Abstract
Increased pressure due to postischemic edema aggravates renal ischemia-reperfusion injury (IRI). Prophylactic surgical decompression using microcapsulotomy improves kidney dysfunction after IRI. Supportive cell therapy in combination with microcapsulotomy might act synergistically protecting kidney function against IRI. The effects of therapeutic endothelial cell application alone and in combination with microcapsulotomy were investigated in a xenogenic murine model of 45-min warm renal ischemia. Renal function and perfusion were determined before as well as 2 and 18 days postischemia by 99mTc-MAG3 imaging and laser Doppler. Histological analysis included H&E stains and immunohistology for endothelial marker MECA-32, cell proliferation marker Ki-67, and macrophage marker F4/80. Histomorphological changes were quantified using a tubular injury score. Ischemia of 45 min led to severe tissue damage and a significant decrease in renal function and perfusion. Microcapsulotomy and cell therapy alone had no significant effect on renal function, while only surgical decompression significantly increased blood flow in ischemic kidneys. However, the combination of both microcapsulotomy and cell therapy significantly improved kidney function and perfusion. Combination therapy significantly reduced morphological injury of ischemic kidneys as determined by a tubular injury score and MECA-32 staining. Macrophage infiltration evidenced by F4/80 staining was significantly reduced. The Ki-67 proliferation index was increased, suggesting a regenerative environment. While microcapsulotomy and cell therapy alone have limited effect on renal recovery after IRI, combination therapy showed synergistic improvement of renal function, perfusion, and structural damage. Microcapsulotomy may create a permissive environment for cell therapy to work.
Keywords
Introduction
The reduction in renal transplantation rejection rates achieved over recent decades has not translated into a commensurate improvement in long-term allograft survival (8). Therefore, increasing attention is being focused on the etiology of late graft loss. It becomes more and more clear that ischemia-reperfusion injury during transplantation not only impacts on acute epithelial and endothelial renal injury but also predisposes to long-term graft failure (4). In the last two decades, reams of substances were shown to reduce acute ischemia-reperfusion injury in various experimental models and were applied in clinical practice on an occasional basis. More recently, cell-based therapy strategies involving endothelial cells, endothelial progenitor cells, and mesenchymal stem cells (MSCs) were successfully developed in animal models, but they have not proven effective in clinical practice yet (6,23,24).
We have recently demonstrated that increased pressure due to postischemic inflammatory edema may aggravate renal ischemia-reperfusion injury and that prophylactic surgical decompression using microcapsulotomy improves kidney dysfunction (13). Since cell therapies as well as surgical decompression have been shown to protect the kidney against ischemic injury, we speculated that cell therapy with endothelial cells in combination with surgical microcapsulotomy might work synergistically.
Materials and Methods
Endothelial Cells for Therapeutic Application
Human umbilical vein endothelial cells (HUVECs) were obtained from American Type Culture Collection (ATCC, Manassas, VA, USA) and cultured according to the product information sheet using Vascular Cell Basal Medium-2 (Lonza, Basel, Switzerland) supplemented with 10% fetal calf serum (FCS; Lonza) and endothelial cell growth kit components including vascular endothelial growth factor (VEGF; Lonza).
Characterization of HUVECs Using Flow Cytometry
HUVECs were incubated for 15 min with Flebogamma (Grifols, Langen, Germany) to prevent unspecific binding of antibodies. Then, cells were stained using antibodies against biotinylated vascular endothelial growth factor receptor 2 (VEGFR2; Relia Tech, Braunschweig, Germany) with streptavidin–allophycocyanin (APC; eBioscience, San Diego, CA, USA) as secondary antibody and cluster of differentiation 31-phycoerythrin (CD31-PE; MACS Miltenyi, Bergisch Gladbach, Germany). Isotypeidentical antibodies served as controls (BD Biosciences, Franklin Lakes, NJ, USA). Each analysis included 50,000 gated events. The threshold was set according to the controls using isotype-identical antibodies. All samples were examined using a FACSCalibur cytometer, and data were analyzed with CELLQuest Pro software (both BD Biosciences).
Animals
Male Balb/C nude (nu/nu) mice, aged 10–12 weeks and weighing 21–24 g, were purchased from Charles River Laboratories (Sulzfeld, Germany). Animals were fed a standard diet and allowed free access to water. All animal experiments were conducted in accordance with institutional guidelines and were approved by the Administrative Panel on Laboratory Animal Care (Government of Upper Bavaria, Germany).
Renal Ischemia-Reperfusion Injury Model
Mice were anesthetized by intraperitoneal injection of a combination of 0.05 mg/kg fentanyl, 0.5 mg/kg medetomidine (Pfizer, Berlin, Germany), and 5 mg/kg midazolam (Ratiopharm, Ulm, Germany) and placed on a heated surgical pad to keep the body temperature constant. The right kidney was exposed through median abdominal incision, and mice were subjected to ischemia by clamping the renal pedicle with a nontraumatic microaneurysm clamp (Braun, Melsungen, Germany), which was removed after 45 min. The incision was closed with a 5–0 suture (Ethicon, Livingston, Scotland, UK) and surgical staples (Hugo Sachs GmbH, March, Germany). Postoperatively, anesthesia was antagonized by SC injection of a combination of 2.5 mg/kg atipamezole (Pfizer), 0.5 mg/ kg flumazenil (Delta Select, Pfullingen, Germany), and 1.2 mg/kg naloxone (Inresa, Bartenheim, France). Mice were randomly divided into four groups receiving either vehicle, microcapsulotomy, HUVECs, or combination therapy. Microcapsulotomy was performed at the lower pole of the kidney by a standardized 0.3 mm (30 gauge) needle (Braun) puncture as previously described (13). The kidney capsule was slightly lifted using a blunt forceps to prevent parenchymal injury. The group size was n = 6 in each group and n = 5 in the control group due to perioperative loss.
Cell Therapy
For cell-based treatment, HUVECs (106/0.2 ml) of an early passage (passage 5) were intravenously injected 24 h after release of the clamp. We selected 24 h as the optimal time point for cell injection following ischemic injury as VEGF protein levels were previously shown to peak after this period and subsequently decline back to baseline levels within 3–4 days (30).
Determination of Renal Function
Kidney function was measured by scintigraphy using 99mTechnetium-mercapto-acetyl-triglycine (99mTc-MAG3), a radioactive compound predominantly excreted by tubular secretion (Technescan MAG3, Covidien, Neustadt/ Donau, Germany). Scans were performed according to a modified protocol from a previous study (26). After hydration with sterile saline and induction of anesthesia using a combination of 0.05 mg/kg fentanyl, 0.5 mg/kg medetomidine, and 5 mg/kg midazolam mice were subjected to whole-body scintigraphy in a triple-headed gamma camera (Philips Prism 3000 XP, Cleveland, OH, USA). The detector head was equipped with a low-energy highresolution (LEHR) collimator, but only one head was used. Intravenous injection of a standardized dose of ~3.7 × 107 Bq per mouse and acquisition in a dynamic planar technique were simultaneously started with a frame time of 5 s and a total scan time of 10 min. The image acquisition magnification was set to four times. 99mTc-MAG3 imaging was performed 4 days before as well as 2 days and 18 days after induction of ischemia-reperfusion injury. After data acquisition, anesthesia was antagonized by SC injection of a combination of 2.5 mg/kg atipamezole, 0.5 mg/kg flumazenil, and 1.2 mg/kg naloxone.
Image Analysis
Image files were analyzed using Hermes Dynamic Study Display software V4.0 (Hermes Gold V2.10, Hermes Medical Solutions, Stockholm/London, Sweden/UK). After standard manual region of interest (ROI) determination of the whole body, kidneys including background regions, left ventricle as well as the site of injection data were exported to Microsoft Excel (Redmond, WA, USA). The ratio of background corrected kidney ROI and the injection site corrected whole-body ROI led to the respective renogram values represented as percentage of injected dose (%ID). The renograms including the peak of the curves and also the flattening of the uptake slope represent a mixture of renal perfusion (i.e., unextracted tracer) and renal extracted tracer rather than precise markers of renal function. This is the reason why we introduced the concept of the fractional uptake rate (FUR). The fractional uptake rate (FUR) was calculated from the scintigraphy data as a measure of renal clearance as previously described (13,14,27).
Renal Perfusion Measurements
After final 99mTc-MAG3 imaging on day 18, blood flow of the ischemic (right) and nonischemic (left) kidney was assessed using a laser Doppler blood flow analyzer (O2C, Lea Medizintechnik, Giessen, Germany) with a microprobe of 2-mm tissue penetration (LFM-2, Lea Medizintechnik). Relative perfusion was obtained as the ratio of blood flow in the ischemic and the healthy kidney.
Histological and Immunohistochemical Analyses
Tissue specimens of the ischemic right and the contralateral nonischemic kidney were collected for histological analysis. After paraffin embedding, histological sections were stained with hematoxylin and eosin (H&E) and immunohistology for cell proliferation marker Ki-67 and endothelial marker mouse endothelial cell antigen 32 [MECA-32; plasmalemma vesicle-associated protein (PLVAP)]. Structural damage was assessed by histological analysis according to a semiquantitative tubular injury score (TIS) (32). For each mouse, at least three high-power fields (400× magnification) were examined. The percentage of tubules that displayed cellular necrosis, loss of brush border, cast formation, vacuolization, and tubular atrophy/dilation were scored as follows: 0, none; 1, <11%; 2, 11–25%; 3, 26–45%; 4, 46–75%; and 5, >75%. Proliferating cells were identified by double staining of nuclei for fluorescein isothiocyanate (FITC)-labeled Ki-67 [Ki-67 purified (Biomeda, Foster City, CA, USA) + secondary polyvalent biotinylated antibody (Zytomed, Berlin, Germany) + streptavidin–FITC (BD Biosciences)] and 4′,6-diamidino-2-phenylindole (DAPI, Vector Laboratories, Burlingame, CA, USA). Images were obtained by fluorescence microscopy (Axioskop 40, Zeiss, Goettingen, Germany) and quantified as Ki-67-positive cells per high-power field. Endothelial cells were stained using horseradish peroxidase-labeled anti-MECA-32 (Developmental Studies Hybridoma Bank, Iowa City, IA, USA). Samples were developed using 3,3′-diaminobenzidine (Sigma Aldrich, St. Louis, MO, USA). Development using 3,3′-diaminobenzidine produces an easily observable dark brown/black color. MECA-32-positive capillary cross sections per high-power field were determined as previously described (22). For the characterization of the inflammatory infiltrate histological sections were stained for macrophage marker F4/80 using horseradish-peroxidase-labeled anti-F4/80 (Serotec, Kidlington, UK) and developed using 3-amino-9-ethylcarbazole (Sigma Aldrich). Quantitative image analysis of histological sections at 50x magnification was performed using ImageJ (ImageJ 1.43u, National Institutes of Health, Bethesda, MD, USA) based on binary images after splitting of color channels for the determination of the labeled area fraction.
Statistical Analysis
Continuous variables were tested for normal distribution using the Kolmogorov-Smirnov test and for equality of variances using Levene's test. For multigroup comparisons, a one-way ANOVA was performed followed by Bonferroni post hoc analysis. Comparisons of two groups were performed using the paired or unpaired Student's t test (two-sided), whichever was appropriate. Our primary hypothesis was that combined therapy using HUVECs and microcapsulotomy preserves kidney function following renal ischemia-reperfusion injury measured by FUR. To test this hypothesis, FUR values were compared between preischemic and postischemic kidneys (day 18) by paired t test. Data are expressed as mean±SEM. Values of p < 0.05 were considered statistically significant.
Results
HUVEC Surface Marker Expression
First, we assessed surface expression of vascular endothelial growth factor receptor 2 (VEGFR2) and endothelial marker CD31 on HUVECs by flow cytometry. Expression of VEGFR2, which enables VEGF-mediated migration to the site of ischemia amounted to 61.8+9.0%. Consistent with their status of committed endothelial differentiation 98.4 + 0.6% HUVECs expressed the marker CD31. The percentage of cells exhibiting surface expression of both CD31 and VEGFR2 was 60.8 ± 8.5% (Fig. 1).

FACS analysis of surface marker expression in human umbilical vein endothelial cells (HUVECs). (A) HUVECs were examined for the expression of the surface marker human cluster of differentiation 31 (hCD31) and human vascular endothelial growth factor receptor 2 (hVEGFR2) during early passages. In the present fluorescence-activated cell sorting (FACS) analysis, the percentage of double-positive-stained cells was 54.96%. (B) Almost all HUVECs expressed endothelial marker hCD31 (98.4±0.6%). HUVECs show a robust expression of hVEGFR2 (61.8±9.0%), all of which the majority exhibited double positive characteristics (60.8±8.5%). Values are depicted as mean value±SEM. APC, allophycocyanin; PE, phycoerythrin.
Renogram Characteristics
Kidneys subjected to 45-min ischemia exhibited reduced maximum height and flattening of the uptake slope in the background-corrected renal curves as compared to baseline (Fig. 2A). Renograms of ischemic kidneys treated with monotherapy using microcapsulotomy (Fig. 2B) or HUVECs (Fig. 2C) showed similar changes. By contrast, combination therapy using microcapsulotomy and cell-based treatment resulted in increased maximum height and raising of the uptake slope at day 18 after initial flattening on day 2 (Fig. 2D).

Renograms of ischemia and therapeutic treatments. Background-subtracted curves are shown for ischemic kidneys treated (A) with vehicle, (B) microcapsulotomy (mc), (C) HUVECs, and (D) combination therapy. Values of the renal curve are depicted as mean value±SEM for each time point. Pre, baseline renal function before surgery; %ID, percent of injected dose.
Combined Microcapsulotomy and Cell Therapy Enables Recovery of Renal Clearance Following Renal Ischemia-Reperfusion Injury
Unilateral ischemia of 45 min resulted in a progressive decrease in FUR 2 days (66.8±18.1%, p = 0.141) and 18 days (45.9±11.5%, p = 0.009) after ischemia in comparison to baseline (Fig. 3A). Neither microcapsulotomy (80.8±20.8%, p = 0.201) nor cell-based therapy (27.3±3.3%, p = 0.184) alone conferred significant recovery of FUR 18 days postischemia as compared to control. Most notably, combination therapy using microcapsulotomy and HUVECs led to significant amelioration of FUR on day 18 (121.2±23.7%, p = 0.024 vs. control).

The fractional uptake rate (FUR) of ischemic and contralateral nonischemic kidneys in a murine model of unilateral renal ischemia-reperfusion injury. (A) Renal ischemia-reperfusion injury led to a gradual decrease in FUR in the control (p < 0.05 day 18 vs. baseline) and the HUVEC-treated group (p < 0.05 day 18 vs. baseline). In ischemic kidneys, no significant improvement of FUR was observed using microcapsulotomy alone, while combination therapy consisting of HUVECs and microcapsulotomy was associated with complete recovery of FUR by day 18 (n.s. vs. baseline). (B) FUR of the contralateral nonischemic kidney remained unchanged in the control and the cell-treated group during follow-up (n.s. day 18 vs. baseline), while microcapsulotomy of the ischemic kidney enabled compensatory FUR increase in the contralateral nonischemic kidney (p < 0.05 day 18 vs. baseline). Data are means±SEM. from n = 6 mice in each group. R, right (ischemic); L, left (contralateral).
Unilateral ischemia of 45 min had no effect on contralateral nonischemic kidneys 18 days after injury (FUR 85.9±13.6%, p = 0.358). Interestingly, while HUVEC therapy alone did not alter renal clearance in contralateral nonischemic kidneys 18 days postischemia (FUR 119.5±30.7%, p = 0.552), microcapsulotomy therapy enabled a compensatory increase in FUR (307.3±96%, p = 0.083), which did not quite reach statistical significance. This effect could also be observed in combination with cell therapy (245.9±39.3%, p = 0.014) where it reached statistical significance (Fig. 3B).
Combined Treatment Improves Kidney Blood Flow After Ischemia-Reperfusion Injury
To demonstrate the potentially underlying mechanisms of our findings, we investigated the regenerative capacity of vascular function in the murine in vivo model by measuring renal blood flow with laser Doppler (Fig. 4). Vascular perfusion showed insufficient restoration after ischemia and was significantly decreased in comparison to nonischemic kidneys at the end point (67.7±3.9%, p < 0.01). The administration of HUVECs alone did not lead to a significant improvement of kidney perfusion following renal ischemia-reperfusion injury (56.6±7.5%, p = 0.293 vs. control). Surgical pressure relief using microcapsulotomy alone led to a significant increase in renal blood flow (82.3±4.0%, p = 0.037). Combined cell and decompression therapy resulted in an even greater enhancement of kidney perfusion recovery (98.5±8.6%, p = 0.020).
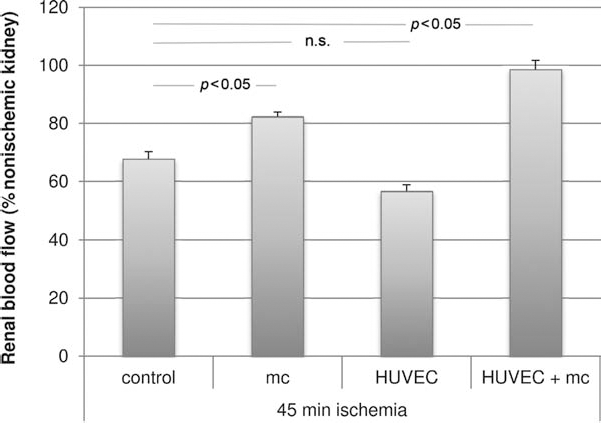
Therapeutically administered HUVECs enhance renal perfusion after ischemia-reperfusion injury only in the presence of microcapsulotomy. Compared to the use of vehicle, the administration of HUVECs following renal ischemia-reperfusion injury did not result in a significant increase in renal blood flow (n.s. vs. control). While surgical pressure relief alone led to a moderate improvement of renal blood flow as compared to control (p < 0.05 vs. control), combined cell-based and decompression therapy conferred a considerable enhancement of kidney perfusion (p < 0.05 vs. control). Data are presented as percentage of the nonischemic kidney (% nonischemic kidney) and depicted as means±SEM.
Histological Analysis Confirms Renoprotective Effects of Combination Therapy
Ischemic kidneys exhibited severe tissue damage as evidenced by tubular atrophy, necrosis, and dilatation, inflammatory infiltration, loss of brush borders, protein cylinders as well as cytoplasmic degeneration in contrast to healthy kidneys (Fig. 5A). Cell-based treatment reduced histological signs of ischemia-reperfusion injury in that tubular necrosis, atrophy, and dilatation were only detectable in scattered areas. However, abundant protein cylinders were still found. Surgical decompression alone preserved renal parenchyma even further and only isolated spots of atrophic changes and cellular infiltration were visible. Strikingly, combined therapy effectively prevented parenchymal atrophy, necrosis, and fibrosis in schemic kidneys. Glomerular structures were well preserved in any group as expected.

Histological analysis confirms renoprotective effects of combination therapy. (A) In reference to the healthy kidney, ischemic kidneys in the control group exhibited severe tissue damage particularly of tubular structures. Renal tissue showed reduced incidence of necrosis and atrophy in ischemic kidneys treated with microcapsulotomy and/or HUVECs. Kidneys exposed to ischemia-reperfusion injury were virtually rescued by combined therapy using HUVECs and surgical pressure relief. (B) The tubular injury score (TIS) correlates with the functional findings. Considerable tissue damage was seen in the control group (TIS 16.8±0.4). Ischemic kidneys showed significant improvement of tubular damage when treated with microcapsulotomy alone (TIS 9.1±1.0, p < 0.05 vs. control), HUVECs alone (TIS 11.9±1.1, p < 0.05 vs. control), and combined cell and decompression (TIS 4.8±0.6, p < 0.01 vs. control). Hematoxylin and eosin (H&E) stain. Original magnification: 200×. Scale bar: 100 μm.
Tubular Injury Score
Semiquantitative assessment of the tubular injury score (TIS) at the end point (Fig. 5B) demonstrated considerable tubular damage in the control group (TIS 16.8±0.4), whereas a significant decrease in tubular injury was seen in ischemic kidneys treated with microcapsulotomy alone (TIS 9.1±1.0, p < 0.05 vs. control) and HUVECs alone (TIS 11.9±1.1, p < 0.05 vs. control). Importantly, ischemic kidneys receiving combined cell and decompression therapy exhibited an even more pronounced improvement of TIS (TIS 4.8±0.6, p < 0.01 vs. control).
Cell Proliferation Marker Ki-67
Ki-67 staining (Fig. 6) at the end point showed a significantly higher proliferation rate after ischemiareperfusion injury in animals treated with microcapsulotomy (5.6±0.2 cells per high-power field, p < 0.01 vs. control) or HUVECs (6.0±0.7 cells per high-power field, p < 0.01 vs. control) in comparison to control kidneys (1.3±0.2 cells per high-power field). Combination therapy exhibited the highest proliferation rates (9.1±1.8 cells per high-power field, p < 0.01 vs. control).

Immunohistological analysis for cell proliferation marker Ki-67. (A) Nuclei were stained using 4,6-diamidino-2-phenylin-dole (DAPI), Ki-67 was labeled with fluorescein isothiocyanate (FITC). (B) Staining for Ki-67 showed increased numbers of proliferating cells in ischemic kidneys treated with microcapsulotomy and/or HUVECs (p < 0.01 vs. control). Original magnification: 400×. Scale bar: 50 μm.
Endothelial Marker MECA-32
The peritubular capillary network in ischemic kidneys of the control group was considerably diminished as compared to nonischemic controls (Fig. 7A). Ischemic kidneys treated with monotherapy using decompression and cell-based treatment, respectively, showed rarefaction of peritubular capillaries. MECA-32 staining in peritubular capillaries was largely preserved in animals receiving a combination therapy of surgical decompression and cell therapy (Fig. 7B).

Immunohistological analysis for endothelial marker MECA-32. (A) Histological sections were stained for mouse endothelial cell antigen (MECA-32) for the detection of peritubular capillaries. (B) While quantification of MECA-32-positive capillary cross sections showed a considerable decrease in peritubular capillaries in ischemic control kidneys, combination therapy enabled significant preservation of the peritubular capillary network (p < 0.01 vs. control). Monotherapy using microcapsulotomy and HUVECs, respectively, was inferior in terms of preservation of vascularity. Original magnification: 400×. Scale bar: 50 μm.
Macrophage Marker F4/80
Macrophage infiltration (Fig. 8) evidenced by F4/80 staining was increased 10-fold in kidneys subjected to 45 min of warm ischemia (27.8±2.4% F4/80 area fraction) as compared to controls (2.2±1.1% F4/80 area fraction). Interestingly, both HUVEC treatment and microcapsulotomy as stand-alone treatments had no clear effect of macrophage infiltration. HUVEC treatment alone slightly increased macrophage infiltration of ischemic kidneys (37.3±1.9% F4/80 area fraction, p = 0.05 vs. control), while microcapsulotomy alone had no effect (27.8±2.4% F4/80 area fraction, n.s. vs. control). However, combination of both therapy options led to an effective reduction of macrophage infiltration (15.0±1.8% F4/80 area fraction, p < 0.05 vs. control).
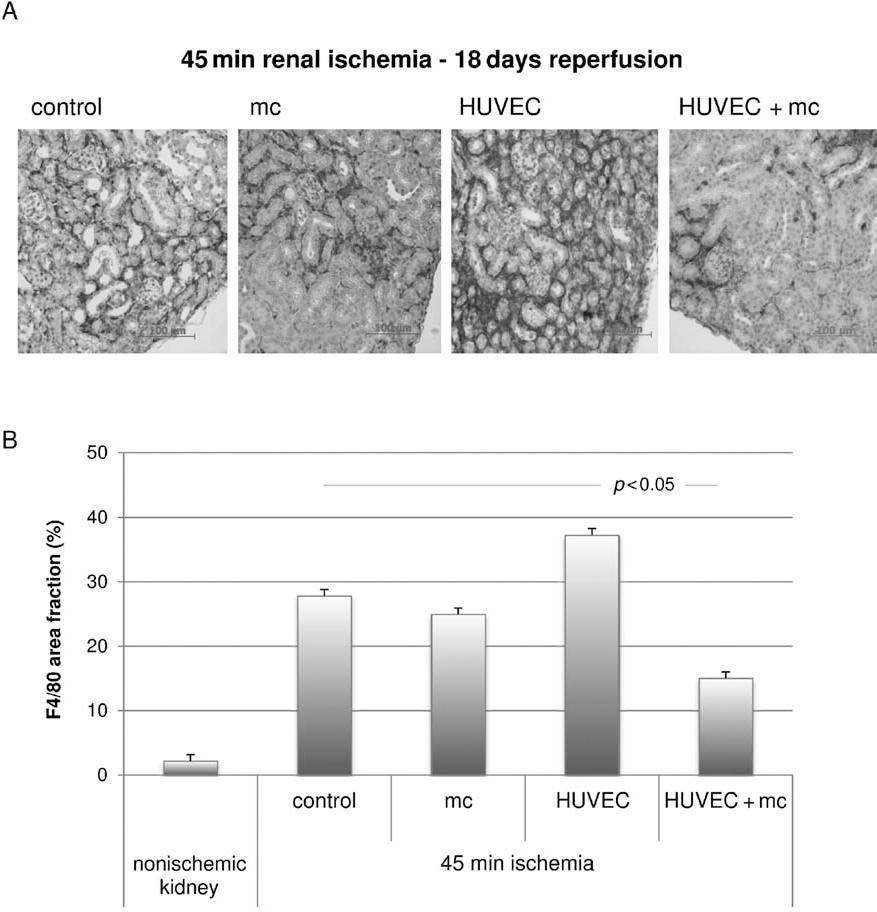
Immunohistological analysis for F4/80 macrophages. (A) Staining for F4/80 showed severe macrophage infiltration in ischemic kidneys treated with HUVECs alone, while microcapsulotomy alleviated the presence of macrophages. The most effective decrease in F4/80 staining was found for combination therapy. Original magnification: 200×. Scale bar: 100 μm. (B) Quantitative analysis based on binary images from histological sections at 50× magnification showed considerably increased staining for F4/80 in ischemic control kidneys (27.8±2.4% F4/80 area fraction) compared to nonischemic kidneys. Ischemic kidneys treated with HUVECs alone exhibited even more pronounced macrophage infiltration (37.3±1.9% F4/80 area fraction, p = 0.05 vs. control). Microcapsulotomy led to a nonsignificant decrease in F4/80 staining (27.8±2.4% F4/80 area fraction, n.s. vs. control), while combined therapy using HUVECS and microcapsulotomy was associated with a significant reduction (15.0±1.8% F4/80 area fraction, p < 0.05 vs. control).
Discussion
Here we show that endothelial cell therapy alone has only modest potential to preserve renal tubular integrity and vascular function in a murine model of renal ischemiareperfusion injury. This may appear contradictory to previous publications showing beneficial effects of cell therapy in kidney injury (6,23,24). However, the results based on serum creatinine levels were obtained early after injury thus depicting rather acute treatment effects than pivotal long-term outcome. Using 99mTc-MAG3 imaging as a high-sensitivity marker for the determination of renal clearance, we were able to present novel insights into more subtle changes in renal function dynamics in a murine model of renal ischemia-reperfusion both early and, most notably, in the long term. Our data demonstrate the limitations of cell therapy in the regenerative setting with respect to renal tubular and vascular function.
Given these results, we sought for an effective therapeutic additive in order to improve recovery of both kidney function and perfusion. Based on our previous findings indicating the existence of an intrinsic renal compartment syndrome following ischemia-reperfusion injury, we developed a surgical decompression strategy in order to achieve relief of increased interstitial pressure due to inflammatory edema (13). Indeed, our data strongly support the contribution of HUVEC-based therapy to enhanced organ function and perfusion only in the presence of microcapsulotomy and vice versa.
Permanently poor vascularization represents a major limitation for epithelial regeneration (25) leading to peritubular rarefaction and, finally, tubulointerstitial fibrosis (5). In the present study, we aimed to support the body's own regenerative capacity after ischemia-reperfusion injury by targeted external cell supply. In this context, the importance of local and circulating endothelial cells and endothelial progenitor cells in tissue regeneration and neovascularization following organ/tissue injury has been conclusively demonstrated (1,29). From the wide range of cell types applicable for therapeutic use, we therefore chose endothelial cells in order to enhance renal function by primarily promoting vascular repair. As veins, capillary venules in particular, represent the primary site of vascular leakage in inflammation (17,18), we applied venous endothelial cells derived from umbilical cord. Using a similar cell type meets recent reports on a largely genetic determination of arteriovenous specification (31). HUVECs exhibit a high proliferative potential and a robust expression of VEGFR2. The latter is of particular importance, as it mediates migration and homing of the cells to sites of ischemia/tissue injury. Moreover, VEGFR2 represents the major signaling receptor in endothelial cells (20). These properties strongly characterize HUVECs as an effective and specific modality to enhance vascular repair. In order to study the characteristics and regenerative capacity of human HUVECs, a murine model using athymic Balb/C nu/nu mice was applied. Moreover, the xenogenic model is an attractive starting point for future studies in terms of cell tracking by human leukocyte antigen (HLA) labeling and ablation of therapeutically applied human cells using diphtheria toxin. Cell tracking studies will be crucial for the understanding of the mode of action. They may help to elucidate how combined therapy using HUVECs and microcapsulotomy exerts its beneficial effects in renal ischemia-reperfusion injury. In particular, it will be interesting to examine which role paracrine/autocrine effects may play. In our hands discrimination of murine/human cells based on endothelial markers was limited by cross reactivity of antibodies.
To further investigate the functional characteristics of ischemia-reperfusion induced vascular damage, we performed laser Doppler perfusion measurements. Importantly, the functional data were supported by the immunohistological results of MECA-32 labeling. Following ischemia-reperfusion injury considerable rarefaction of peritubular capillaries and decreased blood flow was found in the control and the cell-treated group and, to a lesser extent, in the microcapsulotomy group. Most notably, enhanced staining for MECA-32 together with increased vascular perfusion was seen in ischemic kidneys treated with combination therapy. In contrast to previous data from our laboratory demonstrating a significant enhancement of renal perfusion by therapeutic application of endothelial progenitor cells (EPCs) (19), we found no significant improvement of renal blood flow in HUVEC-treated ischemic kidneys. The discrepancy may be explained by differential production of VEGF that is pivotal for the maintenance of the vascular system in adults (16,21), embryonic vasculogenesis (28), and postnatal angiogenesis (10,11). Aortic cells and EPCs are known to release VEGF, while venous cells lack VEGF expression (3,16).
In contrast to cell-based treatment alone, applying microcapsulotomy enabled the recovery of perfusion in ischemic kidneys. However, the beneficial effects only translated into a significant increase in tubular function in association with cell therapy. The use of a different mouse strain may explain differences seen in our previous work demonstrating functional improvement of both tubules and vasculature after microcapsulotomy (13). Notably, Balb/C nu/nu mice in the present study were more susceptible to renal ischemia-reperfusion injury than their wild-type counterparts, as seen from the histological analysis. This is in line with recent studies on the pathophysiological role of B-cells and the protective effects of regulatory T-cells in ischemia-reperfusion injury (9,15).
The synergism between microcapsulotomy and cell therapy is not easy to explain. We hypothesize that successful cell therapy may be linked to a permissive microenviroment. Decompression of inflammed kidneys may generate an enviroment for cell therapy to work. The fact that microcapsulotomy provides a regenerative milieu for HUVECs is suggested by a high Ki-67 labeling in these kidneys. However, HUVECs applied in a state of high intrarenal pressure may have adverse effects. By causing microembolism in peritubular capillaries, they may even aggravate ischemia/reperfusion (I/R) damage. Macrophage infiltration seems to be closely related to the extent of kidney damage and may represent the transition of an acute to a more chronic state of inflammation (2).
Our results further raise the question of what triggers the compensatory functional enhancement of the nonischemic contralateral kidney after I/R injury of the ipsilateral kidney. Only animals treated with microcapsulotomy showed a compensatory functional enhancement of kidney function while in controls kidney function of the contralateral kidney was adversely affected by I/R of the ipsilateral kidney. We speculate that reno-renal reflexes and/or hemodynamic changes may play a role.
The present study suggests that a multitargeted approach may be necessary to effectively prevent renal ischemia-reperfusion injury. In particular, providing the ideal composition of cell types for cell therapy represents an important issue. This may also include recently reported mRNA microvesicles derived from human bone marrow MSCs as a new therapeutic approach for kidney injury (7,12). Further studies including cell tracking experiments will be necessary to fully understand the prerequisites that are necessary for successful clinical application of cell-based therapies.
Footnotes
Acknowledgments
We are grateful to Heidrun Zankl, Department of Nuclear Medicine, University of Munich, for her excellent technical assistance. We thank Jana Mandelbaum and Dan Draganovic for their help with MECA staining. We are indebted to Cyril Nimmon, Chiang Mai, Thailand and Markus Diemling, Hermes Medical Solutions, Stockholm, Sweden for their continuous support regarding the kidney function quantification methods. We thank Prof. Ulrich Mansmann, Division of Biometrics and Bioinformatics, IBE, University of Munich, for his expert opinion and advice in solving statistical problems. The present report includes data that were generated during the doctoral thesis of Andreas Meyer at the University of Munich Medical School, Germany. This work was supported by the FöFoLe research grant of the University of Munich. The authors declare no conflict of interest.
