/well. After 16 h, DRG were cocultured with BM-MSCs incubated in Iscove's modified Dulbecco medium (IMDM, Cambrex) supplemented with 10% FBS (40,000 cells/insert) using a 1.0-μm-pore Transwell system for 24 h. Control neurons were not exposed to BM-MSCs. To analyze neurite outgrowth, fixed DRG neurons were permeabilized in 0.2% Triton X-100 (Sigma) and neurites were visualized using the mouse anti-b-tubulin III antibody (1:2,000, Promega, Madison, WI, USA) and anti-mouse Alexa Fluor 568 (1:1,000, Molecular Probes, Madrid, Spain). All neurites with a length greater than the cell body diameter were measured using ImageJ/NeuronJ (NIH, Bethesda, MD, USA/E, Meijering, Biomedical Imaging Group, Rotterdam, Netherlands). Measurements were done in over 80 neurons per condition.
BDNF Quantification by Enzyme-Linked Immunosorbent Assay (ELISA)
BM-MSCs were grown until 80–90% confluence in T25 flasks (VWR International) with 4 ml of IMDM without supplements, and 100 μl was used for BDNF quantification using the Emax Immunoassay System (Promega) according to the manufacturer's instructions.
5-Bromo-2-Deoxyuridine (BrdU) Proliferation Assay
Primary Schwann cells or TwS1 cells were incubated for 24 h with BM-MSC conditioned medium and treated with 10 μM BrdU (Sigma) during the last 7 h of incubation. Fixed cells were treated with 2 M HCl (Fluka/Sigma) for 20 min, incubated with monoclonal mouse anti-BrdU (1:10, DakoCytomation, Glostrup, Denmark) for 1 h and anti-mouse Alexa Fluor 568 (1:1,000, Molecular Probes) for 30 min. The percentage of BrdU+ cells was calculated from over 500 cells per condition.
Statistical Analysis
Results are presented as mean (±standard error of the mean, SEM). All analyses were done using the Student's t test. In case of multiple comparisons, Student's t test with Bonferroni correction was used. A value of p < 0.05 was considered statistically significant (*p < 0.05, **p < 0.01, ***p < 0.001).
Results
Primary BM-MSCs Express Active GALC, the Defective Enzyme in KD
The multipotency of the isolated BM-MSCs was supported by the expression of stemness-associated markers such as nucleostemin and nanog (Fig. 1A) and by their capacity to differentiate in vitro into mesenchymal lineages, namely, adipocytes (Fig. 1B, upper) and osteo-blasts (Fig. 1B, lower). Moreover, BM-MSCs expressed the messenger RNA (mRNA) for the defective enzyme in KD, GALC (Fig. 1A), and produced the active form of the enzyme with an enzymatic activity of 6.9 ± 1.4 nmol/h per milligram protein, which is within the range of Schwann cells (8.1 nmol/h per milligram protein) and human fibroblasts (1.5–4.5 nmol/h per milligram protein) (35), and therefore they are an attractive cell source for therapeutic application in KD.
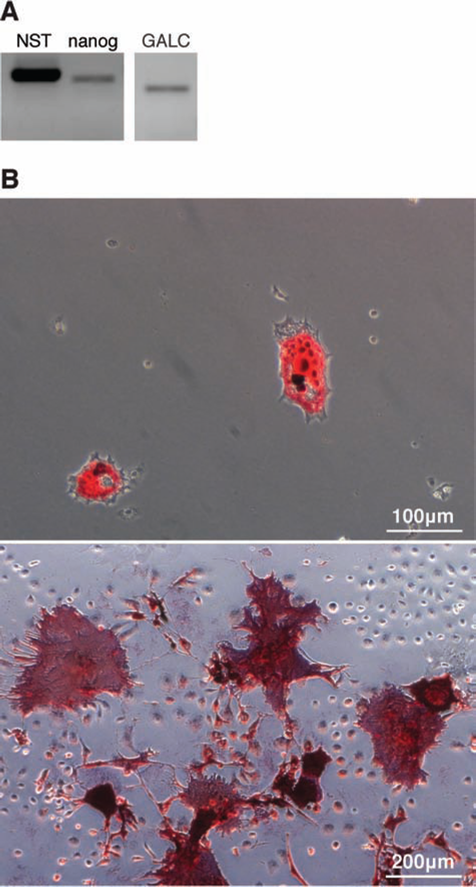
Primary BM-MSCs express active GALC, the defective enzyme in KD. (A) RT-PCR analysis of bone marrow-derived murine mesenchymal stromal cell (BM-MSC) expression of the stemness-related markers nucleostemin (NST) and nanog and of the defective enzyme in KD (galactocerebrosidase, GALC). (B) Differentiation of BM-MSCs into adipocytes (top) and osteoblasts (bottom) visualized by oil red O staining and alkaline phosphatase reaction, respectively.
BM-MSCs Graft Twitcher Sciatic Nerves and Express Active GALC Upon Transplantation
Although BM-MSCs are generally considered non-immunogenic, recent evidence has suggested that MSCs are not intrinsically immune privileged and that under the settings of allogeneic transplantation, as is the case in this study, these cells may induce rejection followed by an immune memory (57). Thus, a mild immunosup-pression with cyclosporin A was chosen as an attempt to enhance BM-MSC graft survival. As BM-MSCs have the ability to home to injured tissues when delivered systemi-cally (3), and as the Twitcher blood–nerve barrier (BNB) is impaired, thus facilitating BM-MSC grafting (32), we searched for EGFP+ cells in the sciatic nerves of transplanted Twitcher mice. EGFP+ cells were found at low density (8.7 ± 2.9 cells/mm2) in the endoneurium of the nerves analyzed (Fig. 2A, left). EGFP+ cells colocalized with the defective enzyme GALC (Fig. 2A, right), thus supporting the in vivo expression of the enzyme by grafted cells. GALC activity in BM-MSCs was determined before transplantation as being 6.9 ± 1.4 nmol/h per milligram of protein. Accordingly, in the sciatic nerves of transplanted Twitcher mice, GALC had a significant 1.8-fold increase in activity when compared to nontransplanted Twitcher nerves, reaching ~42% of the activity of GALC in WT nerves (Fig. 2B). Even though functional GALC was present in the sciatic nerve of the transplanted Twitcher mice, no decrease in psychosine accumulation was found in their nerves (Fig. 2C). Interestingly, upon in vitro coculture of TwS1 cells with BM-MSCs in a Transwell system, no detectable increase in GALC enzymatic activity was observed in TwS1 cell homogenates (Fig. 2D). Moreover, when the GALC activity was measured in the extracellular milieu, the amount of secreted GALC was low (0.04 nmol/h per milliliter), which probably underlies the lack of cross-correction.

BM-MSCs graft Twitcher sciatic nerves and express active GALC upon transplantation. (A) Anti-enhanced green fluorescent protein (EGFP) (left; positive cells highlighted by arrows) colocalization with anti-GALC (right, positive cells highlighted by arrows) immunohistochemistry in Twitcher sciatic nerves (n = 3 transplanted Twitcher mice; age range 35–39 days); counterstaining performed with hematoxylin and eosin. (B) GALC activity (nmol/h per mg protein) and (C) psychosine levels (pmol/3 mg of wet weight tissue) in the sciatic nerve of wild-type (WT) and Twitcher mice either transplanted (Twi + MSC) or nontransplanted (Twi) (n = 6 for each group; age range: WT 39–41 days; Twi 38–42 days; Twi + MSC 40–42 days). (D) GALC activity (nmol/h per mg protein) measured in MSC and TwS1 cell homogenates cultured in the presence (TwS1 + MSC) or absence (TwS1) of BM-MSCs in a Transwell system.
BM-MSCs Rescue the Axonal Pathology of Twitcher Mice Through a Neurotrophin-Dependent Mechanism
Delivery of immortalized BM-MSCs to Twitcher mice has a major impact on axonal regeneration/survival; we therefore evaluated whether primary BM-MSC transplantation was related to an increased axonal density in this mouse model. The density of unmyelinated axons was 3.1-fold increased in transplanted Twitcher mice when compared to nontransplanted animals, reaching 51% of the density of WT mice (Fig. 3A). A similar trend was observed for the density of myelinated axons, as transplanted Twitcher mice had a 1.2-fold increase in myeli-nated fibers when compared to nontransplanted mutants (Fig. 3B). Despite the increase in myelinated axons, no difference in g-ratio was found (data not shown), therefore demonstrating that supporting myelination is not the primary mechanism of action of transplanted BM-MSCs. Of note, there was no significant difference in the cross-sectional nerve areas between BM-MSC transplanted and nontransplanted Twitcher mice (0.22 ± 0.03 vs. 0.25 ± 0.04 mm2, respectively, p=0.17), excluding a possible effect of transplantation on nerve edema. In Twitcher peroneal nerves, a ratio of 3.2 for unmyelinated/myelinated axons was previously reported (20), whereas in our work this ratio in the sciatic nerve was 2.7. This slight variation is probably due to the different nerves assessed in the two studies. It is interesting to note that following BM-MSC delivery, this ratio increased to 7.1, which reinforces the preferential targeting of unmyelinated axons over myelinated ones.

BM-MSCs partially rescue the axonal pathology of Twitcher mice. Density of (A) unmyelinated and (B) myelinated axons of wild-type (WT) and Twitcher sciatic nerves either nontransplanted (Twi) or transplanted with BM-MSCs (Twi + MSC). For unmy-elinated axon density assessment, the following animals were analyzed: Twi + MSC (n = 5, age range 38–44 days), Twi (n = 3, age range 39–44 days), WT (n = 4, age range 38–44 days). For myelinated axon density assessment, Twi + MSC (n = 4, age range 39–44 days), Twi (n = 4, age range 40–44 days), and WT mice (n = 3, age range 38–44 days) were used. Representative electron microscopy photomicrographs of ultrathin sciatic nerve sections with arrows highlighting unmyelinated axons are below (A), and representative sciatic nerve sections stained with p-phenylenediamine (PPD) are shown below (B). (C) Density of cleaved caspase-3-positive motor neurons in the spinal cords of Twi (n = 3, age range 34–36 days) and Twi + MSC (n = 2, age range 35–39 days). Representative spinal cord sections are shown, with an arrow highlighting an immunostained motor neuron in Twi (left). (D) Mean length of the longest neurite of Twitcher dorsal root ganglia (Twi DRG) neurons grown in the absence or presence of BM-MSCs (Twi DRG + MSC). Representative photomicrographs of Twi DRG neurons and Twi DRG + MSC stained with β-tubulin III are shown. (E) RT-PCR analysis of the expression of brain-derived neurotrophic factor (BDNF), neurotrophin 3 (NT3), and nerve growth factor (NGF) by BM-MSCs. (F) BDNF mRNA levels measured by qPCR in TwS1 cells growing in the presence of BM-MSCs (TwS1 + MSC) or in the absence of BM-MSCs (TwS1).
The sciatic nerve contains the axons from motor neurons besides the axons from DRG neurons; we therefore evaluated how transplant influences these motor neurons. Recently, it has been described that caspase-3 is activated in Twitcher mouse neurons (41); thus, we performed immunohistochemistry for active caspase-3 in the spinal cord. A decrease in cleaved caspase-3 was detected in motor neurons from transplanted Twitcher mice when compared to nontransplanted Twitcher mice (3.9 ± 1.7 vs. 11.4 ± 0.4 cleaved caspase-3-positive motor neurons per square millimeter, respectively) (Fig. 3C), further sustaining a supportive role of BM-MSC transplantation and suggesting that it is not restricted to the PNS.
In accordance with a protective role of BM-MSCs in vivo, when Twitcher DRG neurons were grown in the presence of BM-MSCs in a coculture system, a 1.5-fold increase in the length of the longest neurite was observed (Fig. 3D). In brief, in Twitcher mice, BM-MSCs have a neuritogenic effect both in vivo and in vitro, instead of favoring myelination. Similar to immortalized BM-MSCs, primary BM-MSCs expressed the neurotrophins BDNF and nerve growth factor (NGF) but not neurotrophin-3 (NT3) (Fig. 3E) (8,32). Moreover, these cells were not only capable of secreting BDNF (23.3 ± 1.1 pg BDNF/ml per 106 cells) but also led to a 2.4-fold increase in the levels of BDNF expressed by TwS1 cells when in coculture (Fig. 3F). Given the important role of neurotrophins in supporting axonal growth/survival, our data suggest that BM-MSCs may exert their effect on neurons, at least in part, through neurotrophin secretion.
BM-MSCs Stimulate the Proliferation of Twitcher-Derived Schwann Cells
Despite the absence of any detectable effect on myeli-nation, BM-MSC transplantation promoted the proliferation of Schwann cell precursors, as transplanted Twitcher mice had an increased GFAP immunostaining in the sciatic nerve (Fig. 4A, right) and a 4.1-fold increased density of PCNA+ cells (Fig. 4B) when compared to nontrans-planted mutants. Accordingly, in vitro, the conditioned medium of BM-MSCs induced the proliferation of both primary Schwann cells and immortalized TwiS1 cells, as detected by scoring BrdU+ cells (Fig. 4C).

BM-MSCs stimulate the proliferation of Twitcher-derived Schwann cells. (A) Glial fibrillary acidic protein (GFAP) immu-nohistochemistry (IHC) in transverse sciatic nerve sections from nontransplanted (Twi) (n = 5, age range 36–38 days) or BM-MSC transplanted (Twi + MSC) (n = 3, age range 36–40 days) Twitcher mice (arrows illustrate GFAP-positive cells); counterstaining performed with hematoxylin. (B) Quantification of proliferating cell nuclear antigen positive (PCNA+) cells, assessed by IHC staining, in sciatic nerve transverse sections of Twi and Twi + MSC (n = 3 for each group; age ranges 34–36 and 35–39 days, respectively). (C) Percentage of bromodeoxyuridine-positive (BrdU+) nuclei in primary Schwann cells (SC) and in a Twitcher-derived Schwann cell line (TwS1) when grown either with or without BM-MSC conditioned medium (CM).
BM-MSCs Promote the Activation of Macrophages in the Sciatic Nerve
Although macrophage invasion is normally considered a pathological feature in KD, these cells can counteract demyelination by promoting clearance of myelin debris (25). BM-MSCs play a strong immunomodulatory effect (55), so we performed Iba-1 IHC to assess the effect of transplantation on macrophage activation. Transplanted Twitcher mice had an increase in active macrophages when compared to nontransplanted mutants (Fig. 5A, right and left, respectively), although no differences were found in the total number of macrophages (Fig. 5B).

BM-MSCs promote the activation of macrophages in Twitcher sciatic nerves. (A) Ionized calcium binding adaptor molecule 1 (Iba-1) IHC in sciatic nerves of nontransplanted Twitcher mice (Twi) (n = 5, age range 36–38 days) and Twitcher mice transplanted with BM-MSCs (Twi + MSC) (n = 3, age range 36–40 days); counterstaining performed with hematoxylin; arrows highlight Iba-1+ cells. (B) Macrophage density in the sciatic nerve of Twi and Twi + MSC (n = 6 each group; Twi: age range 39–41 days and Twi + MSC: age range 37–40 days).
Discussion
Current therapies for KD are not successful in correcting the neuropathology in the PNS. The present study was designed to test whether primary BM-MSCs are capable of targeting the Twitcher PNS. We show that primary BM-MSCs rescue the axonal pathology of Twitcher mice, and thus our study reinforces the importance of considering BM-MSCs, in synergy with other therapies, as possible candidates for clinical application in KD patients. It is generally accepted that treatments for KD should be started on asymptomatic newborn babies with KD, as transplants in asymptomatic carriers appear to offer the highest potential for correction. However, most of the KD patients referred to the clinics are already symptomatic, as newborn screening for KD is very limited. Moreover, up to now, the timing of HSCT in KD remains experimental. As such, testing therapeutic strategies aiming at the correction of KD in symptomatic patients is still a valid approach despite the fact that transplantation in asymptomatic Twitcher mice would certainly result in an improved therapeutic outcome.
KD is a classical demyelinating disorder, and less attention has been given to the concomitant neuronal/ axonal degeneration, generally referred to as a consequence of myelin loss. However, recent reports suggest that the reason underlying the failure of current therapies for KD is the fact that they neglect axonal loss as they are mainly focused on endorsing remyelination (12). In this context, add-on therapies with BM-MSCs are particularly interesting for KD because, as we show here, these cells impact specifically axonal regeneration and/or survival when delivered systemically in a mouse model of KD. In the settings of this study, the optimal dosage of cyclosporine should be further assessed as it is possible that mild immunosuppression has little effect on the survival of allogeneic MSC grafts (44,50). Furthermore, this study does not exclude that cyclosporin A might be involved in some of the differences observed between transplanted and nontransplanted Twitcher mice as non-immunosuppressed animals have not been included in this study. In some clinical studies, MSCs have proven to be safe and have been suggested to enhance engraftment of HSCs and/or to reduce the occurrence of graft-versus-host disease after allogeneic transplantation (10,47). Cotransplantation of BM-MSCs and HSCs has already been applied to patients with metachromatic leukodystro-phy and Hurler syndrome, and it was shown that in patients with metachromatic leukodystrophy BM-MSCs enhanced the performance of HSCs, thus supporting the therapeutic efficacy of using coinfusions of BM-MSCs and HSCs to treat KD (22). Although the assessment of a putative effect of BM-MSCs in the CNS of Twitcher mice was beyond the scope of our study, future research should also address whether these cells are capable of promoting a decrease in CNS pathology in this model.
Although their overall action was similar to that of the previously studied BM-MSC line, primary BM-MSCs were more potent in rescuing the axonal pathology of Twitcher mice. A higher density of unmyelinated axons was obtained with primary BM-MSC transplantation (3.1-fold increase) than with the BM-MSC line (2.4fold increase) (32). It is noteworthy that in both studies BM-MSC transplantation had a stronger effect in supporting unmyelinated axons than myelinated ones (for primary BM-MSCs, transplantation led to an increase of 3.1-fold of the density of unmyelinated axons, whereas the density of myelinated axons was increased by 1.2fold). A putative effect of BM-MSC transplantation on the dying back neuropathy that occurs in Twitcher mice (2) remains to be further explored and would probably be studied in more detail in longitudinal nerve sections. Moreover, the possible functional impact of BM-MSCs remains to be determined and could be assessed in the future by performing nerve conduction velocity studies. In addition to a stronger protective effect toward axonal loss, primary BM-MSCs also promoted a higher increase in the proliferation of Schwann cell precursors when compared to the immortalized BM-MSC line previously assessed (4.1-fold vs. 3.2-fold) (32).
In contrast to immortalized BM-MSCs, primary BM-MSCs induced an increase in active GALC in the nerves of Twitcher mice. This is probably due to the fact that fivefold more primary BM-MSCs grafted the sciatic nerve when compared to immortalized MSCs. Thus, although 2.5-fold more immortalized BM-MSCs were transplanted, the fact that more primary MSCs (with a twofold increased GALC activity) were capable of grafting probably underlies the increased GALC activity in animals that received primary cells. Despite the increment in GALC activity to about 40% of the normal values, psychosine accumulation did not decrease. It is usually accepted that values as low as 1–6% of the active enzyme, at least in the CNS, are sufficient to rescue the phenotype in animal models of lysosomal storage disorders (7,17,18). However, in this particular setup, it seems that, at least in vitro, BM-MSCs are unable to perform cross-correction given the low levels of active enzyme detected in the extracellular medium. It has also been reported that in the case of GALC, the cross-correction of various cell populations of the CNS is differently achieved and that neuronal cells are favored when compared to oligodendrocytes (28). Hence, as glia preferentially accumulate psychosine over neurons (2), and as Schwann cells are the major components of the sciatic nerve, the secreted GALC would be mainly taken up by axons and transported throughout these cells (34) rather than being internalized by Schwann cells. In this case, psychosine accumulation in Schwann cells would not be reverted, and psychosine levels would remain high in the tissue. It is also possible that, as in our experimental setup, Twitcher mice were sacrificed an average 14 ± 2 days after transplantation; longer time points are needed to observe a reduction in psychosine levels as supported by in vivo (12) and in vitro assays (51), where the enzymatic activity of GALC is reconstituted much earlier than psychosine is reverted.
In addition to neuroprotection, MSCs are well known to have a strong immunomodulatory effect. In KD, macrophage invasion occurs along with demyelination, although the role of inflammation in the context of KD is still not fully understood. In a recent study in which neonatal Twitcher mice (PND 3–4) were injected intra-cerebroventricularly with BM-MSCs, the authors saw a marked decrease in macrophage infiltration and microglia activation as well as a downregulation in several proin-flammatory cytokines (36). In contrast, in the present study, we found an increased activation of macrophages in the PNS of BM-MSC-transplanted Twitcher mice. This increased activation of macrophages may be beneficial as impaired remyelination occurs in macrophage-deficient Twitcher mice (25).
In conclusion, this study supports a paracrine effect of primary BM-MSCs affecting neurons, Schwann cells, and macrophages that will culminate in the promotion of axonal regeneration and/or survival. In vitro, secretion of neurotrophins, namely, BDNF, might be at least in part responsible for the BM-MSC paracrine action. However, in our previous study with immortalized BM-MSCs, we found that, in vivo, the cell line was capable of surmounting the effect of K252a, an inhibitor of tyrosine kinase (Trk) receptors, and was still able to promote axonal regeneration (19,32). Future studies should further dissect in vivo the mechanism of action of primary BM-MSCs when systemically delivered to the KD mouse model and evaluate the success of a combinatory HSC-MSC therapy to further ensure translational success in humans. To this end, it would be interesting to perform novel studies in Twitcher mice combining the use of MSCs with HSCs or even with neural stem cells.
Footnotes
Acknowledgments
The authors would like to thank Dr. Ana Filipa Gonçalves (Institute of Physiology, University of Zurich, Switzerland) for setting up the initial conditions for BM-MSC isolation and genotyping of Twitcher mice, Rodolfo Águas (IBMC) for help in the characterization of BM-MSCs, Dr. Cristropher Eckman (Mayo Clinic College of Medicine, FL, USA) for kindly providing the anti-GALC antibody, Dr. Frederico Silva (IBMC) for immunopurifying the anti-EGFP antibody, Dr. Clara Sá Miranda and Daniel Caiola (IBMC) for help in GALC activity measurements, Dr. Bárbara Macedo (IBMC) for psychosine measurements, and Dr Pedro Brites (IBMC) for help in PPD staining, g-ratio measurement, and revision of the manuscript. This work was supported by the European Leukodystrophy Association (ELA Foundation No. 2010-042C5A) to M.M.S. C.A.T. was supported by Programa Ciência, funded by POPH-QREN and MCTES, and C.O.M. and M.A.L. by Fundação para a Ciência e Tecnologia (SFRH/BD/29768/2006 and BPD/34811/2007). The authors declare no conflict of interest.
