Abstract
Neural transplantation of GABA-producing cells into key structures within seizure-suppressing circuits holds promise for medication-resistant epilepsy patients not eligible for resection of the epileptic focus. The substantia nigra pars reticulata (SNr), a basal ganglia output structure, is well known to modulate different seizure types. A recent microinjection study by our group indicated that the subthalamic nucleus (STN), which critically regulates nigral activity, might be a more promising target for focal therapy in epilepsies than the SNr. As a proof of principle, we therefore assessed the anticonvulsant efficacy of bilateral and unilateral allografting of GABA-producing cell lines into the STN using the timed intravenous pentylenetetrazole seizure threshold test, which allows repeated seizure threshold determinations in individual rats. We observed (a) that grafted cells survived up to the end of the experiments, (b) that anticonvulsant effects can be induced by bilateral transplantation into the STN using immortalized GABAergic cells derived from the rat embryonic striatum and cells additionally transfected to obtain higher GABA synthesis than the parent cell line, and (c) that anticonvulsant effects were observed even after unilateral transplantation into the STN. Neither grafting of control cells nor transplantation outside the STN induced anticonvulsant effects, emphasizing the site and cell specificity of the observed anticonvulsant effects. To our knowledge, the present study is the first showing anticonvulsant effects by grafting of GABA-producing cells into the STN. The STN can be considered a highly promising target region for modulation of seizure circuits and, moreover, has the advantage of being clinically established for functional neurosurgery.
Keywords
Introduction
Neural transplantation is one therapeutic approach experimentally investigated to treat patients with intractable epilepsy, in which surgical resection of the epileptic focus is not feasible (8,10,25,53,60,77,78,80,85). In epilepsy, neural transplantation aims to correct a presumed imbalance between excitatory and inhibitory neurotransmission. Thus, seizures might be suppressed by grafting cells, which continuously release an inhibitory transmitter such as γ-aminobutyric acid (GABA). In this context, pharmacological microinjection studies using drugs that locally increase GABAergic inhibition show good predictive validity for anticonvulsant efficacy in response to focal manipulation and are thus helpful to define promising brain targets for grafting studies with GABA-producing cells.
Apart from transplantation of appropriate cells into the seizure focus, more remote brain structures such as the basal ganglia are considered promising target structures for focal therapies of epilepsies because of their capability to modulate different experimental seizure types with different location of the seizure focus (35). This is especially true for the substantia nigra pars reticulata (SNr), a basal ganglia output structure that has been shown to be a common final path for the propagation and modulation of seizure activity (20,35,46). There is ample evidence that inhibition of the SNr by bilateral microinjection of drugs, which increase GABAergic or decrease glutamatergic function, or by bilateral grafting of GABA-producing cells suppresses or reduces a large number of different seizure types (15,20,22,24,35,46,48,52,55,59,63,82,86).
Among other afferents, the activity of the SNr is strongly regulated by monosynaptic glutamatergic (excitatory) input from the subthalamic nucleus (STN). Accordingly, microinjection of the GABAA receptor agonist muscimol into the STN decreased both metabolic and electrophysiological activity in the SNr (29). Inhibition of the STN (i.e., indirect inhibition of the SNr) therefore was expected to result in similar seizure suppression and modulation as direct inhibition of the SNr. Indeed, microinjection of muscimol into the STN was shown to protect against acute seizures induced by flurothyl (88) and bicuculline (26). Furthermore, microinjection of muscimol into the STN was shown to suppress generalization of amygdala-kindled motor seizures (22) as well as the occurrence of spontaneous nonconvulsive seizures in a genetic model of absence epilepsy (23).
In a direct comparison of the magnitude of anticonvulsant effects induced by microinjection of the antiepileptic drug vigabatrin, an irreversible inhibitor of GABA degradation, into several basal ganglia regions, we recently have shown that the STN seems to be a more promising target region for focal therapeutic manipulation than different subregions of the SNr (12). Apart from this important observation, the STN has the advantage of being clinically established for functional neurosurgery, as it is targeted for high-frequency stimulation in patients suffering from intractable epilepsy and Parkinson's disease (1–3,17,50,84). Thus, experimental as well as clinical data render the STN highly attractive for further focal therapies, such as neural transplantation in epilepsies.
To our knowledge, no studies aiming to suppress the STN by grafting of GABA-producing cells have been performed so far in experimental models for epilepsy. As a first proof-of-principle study, we therefore investigated transplantation of GABA-producing cells into the STN using an acute seizure model in the rat. To assess the duration of putative anticonvulsant effects, we evaluated seizure threshold increases at several time points after cell grafting into the STN.
In the present study, we allografted immortalized GABAergic cells (M213-2O) as well as human glutamic acid decarboxylase (hGAD)-overexpressing cells (Clone-4), both originating from rat striatal cells (18,40,41), into the STN. The hGAD-overexpressing cells have been shown to produce and release GABA at much higher amounts, up to 50-fold, than the parent cell line M213-2O (18,40,57). Previous studies by our group (63) as well as by the group of Magda Giordano (15,16) revealed anticonvulsant effects by bilateral grafting of these GABA-producing cell lines into the SNr in different epilepsy and seizure models.
The previous muscimol studies mentioned above (26,88) provided conflicting results whether unilateral or only bilateral manipulation of the STN is sufficient to induce anticonvulsant effects. Therefore, we paid attention not only to bilateral but also to unilateral transplantation of GABA-producing cells into the STN. Grafting bilaterally outside the STN as well as transplantation of control cells, which do not produce or release GABA (41), served as controls.
Materials and Methods
An overview of the study design is illustrated in Figure 1.
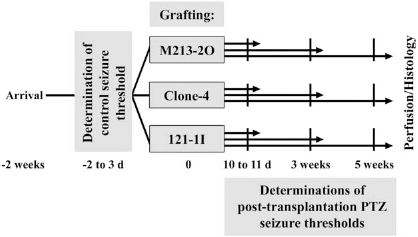
Time line representing the sequence of procedures conducted during the present study. Three different cell lines were transplanted: γ-aminobutyric acidergic (GABAergic) M213-2O cells, GABA-producing Clone-4 cells that overexpress the hGAD, and non-GABA-producing 121-1I control cells that do not express GAD. Refer to the text for details. PTZ, pentylenetetrazole.
Animals
For the present experiments, 51 adult female Wistar rats (Harlan-Laboratories, Horst, Netherlands) were purchased at a body weight of 200–220 g. The rats were housed in groups of three to five in an animal husbandry facility without males in order to keep them asynchronous with respect to their estrous cycle (47). All rats were kept under controlled environmental conditions with a 12-h/12-h light/dark cycle, with light on at 6:00 a.m., for at least 1 week before the experiments. Standard laboratory chow (Altromin 1324 standard diet) and tap water were allowed ad libitum. All experiments were done in compliance with the European Communities Council Directive of November 24, 1986, and were formally approved by the animal subjects review board of our institution.
Seizure Model Used for Assessing the Anticonvulsant Effect After Transplantation Into the STN
For determination of seizure thresholds before and after transplantation of different cell lines into the STN, the timed intravenous pentylenetetrazole (PTZ) infusion seizure test was performed as described in detail recently (51,70). This test can be repeatedly performed in the same rat at intervals of about 48 h, thus allowing the study of alterations in seizure threshold in individual rats (51). To prevent a kindling effect, in the present study the number of PTZ seizure threshold determinations was limited to a maximum of four (with intervals of at least 10 days) in the same rat. One PTZ seizure threshold was determined 2 to 3 days before transplantation (control seizure threshold), so that each rat served as its own control. After transplantation, one to three PTZ seizure thresholds were determined. Based on previous transplantation experiments with GABA-producing cells (39,63), the following time points after transplantation were chosen: 10–11 days, 3 weeks, and 5 weeks (not all time points were examined in all animals).
In all rats, the PTZ seizure threshold was determined by infusion of a 0.8% solution of PTZ in saline (Caesar & Loretz, Hilden, Germany) via a 24-gauge needle (Terumo®Europe n.V., Leuven, Belgium) into the lateral tail vein of conscious, unrestricted rats. The needle was secured to the tail vein by a piece of adhesive tape and the animal was permitted to move freely inside a Makrolon® cage type III (EBECO®, Castrop-Rauxel, Germany). The needle was connected to a syringe by a flexible polyethylene tubing (Kleinfeld Labortechnik, Gehrden, Germany) and the PTZ solution was infused at a constant rate of 1.0 ml/min using an infusion pump (PHD 2000 Infusion, Harvard Apparatus, Holliston, MA, USA). Infusion was terminated immediately following the onset of the first clonic seizure. The PTZ seizure threshold was calculated in milligrams PTZ per kilogram based on the time needed to induce this clonus, the body weight of the animal, the rate of PTZ infusion, and the PTZ concentration. In all rats, the body weight was recorded shortly before PTZ seizure threshold determinations. Additionally, we calculated the threshold for the first myoclonic twitch, which always preceded the clonic seizure. In addition to calculating the PTZ dose to induce the first clonic seizure (and myoclonic twitch), all seizures or other abnormal behaviors occurring after this endpoint were recorded. For this purpose, rats were closely observed until they resumed normal behavior, which typically was about 1–2 h after PTZ infusion.
Due to deaths of animals after PTZ infusion or paravenous infusion, some animals were excluded from final analyses.
Transplanted Cell Lines
The following cell lines, which were kindly provided by Dr. William J. Freed (NIDA IRP, NIH, DHHS, Baltimore, MD, U.S.A.), were used for the present transplantation study:
M213-2O cells (GABAergic cell line): The parent cell line M213-2O was derived from embryonic day 14 (E14) rat striatum and was immortalized with the temperature-sensitive mutant 58 (tsA58) allele of simian virus 40 (SV40) large T antigen. Among other neuronal characteristics, the parent cell line has GABAergic properties (40,41). The cells were described to contain GABA in an amount of 2.44 ± 0.57 μmol/g protein (40). The cells express GAD constitutively in vitro as well as after transplantation into the rat brain (41,63).
M213-2O hGAD67 cells (CL4 cells, hGAD-overexpressing cell line; hGAD, human GAD): The CL4 cell line was obtained from the parent cell line by additional transfection with the hGAD67 cDNA using an episomal, Epstein-Barr virus-based plasmid vector (pREP10) (18). This transfection resulted in higher GABA synthesis (44-fold) and higher GABA release (50-fold at 30 min after 50 mM KCl) than the parent cell line and in the expression of hGAD additionally to rat GAD (18).
121-1I cells (non-GABAergic; control cell line): The control cell line 121-1I was also derived from embryonic rat striatum and immortalized with the tsA58 allele of SV40 large T antigen. It has an epithelial-like morphology, shows no GAD expression, and (unlike the GABA-producing cells described earlier) is positive for vimentin (41).
Cell Cultivation
Cells were grown at 37°C (95% humidity, 5% CO2) in DMEM/F12 1:1 [which contains L-glutamine and 15 mM 4-(2-hydroxyethyl)piperazine-1-ethanesulfonic acid (HEPES)] (Invitrogen, Darmstadt, Germany) with 10% fetal calf serum (Linaris, Wertheim-Bettingen, Germany) and 1% penicillin-streptomycin (Biochrom, Berlin, Germany). Confluences of 90–100% were reached after 3–4 days. CL4 cells were selected by hygromycin B (200 μg/ml medium) (Carl Roth, Karlsruhe, Germany) after two passages and up to three passages before transplantation.
Cell cultures were used to verify the expression of GAD67. Therefore, cells plated on cover slips (Gerhard Menzel GmbH, Braunschweig, Germany) coated with 0.01% poly-d-lysine (Sigma-Aldrich Chemie GmbH, Steinheim, Germany) at passages 10 to 21 were washed with phosphate-buffered saline (PBS; AppliChem GmbH, Darmstadt, Germany) at a confluence of 60–70% (after 4 to 5 days) and then fixed with 4% paraformaldehyde (Carl Roth) for 10 to 20 min. Cells were then washed with Tris-buffered saline (TBS, pH 7.6; AppliChem), blocked for 1 h [blocking solution: TBS with 2% bovine serum albumin (BSA; Sigma-Aldrich), 0.3% Triton X-100 (Sigma-Aldrich), and 5% goat serum (Dianova GmbH, Hamburg, Germany)], and then incubated with carrier solution for 10 min (TBS with 1% BSA, 0.3% Triton X-100, and 1% goat serum). Cells were then incubated at room temperature for 1 h with the primary antibody (mouse anti-GAD67, MAB5406, 1:1000, monoclonal; Millipore, Schwallbach, Germany) diluted in carrier solution. The cells were washed with TBS and incubated for 10 min with carrier solution and then for 60 min with the secondary antibody (biotinylated goat anti-mouse IgG, 1:500; Dianova) diluted in carrier solution. Cells were then washed with TBS, incubated for 60 min at room temperature with fluorescein-labeled streptavidin [cyanine 3 (Cy3); 1:500 in TBS; Sigma-Aldrich], and finally washed again with TBS. Following incubation with bisbenzimide (5 μM, 30 min; Hoechst 33342; Sigma-Aldrich) to label cell nuclei, cells were washed with PBS and distilled water. Cover slips were mounted onto glass slides with Mowiol (EMD Biosciences Inc., La Jolla, CA, USA).
At the day of transplantation, cells at confluences of 85–95% were dissociated with trypsin-ethylenediaminetetraacetic acid (EDTA; Biochrom AG), pelleted, and for some animals preincubated with bisbenzimide (see the text), washed, pelleted, and resuspended in DMEM/ F12 1:1 (without serum and antibiotics). The cell suspension was kept at room temperature (occasionally held on ice) until transplantation at a concentration of 80,000 cells/800 nl. Viability of cells was at least 90% before transplantation as determined by the trypan blue exclusion assay (Sigma-Aldrich).
For later histological verification of graft survival and location, cells were prelabeled with bisbenzimide. Therefore, cells were preincubated with 5 μM fluorochrome bisbenzimide for 30 min prior to the washing step as previously reported (32,41,54). Preincubation with bisbenzimide was previously shown to not change the viability or morphology of the presently used GABA-producing cell lines (16). In order to ensure that the prelabeling of cells also does not affect seizure thresholds, cells were prelabeled only for 16 animals.
Transplantation of Cells
Forty-nine rats from the initially purchased 51 animals could be used for bilateral transplantation of cells. The remaining 2 rats died after determination of the control PTZ seizure threshold. Two to 3 days after determination of the control PTZ seizure threshold, rats were randomly assigned for grafting of either M213-2O cells (n = 20), CL4 cells (n = 14), or 121-1I cells (n = 15). One animal per group could not be used for final evaluation due to incorrect transplantation procedure or unintended paravenous injection of PTZ, so that the final animal numbers for evaluation of data were as follows: M213-2O cells, n = 19; CL4 cells, n = 13; or 121-1I cells, n = 14 (Table 1).
Summary of the Transplantation Procedures and Sample Sizes

γ-Aminobutyric acidergic (GABAergic) M213-2O cells, human glutamic acid decarboxylase (hGAD)-overexpressing Clone-4 cells, and control 121-1I cells were transplanted with a glass microcapillary or with a Hamilton syringe, were unlabeled or labeled with bisbenzimide (5 μM), and were localized bilaterally within the STN, unilaterally within the STN, or bilaterally outside the STN. Respective sample sizes (n) are given. STN, subthalamic nucleus.
For cell grafting, animals were anesthetized with isoflurane (CP-Pharma, Burgdorf, Germany) via an inhalation mask adjusted to a stereotaxic frame (David Kopf Instruments, Tujunga, CA, USA). The animals inhaled isoflurane (3%) until surgical stage was reached. Afterward, isoflurane was reduced to 1.5% for maintenance of anesthesia. Animals were mounted to the stereotaxic device and prepared for transplantation of the three cell lines. In addition to isoflurane, the rats received local anesthesia of the scalp (2% tetracaine hydrochloride, Caesar & Loretz) and the periost (0.25% bupivacaine hydro-chloride; Carbostesin®, AstraZeneca, Wedel, Germany). Deposits of 80,000 cells in 800 nl were bilaterally grafted (one deposit per hemisphere) aimed at the STN using the following stereotaxic coordinates in millimeters relative to bregma according to the atlas of Paxinos and Watson (65): posterior, – 3.6; lateral, ± 2.6; and ventral, – 7.9 mm. These coordinates were based on preliminary experiments in age-matched Wistar rats.
We have successfully used a microtransplantation strategy with glass microcapillaries in our previous transplantation studies in which we targeted the SNr or the piriform cortex (39,52,63). However, the volume of the presently targeted STN is much smaller. This limited space may prevent the absorption of all transplanted cells. To compare different transplantation methods concerning their suitability for grafting a cell suspension into a small and densely packed brain region such as the STN, transplantation was therefore performed by using either of two different systems. Both systems were rinsed with ethanol (70%) followed by distilled water and sterile PBS before about 2 μl of the resuspended cells was filled in. Permeability of both systems was controlled after rinsing and again immediately before insertion into the brain.
Transplantation via Glass Microcapillary
Here we used a method adapted from Nikkhah et al. (61) and Nolte et al. (63) to ensure airtight connections between a 5-μl Hamilton syringe (Reno, NV, USA), a rubber connector (Kleinfeld Labortechnik GmbH, Gehrden, Germany), and a glass microcapillary (Drummond Scientific Company, Broomall, PA, USA). The glass microcapillary (without filament) was pulled by means of a horizontal micropipette puller (PC-84 Sachs-Flaming, Sutter Instruments, San Rafael, CA, USA). The tip was then broken to a diameter of 50 to 70 μm under microscopic control. Ten seconds after insertion of the microcapillary, 800 nl of cell suspension was infused over a period of 30 s. The microcapillary was left in place for 10 s after the end of injection and controlled for permeability after removal from the brain.
Transplantation via Hamilton Syringe
Here we used a 5-μl Hamilton syringe (outer tip diameter, 570 μm; inner tip diameter, 310 μm) without microcapillary to provide space for the grafted cells. Immediately after insertion of the Hamilton syringe, 400 nl of cell suspension was infused over a period of 30 s and then left in place for 2.5 min before another 400 nl was infused over 30 s. The Hamilton syringe was left in place for 2.5 min after the end of injection and controlled for permeability after removal from the brain.
After suturing (Johnson-Johnson Intl., New Brunswick, NJ, USA) the scalp, isoflurane anesthesia was terminated and animals were closely observed for any behavioral alterations after recovering from anesthesia. Following a recovery period of 10 to 11 days, the first posttransplantation seizure threshold was determined followed by two further seizure threshold tests (3 weeks and 5 weeks after transplantation) (Fig. 1).
The experimenter (A.H.) performing the rat experiments was blinded to the type of transplanted cell line throughout the study, that is, at the time of transplantation and subsequently during determination of PTZ seizure thresholds.
Histological Analysis
At different time points after transplantation (i.e., after different numbers of seizure threshold determinations) (Fig. 1), the rats were deeply anesthetized with chloral hydrate (AppliChem) and transcardially perfused with 100–150 ml of 0.01 M PBS (pH 7.6) followed by about 240 ml of 4% paraformaldehyde in 0.2 M phosphate buffer (PB) (pH 7.6). The brains were removed and cryoprotected in 10% sucrose (ALDI Einkauf GmbH & Co. OHG, Essen, Germany), followed by 30% sucrose 1–3 days later, in 0.1 M PB (pH 7.6) and stored at 4°C. Three series of coronal sections were cut at 40 mm on a freezing microtome (Leica Microsystems GmbH, Wetzlar, Germany). One series was Nissl stained with thionine (Sigma-Aldrich) for verification of transplantation sites. The remaining two series were frozen at – 20°C until free-floating processing for immu-nohistochemistry to detect glial fibrillary acidic protein (GFAP) was performed, or they were kept as reserve.
For immunohistochemistry the brain sections were treated as follows (between the following steps, sections were washed in 0.05 M TBS, pH 7.6). Sections were incubated for 30 min in 0.5% hydrogen peroxide solution (Riedel de Häen®, Hannover, Germany) to block endogenous peroxidase activity and then preincubated in blocking solution for 60 min: TBS with 2% BSA, 0.3% Triton X-100, and 10% normal rabbit serum (Dianova). Thereafter, samples were directly transferred into “carrier” solution (TBS with 1% BSA, 0.3% Triton X-100, and 1% rabbit serum) with the primary antibody (mouse anti-GFAP, G3893, 1:1000; Sigma-Aldrich; monoclonal) at 4°C for 20 h for detection of GFAP. The next day, sections were again incubated in carrier solution for 10 min and then were directly exposed to the matching secondary antibody (rabbit anti-mouse IgG; 1:500; Dianova) conjugated to biotin for 90 min at room temperature. The antibody was dissolved in carrier solution. Sections were then incubated in horseradish peroxidase-labeled avidin (Vectastain® ABC Kit; Biozol Diagnostica, Eching, Germany) for 90 min at room temperature followed by the nickel-intensified 3,3,-diaminobenzidine (DAB) reaction (0.05% DAB and 0.6% ammonium nickel sulfate in TBS, both from Sigma-Aldrich) in the presence of 0.01% hydrogen peroxide for 10 min at room temperature. Finally, sections were mounted on gelatin-coated glass slides (Gerhard Menzel), air-dried, treated with toluol (AppliChem) for 1 min, and coverslipped with Entellan (Merck, Darmstadt, Germany) followed by microscopic analysis. Samples with omission of primary antibody served as negative controls.
Comparable to (but clearly less pronounced than) previous findings after grafting into the SNr in amygdala-kindled rats (63), transplantation of the hGAD-overexpressing cell line (Clone-4) caused tissue reactions within the host brain (see the Results section). The extent of the presently observed tissue reactions was scored microscopically by using thionine-stained sections (1, no tissue reaction; 2, equivocal; 3, clear, but spatially restricted to the grafting site; 4, clear and infiltrating tissue around graft; 5, pronounced and expansive tissue reaction including malacia). The score was used for verification of putative correlations of tissue reactions with time after transplantation, transplantation system used, or seizure threshold. To increase sample sizes for calculation of putative differences in tissue reaction sizes at different times after transplantation, data from bilaterally and unilaterally localized tissue reactions within the STN and from grafting outside the STN were pooled. The time point of 3 weeks was not included into this comparison because only two animals were euthanized at this time point.
Statistical Analysis
All seizure threshold data were normally distributed as verified by the Kolmogorov-Smirnov and the D'Agostino and Pearson tests. Seizure threshold data were analyzed by one-way ANOVA followed by Dunnett's test for multiple comparisons. Seizure thresholds were not determined at all time points in all animals (Fig. 1), and so we did not use a paired test. For evaluation of a putative influence of the preincubation of cells with bisbenzimide or of the transplantation system on the seizure thresholds, a two-way ANOVA followed by Bonferroni test for multiple comparisons was used. For evaluation of putative correlations between the extent of tissue reactions within the host brain in response to grafting of hGAD-overexpressing cells and the time after transplantation or the transplantation system used, respectively, the Mann-Whitney U test was used. For evaluation of putative correlations between the extent of the tissue reactions and the seizure thresholds after grafting, the Spearman correlation test was used. All statistical analyses were performed with the Prism 5.03 software from GraphPad (La Jolla, CA, USA). All tests were used two-tailed and a value of p ≤ 0.05 was considered significant.
Results
Cell Cultivation
Confirming earlier data (41,57), phase-contrast examination showed multipolar morphology of the cultured GABAergic cells (M213-2O), hGAD-overexpressing cells (Clone-4), and control cells (121-1I) (Fig. 2). Also confirming previous data (15,18,41,57), GAD67 was differentially expressed in the two GABA-producing cell lines, with much stronger immunofluorescence for GAD67 in the hGAD-overexpressing cells (Fig. 2). In contrast, GABAergic M213-2O cells showed only moderate staining with anti-GAD67 (Fig. 2). As expected, GAD67 was not detected in the control cells 121-1I (Fig. 2).
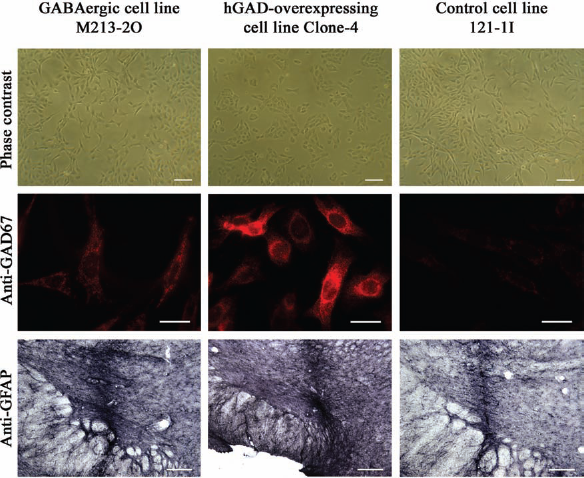
Morphology and GAD67 immunofluorescence of cultured cells and astrogliosis after transplantation into the STN. Phase contrast microscopy revealed that cultured cell lines (GABAergic M213-2O cells, hGAD-overexpressing Clone-4 cells, and control cells 121-1I) showed multipolar morphology (first row; scale bars: 100 μm). In vitro immunofluorescence revealed moderate GAD67 expression (red) in M213-2O cells, intense GAD67 expression in the hGAD-overexpressing Clone-4 cells, and no GAD67 staining in the control cells (second row; scale bars, 20 μm). Although the hGAD-overexpressing Clone-4 cells caused tissue reactions after grafting (see the text), the astrogliosis around the track was comparable for all three cell lines 5 weeks after transplantation into the subthalamic nucleus (STN). This is shown by immunohistological detection of glial fibrillary acid protein (GFAP) in coronal sections of the brain (third row; glass microcapillary system; scale bars, 200 μm). Diaminobenzidine was used for chromogenic reaction.
Localization of Transplanted Cells
As mentioned before, the final animal numbers for evaluation of data were as follows: M213-2O cells, n = 19 animals; CL4 cells, n = 13 animals; or 121-1I cells, n = 14 animals. Viability and localization of transplants were analyzed at 10/11 days, 3 weeks, or 5 weeks after implantation in individual rats (Fig. 1). Localizations of transplants were bilaterally within the STN, unilaterally within the STN, or bilaterally outside the STN. For respective animal numbers, refer to Table 1.
In cases in which the grafts were placed unilaterally outside the STN, the transplants were found dorsal or anteriodorsal to the STN (within the zona incerta; n = 8 for M213-2O grafts, n = 4 for Clone-4 grafts, n = 4 for 121-1I grafts), ventral to the STN (within the cerebral peduncle; n = 1 for M213-2O grafts), lateral to the STN (n = 1 for Clone-4 grafts, n = 1 for 121-1I grafts), or anterior to the STN (n = 1 for M213-2O grafts). In one case (M213-2O cells), the graft could not be localized in one hemisphere anymore and was therefore considered as unilaterally misplaced. In cases in which the grafts were bilaterally placed outside the STN, the transplants were found dorsal to the STN (bilaterally within the zona incerta; n = 3 for M213-2O grafts, n = 2 for 121-1I grafts), ventral to the STN (bilaterally within the cerebral peduncle; n = 1 for Clone-4 grafts), bilaterally lateral to the STN (n = 1 for 121-1I grafts), or were found at different locations on both sides (n = 1 in medial lemniscus left and cerebral peduncle right for Clone-4 grafts, n = 2 in zona incerta left and lateral to STN right for Clone-4 grafts). In one case (Clone-4 cells), the grafts could not be localized in both hemispheres anymore and were therefore considered as bilaterally misplaced.
Within the STN, Nissl staining revealed areas containing nests of implanted cells of the GABA-producing cell lines and the control cell line, respectively (Figs. 3–5). Even when the transplant was correctly placed within the STN, graft areas typically extended into areas surrounding it, probably due to the limited space within the densely packed STN, which prevents the absorption of all transplanted cells. This observation was independent of the transplantation system used; that is, neither of the two transplantation methods was advantageous in this respect. The extension of grafted cells into areas surrounding the STN was particularly obvious after labeling of the cells with bisbenzimide and is exemplarily shown in Figure 3, in which M213-2O cells were localized not only within the STN but also below (within the cerebral peduncle).

Coronal rat brain sections (Nissl stained or bisbenzimide fluorescence) showing the STN implanted with a cell suspension of GABAergic cells (M213-2O). Rectangles depict areas that are shown in higher magnification below, respectively. The sections indicated as “Merged” show merged pictures of the respective thionine (Nissl) and bisbenzimide staining given above. The M213-2O cells shown here exemplarily were transplanted using the Hamilton syringe system. Rats were perfused at 10/11 days (10d), 3 weeks (not illustrated), or 5 weeks (5wk) after grafting. Vital grafts were visible at any time point after transplantation. Note that viable cells typically were not restricted to the STN but extend to other brain areas such as the cerebral peduncle (cp; left example). Scale bars: 500 μm on panels with lower magnification and 200 μm on panels with higher magnification.

Coronal rat brain sections (Nissl stained or bisbenzimide fluorescence) showing the STN implanted with a cell suspension of GABA-producing cells overexpressing hGAD (isoform 67; Clone-4). Rectangles depict areas that are shown in higher magnification below, respectively. The section indicated as “Merged” shows a merged picture of the respective thionine and bisbenzimide staining given above. The hGAD-overexpressing cells shown here exemplarily were transplanted using either the glass microcapillary system (upper left) or the Hamilton syringe system (lower left and right). Rats were perfused at 10/11 days (10d), 3 weeks (not illustrated), or 5 weeks (5wk) after grafting. Note the more or less pronounced tissue reactions caused by grafting of the hGAD-overexpressing cells. Nevertheless, viable cells still were detected 5 weeks after transplantation. Scale bars: 500 μm on panels with lower magnification and 200 μm on panels with higher magnification. cp, cerebral peduncle.

Coronal rat brain sections (Nissl stained or bisbenzimide fluorescence) showing the subthalamic nucleus (STN) implanted with a cell suspension of control cells (121-1I). Rectangles depict areas that are shown in higher magnification below, respectively. The section indicated as “Merged” shows a merged picture of the respective thionine and bisbenzimide staining given above. The control cells shown here exemplarily were transplanted using either the glass microcapillary system (upper left) or the Hamilton syringe system (lower left and right). Note that tracks left after using the Hamilton syringe system were easily visible (lower left), whereas tracks left by the glass microcapillary system were hardly visible even at 10/11 days after grafting (upper left). Rats were perfused at 10/11 days (10d), 3 weeks (3wk), or 5 weeks (not illustrated) after grafting. Vital grafts were visible at any time point after transplantation. Scale bars: 500 μm on panels with lower magnification and 200 μm on panels with higher magnification.
Depending on the transplantation system used (glass microcapillary or Hamilton syringe), a more or less pronounced tissue damage along the implantation track was observed (representative examples are shown in Fig. 5). Whereas the track of the glass microcapillary was hardly visible even at 10 or 11 days after transplantation in Nissl-stained sections, direct use of a Hamilton syringe typically left a track, which was easily visible also at later time points (e.g., for 3 weeks after transplantation is shown in Fig. 5). Nevertheless, the track was always easily visible (even when the glass microcapillary was used) after immunohistological staining for GFAP (Fig. 2). Comparable to previous findings (63), the borders of implants as well as the needle tracts were marked by astrogliosis after transplantation of GABA-producing cells as well as after grafting of 121-1I control cells (Fig. 2). This was independent of the grafted cell line (Fig. 2) and the transplantation system used (not shown).
Viability of Transplanted Cells
Preincubation of the cells with bisbenzimide was previously shown not to influence viability and morphology of the presently used GABAergic cell lines (16). Using preincubation of cells from the three cell lines with bisbenzimide, we detected grafted cells up to the end of our experiments, that is, up to 5 weeks after transplantation (Figs. 3–5). In other words, cells of the three cell lines were viable at least for 5 weeks after transplantation. Although not verified in the present study, the GABA-producing cells (but not the control cells) were repeatedly shown before to express GAD at different times after transplantation into the rat brain. Thus, M213-2O cells have been shown to survive up to 15 weeks after grafting into cortical cavities or into the striatum and to express GAD immunoreactivity in vitro and up to several weeks after transplantation into different brain regions (15,18,40,41,63). Expression of the hGAD67 transgene has been shown in cultures of hGAD-overexpressing cells as well as in mesencephalic tissue of rats up to 12 weeks after transplantation of the cells into the SNr (15,16).
Comparable to (but clearly less pronounced than) previous findings after grafting into the SNr in amygdala-kindled rats (63), transplantation of the hGAD-overexpressing cell line (Clone-4) caused tissue reactions within the host brain (Fig. 4). These tissue reactions appeared as cell accumulations, which were not restricted to the STN but additionally involved surrounding areas (Fig. 4). The tissue reactions caused by grafting of hGAD-overexpressing cells were characterized in detail previously (63). They involved infiltrating inflammatory cells and neuronal loss without tumor formation (63). In this previous study, no surviving Clone-4 cells were detected 4 to 8 weeks after transplantation. Using preincubation of the cells with bisbenzimide, we now show that even in rats showing a more pronounced tissue reaction, viable transplanted cells could be observed within the STN at least up to 5 weeks after grafting (Fig. 4).
There was no difference between the extent of tissue reactions 10/11 days versus 5 weeks after grafting of Clone-4 cells (p = 0.516, Mann-Whitney U test). In other words, the tissue reaction did not become more pronounced until 5 weeks after transplantation, but rather did develop in the initial time window after grafting, that is, anytime within the first 10 to 11 days after grafting, and then remained stable for the following weeks. Also, the transplantation system used (glass microcapillary vs. Hamilton syringe) did not influence the extent of the tissue reactions (p = 0.154, Mann-Whitney U test). Again, data from bilaterally and unilaterally localized tissue reactions within the STN and from grafting outside the STN were pooled to increase sample sizes. Animals from all time points were included here. Finally, for the calculation of putative correlations between tissue reaction sizes and seizure thresholds, only data from graft localizations bilaterally or unilaterally within the STN (mean scores of both hemispheres) were included. The extent of the tissue reactions was not correlated with the seizure thresholds tested at 10/11 days after transplantation (r = 0.631, p = 0.139, myoclonic twitch; r = 0.703, p = 0.088, clonus; Spearman correlation). Other time points were not tested because the tissue reactions did not change over time (see above).
Behavioral Adverse Effects of Cell Transplantation
Independent of the grafted cell line and of anticonvulsant effects (see the text), in general no apparent side effects on behavior of transplanted rats were observed. With one exception, the animals behaved normally after transplantation and at the time of test trials. In one rat, which received M213-2O cells, a transient period of backward walking was observed 1 day after transplantation. Interestingly, and in line with previous observations (63), even in animals showing pronounced tissue reactions after grafting of hGAD-overexpressing cells, no apparent adverse effects were observed. Finally, although unilateral microinjection of muscimol into the STN of rats has been shown to cause contralateral postural asymmetry (26), unilateral grafting of GABA-producing cells into the STN did not cause similar postural side effects.
Seizure Thresholds Before Transplantation
For transplantation, rats were randomly assigned to three groups, that is, for transplantation of M213-2O cells, hGAD-overexpressing CL4 cells, and 121-1I cells. As could be expected from a random assignment procedure, there were no significant differences of seizure thresholds between the three groups (p = 0.41 for the first myoclonic twitch and p = 0.20 for clonus; oneway ANOVA). PTZ seizure thresholds for induction of the first myoclonic twitch before transplantation were 16.54 ± 0.48 mg/kg for rats that received GABAergic M213-2O cells, 17.52 ± 0.77 mg/kg for rats that received hGAD-overexpressing CL4 cells, and 17.68 ± 0.87 mg/kg for rats that received the control cells 121-1I. PTZ seizure thresholds for induction of clonic seizures before transplantation were 19.83 ± 0.47 mg/kg for rats that received GABAergic M213-2O cells, 21.23 ± 0.86 mg/kg for rats that received hGAD-overexpressing CL4 cells, and 21.46 ± 0.90 mg/kg for rats that received the control cells 121-1I.
Effects of Transplantation on Seizure Thresholds
PTZ seizure thresholds (in milligrams per kilogram body weight) for induction of the first myoclonic twitch (Fig. 6) and of clonic seizures (Fig. 7) were determined before and at different time points after transplantation of GABAergic M2132O cells, hGAD-overexpressing Clone-4 cells, and control cells 121-1I, respectively.
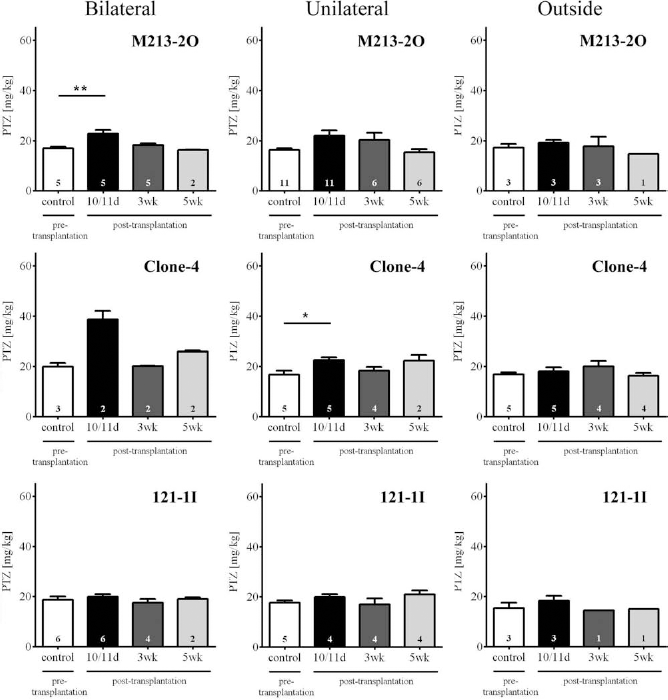
PTZ seizure thresholds for induction of first myoclonic twitch. Means and SEM of PTZ seizure thresholds for induction of the first myoclonic twitch before and at different time points after transplantation of GABAergic cells (M213-2O), hGAD-overexpressing cells (Clone-4), and non-GABAergic cells (121-1I) localized bilaterally or unilaterally within the subthalamic nucleus (STN) or bilaterally outside the STN. Sample sizes are given within the bars. Seizure thresholds significantly increased 10/11 days (10/11d) after bilateral transplantation of GABAergic M213-2O cells into the STN when compared with the pretransplantation value (p = 0.004, one-way ANOVA; **p < 0.01, q = 4.141, Dunnett's test). The anticonvulsant effect was transient, because seizure thresholds had fallen back to pretransplantation levels 3 weeks (3wk) and 5 weeks (5wk) after grafting. Bilateral grafting of hGAD-overexpressing cells also increased seizure threshold (sample size too low for statistical verification). Unilateral grafting of GABAergic M213-2O cells into the STN significantly altered seizure thresholds (p = 0.05, one-way ANOVA). The post hoc Dunnett's test, however, failed to detect significant differences. Unilateral grafting of hGAD-overexpressing cells into the STN significantly raised seizure thresholds 10/11 days after transplantation (p = 0.036, one-way ANOVA; *p < 0.05, q = 3.061, Dunnett's test). Bilateral grafting outside the STN as well as grafting of control cells did not change seizure thresholds, emphasizing the site specificity (STN) and cell specificity (GABA-producing cells) of the observed anticonvulsant effects.
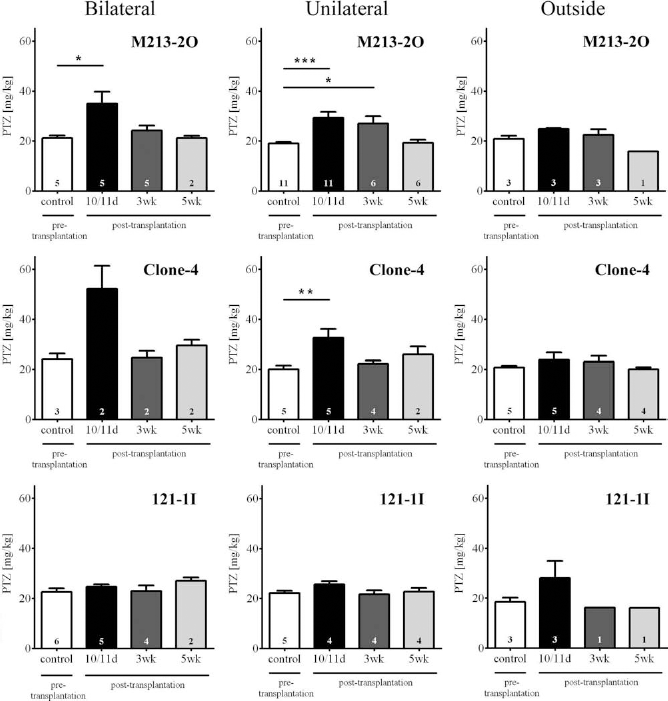
PTZ seizure thresholds for induction of clonic seizures. Means and SEM of PTZ seizure thresholds for induction of clonic seizures before and at different time points after transplantation of GABAergic cells (M213-2O), hGAD-overexpressing cells (Clone-4), and non-GABAergic cells (121-1I) localized bilaterally or unilaterally within the subthalamic nucleus (STN), or bilaterally outside the STN. Sample sizes are given within the bars. Seizure thresholds significantly increased 10/11 days (10/11d) after bilateral transplantation of GABAergic M213-2O cells into the STN when compared with the pretransplantation value (p = 0.021, one-way ANOVA; *p < 0.05, q = 3.358, Dunnett's test). The anticonvulsant effect was transient because seizure thresholds have fallen back to pretransplantation levels 3 weeks (3wk) and 5 weeks (5wk) after grafting. However, unilateral grafting of M2132O cells into the STN raised seizure thresholds for up to 3 weeks (p = 0.0007, one-way ANOVA; ***p < 0.001 at 10/11 days and *p < 0.05 at 3 weeks after grafting, q = 4.139 and q = 2.721, respectively, Dunnett's test). Bilateral grafting of hGAD-overexpressing cells also increased threshold for induction of clonic seizures (sample size too low for statistical verification). Unilateral grafting of hGAD-overexpressing cells into the STN significantly raised seizure thresholds 10/11 days after transplantation (p = 0.019, one-way ANOVA; **p < 0.01, q = 3.621, Dunnett's test). Bilateral grafting outside the STN as well as grafting of control cells did not change seizure thresholds, emphasizing the site specificity (STN) and cell specificity (GABA-producing cells) of the observed anticonvulsant effects.
Anticonvulsant Effects by Bilateral Grafting Into STN
Bilateral transplantation of the GABAergic cell line M213-2O into the STN caused a significant increase (34%) in the mean threshold for induction of the first myoclonic twitch 10/11 days after grafting compared to the value obtained before transplantation (Fig. 6). Similarly, the mean threshold for induction of clonic seizures was significantly raised (66%) 10/11 days after grafting of M213-2O cells (Fig. 7). These anticonvulsant effects were transient as they were not observed 3 weeks or 5 weeks after grafting (Figs. 6 and 7).
A similar trend for seizure threshold increase was seen by bilateral transplantation of the hGAD-overexpressing cells, although the low sample size for bilateral grafts did not allow statistical verification (Figs. 6 and 7). Nevertheless, the mean seizure thresholds were strongly raised by 95% (myoclonic twitch) and 117% (clonus), respectively, 10/11 days after grafting of hGAD-overexpressing cells. When data from bilateral and unilateral grafting of hGAD-overexpressing cells into the STN were pooled together for statistical verification, the threshold for the first myoclonic twitch was significantly raised 10/11 days after grafting (not illustrated; p = 0.009, one-way ANOVA; p < 0.01, q = 3.476; Dunnett's test). Similarly, the threshold for clonic seizures was significantly raised 10/11 days after grafting of hGAD-overexpressing cells into the STN (pooled data from bilateral and unilateral grafting; not illustrated; p = 0.002, one-way ANOVA; p < 0.01, q = 4.271, Dunnett's test).
At no time point were seizure thresholds significantly changed by grafting of the non-GABAergic control cells (121-1I) (Figs. 6 and 7).
Anticonvulsant Effects by Unilateral Grafting Into STN
Even unilateral transplantation of the GABAergic cell line M213-2O into the STN raised the mean threshold for induction of the first myoclonic twitch by 35% 10/11 days after grafting compared to the value obtained before transplantation (Fig. 6). With respect to the threshold for induction of clonic seizures, a significant and longer-lasting anticonvulsant effect was observed by unilateral grafting of GABAergic M213-2O cells into the STN. The mean threshold for induction of clonic seizures was significantly raised 10/11 days (54%) and 3 weeks (42%) after grafting of M213-2O cells (Fig. 7). Although longer lasting, this effect was transient, because 5 weeks after unilateral grafting of M213-2O cells, the seizure threshold had fallen back to the pretransplantation level (Fig. 7).
Comparably, unilateral transplantation of hGAD-overexpressing cells into the STN significantly raised the mean seizure thresholds by 34% (myoclonic twitch) (Fig. 6) and 62% (clonus) (Fig. 7), 10/11 days after grafting. These effects were also transient and were not detectable 3 or 5 weeks after bilateral grafting into the STN (Figs. 6 and 7).
As with bilateral grafting into the STN, at no time point were seizure thresholds significantly changed by unilateral grafting of the control cells into the STN (Figs. 6 and 7).
Lack of Anticonvulsant Effects by Grafting Bilaterally Outside the STN
None of the used cell lines caused a significant change in seizure thresholds (neither myoclonic twitch nor clonus and at no time point after grafting) when transplanted bilaterally outside the STN (Figs. 6 and 7). Grafts outside the STN were located within the zona incerta, cerebral peduncle, medial lemniscus, or lateral to the STN. This observation emphasizes the site specificity of the above-described anti-convulsant effects in response to grafting into the STN.
Comparison to Previous Data From Bilateral Grafting Into SNr
We previously performed a transplantation study in which we used the same GABAergic M213-2O cell line and the same control cell line 121-1I as in the present study. In this previous study, we grafted the cells bilaterally into the SNr of fully amygdala-kindled rats and observed an increased generalized motor seizure threshold (GST) 9–11 days after grafting (63). Although different from the presently used acute seizure model, we were interested in comparing the anticonvulsant efficacy observed in the previous and the present study (Fig. 8). For this purpose, we calculated the relative seizure threshold increases by grafting of M213-2O cells into the SNr and STN, respectively, in comparison to the mean relative seizure thresholds after grafting of the control cell line. This was done for better comparison across the two studies because grafting of the control cell line slightly (although not significantly) changed seizure thresholds in both studies to a different level.
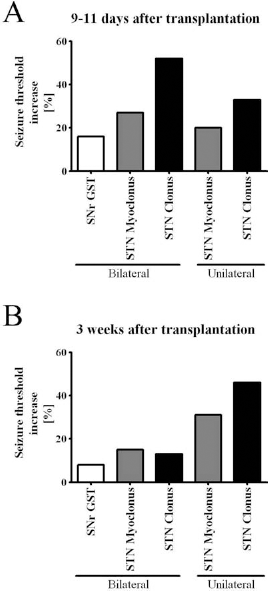
Comparison of a previous (63) and the present transplantation study. We used the same GABAergic M213-2O cell line and the same control cell line 121-1I in the previous (63) and in the present study. In the previous study, we grafted the cells bilaterally into the substantia nigra pars reticulata (SNr) of fully amygdala-kindled rats and observed an increased generalized motor seizure threshold (GST) 9–11 days after grafting (63). For better comparison across the two studies, we calculated the relative seizure threshold increases by grafting of M213-2O cells into the SNr and the subthalamic nucleus (STN), respectively, in comparison to the mean relative seizure thresholds after grafting of the control cell line. Only time points were included in the evaluation, at which significant changes in seizure thresholds compared to pretransplantation values occurred in at least one of the two studies (A, 9–11 days after grafting; B, 3 weeks after grafting). Although different models were used in the two studies, the data indicate more pronounced anticonvulsant effects by bilateral and even by unilateral grafting into the STN than by bilateral grafting into the SNr.
In the previous study, in which we transplanted the GABAergic cell line M213-2O bilaterally into the SNr of kindled rats, we observed that 9–11 days after grafting the GST was elevated by 16% compared to grafting of the control cell line 121-1I (63) (Fig. 8). However, in the present study the PTZ seizure threshold was raised by 27% and 52% (myoclonic twitch and clonus, respectively) at the same time point after grafting (10/11 days) of M213-2O cells bilaterally into the STN and compared to grafting of the control cell line 121-1I. Even unilaterally localized M213-2O grafts within the STN still raised the PTZ seizure threshold by 20% and 33% (myoclonic twitch and clonus, respectively) 10–11 days after grafting when compared to grafting of the control cell line 121-1I (Fig. 8). Three weeks after transplantation, we previously (63) did not observe significant changes in GST anymore (8% elevation of seizure threshold compared to grafting of control cell line) (Fig. 8). In the present study, however, the PTZ seizure threshold was still raised by 15% and 13% (myoclonic twitch and clonus, respectively) by bilateral grafting into the STN and even by 31% and 36% (myoclonic twitch and clonus, respectively) by unilateral grafting into the STN compared to grafting of the control cell line 121-1I (Fig. 8). Although different models were used in the two studies, the data indicate stronger and more long-lasting anticonvulsant effects by grafting into the STN than by grafting into the SNr.
Lack of Robust Influence of Methodological Parameters on Observed Anticonvulsant Effects
Although the transplantation system used (glass micro-capillary or Hamilton syringe) caused different lesion sizes, which was visible by the track left after grafting (Fig. 5), at no time point after grafting it did influence anticonvulsant efficacy (not illustrated). Furthermore, the preincubation of the cells with bisbenzimide also did not influence anticonvulsant efficacy of the GABA-producing cell lines (not illustrated). The only significant difference was a higher seizure threshold (myoclonic twitch and clonus) 3 weeks after grafting of bisbenzimide-labeled control cells compared to unlabeled control cells (p = 0.012, myoclonic twitch and p = 0.004, clonus, two-way ANOVA; p < 0.05 for myoclonic twitch and clonus, Bonferroni test). The lack of robust influence on anticonvulsant efficacy by bisbenzimide labeling of the cells is in line with a previous study showing that treatment with bisbenzimide does not change the viability or morphology of the presently used GABA-producing cell lines, neither immediately nor 4 weeks after labeling (16).
Discussion
Using an acute seizure model that allows repeated seizure threshold determinations in individual rats, the main findings of the present study were (a) that anticonvulsant effects can be induced by bilateral transplantation of GABAergic M213-2O cells and hGAD-overexpressing cells into the STN, (b) that this anticonvulsant effect was observed even after unilateral transplantation into the STN, and (c) that this anticonvulsant effect was stronger and (d) more long-lasting than transplantation of the same cell line into the SNr of amygdala-kindled rats (63). Neither transplantation of a control cell line nor transplantations outside the STN-induced anticonvulsant effects, emphasizing the site specificity (STN) and cell specificity (GABA-producing cells) of the observed anticonvulsant effects. To our knowledge, the present study is the first showing anticonvulsant effects by grafting of GABA-producing cells into the STN.
Mechanisms of Anticonvulsant Action
M213-2O cells and hGAD-overexpressing cells have been shown to produce and release GABA in vitro, to survive up to several weeks after grafting into cortical cavities or into the striatum, and to express GAD immunoreactivity in vitro and up to several weeks after transplantation into different brain regions (15,18,40,41,57,63). Thus, it can be assumed that grafting of these cells into the STN results in an increased GABAergic inhibition of the STN. The inhibition caused by transplantation of GABA-producing cells in the present study may be caused by functional integration into the host neural circuitry via establishment of synaptic contacts or, more likely, by tonic, nonsynaptic release of GABA.
It is important to note that grafting of GABA-producing cells into the STN probably does not completely mimic microinjection of drugs that increase GABAergic function such as the GABAA receptor agonist muscimol. Dybdal and Gale (26) showed that unilateral microinjection of muscimol into the STN of rats caused contralateral postural asymmetry, whereas unilateral grafting of GABA-producing cells into the STN in the present study did not cause similar postural side effects. This contrast might be explained by a different time course and strength of GABAergic inhibition induced by the two different methods, that is, microinjection of a direct GABAA receptor agonist versus grafting of GABA-producing cells. Nevertheless, our observed anticonvulsant effects in the intravenous PTZ seizure threshold test are in line with microinjection studies with muscimol, showing anticonvulsant effects by pharmacological inhibition of the STN in different epilepsy and seizure models, including generalized nonconvulsive seizures (23), flurothylinduced seizures (88), amygdala-kindled seizures (22), and bicuculline-induced seizures (26). Thus, as already known for the SNr, the STN also seems to be relatively nonselective as to the type of seizure it can influence. However, previous data indicate that inhibition of the STN may not have the same range of anticonvulsant effects as observed after inhibition of the SNr, because unlike results obtained after inhibition of the SNr, inhibition of the STN failed to protect against generalized tonic brain stem seizures in an electroshock model (79).
Anticonvulsant Circuitry
For epilepsy patients with multiple foci or without a clear focal onset, therapeutic manipulation of key structures within seizure-suppressing circuits may be a promising strategy. Studies on different seizure or epilepsy models imply that especially the SNr may be of particular interest in this respect, because bilateral microinjection of drugs that either increase GABAergic inhibition or reduce glutamatergic excitation in the SNr prevents or attenuates different experimentally induced seizure types (20, 24,33,35,37,46,48,55,59,82). Accordingly, using [3H]2-deoxyglucose mapping, the SNr has been shown to be activated during different experimentally induced generalized seizures including PTZ-induced convulsions (4). Direct or indirect inhibition of the SNr may interfere with such hyperactivity.
The STN occupies an important position to regulate basal ganglia outflow. It is composed of glutamatergic projection neurons, which are under GABAergic control mainly by the globus pallidus (globus pallidus externus in primates) (6). The STN also receives monosynaptic afferents from the cerebral cortex, thalamus, pedunculopontine nucleus, and SNr and projects back to the globus pallidus (externus), striatum, and pedunculopontine nucleus. Importantly, the STN projects monosynaptically to the SNr and the entopeduncular nucleus (globus pallidus internus in primates). Bilateral pharmacological inhibition of the STN either by GABA agonists or glutamate antagonists has been shown to decrease activity of SNr neurons (29,73) and, accordingly, to be anticonvulsant in different seizure or epilepsy models (22,26,88). Similar to microinjection of GABA mimetic drugs into the STN, transplantation of GABA-producing cells into the STN thus is likely to indirectly inhibit ipsilateral SNr neurons, which then results in disinhibition of downstream neurons within the dorsal midbrain and the pedunculopontine nucleus (35,36,62,71,72).
In our previous study, in which we transplanted the GABAergic cell line M213-2O bilaterally into the SNr of kindled rats, we also observed anticonvulsant effects. The GST was transiently increased for about 9 to 11 days (63). In the present study, even stronger anticonvulsant effects were observed up to 3 weeks after unilateral grafting into the STN. In other words, although different seizure/epilepsy models were used in the two transplantation studies, our present data indicate that inhibition of the STN may be more strongly anticonvulsant than a direct inhibition of the SNr. This is supported by recent pharmacological data by our group showing that bilateral microinjections of the GABA-elevating drug vigabatrin into the STN in the same acute seizure model as used presently was more effective to raise seizure thresholds than comparable microinjections into subregions of the SNr (12). Thus, apart from an effect mediated by the SNr, further mechanisms can be assumed to contribute to the anticonvulsant effects observed by transplantation of GABA-producing cells into the STN. There are several plausible mechanisms as described in the following.
Decreasing the excitatory (glutamatergic) drive of the STN to the ipsilateral entopeduncular nucleus, which then may influence seizure circuits via the habenula and other limbic structures, is likely to be involved in STN-evoked anticonvulsant effects (26,35). Also, the STN may mediate its anticonvulsant effects by changing the activity of the striatum. Furthermore, a more recently described direct subthalamocortical loop circuit in rats (19) may contribute to mediate anticonvulsant manipulations of the STN. Finally, an effect via parts of the zona incerta, which is located in close vicinity to the STN, is conceivable. It is important to note that occasionally an expansion of the graft into brain regions adjacent to the STN (zona incerta, cerebral peduncle) was observed. However, it is unlikely that cells located outside the STN contributed to the observed anti-convulsant effects, because grafts, which were completely placed outside the STN, did not raise seizure thresholds, emphasizing the site specifity of the observed anticonvulsant effects. Nevertheless, the zona incerta is highly connected with nearly all neural centers including the basal ganglia and various brainstem nuclei. It was suggested to have a global role in controlling visceral activity, influencing arousal, shifting attention, and maintaining posture and locomotion (58). The zona incerta has mainly glutamatergic projections to the SNr and the pedunculopontine nucleus (45) and it projects to the superior colliculus mainly from the ventral part, in which staining of GABAergic neurons is most intense within the zona incerta (58). However, its role in seizure modulation seems to be rather inconsistent (13,44), probably due to its complex chemoarchitecture and connectivity and its diverse subsections.
During the past decade, deep brain stimulation of appropriate brain regions has been used to treat several neurologic disorders including epilepsy (1,2,84). High-frequency stimulation of the STN in humans was suggested not only to suppress subthalamic neural activity, but also to activate fibers of passage as well as collateral projections, and finally to antidromically activate cortico-subthalamic projections (11,43,50). The latter mechanism may explain why anticonvulsant effects using high-frequency stimulation can also be achieved in some cases with unilateral stimulation of the STN in humans (17,35). However, as described above, the mechanisms by which high-frequency stimulation of the STN is anticonvulsant are thought to be somehow different from those after manipulation by microinjection of drugs or grafting of appropriate cells.
Anticonvulsant Effect by Unilateral Transplantation Into STN
With respect to the SNr, bilateral manipulation is necessary to prevent or attenuate convulsive and/or electro-graphic seizures (20,21,33,34,48,87). However, apparently conflicting data exist concerning anticonvulsant effects induced by unilateral manipulation of the STN. Whereas Dybdal and Gale (26) showed that bilateral, but not unilateral, microinjection of muscimol into the STN is effective to protect against seizures induced by bicuculline, Velísková and colleagues (88) showed anticonvulsant effects also after unilateral muscimol injection into the STN using the flurothyl seizure model, which is in line with our observation that unilateral transplantation of GABA-producing cells into the STN is sufficient to induce anticonvulsant effects in the PTZ seizure threshold test. However, comparable to our present transplantation study, muscimol was actually not microinjected unilaterally to the STN in the study by Velísková et al. (88), but rather was injected bilaterally with a unilateral misplacement in the vicinity of the STN. Indeed, Velísková et al. (88) discussed that the anticonvulsant effects induced by unilaterally correct muscimol injection into the STN may be due to this direct effect within the STN in one hemisphere plus the spread of muscimol from adjacent regions to the STN within the misplaced hemisphere. Similarly, we cannot completely exclude that part of the anticonvulsant effects we observed after only unilaterally correct placement of the graft was caused by a combination of the correct placement in the STN within one hemisphere and diffusion of GABA released from graft cells transplanted outside, but in the vicinity of the STN, within the other (misplaced) hemisphere. Further studies with unilateral transplantation of cells (instead of bilateral grafting with unilateral misplacement as in the present study) will clarify this issue.
However, due to limited diffusion properties of GABA and relatively fast elimination from extracellular space via reuptake and efflux, it seems rather unlikely that GABA released from transplanted cells is able to overcome larger distances (from the vicinity of the STN to the STN). Thus, other mechanisms apart from diffusion of GABA from adjacent regions to the STN and also apart from antidromic activation of cortico-subthalamic projections described for high-frequency stimulation as mentioned here seem more likely to contribute to the anticonvulsant effects observed after unilateral grafting of GABA-producing cells into the STN. (a) Although the STN is not known to have bilateral efferents, the SNr has been described to project bilaterally to the superior colliculus (38). While this observation does not explain the lack of anticonvulsant effects by unilateral inhibition of the SNr, it may explain in part the anticonvulsant effects observed in the present study after unilateral grafting into the STN. (b) As described before, the possibilities of the STN to mediate anticonvulsant effects may be more widespread than those of the SNr. (c) As mentioned earlier, a recently described direct subthalamocortical loop circuit in rats (19) may contribute to mediate anticonvulsant manipulations of the STN, presumably via commissural pathways such as those in the anterior commissure. (d) Projections from the zona incerta are often bilateral in rats, such as efferents to the thalamus and brain stem (58,67,74) and, therefore, are thought to influence brain activity in both hemispheres.
Explanations for Transient Effects
Despite long-term survival of grafted cells, previous studies (15,39,52,63,86) as well as the present study revealed only transient anticonvulsant effects by transplantation of different GABA-producing cell types into basal ganglia or related regions for a number of conceivable reasons. (a) A limited survival of grafted cells, which was not determined in the present study, most likely accounts for the transient effects. Future studies will have to include quantitative measures of cell survival and of GAD and GABA production in vivo at multiple time points after grafting to clarify the relation with the duration of anticonvulsant efficacy. Indeed, pretreating of hippocampal fetal cells with neurotrophic factors and a caspase inhibitor has been shown to result in an increased graft survival and in longer-lasting anticonvulsant effects after transplantation into the hippocampus in chronically epileptic rats (69). Whether this pretreatment strategy is similarly successful with immortalized cells needs to be investigated. (b) Although moderate cell loss can be induced in limbic brain structures by PTZ kindling, that is, by higher numbers of repeated PTZ injections, Franke and Kittner (31) did not show significant cell loss (although some first-damaged neurons were found) in adult rat hippocampal formation after six PTZ stimulations with a frequency of three stimulations per week and a fixed dose of 35 mg/kg intraperitoneally. To our knowledge, there are no studies investigating putative neurodegeneration in STN after single or repeated PTZ injections or in cells grafted into this basal ganglia structure. Franke and Kittner (31) qualitatively showed that even after 5 weeks of PTZ kindling with three PTZ injections per week only rare cell damage could be observed in the substantia nigra. It is therefore rather unlikely that the low number of PTZ tests used in our present study would cause significant cell loss. (c) A decline in synthesis and/or release of GABA by the grafted cells over time as well as compensatory downregulation of GABA receptors in response to chronic exposure to GABA may contribute to the diminishing anticonvulsant effects (30). (d) Also, an upregulation of N-methyl-d-aspartate (NMDA) receptors within the SNr, as has been described 2 weeks after lesion of the STN (68), may lead to enhanced sensibility of SNr neurons to the remaining excitatory influence and may then counteract the inhibition of the STN. (e) Network changes may limit the anticonvulsant effects because the STN is reciprocally connected to the SNr (42). Thus, indirect inhibition of the SNr by manipulation of the STN may result in a delayed disinhibition of the STN. (f) Finally, methodological issues may influence duration of anticonvulsant effects after grafting. Optimization of the transplantation protocol, such as appropriate cell suspension concentration and volume, was suggested to improve cell survival rate after transplantation (83), although transplanted cell number may not necessarily affect GABA tissue levels after grafting (15). Also, the presently used transplantation system (glass microcapillary vs. Hamilton syringe system) did not robustly influence efficacy or duration of anticonvulsant effects.
Anticonvulsant Effects and Tissue Reactions After Grafting of hGAD-Overexpressing Cells Into the STN
The anticonvulsant effects observed after transplantation of the hGAD-overexpressing cell line into the STN are in line with previous studies showing anticonvulsant effects after bilateral grafting of this cell line into the SNr or the inferior colliculus in different seizure or epilepsy models (15,16,75). This anticonvulsant effect of Clone-4 cells can be attributed to their ability to produce high amounts of GABA (15,18) and to express the hGAD67 transgene in culture as well as in mesencephalic tissue of rats up to 12 weeks after transplantation into the SNr (15,16). It is worth noting that Mejía-Toiber and colleagues (57) found that the hGAD-overexpressing cells release GABA upon glutamate activation. This is likely to add to the presently observed anticonvulsant effects after transplantation into the STN, which receives glutamatergic input from the cortex (5,64).
On the other hand, we observed a more or less pronounced tissue reaction within the host brain after transplantation into the STN of naïve rats. It is thus conceivable that part of the anticonvulsant effects we observed even after unilateral grafting of hGAD-overexpressing cells into the STN is attributable simply to a lesion effect. However, although bilateral excitotoxic lesion of the STN has been shown to suppress spontaneous absence seizures in rats at least in part, unilateral lesion failed to induce anticonvulsant effects (89). Later studies also failed to reveal suppressive effects by bilateral STN lesion on the duration of electroshock-induced tonic hindlimb extensions (79) or by unilateral STN lesion on bicuculline-induced seizures (9). Furthermore, although STN-lesioned rats were previously described to show startle-like behavior in response to handling (79,89), we never observed similar side effects after grafting of hGAD-overexpressing cells, indicating that our observed tissue reactions do not simply mimic lesion of the STN. Finally, our observation that the extent of the tissue reaction is not correlated with the seizure threshold also argues against a simple lesion effect induced by grafting of the hGAD-overexpressing cells.
In a previous transplantation study by our group, we observed even stronger tissue reactions and, at the same time, a lack of anticonvulsant effects of transplantation of hGAD-overexpressing cells into the SNr of amygdala-kindled rats (63). Furthermore, no surviving grafted cells were detectable 4 to 8 weeks after transplantation into the SNr in the previous study (63). In contrast, in the present study, we observed surviving grafted cells up to 5 weeks after transplantation.
In the previous study, we hypothesized that the graft itself as well as the condition of the host contributed to the tissue reactions because inflammatory tissue reactions in response to Clone-4 cells were observed only in kindled (63) but not in nonkindled rats (15,16,63,75). Indeed, although we now also observed tissue reactions in naïve rats, these were less pronounced than those previously observed in kindled rats and did not impede the survival of grafted cells. Similar to our previous study (63), the hGAD-overexpressing but not the M213-2O parent cell line induced tissue reactions. Thus, the graft itself contributes to the host reaction.
Because only the hGAD-overexpressing cells induced tissue reactions, it seems likely that a protein produced by these cells was responsible. We believe that hGAD itself is the most likely candidate rather than the Epstein-Barr virus nuclear antigen 1 (EBNA-1) protein or the hygromycin selection marker proteins, which are also produced by the hGAD-overexpressing cell line. hGAD proteins have a strong immunogenic potential, which has previously been described at least for humans. Although most often associated with an autoimmune response to hGAD in insulin-dependent diabetes, an immunogenic role of hGAD proteins has also been identified in several neurological autoimmune disorders including stiff-person syndrome and some types of epilepsy (7,14,27,28,49,56,66,76,81).
Methodological Issues
Although neither PTZ seizure thresholds nor the extent of tissue reactions in response to grafting of the hGAD-overexpressing cells were differentially influenced depending on the used transplantation system (glass microcapillary or Hamilton syringe), we consider the glass microcapillary system as the preferred system because of the less marked damage along the implantation track. Future studies should include quantitative measures of grafted cell survival along the needle or capillary track to further elucidate putative differences between the two transplantation systems.
A study by Castillo and colleagues (15) did not indicate any leakage of bisbenzimide from grafted cells to host cells 15 days after transplantation, as was investigated by direct intracerebral injection of different concentrations of bisbenzimide, including the one used in the present study, and by transplantation of cells lysed before grafting. Thus, the transplanted cells in our study are likely to migrate during the grafting procedure or afterward into loosely packed areas surrounding the STN, such as the cerebral peduncle.
Together with previous reports that preincubation with bisbenzimide does not change the viability or morphology of the presently used GABA-producing cell lines (16), our observation that this preincubation did not influence anticonvulsant efficacy of the GABA-producing cell lines renders the labeling of the cells with bisbenzimide a reliable method to detect viable cells over several weeks after grafting in seizure threshold tests.
Conclusion
The present data prove that anticonvulsant effects can be achieved by grafting of GABA-producing cells into the STN. Because of higher clinical relevance, further studies will investigate if transplantation of GABA-producing cells into the STN is also able to suppress seizure activity in animal models of pharmacoresistant seizures. Nevertheless, the present data indicate that anticonvulsant effects obtained even after unilateral transplantation of GABA-producing cells into the STN may be more pronounced and more long-lasting than similar bilateral transplantations of the same cells into the SNr (63). Together with recent pharmacological data (12), the STN can be considered a highly promising target region for modulation of seizure circuits and, moreover, has the advantage of being clinically established for functional neurosurgery.
Footnotes
Acknowledgments
We are very grateful to Dr. William J. Freed (NIDA IRP, NIH, DHHS, Baltimore, MD, USA) for kindly providing the striatal cell lines and for helpful comments on the manuscript. We thank Edith Kaczmarek, Michael Weißing, Franziska Kaiser, and Doris Möller for skillful technical assistance. The assistance of Laura Gey and Hannah Scheiblich is also gratefully acknowledged. The study was supported by a grant (FOR 1103/GE 1103/7-1) from the German Research Foundation (Bonn, Germany). A.H. received a scholarship from the Jung-Stiftung für Wissenschaft und Forschung (Hamburg, Germany). S.B. received a scholarship from the Studienstiftung des deutschen Volkes (Bonn, Germany). The authors declare no conflicts of interest.
