Abstract
The transplantation of porcine islets of Langerhans to treat type 1 diabetes may provide a solution to the demand for insulin-producing cells. Porcine islets encapsulated in agarose–agarose macrobeads have been shown to function in nonimmunosuppressed xenogeneic models of both streptozotocin-induced and autoimmune type 1 diabetes. One advantage of agarose encapsulation is the ability to culture macrobeads for extended periods, permitting microbiological and functional assessment. Herein we describe optimization of the agarose matrix that results in improved islet function. Porcine islets (500 IEQs) from retired breeding sows were encapsulated in 1.5% SeaKem Gold (SG), 0.8% SG, or 0.8% Litex (Li) agarose, followed by an outer capsule of 5% SG agarose. Insulin production by the encapsulated islets exhibited an agarose-specific effect with 20% (0.8% SG) to 50% (0.8% Li) higher initial insulin production relative to 1.5% SG macrobeads. Insulin production was further increased by 40–50% from week 2 to week 12 in both agarose types at the 0.8% concentration, whereas islets encapsulated in 1.5% SG agarose increased insulin production by approximately 20%. Correspondingly, fewer macrobeads were required to restore normoglycemia in streptozotocin-induced diabetic female CD(SD) rats that received 0.8% Li (15 macrobeads) or 0.8% SG (17 macrobeads) as compared to 1.5% SG (19 macrobeads). Islet cell proliferation was also observed during the first 2 months postencapsulation, peaking at 4 weeks, where approximately 50% of islets contained proliferative cells, including β-cells, regardless of agarose type. These results illustrate the importance of optimizing the microenvironment of encapsulated islets to improve islet performance and advance the potential of islet xenotransplantation for the treatment of type 1 diabetes.
Introduction
Type 1 diabetes mellitus affects approximately 3 million people in the US and is increasing at a rate of 3% per year in children under the age of 14 around the world (18,22). Patients with type 1 diabetes experience a specific destruction of the insulin-producing β-cells within the islets of Langerhans that are scattered throughout the pancreas. The exogenous administration of insulin by way of daily, often multiple, subcutaneous insulin injections remains the current standard of care for most patients. Whole pancreas transplantation can also provide for the immediate physiological delivery of insulin as well as other factors regulating body metabolism. The downside of this more recent approach, however, is the necessity of lifelong immunosuppressive therapy that is associated with an increased potential for severe long-term complications including kidney dysfunction, anemia, and the development of cancer (26). Moreover, the number of suitable human cadaveric pancreases for transplantation is insufficient to treat the ever-increasing number of type 1 diabetic patients, with only 1,000 donor pancreases considered acceptable and consequently used for transplantation in 2011 (51).
The transplantation of only the islets of Langerhans, following isolation from the pancreas, is an attractive option to restore lost insulin-producing cells in diabetic patients. Although long-term insulin independence remains elusive following islet transplantation, results over the last 12 years have shown recognizable clinical benefits following islet transplantation, including improved health-related quality of life, short-term insulin independence, improved glucose regulation, and the virtual elimination of hypoglycemic events (5,47). Recurrent disease is thought to account for the eventual loss of transplanted islet function and will need to be solved if this procedure is to become a viable option (17,34,40). As with whole pancreas transplantation, the lack of a sufficiently large human islet donor population and the requirement for lifelong immunosuppression also limit the usefulness of this approach.
The use of animal-derived islets of Langerhans could resolve the islet supply issue, whereas immunoisolation of the islets, via encapsulation, may ameliorate the need for immunosuppression. Pigs (Sus scrofa domesticus) are the most suitable species for pancreas donation based on the facts that human and porcine insulin differ by only a single amino acid, the supply of porcine pancreases is virtually unlimited, the lack of ethical issues with the use of this species, and the extensive history of porcine insulin use in the clinic (2). Currently, the favored approach to islet encapsulation involves the microencapsulation of single islets in alginate hydrogels cross-linked by a cation such as calcium or barium (9), which can be layered with poly-l-lysine (28,41) or poly-l-ornithine (7,11) to provide strength and selective permeability. An increasing number of encapsulation techniques, however, are being developed such as conformal coating with hydroxyethyl methacrylate-methyl methacrylate polymer, polyethylene glycol, fabricated devices [reviewed in (25)], and even coating islets with living cells (46). We have taken a somewhat different approach of encapsulating several hundred porcine islets together in agarose–agarose macrobeads and have reported the ability of these islet macrobeads to regulate blood glucose in several preclinical animal models (13,19–21). Importantly, our preclinical studies in rats have not found any evidence of porcine islet tissue sensitivity or viral transmission, thus supporting the macrobeads as a safe approach to xenogeneic islet transplantation (12,13).
An exceptional feature of the islet macrobead is the capacity of the encapsulated islets to survive in culture for more than 12 months (13). During the first several weeks of macrobead culture, a reliable increase in insulin production is observed, which eventually plateaus and stabilizes over the first 2–3 months. We set out to understand the natural history of this increased insulin production and to thereby optimize encapsulation conditions to maximize the functional capacity of the islet macrobeads. In particular, our experience with the agarose encapsulation of various tumor cell lines suggested that different cell types may require differing agarose concentrations for maximum viability and the formation of tumor colonies (44). We hypothesized that varying either the agarose type and/or concentration would affect islet viability and insulin production. In this article, we report an optimized agarose encapsulation procedure that minimizes β-cell death during the peri-isolation period while allowing β-cell proliferation and an increase in initial and long-term insulin production. This, in turn, makes the macrobeads more efficient, resulting in the requirement for fewer macrobeads, and thus islets, for the restoration of normoglycemia.
Materials and Methods
Porcine Islet Isolation and Encapsulation
Donor islets were prepared from Newsham sows that were over 2 years of age and had a history of multiple parities. The animals were from various sources taken from the production line at Bob Evans Farms, Inc. in Xenia, OH, USA. Islet counts were expressed as islet equivalents (IEQs) based on a standard islet size of 150 μm, and 500 IEQs were encapsulated in agarose–agarose macrobeads as previously described, with the following modifications (12). Briefly, retrieved pancreases were perfused with a collagenase/neutral protease [Collagenase P used at 1.0 g/L or Liberase mammalian tissue free (MTF)/thermolysin used at 7.5 U/g pancreas; Roche, Indianapolis, IN, USA] and 0.01 g/L DNase I (Sigma-Aldrich, St. Louis, MO, USA) or 2.5 mg/pancreas Pulmozyme (Genentech, South San Francisco, CA, USA) solution prepared in Hanks balanced salt solution (HBSS) or cold storage purification stock solution (Mediatech, Inc., Manassas, VA, USA). Following quantification, islets were separated into 2,000-IEQ aliquots before being resuspended in 0.5 ml of either 1.5% SeaKem Gold (SG), 0.8% SG, or 0.8% Litex (Li) agarose (Lonza Rockland, Inc., Rockland, MD, USA) prepared in minimal essential medium plus 25 mM 4-(2-hydroxyethyl)piperazine-1-ethanesulfonic acid (HEPES) buffer (Sigma-Aldrich). The SG agarose has previously been found to support islet encapsulation at the 1.5% concentration (12), whereas the Litex agarose has proven suitable at a concentration of 0.8% for the encapsulation and transplantation of tumor-inhibitory cells in a clinical trial of cancer macrobead therapy (44,45). The islet–agarose suspension was expelled beneath the surface of sterile mineral oil (Sigma-Aldrich) to produce four 0.125-ml spherical beads of approximately 5–6 mm in diameter, each containing 500 IEQs. The first-coat macrobeads were then cultured at 37°C in a humidified atmosphere of 5% CO2. After 5–7 days, a second coat of 5% SG agarose was applied, giving each macrobead a final diameter of 8–9 mm. The second coat of agarose provides strength to the macrobead, covers any islet tissue that may not be fully encapsulated, and reduces the average pore size of the agarose to afford at least some immunoprotection following implantation. We have also noted reduced in vitro islet viability and insulin production in macrobeads lacking a second coat (unpublished observations).
Macrobeads were then cultured at 37°C in a humidified atmosphere of 5% CO2 until collected for histology or implantation. Culture medium [Roswell Park Memorial Institute (RPMI; Invitrogen) containing 11 mM glucose and supplemented with 2.5% heat-inactivated porcine serum (Biologos, Montgomery, IL, USA) and 1% antibiotic/antimycotic (Life Technologies Corporation, Grand Island, NY, USA)] was changed weekly and 24-h postchange media samples were taken weekly for porcine insulin ELISA assays (Mercodia, Inc., Uppsala, Sweden). This sampling procedure was followed for macrobeads assessed both prior to implantation and after retrieval at necropsy.
Quantification of Islet Cell Death by Terminal Deoxyuridine Nick-End Labeling (TUNEL) Assay
Three independent islet isolations were conducted to generate macrobeads for this study. Two porcine islet macrobeads from each agarose group, maintained in culture as described above, were collected at 24 h (first coat only) and 1, 2, 4, 6, and 8 weeks postisolation and fixed in 10% neutral buffered formalin (NBF; VWR, West Chester, PA, USA). Tissue sections were stained following the manufacturer's instructions using the ApopTag Plus Peroxidase In Situ Apoptosis kit (Millipore, Billerica, MA, USA) and counterstained with hematoxylin (Vector Laboratories Inc, Burlington, CA, USA) to visualize cells with damaged DNA. For quantification, two islets from each of two macrobead halves (n = 12 total islets per agarose per time point) were randomly selected and photographed, and each nucleus within the islets was scored as positive or negative for TUNEL staining by two independent, blinded scorers. Results were then compared by a third scorer, and only nuclei that were scored the same by both initial scorers were included in the final totals. A range of 348–1,164 nuclei (average of 653 nuclei) were scored for each first-coat agarose group per time point.
Quantification of Islet Cell Proliferation by Bromodeoxyuridine (BrdU) Incorporation
These experiments were conducted using the same three islet isolations as for the TUNEL staining described above. Two cultured macrobeads from each agarose group were collected and fixed in 10% NBF at 24 h (first coat only), 1, 2, 4, 6, and 8 weeks postisolation after incubation in BrdU (GE Healthcare, Waukesha, WI, USA) for 24 h (24-h time point) or 6–7 days (1- to 8-week time points). Tissue sections were stained for BrdU incorporation with rat anti-BrdU antibody (1:200, 5 μg/ml; Abcam, Cambridge, MA, USA), Vectastain Elite anti-rat IgG ABC Kit (Vector Laboratories Inc, Burlington, CA, USA), Vector VIP Peroxidase Substrate Kit (Vector Laboratories Inc.), and counterstained with hematoxylin. For quantification, the total number of islets in each macrobead cross section was counted and each islet was scored as positive or negative for BrdU staining. An islet was counted in the total if it contained ≥5 distinct, contiguous nuclei and scored as positive for proliferation if it contained at least one BrdU-positive nucleus. Each cross section was scored by two independent scorers and their scores combined for statistical analysis. A range of 135-215 (average 172) islets were scored for each first-coat agarose group per time point.
Identification of Proliferative and Apoptotic Islet β-Cells by Immunofluorescent Staining for Ki67, Cleaved Caspase-3, and Insulin
These experiments were conducted using the same three islet isolations as for the BrdU and TUNEL staining described above. For Ki67/insulin double staining, porcine islet macrobeads from the 4-week postisolation time point were used based on the peak in proliferation observed at that time point in our BrdU incorporation experiments. Tissue sections were stained with rabbit anti-Ki67 (1:200, 3 μg/ml; Abcam) and guinea pig anti-insulin (1:200, 77.5 μg/ml; Dako, Carpinteria, CA, USA) antibodies followed by fluorescent secondary antibodies Alexa Fluor 488 Goat Anti-Guinea Pig IgG and Alexa Fluor 555 Goat Anti-Rabbit IgG (1:1000, 2 μg/ml; Life Technologies). Prolong Gold Antifade Reagent with 4′,6-diamidino-2-phenylindole (DAPI) (Life Technologies) was used to visualize nuclei.
For cleaved caspase-3/insulin double staining, porcine islet macrobeads from the 1-week postisolation time point were used based on the elevated level of TUNEL staining previously observed at that time point. Tissue sections were stained as for Ki67/insulin, substituting rabbit anti-cleaved caspase-3 (1:100, 0.63 μg/ml; Cell Signaling Technology, Danvers, MA, USA) for the Ki67 antibody.
For quantification, Ki67-positive or cleaved caspase-3-positive cells (45-57 and 75-141 cells per agarose type, respectively) were identified and subsequently scored for the presence or absence of insulin staining. To determine the overall prevalence of insulin-positive cells, all of the cells in islets found to contain a Ki67-positive or cleaved caspase-3-positive cell (858-1,337 and 441-942 cells per agarose type, respectively) were counted and scored for the presence/absence of insulin reactivity.
Animals and Diabetes Induction
Adult male and female Sprague-Dawley [Crl:CD(SD)] rats were purchased from Charles River Laboratories (Wilmington, MA, USA) and received at 6 weeks of age. After a 1- to 2-week acclimation period, animals designated as insulin-only controls or as macrobead implant recipients were administered 65 mg/kg streptozotocin (STZ; VWR) via the tail vein under isoflurane (AErrane®, Baxter Healthcare Corporation, Deerfield, IL, USA) anesthesia. Diabetes was clinically confirmed with nonfasting blood glucose values over 400 mg/dl for 3 consecutive days (Accu-Chek ComfortCurve BG monitor and Chemstrips; Roche Diagnostics). Animals that did not exhibit clinical diabetes within 2 weeks after initial treatment were administered a second dose of STZ, after which all animals became diabetic. Diabetic rats were treated daily with subcutaneous injection of insulin (PZI insulin, Blue Ridge Pharmaceuticals, Inc., Greensboro, NC, USA). For macrobead-implanted rats, administration of exogenous insulin was discontinued at the time of implant for the duration of the study. This study was reviewed and approved by The Rogosin Institute's Animal Care and Use Committee (IACUC). All procedures and use of animals were in compliance with the Guide for the Care and Use of Laboratory Animals (36). Rats were received and maintained at The Rogosin Institute–Xenia Division animal facility that holds full accreditation status awarded by the Association for Assessment and Accreditation of Laboratory Animal Care, International (AAALAC, Int.).
Porcine Islet Macrobead Implantation
Upon receipt, rats were randomly assigned to experimental groups as follows (n = 8 males and n = 8 females per group): control (no treatment), STZ plus insulin, STZ plus 1.5% SG macrobeads, STZ plus 0.8% SG macrobeads, or STZ plus 0.8% Li macrobeads. Both male and female recipients were used to assess any gender-specific effects of porcine islet macrobead therapy. Diabetic animals at 12–13 weeks of age were implanted with macrobeads as previously described with the day of implant designated as study day 0 for all subjects (13). Briefly, anesthetic induction was achieved and maintained with isoflurane, and macrobeads were gently placed into the peritoneal cavity via a midline incision. For animals receiving a 1× dose, the number of macrobeads implanted was individually determined per animal based on the highest exogenous insulin dose received over 3 days prior to implant, divided by the average insulin production per macrobead based on the most recent 4-week average in vitro insulin production. For males receiving a 1.7× dose, the number of macrobeads was determined in the same way and that total multiplied by 1.7.
Clinical Observations
Clinical observations from individual animals were recorded daily. Observations included alertness, body coat appearance and body posture, presence of porphyrin staining, suspected signs of pain, distress, or injury, and signs of dehydration. Body weight and nonfasting tail vein blood glucose were measured weekly (control animals), three times per week (macrobead-implanted animals, after maintaining normoglycemia for 5 consecutive days), or daily (insulin-treated animals and hyperglycemic macrobead-implanted animals).
Necropsy
Complete necropsies were performed on all animals at study days 89–92. The pancreas from each animal was collected and fixed in 10% NBF for histopathology and immunohistochemistry. Tissue sections from retrieved pancreas were stained for the presence of insulin (guinea pig anti-insulin; 1:400, 38.75 or 65.3 μg/ml; Dako; Affinipure donkey anti-guinea pig IgG AP conjugate, 1:500, 1.2 μg/ml; Jackson ImmunoResearch, West Grove, PA, USA; Vector Red Alkaline Phosphatase Substrate Kit, Vector Laboratories, Inc.; hematoxylin) and glucagon (monoclonal mouse anti-glucagon, 1:400, 25.75 μg/ml; Sigma-Aldrich; Affinipure donkey anti-mouse IgG AP conjugate, 1:100, 6 μg/ml; Jackson ImmunoResearch; Vector Red Alkaline Phosphatase Substrate Kit; hematoxylin) to confirm β-cell death following STZ treatment. Implanted macrobeads were collected in a sterile fashion and randomly selected and fixed in 10% NBF for histopathology and immunohistochemistry or returned to in vitro culture for up to 8 weeks to assess insulin production by ELISA.
Statistics
Statistical analyses were performed by the Center for Biostatistical Services in the Department of Environmental Health at the University of Cincinnati College of Medicine. An area under the curve model was used for comparison of groups in Figure 1, with statistical analysis performed by one-way ANOVA and Tukey's HSD test to detect significant pairwise differences. One-way ANOVA and Tukey's HSD test were also used for comparison of groups in Figures 2 and 3. The data presented in Figure 4 were analyzed using Fisher's exact test followed by Bonferroni correction for multiple comparisons. The data presented in Table 1 were analyzed by one-way ANOVA and Tukey's HSD test. Area under the curve was used for comparison of groups in Figure 5, with statistical analysis by one-way ANOVA and Tukey's HSD test. There is no statistical analysis associated with Figure 6. Area under the curve was used for comparison of groups in Figure 7, with statistical analysis by one-way ANOVA and Tukey's HSD test. All data, with the exception of Figure 4, are presented as mean ± SD.
Porcine Islet Macrobead and Daily Insulin Requirements in Implanted Animals


Long-term in vitro insulin production by adult porcine islet macrobeads (MB). Each data point represents the mean ± SD milliunits of insulin produced per macrobead per 24 h (mU/MB/24hrs) from 10 independent islet isolations. For statistical analysis, total area under the curve from weeks 2 through 12 was calculated for each agarose type [1.5% SeaKem Gold agarose (SG) vs. 0.8% SG, p<0.05; 1.5% SG vs. 0.8% Li, p<0.0001; 0.8% Litex agarose (Li) vs. 0.8% SG, p<0.0001].
Results
Improved Insulin Secretion with Lower Agarose Concentration and Use of Litex Agarose
We evaluated the effect of changes in first-coat agarose concentration and type on in vitro insulin secretion. During a 12-week period following isolation, islets encapsulated in a first coat of 0.8% SG or 0.8% Li agarose (n = 10 independent isolations per agarose group) produced insulin at a consistently and significantly higher rate than islets encapsulated in 1.5% SG (p < 0.05 and p < 0.0001, respectively) (Fig. 1). In addition, insulin production was significantly increased in 0.8% Li-encapsulated islets in comparison to 0.8% SG-encapsulated islets (p < 0.0001).
Equivalent Islet Cell Damage and Proliferation Regardless of Agarose Concentration and Type
To investigate the source of the differential insulin production, we measured the prevalence of cellular DNA damage and proliferation within the encapsulated islets. DNA damage was visualized by immunohistochemistry using the TUNEL assay. Figure 2A shows representative images taken from islet macrobeads collected at various times postisolation. At 24 h postisolation, TUNEL staining is present in the nuclei (arrows) of many islet cells, but this DNA damage is almost entirely resolved by 2 weeks with minimal histological evidence of cell damage or death. Quantification of TUNEL-positive cells (Fig. 2B) confirmed that DNA damage was extensive in islets during the first 24 h postisolation and remained elevated for the first 2 weeks after encapsulation. Beyond 4 weeks, cell death was present at a greatly reduced level and was negligible by 8 weeks of age, with only 3.8% (1.5% SG), 0.6% (0.8% SG), and 2.1% (0.8% Li) of nuclei staining positive. There were no significant differences in numbers of nuclei displaying DNA damage found between groups at any of the assayed time points, suggesting that differences in the level of initial cellular DNA damage do not contribute meaningfully to the observed disparity in macrobead insulin production between groups.
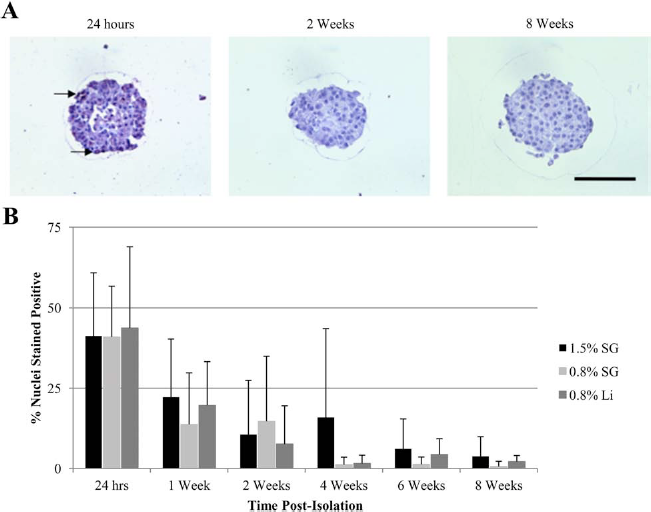
DNA damage in agarose-encapsulated adult porcine islets assessed by TUNEL staining. (A) Representative islet images from 24 h, 2 weeks, and 8 weeks postisolation. All images are of islets encapsulated in 0.8% Li agarose taken from the same isolation. Arrows indicate terminal deoxyuridine nick-end labeling (TUNEL)-positive nuclei. Scale bar: 100 μm. (B) Data represent the mean percentage ± SD of TUNEL-positive nuclei per islet in 12 randomly selected islets per agarose type, per time point.
Cell proliferation in encapsulated islets, as measured by BrdU incorporation, provided a complementary approach for evaluating differences in islet function between groups. BrdU-positive nuclei were present in islets from all three agarose configurations (Fig. 3A). Regardless of first-coat agarose type, the number of proliferating islet cells steadily increased over the first month postisolation (Fig. 3B), peaking at 4 weeks with approximately 50% of encapsulated islets exhibiting at least one BrdU-positive cell. Similar to the above results with TUNEL staining, differences in the number of proliferating islet cells were minimal between groups with no significant differences found between groups at any time point (Fig. 3B).

Proliferation in adult porcine islet macrobeads (MB) as assessed by bromodeoxyuridine (BrdU) incorporation. (A) Representative images from islets encapsulated in 1.5% SG (6 weeks postisolation), 0.8% SG (1 week), or 0.8% Li agarose (1 week). Arrows indicate BrdU-positive nuclei. Scale bar: 100 μm. (B) Data represent the mean percentage ± SD of BrdU-positive islets per section.
Agarose Encapsulation of Islets Exerts Differential Effects on β-Cell Apoptosis Dependent on Agarose Concentration and Type and Supports β-Cell Proliferation
Although no significant overall differences were found in the degree of TUNEL staining or BrdU incorporation between agarose types, the possibility remained that the particular type of islet cell being affected differed between groups. Using immunofluorescent staining for the apoptotic marker cleaved caspase-3 (CC3) and the cell proliferation marker Ki67 in combination with glucagon (α-cells) and insulin (β-cells) staining, we were able to determine whether these specific cell types were differentially represented among apoptosing and/or proliferating cells (Fig. 4).

Confirmation of apoptosis and proliferation in encapsulated porcine β-cells. (A) Immunofluorescent staining for cleaved caspase-3 (red) and insulin (green) in a 1-week-old porcine islet macrobead encapsulated in 0.8% SG agarose. Arrow indicates an apoptotic β-cell identified by dual cytoplasmic staining for cleaved caspase-3 and insulin. Scale bar: 25 μm. (B) Data represent the percentage of insulin-positive β-cells among the overall islet population (All Islet Cells) versus the percentage of β-cells among the subpopulation of apoptotic (Cleaved Caspase-3+) islet cells at 1 week postisolation. (C) Immunofluorescent staining for Ki67 (red) and insulin (green) in a 4-week-old porcine islet macrobead encapsulated in 1.5% SG agarose. Arrows indicate proliferative β-cells identified by dual staining for nuclear Ki67 and cytoplasmic insulin. (D) Data represent the percentage of insulin-positive β-cells among the overall islet population (All Islet Cells) versus the percentage of β-cells among the subpopulation of proliferative (Ki67+) islet cells at 4 weeks postisolation. p values for statistically significant pairwise comparisons are presented in the chart.
We found that glucagon-positive cells make up only between 2% and 6% of all cells at 1 week postisolation, with that number rising slightly to between 4% and 7% at week 4 (minimum 491 cells scored per agarose per time point). Of the CC3-positive cells present at week 1, none were found to be positive for glucagon in either the 1.5% SG (0/160 cells scored) or 0.8% SG (0/91) macrobeads, whereas 2.2% (2/93) were found to be double-positive for CC3 and glucagon in the 0.8% Li macrobeads. At week 4, equally low numbers of glucagon-positive cells were found to be proliferating, with α-cells constituting only 2.3% (1/43), 1.7% (1/58), and 3.6% (2/55) of Ki67-positive cells in the 1.5% SG, 0.8% SG, and 0.8% Li macrobeads, respectively. Overall, α-cells made up a small proportion of the islet mass in our macrobeads and were represented among the apoptotic and proliferative cell populations in proportion to their overall numbers.
Conversely, insulin staining was prevalent at both the week 1 (28–39% of all cells) and week 4 (54–68%) time points in all three agarose groups (Fig. 4) (minimum 441 cells scored per agarose per time point). At week 1, where cell damage was still elevated based on our TUNEL assays but the islets were further removed from the stress of isolation and encapsulation, we found that the percentage of β-cells among the apoptotic population of CC3-positive cells was highly dependent on first-coat agarose type and concentration (Fig. 4A and B). The percentage of apoptotic β-cells was proportional to their overall numbers in 0.8% Li macrobeads (45.2% vs. 37.9%), whereas β-cells were highly overrepresented in the apoptotic cell population in 1.5% SG (80.9% vs. 39.2%, p < 0.0001) and 0.8% SG (62.7% vs. 28.1%, p < 0.0001) macrobeads relative to their overall numbers (Fig. 4B).
At 4 weeks postisolation, where islet cell proliferation peaked in our BrdU incorporation assays, we specifically evaluated the proliferation of β-cells to ascertain any differences between agarose types (Fig. 4C and D). The overall proportion of β-cells was significantly higher in 1.5% SG (66.1%) and 0.8% SG (68.1%) when compared to 0.8% Li (54.0%) macrobeads (p < 0.0001 for both comparisons). Whereas β-cell representation among the Ki67-positive population of proliferating cells was proportional to overall β-cell numbers in 1.5% SG macrobeads (64.6% vs. 66.1%), β-cells were less likely to be proliferating than would be expected based on their total proportion in 0.8% SG (49.1% vs. 68.1%, p < 0.05) and 0.8% Li (37.8% vs. 54.0%, not significant) macrobeads.
Improved Macrobead Function Decreases the Number of Macrobeads Required to Reverse Hyperglycemia
To determine if increased macrobead insulin production observed in vitro translated to improved blood glucose control in vivo, we implanted islet macrobeads from each of the three agarose groups into STZ-induced diabetic rats. Animals were implanted with a number of macrobeads producing insulin (in vitro) that was equivalent to individual preimplant exogenous insulin requirements (1× dose groups), or 1.7 times their preimplant requirements (1.7× male group). Average preimplant exogenous insulin requirements for each experimental group are presented in Table 1 under the heading “Insulin (U/day).” No significant differences in preimplant insulin requirements were noted between animals receiving different agarose types within groups (1× females, 1× males, or 1.7× males). As shown in Table 1 (third column, MB/U Insulin) the number of islet macrobeads required to deliver 1 U of insulin was greatest for macrobeads made with 1.5% SG agarose followed by macrobeads made with 0.8% SG agarose. Macrobeads made with 0.8% Li agarose produced the most insulin per macrobead. All pairwise comparisons within gender were significant after correcting for multiple comparisons, with p < 0.0001 regardless of macrobead dose (1× or 1.7×).
Following implantation, animals were maintained for 90 days and blood glucose and body weight were regularly monitored. Among female test subjects in the 1× dose group, macrobeads from all three agarose groups provided an immediate and sustained normalization of blood glucose levels when compared to exogenous insulin-treated controls (Fig. 5A). Measuring area under the curve, there were no significant differences in blood glucose levels found between the three macrobead implant groups. Significant differences were only found between the insulin-treated and macrobead-implanted groups (1.5% SG vs. insulin, p < 0.0005; 0.8% SG vs. insulin, p < 0.0001; 0.8% Li vs. insulin, p < 0.001). Male rats implanted with a 1× dose of islet macrobeads showed an immediate, albeit temporary, improvement in blood glucose regulation, which returned to preimplant levels by approximately 30 days postimplant (Fig. 5B).

Daily blood glucose measurements in diabetic rats implanted with porcine islet macrobeads. Each data point represents the average of 8 animals treated with insulin or implanted with 1.5% SG, 0.8% SG, or 0.8% Li agarose MB. (A) Female rats implanted with a 1× MB dose. (B) Male rats implanted with a 1× MB dose. (C) Male rats implanted with a 1.7× MB dose. For statistical analysis, total area under the curve from day 1 through day 90/92 was calculated for each agarose type and the insulin treatment control and compared within sex and dose groups (1× Females: 1.5% SG vs. insulin treatment, p<0.0005; 0.8% SG vs. insulin treatment, p<0.0001; 0.8% Li vs. insulin treatment, p<0.001; all other comparisons were not significant).
To address the possibility that male rats received an insufficient dose of islet macrobeads, a 1.7× macrobead dose was added to the study. The 1.7× dose groups responded more gradually to the macrobead implant compared to the 1× dose male rats, with average blood glucose readings declining over the first 2 weeks postimplant (Fig. 5C). Blood glucose levels began to rise approximately 3 weeks postimplant, but did not reach insulin-treated control levels until 60–70 days postimplant, and were still below preimplant levels when the animals were necropsied on day 92. Nonetheless, neither the 1× nor 1.7× dose implanted males exhibited significantly better blood glucose regulation over the entirety of the postimplant observation period than the insulin-treated control group.
Pancreas Histology and Explanted Macrobead Function Confirm Graft Efficacy in Macrobead-Implanted Animals
To rule out the possibility of residual pancreatic islet function contributing to implant success, the pancreases of all study subjects were collected at necropsy. Histological examination of representative pancreas sections from control and experimental animals demonstrated that STZ treatment was extremely effective, with no differences in islet structure or cellular composition observed between insulin-treated and macrobead-implanted animals (Fig. 6). Compared to those of untreated control animals, the islets of all STZ-treated animals were highly disorganized, with dramatically reduced insulin staining. Insulin staining was observed in only a few cells per islet, with a greater proportion of glucagon-positive cells scattered throughout the islets. When combined with the fact that all of the male recipients of islet macrobeads and one of the female recipients developed recurrent hyperglycemia, it is unlikely that residual pancreatic islet function contributed to glycemic control in STZ-treated animals.

Insulin and glucagon staining of representative rat pancreas retrieved at necropsy and corresponding nonfasting blood glucose values. Sections were stained with insulin and glucagon antibodies and counterstained with hematoxylin. Blood glucose levels were measured for up to 90 days postimplant. Within each row, pancreas sections and blood glucose values are from the same animal. Scale bars: 100 μm.
Porcine islet macrobeads were also collected from all implanted animals at necropsy to assess their structural integrity and functional capacity. Gross examination of recovered macrobeads indicated that cracking or breaking was extremely rare, with only two macrobeads exhibiting signs of damage (one each from the 0.8% SG and 0.8% Li groups) out of 1,540 macrobeads recovered. Five macrobeads from each animal recovered at necropsy were returned to in vitro culture and followed for 8 weeks to measure their capacity to produce insulin after 90 days in vivo. Using an area under the curve model, no significant differences were found in postimplant insulin production between agarose groups by gender. However, macrobeads retrieved from 1× dosed female rats produced as much as 50% of preimplant insulin levels at 1 month postrecovery before dropping to approximately 20 mU/macrobead by 8 weeks (Fig. 7A). Interestingly, macrobeads recovered from 1× dosed males displayed extremely low levels (approximately 10 mU/macrobead) of postimplant insulin production (Fig. 7B), whereas macrobeads recovered from 1.7× dosed male rats produced approximately 25% (1.5% SG) and 70% (0.8% SG and 0.8% Li) of preimplant insulin levels over the 8-week observation period (Fig. 7C). Despite apparent differences in average insulin production between agarose groups retrieved from 1.7× dosed males (Fig. 7C), these results were again not statistically significant.
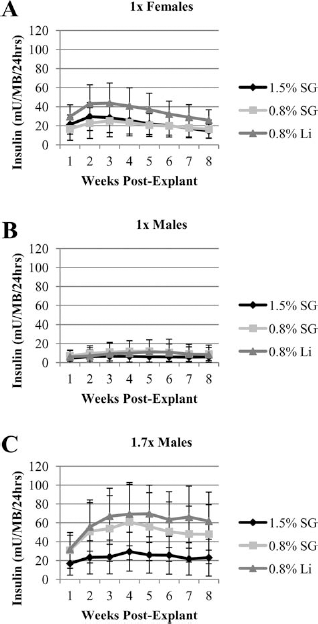
Function of porcine islet macrobeads (MB) retrieved at necropsy. (A) Female rats implanted with a 1× MB dose. (B) Male rats implanted with a 1× MB dose. (C) Male rats implanted with a 1.7× MB dose. Each data point represents mean ± SD of 40 retrieved MB per group. For statistical analysis, total area under the curve from week 1 through week 8 was calculated for each agarose type and compared within sex and dose groups. No significant differences were found.
Discussion
In these studies we have shown that the concentration and type of agarose used to encapsulate porcine islets influences both the in vitro and the in vivo functional capacity of the islets. Regardless of agarose concentration or type, an initially high level of DNA damage at 24 h postencapsulation (30–40% of encapsulated cells) gradually decreased to a low level (< 5%) by 8 weeks. An increase in cell proliferation, which peaked at 4 weeks postencapsulation (approximately 50% of islets with BrdU-positive cells) and then declined over the following 4 weeks, was observed for all agarose conditions. The level of in vitro insulin production correlated with the ability of the islet macrobeads to control blood glucose in vivo with the highest insulin-producing islet macrobeads able to restore normoglycemia in diabetic female rats using 19% (0.8% SG) or 42% (0.8% Li) fewer macrobeads relative to the 1.5% SG group. Moreover, implanted islet macrobeads were shown to significantly improve blood glucose control over standard insulin therapy. In fact, for female recipients, the porcine islet macrobeads were shown to restore normoglycemia long term in a fully xenogeneic and immunocompetent rat model with no immunosuppression.
Differences between the two types of agarose could explain some of the disparity in islet function. Although SG agarose is derived from Gelidium species of seaweed and Litex is processed from Gracilaria species, both share the same basic chemical composition consisting of 1,3-linked β-d-galactopyranose and 1,4-linked 3,6-anhydro-a-l-galactopyranose. Gelidium species will usually yield agarose with higher gel strength and naturally have higher melting and lower gelling temperatures as compared to Gracilaria species. The manufacturing process is designed to further refine these desired properties. For example, SG is a highly purified, high gel strength agarose designed to be used at very low concentrations and in applications where residual charges or contaminants are of concern. The Litex agarose is also highly purified and is processed to control viscosity for ease of use in manufacturing beads. These different properties of agarose will therefore affect how they function in similar applications (personal communication, Maxine Dumais, Lonza Rockland, Inc.).
Given that a decrease in the SG agarose concentration from 1.5% to 0.8% reduces agarose rigidity and resulted in improved islet performance, we consider a change in the mechanical stresses exerted on the islets to be an important factor in determining relative islet function. In fact, it is now well established that the physical forces exerted on cell cultures impact cell fate and function. For example, mesenchymal stem cells have been shown to develop into either bone, muscle, or brain precursor cells depending on the stiffness of the culture substrate (10). We have also observed the need to adjust agarose concentrations in order for different human tumor cell lines to form tumor colonies in Litex agarose–agarose macrobeads (44), thus demonstrating that the rigidity of the encapsulation substrate plays an important role in the survival and proliferation of different cell types.
The observation that agarose-encapsulated porcine islet cells undergo DNA damage and apoptosis is not surprising. The process of islet isolation necessarily disrupts islet cell-cell interactions and extracellular matrix connections along with vascular and neural disruptions, leading to islet cell damage. Additionally, isolated islets rapidly die in culture in a process termed anoikis, resulting from the lack of an extracellular matrix (49). The provision of an extracellular matrix or the encapsulation of islets immediately postisolation can greatly extend the survival of isolated islets (3,4,6,20,23,31,35,39,42,48,52,54). In the case of macrobeads, it seems likely that the first agarose coat provides the required extracellular scaffold that mimics the native pancreas and prolongs islet survival in vitro. It is likely that the second coat of agarose must also be optimized in order to obtain the optimal balance between immunoprotection and nutrient diffusion. In particular, the oxygen diffusion limitations associated with increased encapsulation thickness could impact long-term in vivo islet function in our study.
Prolonged survival time of macroencapsulated porcine islets in vitro provides an opportunity to improve, stabilize, and evaluate islet function as we have previously reported (13). In the present study, the initial islet damage, as assessed by TUNEL staining, is likely a result of the isolation procedure and rapidly subsides. The fact that 30–40% of islet cells demonstrate DNA damage at 24 h postencapsulation, yet no evidence of massive cell death is observed microscopically at week 2, suggests a capacity of the islets to repair such damage. Interestingly, whereas TUNEL staining indicated no differences in overall islet DNA damage between agarose groups, direct assessment of apoptosis specifically in insulin-producing β-cells produced a different outcome. Whereas β-cells were present in the apoptotic cell population in proportion to their overall numbers in 0.8% Li macrobeads, they were found to be highly overrepresented among the apoptotic population of cells from 1.5% SG and 0.8% SG macrobeads at 1 week postisolation. This result is of interest because it provides important insight into the mechanism of differential in vitro insulin production observed in the current study. Specifically, these data support the notion that the initial and consistently lower insulin production obtained from 1.5% SG and 0.8% SG macrobeads may be a direct result of the elevated and disproportionate loss of β-cells from these macrobeads relative to islets encapsulated in 0.8% Li agarose.
The present work also demonstrates the proliferative capacity of macroencapsulated porcine islet cells. The overall proportion of β-cells as well as the share of β-cells among the proliferative population was increased in the 1.5% SG and 0.8% SG compared to the 0.8% Li group. These differences may be indicative of increased β-cell proliferation in the 1.5% SG and 0.8% SG macrobeads in an attempt to recover the excess β-cell mass lost in the initial postisolation period relative to the lower level of β-cell apoptosis observed in 0.8% Li macrobeads.
Other groups have reported the proliferation of various insulin-producing cells including porcine (37) and human (1,27) pancreatic precursor cells, β-cell lines (15,29), and islets following encapsulation (8). These reports all involve microencapsulation of single or only a few islets per device, with the exception of Hayek's group where approximately 50 islets were encapsulated in fibrin gels (1). We believe our article is the first report of macroencapsulated adult porcine islets that retain the ability to both repair DNA damage and undergo proliferation during culture. The ultimate failure of the 1.5% SG and 0.8% SG macrobeads to approach the insulin production capacity of the 0.8% Li macrobeads may indicate that the initially high levels of apoptosis outstrip the finite proliferative capacity of encapsulated β-cells, resulting in a lower steady-state insulin output in our mature macrobeads.
Although normoglycemia was restored long term in diabetic female rats, normoglycemia was not sustained in diabetic male recipients implanted with porcine islet macrobeads. Consistent with previous studies, an increase in random nonfasting blood glucose was observed to parallel an increase in body weight for male recipients following islet macrobead implantation (13). Although male and female rats received approximately the same macrobead dose, implanted male rats gained an average of 104 g (28% increase in body weight), whereas female rats gained an average of only 43 g body weight (an 18% increase). An improvement in blood glucose was observed following implantation of a 1.7× dose of macrobeads in male recipients, suggesting that glycemic control in male recipients may require an even higher dose of islet macrobeads. Notably, we did not observe any untoward effects of implanting 70% more islet macrobeads into the abdominal cavity of diabetic rats, as compared to rats that received only a 1× macrobead dose. Indeed, the retrieved macrobeads from male rats that received a 1.7× dose of macrobeads outperformed retrieved macrobeads from male rats that received a 1× dose when comparing insulin production from macrobeads retrieved at necropsy. This suggests that the higher dose of macrobeads prolongs islet function, perhaps by providing a “critical mass” of islets (53) in order to avoid exhaustion of an insufficient number of transplanted islets (43). Importantly, the improved function of the retrieved macrobeads from the 1.7× dose animals also suggests that the graft failure in 1× dose males was not due to nutritional limitations of the peritoneal cavity.
Another possible explanation for the discrepant outcome between male and female rats implanted with porcine islet macrobeads, which are derived from retired breeder sows, may be the islet response to gender-specific hormones. As previously shown for estrogens through the nuclear receptors ERα and ERβ and the G protein-coupled membrane receptor GPR30, activation by their ligand, mainly 17b-estradiol, serves to protect islets from apoptosis, enhance insulin production and secretion, and protect against the development of diabetes (50). Moreover, exposure to high levels of androgens has been indirectly linked to insulin resistance, β-cell failure, and development of diabetes in women with polycystic ovarian syndrome (14,16) as well as in rodent models (30,32,38). On the basis of our results and these published studies, it is possible that low estrogen and/or increased androgen levels contribute to graft failure in male rats implanted with female porcine islets.
It may also be that nonspecific inflammation in the abdominal cavity following the implantation of the islet macrobeads contributed to a reduction in functional islet mass. It has previously been shown that inhibiting the inflammatory response in isolated human islets improves insulin release (33) and restores normoglycemia in new-onset type 1 diabetic nonobese diabetic (NOD) mice (24). It would be difficult to recognize inflammation in retrieved islet macrobeads given the lack of both vascularization and immune cell infiltration. As previously observed in other porcine islet macrobead in vivo studies, we did not observe signs of significant inflammation in the peritoneal cavity or on the macrobead surface at necropsy (13). Still, given the reduction in insulin production postretrieval, we consider it likely that some degree of inflammation is present in the immediate postimplant period and that the addition of anti-inflammatory treatment may be helpful.
The data reported in this article thus emphasize the importance of optimizing the extracellular environment of encapsulated islets, which is critical for the clinical success of this potential therapy. A higher degree of insulin production per macrobead directly translates into a reduced number of macrobeads required to meet preimplant exogenous insulin requirements. For example, an exogenous insulin requirement of 40 U per day could be provided with an average porcine islet isolation yield of 200,000 islet equivalents and an average insulin production of 100 mU/macrobead, enabling a single donor porcine pancreas to meet the insulin requirements of most patients. This may be significant should multiple implantations be required over the life of a patient and avoids the need to retrieve any previously implanted macrobeads in order to make room for subsequent implants. In our experience with similarly sized macrobeads implanted into the peritoneal cavity to treat human cancer patients, we have found the abdominal cavity able to accommodate at least 2,000 macrobeads with no patient-reported adverse effects (ClinicalTrials.gov Identifier NCT00283075 and NCT01053013). Full capacity of the human intraperitoneal space is estimated to accommodate at least 15,000 macrobeads. This study thus advances the potential of agarose–agarose porcine islet macrobeads for the treatment of type 1 diabetes mellitus.
Footnotes
Acknowledgments
The authors acknowledge the financial support of Metromedia Bio-Science LLC, the assistance of Bob Evans Farms, Inc., and the contributions of Brian Doll, Deborah Hoffer, Ashley Ewing, and colleagues at The Rogosin Institute. We also thank Dr. Changchun Xie and Wendy Weng at the University of Cincinnati Center for Biostatistical Services for their assistance with statistical analysis of the data. The authors declare no conflict of interest.
