Abstract
Aging is accompanied by various forms of immune dysfunction, leading to an increase in frequency of infections and the development of malignant tumors in mice and humans. Sirt1 has been implicated in processes as varied as metabolism, differentiation, cancer, and the stress response and aging. Senescence-accelerated mice prone 10 (SAMP10) show not only spontaneously occurring brain atrophy, with deficits in learning and memory, but also emotional disorders. We attempted in this study to clarify the deficits and found that the percentage of CD4/TNF-α T-cells in the spleen of 24-week-old (but not 6-week-old) SAMP10 to be significantly reduced. The thymus was significantly lighter, and the percentage of CD4+CD8+ cells was significantly lower in the 24-week-old SAMP10 than 6-week-old SAMP10. Microarray analyses indicated that genes related to transcription coactivator activity, growth factor activity, hormone activity, cytokine activity, receptor activity, and regulation of the immune system were downregulated in the thymus of 24-week-old SAMP10. Real-time PCR analysis showed that the expression of KGF, Aire, and Sirt1 was decreased on the thymic epithelial cells (TECs) of 24-week-old SAMP10. However, these parameters improved after the mice were treated with intrabone marrow–bone marrow transplantation. This is the first report of age-related changes in immune system dysfunction in 24-week-old SAMP10 and the first to show that dysfunction on the TECs of 24-week-old SAMP10 was modulated by allogeneic bone marrow cells.
Keywords
Introduction
Aging is accompanied by pleiotropic changes in the immune system that lead to a dysfunction in the cellular and humoral immune responses (30,49). The thymus, which is the source of mature T lymphocytes, involutes steadily with increasing age, resulting in a decreased release of new naive T-cells to the periphery (6), thereby affecting the adaptive immunity. Forkhead box N1 (Foxn1) is an epithelial cell autonomous gene, which is expressed in the epithelia of the thymus, skin, and mammary gland, and which regulates thymic organogenesis in the thymus (33,34). It is also involved in the successful generation of the T-cell immune system (3). A small population of medulla thymic epithelial cells (mTECs) express autoimmune regulator (Aire), which is crucial in the induction of T-cell tolerance toward tissue-restricted antigens (5,14). Some reports have indicated that both cortical TECs (cTECs) and mTECs are derived from common progenitors in ontogeny (9,15,39).
Sirt1 is a class III histone deacetylase within the sirtuin (Sirt) family of related proteins that is uniquely dependent on nicotinamide adenine dinucleotide (NAD+) for catalysis. Sirt1 has been implicated in processes as varied as metabolism, differentiation, cancer, stress response, and aging (11). Sirt1 negatively regulates T-cell activation and plays a major role in clonal T-cell anergy in mice. Loss of Sirt1 function results in abnormally increased T-cell activation and a breakdown of cluster of differentiation 4 positive (CD4+) T-cell tolerance (48).
The senescence-accelerated mouse prone (SAMP) strain shows relatively strain-specific age-associated pathologies. These characteristic pathological phenotypes are similar to the age-associated disorders often observed in older people. The strains exhibit an early onset of age-related decline in the peripheral immunity such as thymic involution, loss of CD4+ T-cells, impaired helper T (Th) cell function, decreased antibody-forming capacity, dysfunction of antigen-presenting cells, decreased natural killer activity, increased autoantibodies, and susceptibility to viral infection (41). Shimada et al. were the first to document a novel murine model for neurodegenerative diseases, the SAMP10 mouse, which is characterized by spontaneously occurring age-related brain atrophy with deficits in learning and memory and emotional disorders (43). However, it is unclear whether the thymus dysfunction develops before the appearance of the neurodegenerative changes in SAMP10.
In the present studies, we examined 6-week- and 24-week-old SAMP10 and found that, in the 24-week-old mice, the percentage of CD4+ T-cells in the peripheral blood had decreased significantly, as had the CD4+ tumor necrosis factor-α-positive (TNF-α+) T-cells. Moreover, the expression of Sirt1, Aire, and keratinocyte growth factor (KGF) on TECs was lower in the 24-week-old SAMP10, though these parameters improved after intrabone marrow– bone marrow transplantation (IBM–BMT), suggesting that stem cell transplantation can improve TEC dysfunction in SAMP10.
Materials and Methods
Animals
Six- and 24-week-old male SAMP10 (H-2Kd) mice and age-matched senescence-accelerated mouse-resistant (SAMR1) mice were purchased from Shimizu Laboratory Supplies (Kyoto, Japan) and maintained in animal facilities under specific pathogen-free conditions. All procedures were performed under protocols approved by the Institutional Animal Care and Use Committee at Kansai Medical University. Body weight and thymus weight were measured at 6 weeks and 24 weeks. The same experiment was repeated three times (n = 6 in each group).
Flow Cytometric Analyses
Peripheral blood was obtained from the tail vein of the recipient mice 30 days after transplantation. The blood was stained with antibodies against fluorescein isothiocyanate (FITC)-CD4 (a marker for helper T-cells), phycoerythrin (PE)-B220 (a marker for B-cells), FITC-CD11b (a marker for monocytes), and PE-Gr-1 (a marker for granulocytes) (BioLegend, San Diego, CA, USA) for 30 min on ice. After washing twice with 2% fetal calf serum/phosphate-buffered saline (FCS/PBS; Life Technologies, Paisley, UK) and using lysing buffer (BD Bioscience, Tokyo, Japan) to lyse red blood cells, the 10,000 events of mononuclear cells acquired were analyzed by FACScan (BD Bioscience). Isotype-matched immunoglobulins (BioLegend) were used as controls.
For analysis of regulatory T (Treg) cells, spleen cells were stained with PE-CD4 (BioLegend) and then stained with forkhead box P3 (FoxP3) using an eBioscience FITC-anti-mouse/anti-rat FoxP3 staining set in accordance with the manufacturer's instructions (eBioscience, San Diego, CA, USA). Spleen cells were used to analyze intracellular cytokines TNF-α, and interleukin 6 (IL-6) using an intracellular cytokine staining kit in accordance with the manufacturer's instructions (BD Biosciences). PE-CD8 (BioLegend) antibody was used to analyze thymocytes.
Thymus Microarray
Thymi of 6-week- and 24-week-old SAMP10 and SAMR1 were used to analyze gene expression by CodeLink™ bioarray analysis (Applied Microarrays, Inc., Tempe, AZ, USA). The chips were scanned using Gene-Pix4000B® (Molecular Devices, Sunnyvale, CA, USA). The microarray images were obtained using CodeLink™ Expression Analysis v5.0 (Applied Micro array, Inc.) software. Microarray data were analyzed using microarray data analysis tool Ver3.2 (Filgen®, Nagoya, Japan).
Immunochemistry
The thymi of SAMP10 and SAMR1 mice were removed at the respective time points. After the tissues had been fixed in 10% formalin (Wako Pure Chemical Industries, Osaka, Japan) for 24 h at room temperature, they were embedded in paraffin. The sections (3 μm thick) were stained with anti-cytokeratin 18 (ready-to-use antibody), cytokeratin 8 (1 μg/ml), and cytokeratin 5/6 (1 μg/ml) (Progen, Queensland, Australia) followed by incubation with horseradish peroxidase-labeled anti-rabbit or anti-mouse antibody (DAKO, Glostrup, Denmark) for 30 min, and then developed with 3,3-diaminobenzidine tetrahydrochloride (DAB; Wako). The stained sections were examined under a microscope.
IBM–BMT
Gamma irradiation was delivered by a Gammacell 40 Exactor (MDS Nordion, Kanata, ON, Canada) with two 137Cs sources. The 24-week-old SAMP10 mice received fractionated irradiation twice a day (5.0 Gy × 2, 4-h interval). One day after the irradiation, whole bone marrow cells (BMCs) from male 9-week-old B6 mice (n = 10; SHIMIZU Laboratory Supplies) were injected into the recipient mice (1 × 107/mouse) by IBM–BMT using our previously described method (28). Syngeneic transplantation from SAMP10 to SAMP10 was used in order to exclude the irradiated effect on the thymus using the same method. The SAMP10 treated with IBM–BMT were examined 16 weeks after IBM–BMT.
Isolation of TECs
TEC isolation was modified from a previously described method (17). Briefly, the thymi were dissected, minced into small pieces, and stirred in Roswell Park Memorial Institute (RPMI) 1640 medium (Nacalai Tesque, Kyoto, Japan) with 2% FCS at room temperature for 10 min to release the thymocytes. Following this, these tissue pieces were digested in 0.2 mg/ml collagenase type 3 (Worthington Biochemical Corporation, Lakewood, NJ, USA), 0.2 mg/ml dispase (Invitrogen Corporation, Grand Island, NY, USA), and 0.125 mg/ml DNase I (Roche, Mannheim, Germany) for 20 min at 37°C with gentle stirring. The digested product was stained with anti-mouse CD45 antibody (BioLegend), and CD45+ cells were depleted using Dynabeads sheep anti-rat IgG (Invitrogen Dynal AS, Oslo, Norway). The cells were collected to isolate RNA after negative selection.
Bone Marrow-Derived Mesenchymal Stromal Cell (BMMSC) Culture
The bone marrow cells were collected from the femurs and tibias of SAMP10 mice that had or had not undergone IBM–BMT. The BMMSCs were cultured using MesenCult media according to the manufacturer's instructions (Stem Cell Technology, Vancouver, BC, Canada). After passage, the cells were further cultured in fresh α modification of minimum essential medium (αMEM; Life Technologies), including 10% FCS and 1% antibiotic– antimycotic solution (Life Technologies). These cells were collected to detect surface markers, including antistem cell antigen 1 (Sca-1), CD90 and CD106 antibodies (BD Bioscience), and CD45 and CD11b (Biolegend) by FACS, and to isolate the RNA.
Real-Time PCR (RT-PCR)
The total RNA was prepared using isolation reagent RNA STAT-60 (TEL-TEST, Inc., Friendswood, TX, USA). cDNA was prepared using PrimeScript® RT reagent Kit (TaKaRa, Shiga, Japan). Reverse transcription-quantitative RT-PCR was analyzed using MightyAmp™ for real time (TaKaRa). The sequences of primers were as follows: glyceraldehyde 3-phosphate dehydrogenase (GAPDH) forward: ACCACAGTCCATGCCATCAC, reverse: TCC ACCACCCTGT TGCTGTA; KGF forward: ATCCTGCC AACTCTGCTCTACAG, reverse: CTTCCCTTTGACAG GA ATCCCCTT; IL-7 forward: ACATCATCTGAGTGC CACA, reverse: CTCTCAGTAGTCTCTTTAG; Aire forward: GGGACTGGTTTAGGTCCACA, reverse: AGGTG GGGATGGAATGCTAC; Foxn1 forward: CTGACCTG GATGCTATCAACC, reverse: CGCAGCTGATGTTGGG CATAGC; Sirt1 forward: CAGAACCACCAAAGCGG AAA, reverse: GGCACTTCATGGGGTATAGA; receptor activator of nuclear factor k-light-chain-enhancer of activated B-cells (NF-kB) ligand (RankL) forward: GCC GAGGAAGGGAGAGAACGAT, reverse: CGCTCGAA AGTACAGGAACAGA; osteoprotegerin (OPG) forward: AAAGCACCCTGTAGAAAACA, reverse: CCGTTTTA TCCTCTCTACACTC.
Statistical Analysis
Statistical significance between experimental groups was determined by the Fisher method of analysis of multiple comparisons (p < 0.05 was regarded as significant). For comparison between treatment groups, the null hypothesis was tested by either a single-factor ANOVA for multiple groups or unpaired t test for two groups. Statistical significance was regarded as significant at p < 0.05.
Results
Body Weight, Thymus Weight, and Lymphocyte Population Analysis
As shown in Figure 1A, the body weight of the 24-week-old SAMP10 mice was significantly greater than that of the 6-week-old SAMP10 (31.5 ± 0.25 vs. 24.5 ± 0.22 g, p < 0.001). Similarly, the body weight of 24-week-old SAMR1 mice was significantly greater than that of the 6-week-old SAMR1 (37.9 ± 0.60 vs. 27.6 ± 0.62 g, p < 0.001). However, as shown in Figure 1B, the thymus was significantly lighter in the 24-week-old SAMP10 than 6-week-old SAMP10 (27.6 ± 2.61 vs. 38.4 ± 2.46 mg, p < 0.05). Also, the thymus was significantly lighter in the 24-week-old SAMR1 than 6-week-old SAMR1 (25.3 ± 1.95 vs. 37.0 ± 2.94 mg, p < 0.05).

Body and thymus weight and percentages of CD4, B220, and CD11b/Gr-1 in the peripheral blood in the SAMR1 and SAMP10 mice at 6 weeks (6w) and 24 weeks (24w) old. Body weight (A) and thymus weight (B). The percentages of cluster of differentiation 4 (CD4)-positive cells (C), B220-positive cells (D), CD11b/Gr-1-positive cells (E). SAMR, senescence-accelerated mice resistant 1; SAMP10, senescence-accelerated mice prone 10.
The lymphocyte populations in the peripheral blood were analyzed by FACS. Figure 1C shows that the percentages of CD4+ cells were significantly lower in the 24-week-old SAMP10 than in the 6-week-old SAMP10 (18.8 ± 1.2% vs. 29.6 ± 1.46%, p < 0.05). Figure 1D shows that the percentages of B220+ cells were also significantly lower (20.9 ± 1.49% vs. 32.8 ± 3.5%, p < 0.05). Figure 1E shows that the percentages of CD11b+/Gr-1+ cells were significantly higher in the 24-week-old mice (44.7 ± 3.73% vs. 29.9 ± 4.08%, p < 0.05). There was no significant difference in the percentage of CD8+ cells between 6-week-and 24-week-old SAMP10 (8.4 ± 2.15% vs. 6.9 ± 0.74%) (data not shown). There were no significant differences in the percentages of CD4+ (Fig. 1C), CD8+, B220+ (Fig. 1D), or CD11b+/Gr-1+ (Fig. 1E) cells between 24-week-and 6-week-old SAMR1 (35.7 ± 2.95% vs. 28.5 ± 1.7%, 10.2 ± 0.85% vs. 7.7 ± 2.4%, 21.3 ± 2.6% vs. 20.8 ± 4.4%, 21.1 ± 3.15% vs. 20.2 ± 1.8%).
Analyses of Cytokine Production, Analyses and Morphology of Thymus
Figure 2A shows that the percentage of TNF-α-producing CD4+ cells was significantly lower in the 24-week-old SAMP10 than in the 6-week-old SAMP10 mice (9.2 ± 1.0% vs. 12.9 ± 0.89%, p < 0.05). Figure 2B shows that the percentage of IL-6-producing CD4+ cells was significantly greater in the 24-week-old SAMP10 than in the 6-week-old SAMP10 (5.3 ± 0.41% vs. 4.1 ± 0.45%, p < 0.05). Interestingly, there were no significant differences in the percentages of TNF-α- and IL-6-producing CD4+ cells between 24-week- and 6-week-old SAMR1 (14.8 ± 0.29% vs. 16.6 ± 0.61%, 4.3 ± 0.37% vs. 7.4 ± 1%) (Fig. 2A and B).

Percentages of TNF-α- and IL-6-producing splenocytes in the SAMR1 and SAMP10. (A) The percentage of tumor necrosis factor α (TNFα)-producing CD4 T-cells in the spleen. (B) The percentage of interleukin 6 (IL-6)-producing T-cells in the spleen. (C) The percentages of CD4+CD8+, CD4-CD8- cells in the thymus of 6-week- and 24-week-old SAMP10 and SAMR1. (D, E) Cytokeratin 18 (CK18) staining, (F, G) CK8 staining, and (H, I) CK5/6 staining in 6-week and 24-week SAMP10. Scale bar: 25 μm.
For analyzing the Treg cells, there was no significant difference in the percentage of CD4+/FoxP3+ cells between 24-week-old SAMP10 and 6-week-old SAMP10 (7.5 ± 1.18% vs. 6.4 ± 0.67%). Similarly, there was no significant difference in the percentage of CD4+/FoxP3+ cells between 24-week- and 6-week-old SAMR1 (4.3 ± 0.7% vs. 7.5 ± 0.10%).
Figure 2C shows that the percentages of CD4+CD8+ thymocytes were significantly lower in the 24-week-than 6-week-old SAMP10 (78.5 ± 0.76% vs. 82.9 ± 0.21%, p < 0.05), while there was no significant difference in the percentages of CD4-CD8- thymocytes (7.8 ± 0.30% vs. 5.9 ± 0.61%). There were no significant differences in the percentages of CD4+CD8+ thymocytes and CD4-CD8- thymocytes between 24-week- and 6-week-old SAMR1 (80.65 ± 0.44% vs. 80.16 ± 1.05%, 9.5 ± 1.73% vs. 8.19 ± 1.32%).
Figure 2D–I show CK18+, CK8+, and CK5/6+ cells in the thymus. There was no significant difference in the expression of cytokeratin between 6-week- and 24-week-old SAMP10 and SAMR1 (data not shown).
Microarray Analysis
Figure 3 shows the results of DNA microarray, which indicated that there were differences between 6-week-and 24-week-old SAMP10. Figure 3A shows that three genes related to transcription coactivator activity [mediator complex subunit 14 (Med14), thyroid hormone receptor-associated protein 3 (Thrap3), and tenascin C (Tnc)] were downregulated, and one gene related to transcription corepressor activity [brain abundant, membrane-attached signal protein 1 (Basp1)] was upregulated. Figure 3B shows that genes related to growth factor activity [fibroblast growth factor 21 (fgf21)], hormone activity [glucagon (gcg), resistin (Retn), and parathyroid hormone-like hormone (Pthlh)], and cytokine activity [secreted phospho-protein 1 (Spp1), chemokine (C-X-C motif) ligand 15 (Cxcl15), cardiotrophin 2 (Ctf2), and chemokine C-C motif ligand 6 (Ccl6)] were all downregulated. Figure 3C shows that most genes related to receptor activity [semaphorin 3d (Sema3d), vomeronasal 1 receptor 199 (Vmnlr199), interleukin 22 receptor α 2 (IL-22rα2), zinc finger protein 369 (Zfp369), triggering receptor expressed on myeloid cells-like 1 (Treml1), cholinergic receptor, nicotinic, β 3 (neuronal) (Chrnb3), asialoglycoprotein receptor 2 (Asgr2), IL-9r, glutamate receptor, ionotropic, d 2 (Grid2), interleukin 6 signal transducer (IL-6st), macrophage receptor with collagenous structure (Marco), coxsackie virus, and adeno-virus receptor (Cxadr) and Sema3f] were downregulated [except for olfactory receptor 975 (Olfr975)]. Figure 3D shows that genes related to chemotaxis [Cxcl13, S100 calcium-binding protein A8 (S100a8) and S100a9] and acute phase response [serum amyloid A3 (Saa3) and regenerating islet-derived 3 g (reg3g)] were downregulated, while one gene related to the response to hypoxia was upregulated [hypoxia upregulated 1 (Hyou1)]. Figure 3E shows two of three genes related to innate immunity [C-type lectin domain family 4, member D (Clec4d) and ring finger and CCCH-type domains 1 (Rc3h1)] were downregulated, while one of three genes was upregulated [histocompatibility 2-blastocyst (H2-Bl)]. Two genes related to regulation of the immune system [orosomucoid 1 (Orm1) and Orm3] were downregulated. One gene related to the inflammatory response [histamine receptor H4 (hrh4)] and one gene related to the humoral immune response [natriuretic peptide receptor 3; longjohn (lgj)] were downregulated. In contrast, one gene related to hormone activity (Gcg), one gene related to receptor activity (Zfp369), one gene related to chemotaxis (S100a8), and one gene related to the acute phase (reg3g) were downregulated; and one gene (Cxcl13) was upregulated in 24-week-old SAMR1 when compared with 6-week-old SAMR1 (data not shown). However, there were no differences in the genes shown in Figure 3 other than those listed above.

Microarray analysis of thymus. (A–E) Results of microarray fold changes for 40 genes differing at least onefold. These genes were related to transcription coactivator activity, transcription corepressor activity, growth factor activity, hormone activity, cytokine activity, chemotaxis, and acute phase response innate immune regulation of immune system, inflammatory response, and humoral immune response. See text for definition of abbreviations.
Analyses of TECs by RT-PCR
Figure 4A–C shows the ratios of KGF/GAPDH (0.57 ± 0.08 vs. 0.98 ± 0.01, p < 0.05), Aire/GAPDH (0.32 ± 0.02 vs. 0.55 ± 0.02, p < 0.05), and Sirt1/GAPDH (3.4 ± 0.03 vs. 4.3 ± 0.24, p < 0.05) for the 24-week-old compared to the 6-week-old mice by RT-PCR. The ratios were significantly lower in the 24-week-old SAMP10 than in the 6-week-old SAMP10 mice. These ratios were improved significantly after BMT (0.77 ± 0.1 vs. 0.57 ± 0.08, 0.42 ± 0.01 vs. 0.32 ± 0.02, 4.2 ± 0.29 vs. 3.4 ± 0.03, p < 0.05, respectively). However, there was no difference in the expression of IL-7 or Foxn1 (data not shown).
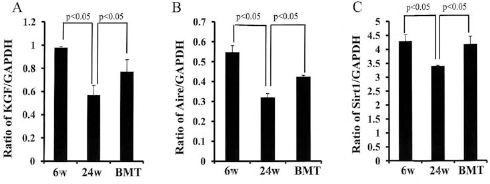
Analyses of TECs by RT-PCR. (A–C) Ratios of keratinocyte growth factor/glyceraldehyde 3-phosphate dehydrogenase (KGF/GAPDH), autoimmune regulator (Aire)/GAPDH, and sirtuin 1 (Sirt1)/GAPDH by RT-PCR. The ratios were significantly lower in the 24-week-old SAMP10 than in the 6-week-old SAMP10 mice. These ratios were improved significantly after intrabone marrow (IBM)–bone marrow transplantation (BMT) compared with the nontreated mice. TECs, thymic epithelial cells.
In contrast, there were no significant differences in the ratios of KGF/GAPDH (1.02 ± 0.22 vs. 1.03 ± 0.13), Aire/ GAPDH (0.70 ± 0.12 vs. 0.84 ± 0.09), or Sirt1/GAPDH (5.82 ± 0.49 vs. 6.33 ± 0.42) between the 24-week- and the 6-week-old SAMR1 mice (data not shown).
Morphology of Bone Marrow and Analyses of BMMSCs by RT-PCR
Many more adipocytes were found in the bone marrow of the 24-week-old SAMP10 (Fig. 5B) than in the 6-week-old SAMP10 mice (Fig. 5A). However, no adipocytes were observed in the bone marrow of 24-week-old SAMP10 treated with IBM–BMT (Fig. 5C). This suggests that donor-derived BMCs (probably MSCs) improved adipogenesis in the bone marrow of treated 24-week-old SAMP10. There is no specific surface marker on BMMSCs, and they were identified as an adherent, fibroblast-like population, and negative for CD11b and CD45, and positive for Scal-1, CD90, and CD106 (data not shown). Figure 5D–F shows the ratios of Sirt1/GAPDH (0.7 ± 0.02 vs. 1.0 ± 0.03, p < 0.05), RankL/GAPDH (1.3 ± 0.07 vs. 0.3 ± 0.06, p < 0.05), and OPG/GAPDH (0.05 ± 0.004 vs. 0.14 ± 0.017, p < 0.05) for 24-week- compared to 6-week-old mice by RT-PCR. The ratios were significantly lower in the 24-week-old SAMP10 than in the 6-week-old SAMP10 mice. These ratios were improved significantly after IBM–BMT (1.2 ± 0.01 vs. 0.7 ± 0.02, 0.1 ± 0.01 vs. 1.3 ± 0.07, 0.11 ± 0.006 vs. 0.05 ± 0.004, p < 0.05, respectively) compared with the nontreated mice.
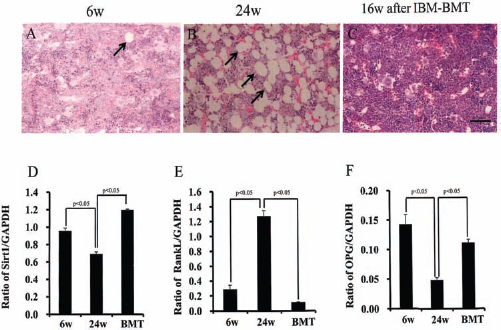
Morphology of bone marrow and analyses of BMMSCs by RT-PCR. (A–C) The morphology of the bone marrow. Many more adipocytes were found in the bone marrow of the 24-week-old SAMP10 (arrows in B) than in the 6-week-old SAMP10 mice (arrow in A). However, no adipocytes were observed in the bone marrow of 24-week-old SAMP10 treated with intrabone marrow– bone marrow transplantation (IBM–BMT) (C). (D–F) Ratios of Sirt1/GAPDH, receptor activator of nuclear factor k-light-chain-enhancer of activated B-cells (NF-kB) ligand (RankL)/GAPDH, and osteoprotegerin (OPG)/GAPDH by RT-PCR. These ratios were improved significantly after IBM–BMT compared with nontreated mice. Scale bar: 25 μm. BMMSCs, bone marrow-derived mesenchymal stromal cells.
Discussion
SAMP10 show age-related behavioral deterioration such as deficits in learning and memory and emotional disorders (45). The inhibitory control of exploration by the hippocampus seems to be damaged and habituation lost in SAMP10 over 7 months of age (42). The immune system modulates the central nervous system (CNS) function and behavioral process (47). The thymus is a source of T-cells, affecting the adaptive immunity, and the immune system plays a central role in modulating learning, memory, and neural plasticity. Under normal quiescent conditions, immune mechanisms are activated by environmental/psychological stimuli and positively regulate the remodeling of neural circuits, promoting memory consolidation, hippocampal long-term potentiation, and neurogenesis. These beneficial effects of the immune system are mediated by complex interactions among brain cells with immune functions (particularly microglia and astrocytes), peripheral immune cells (particularly T-cells and macrophages), neurons, and neural precursor cells (47). However, the thymus continues to grow between birth and puberty and then begins to atrophy. We therefore chose SAMP10 that were younger than 7 months (24-week-old SAMP10) to examine the age-related change in the immune system dysfunction in SAMP10 mice.
IBM–BMT is a method of bone marrow transplantation first established by our group (28). Whole bone marrow cells from donors are injected directly into the bone cavity of recipients, and hematopoietic stem cells (HSCs) and MSCs are both transferred. IBM–BMT is a better method for allogeneic BMT than intravenous-BMT (IV-BMT) because i) no graft-versus-host disease (GVHD) develops even when whole bone marrow cells [including a small number (<6%) of T-cells] are injected, and ii) hemopoietic recovery is rapid. Both HSCs and MSCs can proliferate and differentiate inside the marrow after IBM–BMT without becoming trapped in the lung (23). MSCs have the ability to modify and influence almost all the cells of the innate and adaptive immune systems; to interfere with and affect cellular proliferation, differentiation, maturation, and function to induce an anti-inflammatory phenotype; and to modulate the immune response mediated by MSC soluble factors, including IL-6, macrophage colonystimulating factor (M-CSF), IL-10, transforming growth factor-β (TGF-β), hepatocyte growth factor (HGF), and prostaglandin E2 (PGE2) (1,37). Thus, IBM–BMT was used in preference to IV-BMT in the experiment.
Our results show that the percentage of CD4+CD8+ double-positive thymocytes was significantly lower in the 24-week-old SAMP10, although there was no significant difference in the expression of cytokeratin between cTECs and mTECs in the thymus. DNA microarrays of thymus showed that most genes were downregulated in the thymus of 24-week-old SAMP10. Microarray analyses of the thymus provided some insight into the potential complexity of thymic maturation and the redundancy of chemokines contributing to thymic development. Normal aging is associated with anatomical and functional changes in both the thymus and bone marrow. The aged thymus shows a disproportionate loss of TECs (18) and disrupted thymic architecture (19), which result in an increased risk of autoimmunity through the escape of potentially self-reactive T-cells from the disrupted thymic microenvironment. The thymic stroma of each region provides the microenvironment required for distinct aspects of T-cell development. cTECs regulate the directional migration and population expansion of T-cell progenitors as well as the positive (and in part negative) selection of CD4+CD8+ double-positive thymocytes (4). Gray et al. have shown that Aire also affects the development of mTECs, with increased numbers, and reduced turnover, of this population in Aire-deficient mice (16). Aire expression has a mild impact on the number of the single-positive (SP) thymocytes normally found in T-cell receptor (TCR) transgenic thymi. This impact may be through effects on positive selection, survival, or migration of SP thymocytes (22).
Our previous report suggested that bone marrow cells contain the precursors of functional TECs and that they can differentiate into TECs, thereby correcting thymic function (44). RT-PCR showed that downregulated KGF, Aire, and Sirt1 on the TECs of 24-week-old SAMP10 was improved after IBM–BMT. Thus, the findings showed that dysfunction of the TECs from 24-week-old SAMP10 was modulated by allogeneic bone marrow cells in this experiment. Future studies will focus on the relationship between Aire and Sirt1 on the TECs using Sirt1-deficient mice.
In the peripheral blood, the significant increases in the percentages of Gr-1+ cells may be the result of the significant decreases in the percentages of CD4+ cells and B220+ cells. The Th cell imbalance, wherein there is a significant decrease in Th1 (TNF-α) cells and a significant increase in Th2 (IL-6) cells, may be connected with the neurodegeneration seen in the older SAMP10. It has been suggested that Th cells also show notable alteration with aging, and the dysregulation of the Th1/Th2 balance may contribute to an increased rate of infections in elderly people (2). The balance between Th1 and Th2 cell responses to an infectious agent can influence both pathogen growth and immunopathology. Many factors influence the differentiation of Th1 and Th2 cells, including the antigen dose and form, the affinity between the peptide antigen and the T-cell receptor (38), the nature and degree of costimulation (8), the presence of antigen-presenting cells (20,46), and the cytokine milieu surrounding the differentiating cells (50).
It is known that Th1-type proinflammatory immune responses that are important for the generation of protection against viral infections are implicated in autoimmune disorders, whereas Th2-type anti-inflammatory responses have generally been shown to inhibit autoimmune disease (12). The effect of aging on the local defense system against respiratory syncytial virus (RSV) infection was investigated using an aged mouse model of the SAMP10. The deficiency in cellular immune response was due to a lack of clonal expansion of CD4 and CD8 T lymphocytes, together with an imbalance in the Th1/Th2 cytokine production in the respiratory tract (31). These results suggest that age-related immune dysfunction, especially defects in cellular immune responses, account for the increased morbidity and mortality in RSV infection of the elderly. However, aging appears to be associated with dysregulated inflammation, as suggested by increased plasma levels of inflammatory cytokines IL-6 and TNF-α, which predict an increased mortality risk independent of other risk factors in the elderly (21,27).
Second, BMMSCs of 6-week- and 24-week-old SAMP10 were examined. Our results showed that the number of adipocytes was decreased significantly in the bone of aged SAMP10 after IBM–BMT. Furthermore, the expression of Sirt1, RankL, and OPG of MSCs was improved after IBM–BMT. These results suggest that IBM–BMT may prevent adipogenesis in the bone of aged SAMP10 by upregulating the expression of Sirt1 on the BMMSCs.
Bone contains three principal cell types that are responsible for maintaining these functions including bone-synthesizing osteoblasts, bone-resident osteocytes, and bone-resorbing osteoclasts and undifferentiated BMMSCs. Remodeling and maintenance of bone tissue are regulated through bone extracellular matrix synthesis by osteoblasts and extracellular matrix resorption by osteoclasts (36). In age-related osteoporosis, adipocytes are increased in the bone marrow (10). Activation of the nuclear NAD-dependent protein deacetylase Sirt1 has been shown to result in mobilization of fat in fully differentiated adipocytes and a decrease in adipocyte formation from preadipocytes (35). Activation of Sirt1 blocked adipocyte development and increased the expression of osteoblast markers (7). RankL, a member of the TNF superfamily, is able to stimulate osteoclasts and can cause the resorption of bone mass in vivo by activating preosteoclasts (24,26). Osteoblasts and adipocytes originate from a common progenitor arising from MSCs, and aging causes a decrease in the number of osteoblasts and an increase in the number of marrow adipocytes (32). Sirt1 modulates RankL, activates NF-kB signaling, and inhibits osteoclastogenesis in bone-derived cells (40). Activation of Sirt1 significantly decreases the number of adipocytes and increases the expression of osteoblast markers during differentiation of MSCs (7). Donor-derived TECs were found in both the medullary and the cortical areas of recipient thymus after IBM–BMT (44). Future studies will focus on how IBM–BMT modulates TEC function in the SAMP10.
Although the weight of the SAMR1 thymus, as a control of SAMP10, decreased significantly with aging, there were no significant differences in the percentages of CD4+CD8+ in the thymus, cytokine levels, or expression of KGF, Aire, or Sirt1 on the TECs between 6-week- and 24-week-old SAMR1. Thus, neurodegenerative changes may be associated with thymus dysfunction in SAMP10. Moreover, although irradiation does not appear to change the kinetics of early T-cell development (29), the thymic microenvironment is altered after the treatment. IL-7 production is decreased in TECs after irradiation (13). Moreover, fms-like tyrosine kinase 3 (Flt3) ligand expression is increased after a sublethal dose of irradiation, suggesting that Flt3+ progenitors may undergo preferential expansion (25). However, the thymus had recovered completely by almost 10 weeks after treatment (51). In this experiment, the TECs of SAMP10 mice were studied 16 weeks after IBM–BMT. To distinguish the effects of the IBM–BMT from the effects of irradiation on the TECs, bone marrow cells from SAMP10 were syngeneically transplanted into SAMP10. There were no significant differences in the expression of Sirt1, KGF, or Aire of the TECs, suggesting that the TEC dysfunction of older SAMP10 may be modulated by allogeneic transplantation of bone marrow cells under our experimental conditions.
In this experiment, we attempted to determine whether thymus dysfunction is related to age-dependent CNS dysfunction in the SAMP10 strain. In future experiments, we will examine whether IBM–BMT can correct the abnormal phenotypes such as brain atrophy, shrinkage, and loss of cortical neurons, impaired learning and memory, depressive behavior, and decreased neurotrophic factors and hippocampal zinc and zinc transporters, etc., in the brain of SAMP10 and also examine the crosstalk between the immune and nervous systems using Sirt1-deficient mice.
In conclusion, this is the first report of age-related changes in the immune system dysfunction of 24-week-old SAMP10. Moreover, the decreased expression of Sirt1, Aire, and KGF in the TECs of 24-week-old SAMP10 was improved after IBM–BMT. Similarly, the abnormal expression of Sirt1, OPG, and RankL on the BMMSCs was also improved after IBM–BMT. These findings showed that the dysfunction on the TECs of 24-week-old SAMP10 was modulated by allogeneic bone marrow cells.
Footnotes
Acknowledgments
We would like to thank Mr. Hilary Eastwick-Field and Ms. Keiko Ando for their help in the preparation of the manuscript. This study was supported by Otsuka Pharmaceutical Company, Ltd., and The Research on Allergic Disease and Immunology Committee from Health and Labour Sciences Research Grants of the Ministry of Health, Labour and Welfare. We thank Ms. Aiko Kitajima for her excellent technical assistance regarding morphology. The authors declare no conflicts of interest.
