Abstract
Stem cell-based therapy shows great potential in stroke patients. Intra-artery infusion exhibits greater biological distribution compared to intravenous delivery. In addition, umbilical cord mesenchymal stem cells (UCMSCs) have several advantages compared with other types of stem cells. The aim of this study was to evaluate the safety and efficacy of UCMSCs delivered by a catheter to a near lesion site for treatment of an infarction in the middle cerebral artery territory. Four patients with stroke (three with ischemic and one with hemorrhagic stroke) in the middle cerebral artery territory were recruited in this study. One single dose of 2 × 107 UCMSCs was infused within 20 min via catheterization in the M1 segment of the middle cerebral artery. The safety and efficacy of this approach were assessed during the in-hospital and 6-month follow-up evaluation. The cell delivery was successfully performed in all of the patients, and no major accidents (stroke or death) were observed. Moreover, no fever or rash was reported. After cellular therapy, two of the three ischemic stroke patients demonstrated improved muscle strength. The improvement of the modified Rankin scale was observed in two patients, both of whom suffered from ischemic stroke at 90 and 180 days after the stem cell therapy. The hemorrhagic stroke patient failed to demonstrate improved muscle strength and did not amend his daily activities. Intra-artery delivery of UCMSCs via catheterization was a feasible and safe approach and may improve the neurological function of ischemic stroke patients with the middle cerebral artery territory infarcts.
Introduction
Stroke is one of the major causes of death and adult disability, which results in a large socioeconomic burden (23). Currently, the only specific therapy that is available and approved for the treatment of acute ischemic stroke is intravenous (IV) administration of the tissue plasminogen activator (rtPA), which must be administrated within 4.5 h after the onset of symptoms (11). Owing to the time limitation and potential adverse effects such as hemorrhagic transformation, only 3–8.5% of stroke patients are eligible to receive rtPA treatment (22). With current technological advances, emerging neurovascular interventions such as stent implantation and mechanical embolectomies may be alternative options for stroke treatment (13,17). However, the results of the Stenting versus Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis (SAMMPRIS) study demonstrated that the 30-day rate of stroke recurrence or death in stented patients was substantially higher than the estimated rate of 5.2% and 9.6%, respectively, based on the registry data (16). Moreover, existing lesions do not benefit from these therapies, and thus it is necessary to find other alternatives.
Previous experimental studies have shown that stem cell therapy is a promising approach for the enhancement of stroke recovery (7,8,14). Inspired by these studies, the feasibility, efficacy, and safety of stem cell therapy were evaluated in stroke patients. Neurological functions of patients improved in the absence of cell-related adverse effects in these phase I/II clinical trials (3,24). Until now, most clinical trials have used intravenously delivered autologous mesenchymal stem cells (MSCs) (3,12,15). However, the biological distribution of IV stem cells in the brain is poor (20). Moreover, several studies compared IV cell delivery and intra-artery administration and found that successful delivery of magnetically labeled bone marrow-derived mesenchymal stem cells (BMSCs) could be readily visualized with a magnetic resonance imaging (MRI) after intra-artery, but not IV, injection (5,20,29). Cellular therapy for acute myocardial infarctions demonstrated that intracoronary infusion of stem cells alone in the infarct-related vessel drove cells across vessel walls in their recruitment to the heart (21). However, autologous BMSCs have several logistical and biological limitations to their therapeutic application. Bone marrow aspiration and cell selection require additional steps for their procurement, and the number and potency of autologous cells varied among patients and declined in response to aging and disease (6).
Compared with BMSCs, umbilical cord-derived mesenchymal stem cells (UCMSCs) have some merits. UCMSCs are more plastic, grow faster (28), more biologically closer to embryonic stem cells (10), produce more cytokines (19), and have lower immunity (26).
Thus, this study applied UCMSCs via intra-artery infusion to treat stroke in the middle cerebral artery territory.
Materials and Methods
Patient Population
From May 2011 to May 2012, four patients with stroke (three ischemic and one hemorrhagic) were recruited for this study (Table 1). The eligibility criteria included (1) examination and assessment by three senior stroke neurologists; (2) a stroke within the past 3 months, which was confirmed by computed tomography (CT; Siemans, Munich, Germany) and/or MRI (Siemans); (3) a lesion localized in the middle cerebral artery territory; and (4) no significant neurological functional improvement after undergoing standard medical treatment until administration of stem cells. Patients were excluded from this study if they demonstrated uncontrolled infection, failure of one of the vital organs, intracranial neoplasms, acute myocardial infarction, pregnancy, and allergy to fetal products or iodine. All of the patients were registered in the Nanjing Stroke Registry, and a trained nurse arranged for the follow-up evaluations (18). The trial was conducted in compliance with current Good Clinical Practice standards and in accordance with the principles set forth under the Declaration of Helsinki (1989). An informed consent was obtained from each patient.
Clinical and Biochemical Characteristics of Patients

Cell Preparation
UCMSCs were provided by the Jiangsu Stem Cell Bank, Jiangsu, China. The cells were derived from fresh umbilical cords (three female and one male), which were obtained from four informed, healthy mothers after normal deliveries. The cords were maintained in preservation solution at 4°C until the stem cells were isolated. After the removal of the trophoblast, the Wharton's gum tissues were separated from the umbilical cord under sterile conditions, and the cords were cut into 1-mm3 pieces that floated in low-glucose Dulbecco's modified Eagle's medium with 10% fetal bovine serum (FBS; Gibco, Gaithersburg, MD, USA). The pieces of cord were then subsequently inoculated into T75 culture flasks (Corning, Corning, NY, USA) with serum-free DMEM at 37°C and 5% CO2. Nonadherent cells were removed after several washes with medium. The medium was replaced every 3 days after the initial plating. When the cells grew to 80% confluence, they were passaged at the ratio of 1:3. The P3-generation cells were used for cell therapy. The cells were diluted with saline and maintained in a completely mononuclear cell suspension (final volume of 20 ml).
The cell viability of the purified UCMSCs was >92% (as determined by a trypan blue assay; Invitrogen, Carlsbad, CA, USA), and each cell preparation was tested for pathogenic microorganisms, including aerobic and anaerobic bacteria (as determined by direct cultivation analysis), and hepatitis B surface antigen, hepatitis B core antibody, hepatitis C virus antibody, human immunodeficiency virus antibodies I and II, cytomegalovirus IgM, and syphilis antibody [as determined by enzyme-linked immunosorbent assays (ELISA); eBioscience, San Diego, CA, USA]. The expression of cluster of differentiation 29 (CD29), CD73, CD90, CD105, CD45, CD34, CD14, and CD79 and human leukocyte antigen (HLA)-DR expression were evaluated by flow cytometry using antibodies from eBioscience. The levels of alanine aminotransferase were determined using an automatic biochemistry analyzer (Clindiag Systems B.V.B.A, Pollare/Ninove, Belgium), and endotoxins were determined by Tachypleus amebocyte lysate analysis (Tachypleus Amebocyte Lysate Assay Company, Xiamen, China) according to the manufacturers' instructions.
Assay of Adipocyte Differentiation Ability of UCMSCs
The UCMSCs were seeded in six-well plates (Corning) at a density of 2 × 104 cells/ml. The cells were cultured in Iscove's modified Dulbecco's medium (Gibco) containing 10% FBS, 1 μM dexamethasone (Kangzheng, Hubei, China), 100 μg/ml 1-A-3-isobutyl-xanthine (Sigma-Aldrich, St. Louis, MO, USA), 50 μg/ml ascorbic acid (Gibco). Half of the medium was changed every 3 days. After 14 days, the cells were stained by oil red O (Invitrogen), and lipid droplet formation was observed under a microscope (Olympus, Tokyo, Japan).
Assay of Osteogenic Cell Differentiation Ability of UCMSCs
Cells were seeded in six-well plates at a density of 2 × 104 cells/ml and cultured in IMDM containing 10 nM dexamethasone, 50 μg/ml ascorbic acid, 7.0 mM β-glycerophosphate (Sigma-Aldrich). Half of the medium was changed every 3 days. After 14 days, Von Kossa staining (Invitrogen) was performed to detect matrix precipitation.
Cell Administration
After local anesthetization with 0.1% lidocaine (Kangzheng), right femoral arterial punctures were performed on the patients. The patient was given 3,000 U heparin (Kangzheng) for anticoagulation to obtain an activated clotting time of two to three times the baseline. Digital subtraction angiography (DSA; Siemens) was performed to visualize the intracranial vasculature prior to injection and to monitor the flow normality and vessel patency (Fig. 1C). A large inner diameter micro-catheter (SL 1018 Boston Scientific, Fremont, CA, USA) was navigated into the M1 portion of the middle cerebral artery proximal to the vessel lesions. Approximately 2 × 107 UCMSCs were diluted in 20 ml saline, and the infusion was done at a rate of approximately 1 ml/min. After the infusion, 10 ml saline was used to wash the catheter, and 20 min later a second angiography was performed to assess for the presence of a vessel occlusion (Figs. 1D and 2).

The digital subtraction arteriography of the cerebral vessels of Patient 3. (A) The posterior–anterior view and (B) the posterior–basal view of cerebral vessels 7 days after the onset of symptoms. There was a complete occlusion in the M2 segment of the left middle cerebral artery. (C) Before injection of stem cells. (D) Twenty minutes after the injection of stem cells. Compared with the before (C) and after time (D), there was no new blockage of cerebral vessels. Scale bar: 1 cm.
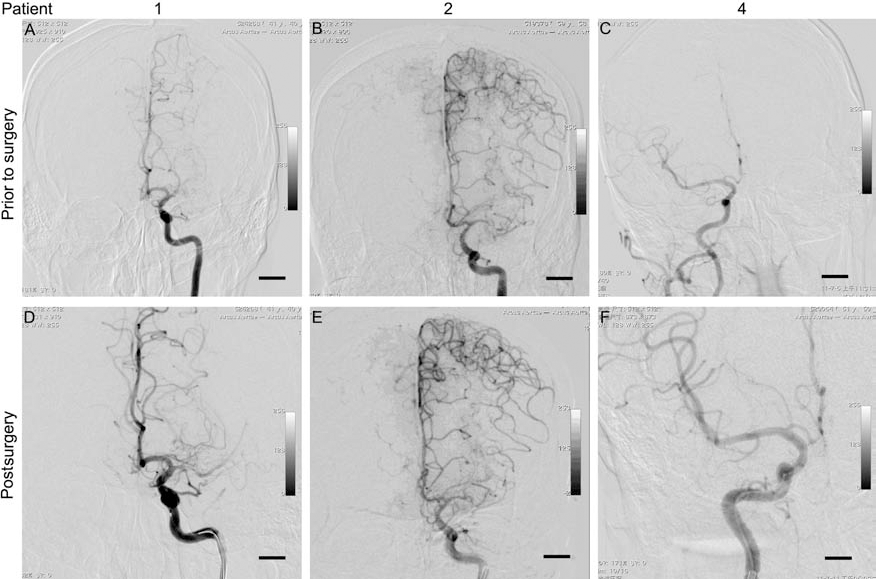
The digital subtraction arteriography of cerebral vessels of Patients 1, 2, and 4. (A–C) Prior to the surgery; (D–F) 20 min after the surgery. Scale bar: 1 cm. No new blockage of cerebral vessels was evident.
Patient Assessment
Within 24 h of admission, the baseline clinical status and laboratory assessments were obtained including a complete blood count, blood biochemical analysis, blood coagulation function test, hemorheological test, serum anti-Treponema and anti-HIV antibody titer, urinalysis, and stool occult blood test. An electrocardiography (Xutian, Shenzhen, China) and chest X-ray photography (GE Healthcare, Wauwatosa, WI, USA) were also included. If the patient did not have the imaging data upon admission, a CT and/or MRI were also recommended. After the stem cell therapy, any uncomfortable complaints such as a fever, rash, and weakness were recorded. Before discharge, the patients' clinical status was reassessed, and Patient 3 had undergone a head MRI to exclude the presence of new lesions. During the 3- and 6-month follow-up evaluations, the clinical statuses were assessed at the outpatient evaluation.
Results
Patient Profile
The baseline demographic and characteristics of the patients enrolled in this study are presented in Table 1. All of the patients were male and relatively young (younger than 60 years old). Patients 1–3 were ischemic stroke patients, and Patient 4 presented with a hemorrhagic stroke. All of the ischemic stroke patients exhibited high-risk issues, such as high plasma glucose (2/3), hyperlipidemia (1/3), hypertension (2/3), smoking (3/3), drinking (2/3), and obstructive sleep apnea syndrome (1/3). The hemorrhagic stroke patient did not have any high-risk issues. The angiography revealed that Patient 1 had a complete occlusion in the M1 segment of left middle cerebral artery and 20% stenosis in the left internal carotid artery; Patient 2 had 99% stenosis in the M1 segment of the left middle cerebral artery, 45% stenosis in the common carotid artery, and 50% stenosis at the origin of the left vertebral artery; Patient 3 presented with a complete occlusion in the M2 segment of the left middle cerebral artery; and Patient 4 had no vessel lesions. All of the ischemic stroke patients received standard medical treatments, and Patient 1 showed resistance to clopidogel treatment. An intracranial stent placement was scheduled in Patient 1; however, it was reconsidered because of the low benefit/risk ratio of the surgery. Both Patient 1 and Patient 2 exhibited multiple vessel lesions, and blood drainage was performed on the hemorrhagic stroke patient (Patient 4).
Safety of UCMSCs Delivered Through Middle Cerebral Artery
The expression of CD29, CD73, CD90, and CD105 by the UCMSCs was higher than 95%. In contrast, CD45, CD34, CD14, CD79, and HLA-DR expression was lower than 2%. The levels of alanine aminotransferase and endotoxins were strictly controlled within 40 IU/L and 5 EU, respectively. The capacity of the UCMSCs to differentiate along adipogenic (Fig. 3A) and osteogenic lineages (Fig. 3B) was confirmed. The potential risks of UCMSCs delivered through the middle cerebral artery are derived from two sources: neurointervention technology and stem cells. We evaluated the vessels by angiography 20 min after stem cell delivery and found no new artery occlusions (Figs. 1D and 2). Before discharge, an MRI was reviewed, which showed no novel ischemic lesions or intracranial hemorrhage in the brain (Fig. 4). At the 6-month follow-up evaluation, there were no fatal complications observed such as a major stroke or death. In addition, no transient ischemic attacks occurred within 180 days of the surgery. Moreover, no fevers or rashes were observed during inhospital and follow-up evaluation.

Differentiating capacity of UCMSCs. After 14 days of culture, the UCMSCs were differentiated along adipogenic (A) stained by oil red O and osteogenic lineages and (B) stained by Von Kossa method. Scale bar: 100 μm.
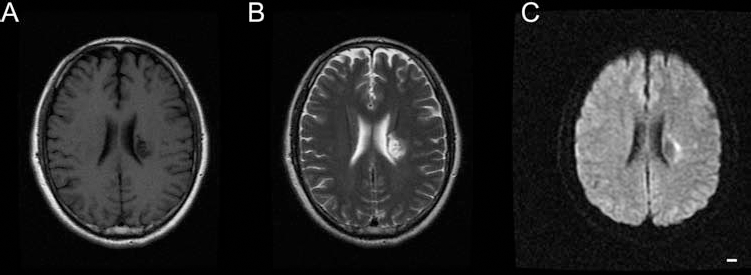
The head magnetic resonance images of Patient 3 before discharge. There was no new infarction in the brain. (A) T1 fluid-attenuated inversion recovery (FLAIR); (B) T2; (C) diffusion-weighted image (DWI). Scale bar: 1 cm.
Efficiency of UCMSCs in Stroke Treatment
Three of four patients presented with muscle weakness prior to the cellular therapy and could not walk independently. After cellular therapy, two of the three patients showed improvement in muscle strength. In detail, Patients 2 and 3 demonstrated improved muscle strength of the upper extremities. Patient 3 also demonstrated improvement of the lower extremities. The modified Rankin scale (mRS) is a commonly used scale for measuring the degree of disability or dependence in the daily activities of stroke patients. Improvement in the mRS scale occurred in two patients, both of whom suffered from ischemic stroke at 90 and 180 days after stem cell therapy. Moreover, the hemorrhagic stroke patient (Patient 4) failed to amend his daily activities (Table 2).
Modified Rankin Score

Discussion
In this small pilot study, we delivered stem cells through perilesioned vessels to treat stroke in the middle cerebral artery territory and found that this approach was safe and provided potential treatment benefits.
Stroke caused by lesions of the middle cerebral artery leads to severe disability. All of the four patients enrolled in this trial were disabled and could not function independently. Three of the patients could not walk, and two of them had difficulty in understanding and speech. The patients were all male and relatively young, which suggest that they may have more family responsibilities, which may indicate their desire to try new approaches. However, standard medical treatment showed less efficiency, and intracranial stenting has a yet undetermined role. Thus, we used stem cells as a treatment option.
Our data showed that the delivery of allogeneic stem cells in the M1 segment of the middle cerebral artery was safe. One major concern with this approach may be that stem cells in the artery can form microemboli, which may induce additional ischemic strokes (27). Thus, we first performed an angiography 20 min after stem cell delivery, which did not reveal any new blockage of blood flow. Although the concern that stem cells placed in the artery may form microemboli is plausible, there is no published data demonstrating that ischemic stroke is caused by microemboli formed from stem cells. To further decrease this risk, we used a relatively small number of stem cells (2 × 107) and diluted the stem cells from 2 ml into 20 ml. Based on mean body mass, 1 × 108 cells/patient is the human dose equivalent to the dose in a rat model (1 × 105 – 3 × 106) (7). Preclinical studies have shown that administration of this number of stem cells is safe (7,8,14). In addition, the head MRI performed prior to discharge did not reveal any new infarcts, and during the follow-up evaluation, there were no emerging local neurological signs. Second, the use of UCMSCs as allogeneic stem cells may induce an immunological response. However, we did not detect any fevers, rashes, or other obvious immune response. Our results were consistent with a previous report, which showed that UCMSCs do not express the HLA-DR and costimulatory molecules CD80 and CD86, which are required for T-cell activation (26).
In this study, our results showed that stem cell therapy improved the neurological function of stroke patients. Ischemic stroke patients showed enhanced improvement compare with hemorrhagic stroke patient, which was consistent with previous studies. Bang et al. used culture-expanded autologous MSCs in patients with ischemic stroke and found that the scores of Bathel index and mRS were improved without any detectable adverse effects (3). In another study, labeled bone marrow mononuclear cells (BMMCs) with 99mTc were injected into a patient with a cerebral infarct within the left middle cerebral artery territory via a catheter, and the patient was scanned with single-photon emission computed tomography (SPECT). The results showed that the uptake and retention of the labeled BMMCs in the territory of the left middle cerebral artery lasted up to 48 h (4). The same group investigated the feasibility of detecting the distribution and homing of BMMCs in patients with chronic ischemic stroke. They provided data indicating cell homing in patient brain at 2 h, and the remaining uptake was mainly distributed to the liver, lungs, spleen, kidneys, and bladder. In acute myocardial infarctions, stem cell-based therapy also demonstrates great potential. In a clinical trial, 20 patients with acute myocardial infarction received intracoronary infusion of stem cells into the infarct artery. Transplantation of progenitor cells was associated with a significant increase in the global left ventricular ejection fraction, which improved the regional wall motion in the infarct zone and profoundly reduced end-systolic left ventricular volumes at the 4-month follow-up evaluation (2). These results were confirmed by other clinical trials in the following years (1,25,30). Taken together, all of these studies suggest that intra-artery stem cells used to treat stroke improved the function of the patients.
In addition to intra-arterial injection, there are many routes to deliver stem cells to the brain, such as direct transplantation, intracerebral parenchyma, intrathecal, IV or intra-artery delivery. Comparatively speaking, intraarterial delivery has many more advantages compared to other methods. Although direct transplantation was the first method developed, it requires transplantation of grafts or stem cells into the desired location (24) and may result in excess injury. One major disadvantage of delivery via an intraparachymal injection is that stem cells scarcely move beyond the injection site, although the stem cells are delivered directly to the brain (9). Intrathecal administration is performed by a lumbar puncture and delivers the cells directly into the cerebrospinal fluid (CSF); however, the CSF may wash away these stem cells. IV delivery of stem cells is the most widely used method because of its limited complications (15), but the biological distribution of stem cells in the brain is poor (20). Intra-arterial administration uses catheterization to implant stem cells into the carotid artery or Willis circle as performed in this study (5). The MSCs labeled with superparamagnetic iron oxide were delivered to animal models of ischemic stroke, and the cell delivery was examined using an MRI. Successful delivery of magnetically labeled MSCs was readily visualized with an MRI after intra-artery, but not IV, injection. These data indicated that intra-arterial administration has a greater biological distribution compared to IV delivery (29).
In our study, we used the UCMSCs to treat stroke compared to other clinical trials, which used BMSCs. As discussed previously, there are some merits in using fetal MSCs compared with BMSCs. First, MSCs derived from neonatal and midgestational fetal tissues are more plastic and grow faster (28). To treat patients in the acute phase of stroke, many studies just isolated stem cells from bone marrow because this approach allowed physicians to get stem cells within 24 h. However, one disadvantage is that the total number of stem cells in the bone marrow is limited. Others have expanded BMSCs in vitro to obtain 108 cells/ patient, although this requires several weeks. The use of standard UCMSCs may overcome these challenges. First, the anticipated number of stem cells could be obtained within 4 h. Second, UCMSCs have been isolated earlier in development from first-trimester blood and hemopoietic organs, and UCMSCs are biologically closer to embryonic stem cells and may thus have advantages over adult bone marrow-derived MSCs (10). Third, UCMSCs produce many cytokines compared to BMSCs (19), which may help repair damaged tissue. Finally, UCMSCs exhibit lower immunological responses because they do not express HLA-DR and costimulatory molecules CD80 and CD86, which are required for T-cell activation (26).
The major limitations of this study include the limited number of enrolled patients and the short duration of the follow-up evaluation, which was only half of a year. In future studies, a larger number of patients are needed to investigate whether intra-artery delivery of UCMSCs improves the neurological function in ischemic stroke patients. Moreover, future studies should use labeled UCMSCs to determine its ability to cross the blood–brain barrier and understand the underlying mechanisms of UCMSCs in ischemic stroke.
Conclusion
Taken together, UCMSCs delivered via catheterization into the Willis circle was a feasible and safe approach, which improved the neurological function of patients with ischemic stroke in the middle cerebral artery territory.
Footnotes
Acknowledgments
This study was supported financially by the National Science Foundation of China (31200817, 81220108008, and 31171016) and Jiangsu Province's Key Medical Center. The authors declare no conflicts of interest.
