Abstract
Parkinson's disease (PD) is associated with increased excitatory activity within the subthalamic nucleus (STN). We sought to inhibit STN output in hemiparkinsonian macaques by transfection with adeno-associated virus (AAV) containing the gene for glutamic acid decarboxylase (GAD). In total, 13 macaques were rendered hemiparkinsonian by right intracarotid 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine injection. Seven animals were injected with AAV-GAD into the right STN, and six received an AAV gene for green fluorescent protein (GFP). Videotaped motor ratings were performed in a masked fashion on a weekly basis over a 55-week period. At 56 weeks, the animals were scanned with 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET). Histological examination was performed at the end of the study. No adverse events were observed after STN gene therapy. We found that the clinical rating scores for the two treatment groups had different patterns of change over time (group × time interaction, P<0.001). On FDG PET, the GAD animals exhibited an increase in glucose utilization in the right motor cortex relative to GFP controls (P<0.001). Metabolism in this region correlated with clinical ratings at end point (P<0.01). Histology confirmed GAD expression in treated animals. These findings suggest that STN AAV-GAD is well tolerated and potentially effective in a primate model of PD. The changes in motor cortical glucose utilization observed after gene therapy are consistent with the modulation of metabolic brain networks associated with this disorder.
Keywords
Introduction
Parkinson's disease (PD) is characterized by a progressive loss of dopaminergic neurons and decline of striatal dopamine. This neurodegenerative process leads to the disinhibition of glutamatergic neurons within the subthalamic nucleus (STN) with a concomitant increase in the activity of excitatory projections to the internal globus pallidus and the substantia nigra pars reticularis (Wichmann and DeLong, 1998; Hamani and Lozano, 2003). Increased pallidal output is responsible for many of the motor symptoms of PD (Brown et al, 2001). A variety of surgical approaches have been introduced to modulate the overactive STN output in PD, including high-frequency deep brain stimulation (Benabid et al, 1996; Lozano and Mahant, 2004) and stereotaxic lesioning (Alvarez et al, 2001; Su et al, 2003) of this structure. Deep brain stimulation represents an increasingly established and successful procedure with improved motor ratings at follow-up periods of up to 4 years (Lozano and Mahant, 2004; Rodriguez-Oroz et al, 2005). Although long-term treatment might not be limited by a major decline in stimulation effects, device-related complications can affect up to 20% of implanted leads (Oh et al, 2002), posing an additional burden on patients with advanced PD who already suffer considerable morbidity.
Recently, gene therapy has been proposed as a novel approach for the treatment of PD (Chen et al, 2005). In general, this therapeutic strategy has focused on the local delivery of genes to enhance dopamine production as well as neurotrophic agents (Bankiewicz et al, 2000; Kordower et al, 2000). Another approach for PD has been suggested using gene therapy to reduce abnormal pallidal output (During et al, 2001). We tested the effect of subthalamic delivery of adeno-associated viral vectors (AAV) encoding glutamic acid decarboxylase (GAD). This enzyme is rate-limiting for the synthesis of GABA, the major inhibitory neurotransmitter in the brain. Our aim was to impart inhibitory GABAergic properties to STN glutamatergic neurons and thereby alter the resulting activity of downstream targets, which influence movement. Direct support for the therapeutic effect of increased GABAergic transmission in the STN is provided by improvements in primate models of PD after a single injection of muscimol, a GABA agonist, into this area (Baron et al, 2002). Human studies have replicated these findings, demonstrating brief resolution of parkinsonian symptoms after a single injection of a GABA agonist into the STN (Levy et al, 2001). Based on this, we reported behavioral improvement after the delivery of AAV-GAD into the STN of a standard rodent model of PD (Luo et al, 2002). This was associated with increased evoked GABA release from the STN, a phenotypic change in STN neuronal firing, and a reduction in nigral dopamine cell death. Several of these findings have recently been independently replicated (Lee et al, 2005), with data suggesting that the degree of motor and physiological improvement may correlate with the level of STN GAD expression after gene transfer. In the current study, we examined the potential of subthalamic AAV-GAD to alter physiology and alleviate motor symptoms in a non-human primate model of parkinsonism.
Materials and methods
Subjects
In total, 13 adult rhesus monkeys (females, 5 to 8 kg) completed the 1-year study. Seven additional animals were originally enrolled but did not complete the study because of death during surgical anesthesia or recovery (n=2), ineffective 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) lesion detected before gene therapy (score ≤9 on the clinical rating scale (CRS) for parkinsonism in primates (Emborg et al, 1998)) (n=3), or inability to complete behavioral testing because of unrelated limb injury (n=2). No animals died, became ill, or were removed because of the gene therapy treatment. The study was performed in accordance with federal guidelines of proper animal care and with institutional animal care approval. Level II biosafety procedures were used.
At baseline, all monkeys behaved normally and were freely moving, exploring their home cage. This condition corresponded to a score of zero on the CRS, a rating instrument that has been previously validated in therapeutic and neuroprotective studies (Kurlan et al, 1991; Emborg et al, 1998, 2004). After baseline testing, all monkeys received an injection of 3 mg of MPTP-HCl into the right carotid artery under sterile surgical conditions (Kordower et al, 2000). At 5 days after MPTP administration, all animals used in the study displayed a crooked arm posture and dragging leg on the side contralateral to the intracarotid injection. (In our experience, monkeys with this behavioral phenotype display severe nigrostriatal dopaminergic depletion and do not recover clinically (Emborg et al, 1998; Emborg-Knott and Domino, 1998; Kordower et al, 2000).) These monkeys were divided into two groups matched according to post-MPTP CRS ratings obtained 5 days after intracarotid injection. Monkeys in one group (Group A: n=7; CRS mean±s.e.: 10.93±0.49) received an injection of AAV-GAD (20 μL of a 3 to 6 × 1012 genomic particles/ml vector stock, together with 5 μL of 20% mannitol) into the ipsilateral (right) STN. Monkeys in the other group (Group B: n=6; CRS: 11.25±0.46) received an injection of AAV with green fluorescent protein (GFP) (same titer as AAV-GAD, similar volumes of vector and mannitol) as a control treatment delivered to the same region. The details of the primate MRI-guided stereotaxic surgery have been presented elsewhere (Kordower et al, 1999, 2000).
Behavioral Testing
The clinical status of the monkeys was assessed weekly for 55 weeks after STN AAV surgery using CRS ratings. They were also tested for fine motor performance as described previously (Emborg et al, 1998; Kordower et al, 2000). All behavioral evaluations were performed by a trained observer (ME), who was blind to the treatment conditions. The presence of dyskinesias, behavioral disturbances, and vomiting was also recorded.
Repeated measures analysis of variance (RMANOVA) was used to determine if CRS scores changed over time and whether patterns of change differed between groups (i.e., group × time interaction). If a significant group × time interaction was found by the RMANOVA, a smoothed spline curve over time was fit on CRS scores across subjects in each group. These two spline curves were then graphically examined to compare the trajectories of the CRS scores for the two groups over time. In addition, for each of the two treatment groups, CRS subscale values for bradykinesia, gross motor skill, balance, and tremor at week 55 were individually compared with baseline values using paired Student's t-tests and considered significant for P<0.05. These analyses were performed using SAS 9.1 (SAS Institute Inc., Cary, NC, USA).
Positron Emission Tomography
At 56 weeks after STN AAV treatment, all animals were scanned under general anesthesia with 18F-fluorodeoxyglucose (FDG) and positron emission tomography (PET). Imaging was performed on a GE Advance tomograph (General Electric Medical Systems, Milwaukee, WI, USA) as described elsewhere (Kordower et al, 2000). A 30-min three-dimensional static emission study was acquired beginning 30 mins after radiotracer injection. Image acquisition was divided into six 5 min segments, with the scanner bed position alternating between two positions separated by 2 mm. For the final analysis, the data acquired during the third and fifth segments were averaged, and those from the fourth and sixth segments were averaged. These frames were then interleaved to provide a single image set, representing the FDG distribution between 40 and 60 mins postinjection, with 70 planes spanning a 14.6 cm axial range.
Scan processing was performed using SPM99 software (Wellcome Department of Cognitive Neurology, London, UK). Images were spatially normalized into a macaque brain template (Cannestra et al, 1997; Black et al, 2001). Images were smoothed with a 4 mm filter. Proportional scaling was used to adjust for differences in global metabolic rate.
Significant changes in glucose metabolism were detected using the general linear model in voxel-by-voxel comparisons generating SPM{t} maps. For the analysis of the main effect of MPTP, scans were cut into individual hemispheres and all hemispheres were flipped to the right side. Within-subject hemispheric differences were identified using the multisubjects and conditions option in SPM, with the untreated hemisphere representing the individual control condition for each animal. To identify voxels that differed across the two AAV treatment groups, we used a two-sample t-test in SPM on whole brain data. This analysis was conducted by an investigator (MC), who was blind to the identity of each treatment group. Additionally, we searched for voxels that correlated with the clinical score at week 55 using this measure as a covariate in SPM. In all analyses, differences were considered significant at a threshold of P=0.05, corrected for multiple comparisons. We also reported metabolic differences at P=0.001 at peak voxel (uncorrected), if related to prior hypotheses. Reported coordinates refer to the stereotaxic coordinate system used in the multimodal nemestrina monkey brain atlas (Cannestra et al, 1997).
Necropsy
At 1 week after FDG PET, the animals were euthanized and their brains processed for histological analysis. Sections through the midbrain and striatum were processed for immunohistochemical staining using antibodies against tyrosine hydroxylase (TH), GAD65, GAD67, and GFP (Emborg et al, 1998; Stephenson et al, 2005). Immunostaining against CD8 (for T-cytotoxic/suppressor cell populations and macaques lymphocytes), CD45 (for lymphocytes, monocytes, and granulocytes), and CD68 (expressed in monocytes/macrophages, dendritic cells, granulocytes, myeloid progenitor cells, and CD34+ hematopoietic bone marrow progenitor cells) was also performed to assess host immune reaction (Kordower et al, 2000).
Counts of TH-ir neurons were performed by a single investigator, masked to the treatment group (JM). Quantitative estimates of the total number of nigral TH-ir neurons in the substantia nigra were determined using an unbiased, stereologic cell counting method as described in detail elsewhere (Emborg et al, 1998). TH-ir neurons within the ventral tegmental area were excluded from this analysis. Neuronal counts were performed using NeuroZoom stereologic software. The density of TH-ir fibers was quantified within the caudate nucleus and putamen using the NIH image 1.60 analysis system. The average of the mean of three defined regions located within the dorsal, mid, and ventral portions of each structure from three equispaced sections was evaluated.
Results
Behavioral Findings
During the 55 weeks after AAV therapy, the animals maintained a constant weight and did not display signs of nausea, vomiting, diarrhea, fever, or infection. Throughout the study, they were cooperative during the testing sessions and were responsive to food stimuli. They also did not develop dyskinesias, hemiballism, or dystonia.
There was no difference between presurgical CRS scores for the AAV-GAD and the AAV-GFP groups (t=0.47, P=0.7, Student's t-test). Analysis of the CRS scores for weeks 0 to 55 revealed that the pattern of change in these ratings over time was different for the two AAV treatment groups (Figure 1). This is supported by RMANOVA, which revealed a significant group × time interaction (F (49, 539)=2.88, P<0.001). This analysis also showed a significant main effect of time (F (49, 539)=3.0, P<0.001) but a nonsignificant main effect of group (F (1, 11)=0.539, P>0.4). Graphical review of the fitted curves for the two groups revealed a gradual decline in CRS scores in the GAD group that continued until reaching a plateau at about week 25. By contrast, the GFP group demonstrated a decline in CRS scores over the first 10 weeks, followed by an increase to approximately 30 weeks with subsequent stabilization. In the GAD group, comparison of CRS subscale values at week 55 with presurgical values revealed significant improvements in bradykinesia (−16%, t=−2.52, P<0.05), gross motor skills (−26%, t=−2.93, P<0.05), and tremor (−36%, t=−3.87, P<0.01). These changes were not significant in the GFP group (changes of −8%, −10%, and −17% in these subscales, P>0.15). For the fine motor task, no significant difference between treatment groups was observed before MPTP administration or after subthalamic AAV delivery (P>0.05 for all time points).
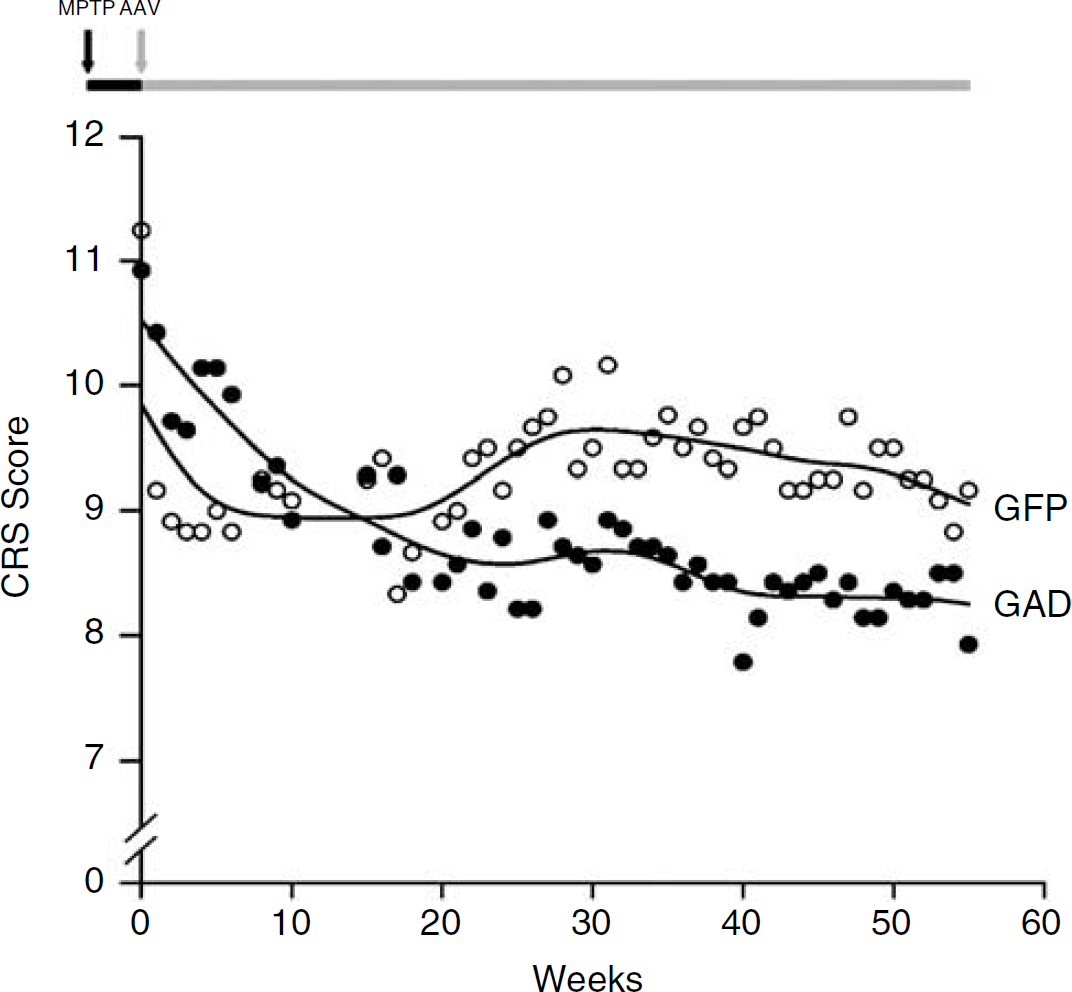
Fitted curves illustrating the time course of CRS scores in unilateral MPTP monkeys receiving STN injection of either AAV-GAD or AAV-GFP. The data are consistent with a significant difference in the trajectories of the GAD and GFP treated animals (see text). (The timeline of the study is illustrated on top. All monkeys were scored zero at baseline before right intracarotid MPTP administration (black arrow). After neurotoxic exposure (black horizontal bar), the monkeys exhibited hemiparkinsonian signs. They were divided into two groups matched according to the CRS ratings obtained 5 days after MPTP administration. At day 5, STN-AAV surgery was performed (gray arrow), corresponding to week 0 on the x-axis. Weekly CRS ratings continued until weeks 55 during post-AAV surgical period (gray horizontal bar). At each time point, mean CRS scores for animals in the GAD and GFP groups are represented by filled and open circles, respectively.)
Positron Emission Tomography
Hemispheric comparison of the whole group (Figure 2) revealed a significant effect of MPTP administration, with relative hypermetabolism in the ipsilateral (right) globus pallidus (x=12, y=12, z=−8; Zmax=7.5; P<0.001, corrected) and in the contralateral cerebellum (x=−16, y=−4, z=−6; Zmax=6.2; P<0.001, corrected). Post hoc analysis of metabolic values in each of these regions did not reveal significant differences between the two AAV treatment groups.
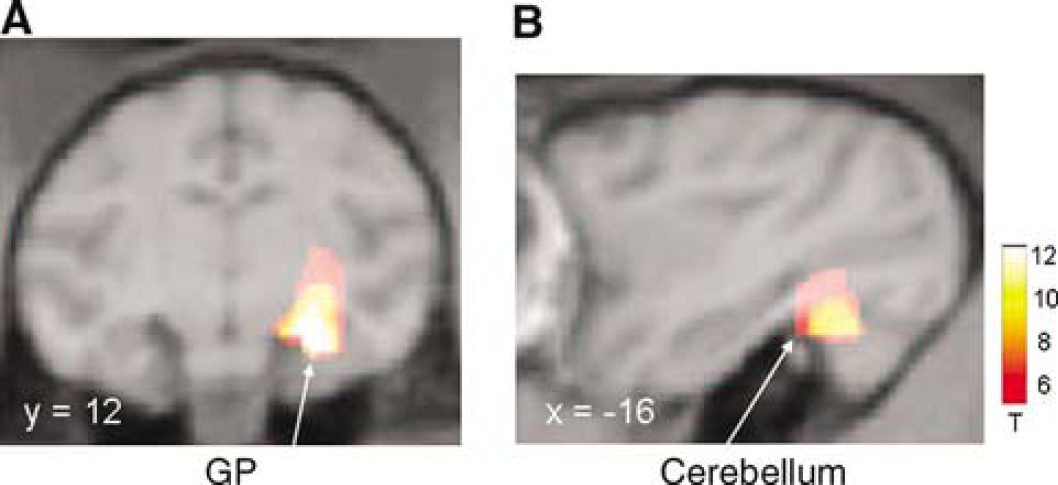
Statistical parametric maps of FDG PET data comparing MPTP-treated hemispheres with contralateral untreated hemispheres (see text). Relative metabolic increases were identified in the (
Comparison of the two AAV groups conducted blind to treatment arm (Figure 3) disclosed a relative increase with GAD treatment that was localized to the ipsilateral motor cortex (gyrus centralis anterior, x=14, y=10, z=26; Zmax=3.5, P<0.001, uncorrected). No significant metabolic reductions were present in the GAD group relative to the GFP controls. No group differences were observed in the vicinity of the STN, and there were no differences in estimates of global metabolic rate. Correlational analysis (Figure 4) disclosed a significant relationship between CRS ratings at week 55 and metabolism in the right motor cortex (x=16, y=14, z=18; T=3.6; P<0.001, uncorrected).
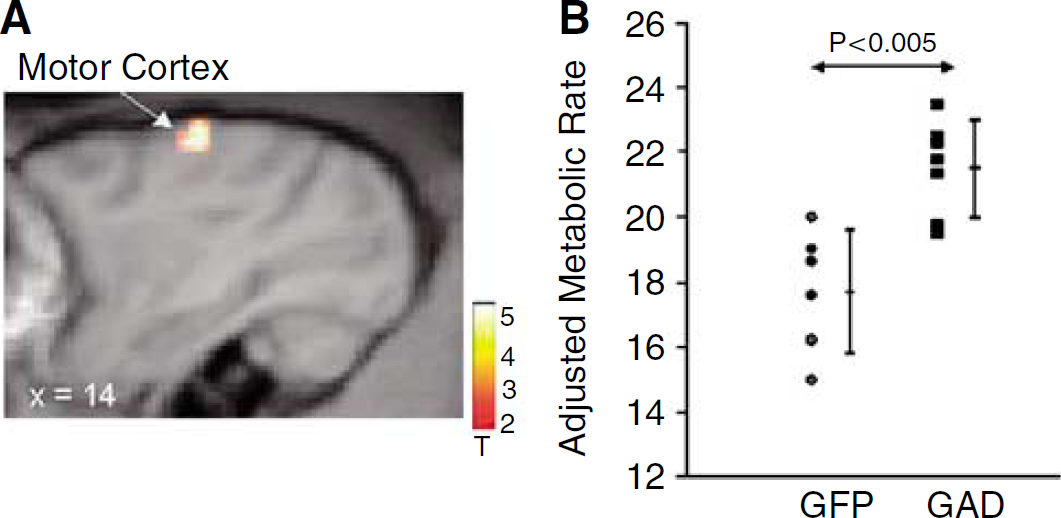
(
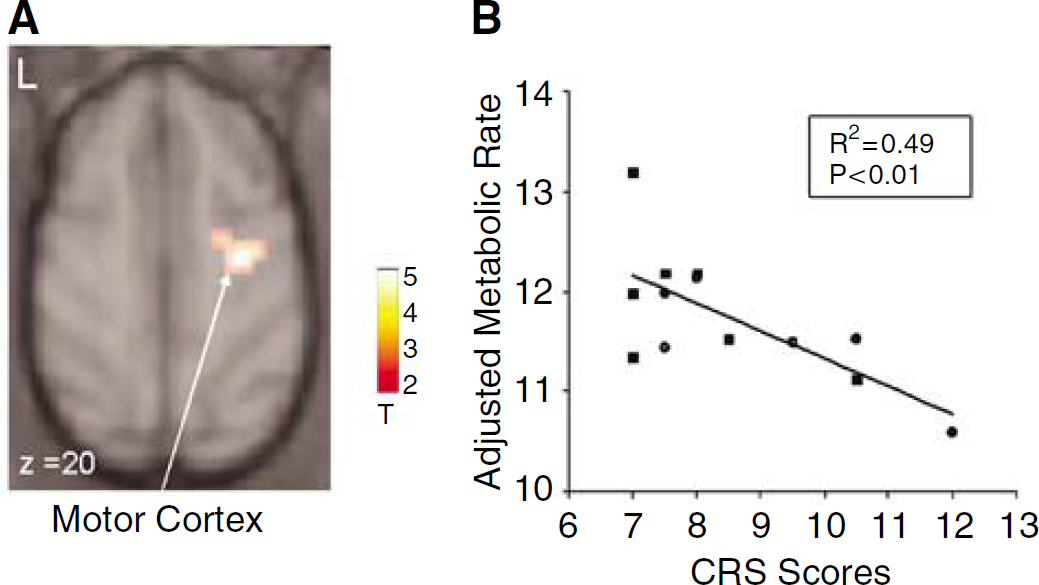
(
Morphology
At 1 year after MPTP and AAV treatment, there was evidence of GAD and GFP expression in the injected STN of the monkeys (Figure 5), with the exception of one GFP control animal. Green fluorescent protein immunoreactivity was observed in the area of the needle tract, in nearby structures such as the substantia nigra and the lenticular fascicle/cerebral peduncles (Figure 5B). While some of the staining was in cell bodies, suggestive of local spread, most of this staining appeared to be in fiber tracts extending out from the STN. Aside from a single GFP-positive cortical neuron in one animal, there was no evidence of axonal transport of virus to other distant structures. Native GAD immunoreactivity within fibers projecting to the STN was very strong, which prevented quantification of STN neurons transduced with the GAD transgene. Although GFP staining was qualitatively observed within the STN of all but one of the GFP animals, great variability was observed with regard to both cell numbers and location within the STN.
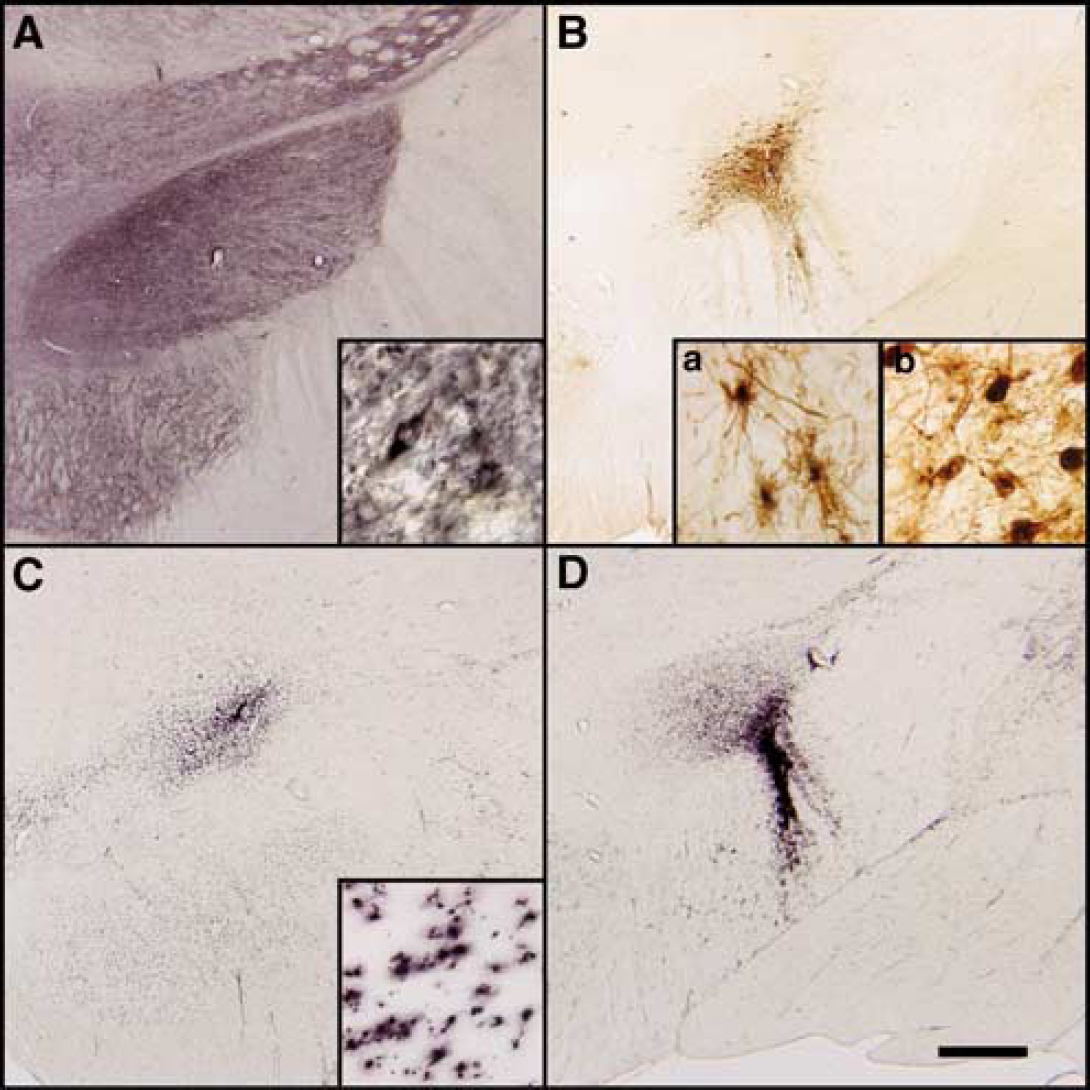
(
CD8 and CD45 immunoreactivity was virtually absent in all the animals (not shown). Mild CD68 immunoreactivity was observed in the area of injection and was most prevalent in GFP-treated monkeys (Figures 5C and 5D). Three monkeys in the GFP group exhibited an unexpected necrosis of the ipsilateral striatum and globus pallidus, associated with strong CD68 immunostaining. We did not find these animals to be influential in the metabolic differences that were noted across treatment groups (Figure 3).
Qualitative observations of brain sections stained for TH revealed unilateral loss of striatal fibers and nigral cells ipsilateral to MPTP administration. Striatal optical density confirmed asymmetrical loss of dopaminergic fibers. Comparison of lesioned and unlesioned hemispheres revealed that AAV-GFP animals had a loss of 72% in the caudate and 76.7% in the putamen, while AAV-GAD had a loss of 74% and 76.6% respectively. Similarly, stereological cell counts of TH positive nigral cells showed a profound loss of cells ipsilateral to the MPTP lesion without significant changes contralaterally. Statistical analysis did not reveal significant differences between treatment groups. This suggests that the degree of lesioning was similar between groups, with no histological evidence of recovery in either group after 1 year.
Discussion
In this study we demonstrate the feasibility of subthalamic GAD gene transfer in a non-human primate model of PD as highlighted by the detection of AAV vector product 1 year after surgery. During the course of the follow-up period, AAV-GAD proved safe, without the development of abnormal involuntary movements or any other adverse event. This therapy affected the course of MPTP-induced motor signs compared with the GFP-treated control animals, as reflected in the relative improvement of the clinical rating scores. In fact, there was a significant improvement in clinical subscores for bradykinesia, tremor, and gross motor skills that was not seen in the GFP controls, further supporting a different effect of the GAD virus on post-MPTP motor deficits compared with controls. The findings at necropsy indicated that these changes were not associated with differences in the integrity of nigrostriatal dopaminergic system as measured by TH immunoreactivity. The behavioral observations were supported by PET imaging results demonstrating that motor cortical glucose metabolism was elevated in the GAD group relative to the GFP controls. Moreover, metabolism in this region correlated significantly with contemporaneous motor ratings.
Our findings support the notion that delivery of AAV-GAD to the STN modulates the excitatory output of this structure, changing the overall neurotransmitter balance in the basal ganglia. The time course of the GAD CRS curve with a progressive improvement over several weeks is consistent with the delay in reaching peak expression found with AAV-mediated gene transfer (Herzog et al, 1997; Thomas et al, 2004). The nadir of the GAD CRS curve at week 25 suggests that either GAD transgene expression reached its peak plateau level at this time point or that the GAD gene transfer is associated with delayed secondary adaptive changes in the physiology of transduced and surrounding cells.
By contrast, the GFP control group has a different trajectory of change in CRS scores over the 1-year follow-up period. These CRS scores showed an unexpected initial decline, followed by an increase until approximately week 30. Subsequently, these values stabilized at a higher level than in the GAD group. The initial clinical improvement in the GFP group cannot be explained by recovery of the dopaminergic system because at post mortem the loss of nigral cells and dopaminergic terminals was similar for both treatment groups. Rather, this decline resulted from the substantial clinical improvement in three monkeys in the GFP control group. At necropsy, these monkeys displayed necrotic lesions in the striatum and globus pallidus, but not in the STN. The cause of these lesions is unknown, although similar areas of tissue destruction have occasionally been noted after intracarotid MPTP injection as has been reported elsewhere (Forno et al, 1995; Emborg et al, 2006). While it is surprising that this only occurred in control animals, there is no evidence to suggest that either GFP or GAD significantly altered this apparent MPTP toxicity. The equivalent number of TH positive nigral neurons and dopaminergic terminals between groups suggests that both the degree of MPTP-induced nigral lesioning as well as the stability of the lesions were no different between groups.
It is likely that the presence of striato-pallidal lesions in these three GFP animals was responsible for the absence of a significant group difference (i.e., main effect) in our data. By selectively improving outcomes in the GFP control group, the pallidal lesions behaved similarly to therapeutic pallidotomy, and worked against the primary hypothesis of relative functional benefit in the GAD treated group. Owing to sample size limitations, we could not perform a formal statistical comparison of the time course of CRS change in the GAD group with that observed in ‘true controls’, that is, the unlesioned GFP subgroup. Nonetheless, a graphical examination of the data (see figure in Supplementary Material) revealed that mean CRS scores for these control animals were consistently higher than that for the GAD group across all time points to week 55. By contrast, the three lesioned GFP monkeys, though initially exhibiting greater clinical improvement, approximated the CRS ratings of the GAD treated animals during the later stages of the study. Indeed, at week 55, the GAD group and the three lesioned GFP animals had similar degrees of clinical improvement (27% and 23% decline in CRS ratings relative to baseline), with comparatively less change (13%) in the unlesioned GFP animals. We note that excluding these animals in post hoc analysis of the imaging data did not alter the finding of relative increases in cortical metabolism in the GAD treated group (see Figure 3B).
In contrast to differences in the time course of the CRS, fine motor testing did not differ in the two AAV treatment groups. This aspect of motor performance may not have been affected by STN gene therapy. It is conceivable that fine motor improvement could be achieved by a more localized delivery of the viral vector within the STN or through the delivery of larger titers of virus. Alternatively, different mechanisms may underlie the features of motor performance captured by the two measures. Indeed, human PD patients treated with STN deep brain stimulation have been shown to have a better response to akinesia of proximal arm movement compared with fine motor finger movements of the hand (Wenzelburger et al, 2003). Consistent with this, we observed significant improvement in subscores for bradykinesia, tremor, and gross motor skills, which was not evident in the GFP control group. This suggests that these specific functions may be more sensitive to the effects of AAV-GAD.
The results of the FDG PET imaging 1 year after gene therapy show significant elevations in ipsilateral motor cortical glucose utilization in the GAD group relative to controls. We attribute this to an increase in the activity of thalamic projections to cortical motor regions, occurring with the modulation of pathological brain networks during antiparkinsonian therapy (Fukuda et al, 2001; Trošt et al, 2006; cf. Eckert and Eidelberg, 2005). This is consistent with previous findings of increased motor cortical activity in PD patients undergoing a variety of stereotaxic procedures for advanced symptoms (Eidelberg et al, 1996; Stefurak et al, 2003; Sestini et al, 2005). Metabolism in that cortical region also correlated with CRS motor ratings at 55 weeks. These findings are also in accord with a recent study of motor cortical stimulation, which resulted in improvement in CRS ratings and regional metabolism in a primate model of parkinsonism (Drouot et al, 2004).
We note that increased metabolism in the motor cortex of the GAD group could have emerged through alterations in the activity of other projections, such as those from the STN directly to this area (Hamani and Lozano, 2003). However, imparting an inhibitory GABA-ergic phenotype to glutamatergic STN-cortical projections does not necessarily change local energy requirements (Chatton et al, 2003). Moreover, this would likely change the nature of the relationship of metabolism to the motor scores. In this case, an increase in cortical inhibition subsequent to GAD therapy would be associated with a positive correlation between glucose utilization and CRS ratings, reflecting greater disability at higher rates of local metabolism. Our finding of a significant negative correlation between these measures relates increasing cortical metabolism to better performance, as observed in PD patients scanned in the rest state (Carbon and Eidelberg, 2002).
We also performed a hemispheric comparison to identify the regional effects of MPTP administration on glucose utilization in the anesthetized primate. In this acute, unilateral MPTP model, metabolic increases relative to the unlesioned hemisphere were present in the ipsilateral GP and contralateral cerebellum. Indeed, elevation in GP metabolism in MPTP primates was found using autoradiographic techniques in a similar unilateral intracarotid model (Palombo et al, 1990), and with in vivo PET imaging after chronic systemic MPTP exposure (Brownell et al, 2003). However, increased cerebellar glucose utilization was not a feature of the prior monkey studies. By contrast, the present findings of pallidal and cerebellar hypermetabolism subsequent to MPTP are consistent with FDG PET findings in PD patients (Eidelberg et al, 1994; Moeller et al, 1999). Importantly, post hoc comparison of local metabolism in these regions did not reveal significant differences between the GAD and GFP groups, indicating that the effect of the neurotoxin was similar in the two STN AAV treatment arms, in accord with the pretreatment CRS ratings and with pathological analysis.
This study was limited by several technical issues. Although the number of animals and length of follow-up here is equivalent to that of other primate gene therapy studies (Bankiewicz et al, 2000; Kordower et al, 2000), the number of animals may still be limiting, especially with the unexpected findings of necrotic lesions in the control group. A further limitation is the surgical technique, which involved MRI localization and stereotactic targeting of the STN. In humans, STN surgery is often performed with electrophysiological mapping to optimize targeting precision. This method was not performed in this study because of the extensive animal training required to perform electrophysiology and surgery in the waking condition. The far smaller size of the overall brain and the STN itself in these animals could lead to even greater variability in the site and amount of vector reaching potentially key subregions of the STN. Finally, complete information concerning the neural pathways affected by gene therapy requires a direct comparison of regional glucose utilization before and after treatment. Nonetheless, despite the limitations of the cross-sectional design that was employed, we identified a significant group difference in cortical metabolism that correlated with contemporaneous behavior descriptors. Further imaging studies incorporating a within-subject design will be needed to delineate the effects of this intervention on metabolic activity in subcortical motor regions (cf. Trošt et al, 2006).
Our observations in the primate suggest that subthalamic delivery of AAV-GAD is safe. All 13 AAV treated animals survived to the 1-year end point without adverse effects. Nonetheless, the possibility of late effects of STN gene therapy cannot be dismissed based on the results of a 1-year study of medication-naïve hemiparkinsonian non-human primates. Additionally, despite likely improvement in the function of basal ganglia—thalamocortical motor pathways after subthalamic AAV-GAD therapy, we could not assess potential treatment effects on the modulation of parallel limbic or cognitive circuits.
In conclusion, the results of these primate studies suggest that subthalamic delivery of AAV GAD is a feasible approach to the symptomatic treatment of parkinsonism. Indeed, these findings provide a useful proof-of-principle for the development of other gene therapies using AAV vectors. Further studies are warranted to optimize the delivery of viral vector, regulate gene expression, and to improve the behavioral outcome of gene therapy for PD and other disorders.
Footnotes
Acknowledgeements
The authors acknowledge the valuable contributions of Dr Jeffrey H Kordower and Dr Martin Lesser to this work. We are thankful to Dr Er-Yun Chen and Erwin Mangubat for assistance in histological processing and analysis. We also thank Mr Sherwin Su for assistance in image analysis and Ms Toni Flanagan and Ms Christine Edwards for their role in manuscript preparation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
