Abstract
Huntington's disease (HD) produces severe neurodegeneration in the striatum leading to disabling motor impairments, including the loss of control of skilled reaching movements. Fetal GABAergic transplants can physically replace the lost striatal cells but with only partial success in functional recovery. Here, we aimed to determine the extent and quality of the repair produced by fetal cell transplantation through an in-depth analysis of reaching behavior in the quinolinic acid-lesioned rat model of HD. Control, quinolinic acid-lesioned plus sham graft, and quinolinic acid-lesioned plus graft groups of rats were assessed in skilled reaching performance prior to and following lesion surgery and 3 months following injection of 400,000 fetal whole ganglionic eminence-derived cells into the striatum. This was compared to their performance in two more rudimentary tests of motor function (the adjusting step and vibrissae-evoked hand-placing tests). Grafted rats demonstrated a significant improvement in reaching success rate (graft +59%, shamTX +3%). Importantly, the quality of reaching behavior, including all components of the movement, was fully restored with no identifiable differences in the normal behavior shown by control rats. Postmortem immunohistochemical examination verified the survival of large intrastriatal grafts, and Fluoro-Gold tracing indicated appropriate outgrowth to the globus pallidus. Our study illustrates for the first time the detailed analysis of qualitative improvement of motor function following brain repair in a rat model of HD. The results demonstrate significant improvements not only in gross movements but also in the skilled motor patterns lost during HD. Fetal GABAergic cell transplantation showed a demonstrable ability to restore motor function to near normal levels, such that there were few differences from intact control animals, an effect not observed in standard tests of motor function.
Keywords
Introduction
Skilled reaching or skilled forehand use in reach-to-eat food paradigms is very valuable in the analysis of forelimb function, disease-specific deficits, and potential repair strategies in both animal models and patients (2, 4, 6, 35, 36, 45, 51, 61, 62). Previous studies suggest homology between human and rodent reaching behavior, making it a task easy to translate into a clinical setting and helpful for preclinical research (30, 36, 37, 51, 63). In particular, the single pellet reaching task offers the possibility of detailed analysis of hand prehension in a quasinatural context (36, 42, 51, 66). In this task, a single pellet is presented to a rat, which then has to reach for it from a raised platform with its preferred hand. The reaching act is filmed and scored afterward, enabling the qualitative examination of reaching patterns and posture, in addition to the commonly used simple reaching success rate. Each reaching movement can be split into several components (23, 42): orientation (toward the pellet), (hand) transport (to the food pellet), grasping (the food pellet), withdrawal (of the hand), and release (of the food pellet into the mouth). Previous studies using the same animal models of the human neurodegenerative disorders Parkinson's disease (PD) (35, 43, 59, 60, 65), stroke (2, 3, 24, 55, 57), and Huntington's disease (HD) (33, 66) revealed reaching deficits particularly in rotatory movements of the distal parts of a limb; hand and wrist supination and pronation were most severely impaired. Also, elbow movements and postural balance were deficient.
Skilled reaching is controlled by neuronal networks in the cerebellum, the cortex, and the basal ganglia. The motor cortex and striatum, however, are the main contributors for the selection and initiation of reaching movements. Therefore, neurodegenerative diseases such as HD, affecting most prominently striatal areas, cause severe disruptions of corticostriatal pathways and obvious impairments in voluntary and target-directed motor behaviors (8, 12, 13, 22, 36, 61, 66).
HD is a progressive neurological genetic disorder resulting in cognitive and psychiatric deficits as well as in abnormal movements, characterized by chorea, dystonia, and impaired fine motor control. This inherited, autosomal dominant disorder (27) causes mutations in the ubiquitously expressed protein huntingtin (HTT), leading to intracerebral aggregation of mutated HTT, which in itself is believed to contribute to neuronal dysfunction, neuronal loss, and eventually a massive atrophy of the striatum.
To reproduce the pathological features of HD, the intrastriatal stereotaxic injection of the excitotoxin quinolinic acid (QA) serves as the most commonly used model causing massive degeneration of medium spiny projection neurons (MSNs) with relative sparing of striatal interneurons, and resulting in striatal tissue atrophy with associated ventricular enlargement. QA-lesioned rats show motor deficits, such as a rotational bias, delayed or absent sensorimotor responses, cognitive deficits, and impaired skilled reaching (17, 19, 61).
Cell replacement therapy has become a promising novel approach to combat cell loss in the brain across various neurodegenerative diseases. Reach-to-eat movements have been examined before and after transplantation in models of PD and HD and have been shown to respond well to the grafts, that is, improving the reaching success rate significantly (11, 18, 22, 28, 29, 41, 44). Interestingly, despite a considerable improvement in the success rate, dopaminergic grafts in a rodent PD paradigm could not repair the qualitative aspects of reaching movements. This has invited speculations about how the graft is being used to improve motor function, whether this is integrative in the basal ganglia circuitry or driving motivation rather than motor ability (35). Rodent experiments have revealed that the homotopic intrastriatal transplantation of embryonic day 15 whole ganglionic eminence (E15 wGE)-derived tissue is most appropriate for functional rewiring of corticostriatal pathways in terms of anatomical integration of the transplants and behavioral recovery. The latter has been shown in several tasks including improvement of reaching success in the staircase test and forelimb akinesia (13, 15, 22, 44).
The present study explores the nature of deficits in (skilled) forelimb use before and after the transplantation of E15 wGE-derived cells in a unilateral rat model of HD. Effects on sensorimotor response and forelimb akinesia were assessed by means of the vibrissae-evoked hand-placing test and the adjusting step test. Skilled reaching was analyzed in the single pellet reaching task. Histological data of graft integration and survival were collected 18 weeks after the transplantation. Sensorimotor response and reaching success rate could be partially restored, whereas reaching patterns (quality of reaching) were completely repaired to control levels.
Materials and Methods
Experimental Design
Thirty-six female Lister Hooded rats received baseline training in the single pellet reaching task, 5 days a week over a period of 4 weeks (Fig. 1). Two days before the lesion surgery, all rats were filmed while performing reaching-for-food movements in the single pellet reaching task, and tested twice in the adjusting step test and in the vibrissae-evoked hand-placing test (baseline behavior). Eleven rats served as nonoperated control (con) and received no surgical procedure during the entire experiment. The remaining rats all received unilateral striatal lesions by stereotaxic injection of quinolinic acid (QA) into the striatum contralateral to the rats' preferred hand. Four weeks after the lesion surgery, all rats were tested daily for 14 consecutive days in the single pellet reaching task and twice in the adjusting step test and in the vibrissae-evoked hand-placing test (postLX behavior). Six weeks after the lesion, the lesioned rats were split into two further groups matched for performance on the single pellet reaching task. Half of the animals received striatal implantation of whole ganglionic eminence (wGE) tissue derived from embryonic day 15 (E15) embryos (graft), whereas the remaining lesioned rats received sham grafts by striatal infusion of culture medium only (shamTX). Thirteen weeks later (postTX behavior), all rats were reassessed in the three behavioral tasks with daily training over 9 consecutive days in the single pellet reaching task followed by 2 days of testing in the adjusting step test and in the vibrissae-evoked hand-placing test. After completion of behavioral testing, all graft rats, four shamTX, and four con rats received intrapallidal stereotaxic infusion of Fluoro-Gold for retrograde labeling of striatopallidal neuronal projections. One week later, all rats were perfused and subjected to histological analyses.

Experimental design. Nonoperated (con), sham-transplanted (shamTX), and transplanted (graft) rats were tested in the adjusting step test, the vibrissae-evoked hand-placing test, and the single pellet reaching task before (baseline behavior) and after (postLX behavior) the intrastriatal injection of quinolinic acid (QA) and 13 weeks (w) after the transplantation of embryonic day 15 whole ganglionic eminence (E15 wGE)-derived cells (postTX behavior). One week before perfusion, rats were stereotaxically injected with Fluoro-Gold (FG) into the globus pallidus. Eighteen weeks after the transplantation, all rats were sacrificed, and their brains were subjected to histological analyses.
All procedures were conducted in accordance with project, personal, and facility licenses issued under the UK Animals (Scientific Procedures) Act, 1986, and subject to local ethical review.
Subjects
Thirty-six adult female Lister Hooded rats (Charles River, Margate, UK) were used in this study. Rats were housed in cages of four, kept in temperature- and humidity-controlled rooms on a 14-h light/10-h dark schedule. All rats were food restricted (12 g per rat per day) during behavioral assessment (outside testing period food was provided ad libitum); water was administered ad libitum at all times. The animals were weighed twice weekly to monitor their body weight. The rats were not allowed to fall below 85% of the free-feeding weights recorded at the start of each food restriction period. Three subjects died during surgery, and two rats were excluded from the study (one with no surviving graft and the other sacrificed early due to emergence of mild seizures). Final group sizes for analysis were as follows: con, 11; shamTX, 10; graft, 10.
Surgery
Striatal Lesions
All shamTX and graft rats were anesthetized with isoflurane (Abbott, Maidenhead, Berkshire, UK; using O2 and N2O as carrier gases) and stereotaxically injected (Stoelting, IL, USA) with 1 μl of QA (0.09 M; Sigma-Aldrich, Dorset, UK) into the striatum contralateral to the rats' preferred hand using a 30-gauge stainless steel cannula (Hamilton, Bonaduz, Switzerland) connected to a microdrive pump (Harvard Apparatus, Holliston, MA, USA). Lesion coordinates (two tracts; two deposits each; 0.25 μl of QA per deposit) were set according to bregma and dura in mm [tract 1: tooth bar (TB) −2.3, anterior/posterior (AP) −0.4, lateral (LAT) ±3.7, dorsoventral (DV) −5.2/ −4.2; tract 2: TB −2.3, AP +1.2, LAT ±2.9, DV −5.2/–4.2]. The injection rate was 0.25 μl/min, and the cannula was left in place for 3 min before slow retraction. A subcutaneous injection of 5 ml of 4% glucose–0.18% saline (Fisher, Loughborough, UK), an intramuscular injection of 0.15 ml of diazepam (Roche, Welwyn Garden City, UK; 5 mg/ml), and recovery in heated cages were provided as immediate postsurgery care. Analgesic treatment was administered 1 day before and 2 days after the procedure [1 g of paracetamol (Tesco, Dundee, UK)/L drinking water].
Striatal Transplantation
Graft tissue was derived from E15 embryos (crown rump length: 12.5 mm), which were harvested by Cesarean section from pregnant Lister Hooded dams and collected in glucose-buffered saline. The brains were removed, and the wGE was dissected, incubated in trypsin, washed, and prepared as dissociated cell suspension in HBSS (Hanks' balanced salt solution; Gibco, Paisley, Scotland, UK) as previously described (21, 58). Cell viability was assessed by trypan blue exclusion (Sigma-Aldrich) in a hemocytometer and revealed that 99% of all cells were viable. Volume of the cell suspension was adjusted to 2 μl of vehicle (HBSS) per wGE.
All lesioned rats received a second stereotaxic injection of either HBSS medium (shamTX) or E15 wGE-derived cells (graft). The animals were anesthetized with isoflurane using O2 and N2O as carrier gases and mounted in the stereotaxic frame (Stoelting). Two microliters of vehicle or cell suspension was infused stereotaxically into the striatum in a single tract (two deposits; 1 μl per deposit so that each graft rat received the equivalent of one wGE in total) using a 10-μl microsyringe with a wide-bore needle (SGE, Ringwood, Australia). Transplantation coordinates were set according to bregma and dura in mm (TB −2.3, AP +0.4, LAT ±3.3, DV −5.0/–4.4). The injection rate was 0.5 μl/min, and the cannula was left in place for 3 min before slow retraction. Each animal received the same schedule of postoperative antibiotics and analgesia as following lesion surgery.
Fluoro-Gold Injection
Five days prior to perfusion, all graft, four shamTX, and four con rats were stereotaxically infused with the retrograde tracer Fluoro-Gold (Fluorochrome Inc., Denver, CO, USA). The animals were anesthetized with isoflurane using O2 and N2O as carrier gases and mounted in the stereotaxic frame. The external segment of the globus pallidus was injected with 193 nl of 1% Fluoro-Gold-saline solution, using a glass capillary (outer diameter 75 μm; Drummond Scientific, Broomall, PA, USA). The injection rate was seven pulses of 27.6 nl over a period of 5 min using a Nanoject microinjector (Drummond Scientific). Injection coordinates were set according to bregma and dura in mm (TB −3.3, AP −1.1, LAT ±3.0, DV −6.0).
Behavioral Assessment
Single Pellet Reaching Task
Skilled reaching behavior of the preferred hand was analyzed by using the reaching-for-food task as illustrated and described fully elsewhere (42, 59, 62). Briefly, rats were tested in a Plexiglas box (13 cm × 45 cm × 40 cm, created in-house), at one end of which was a slit through which the rats could reach with their preferred hand for a single food pellet (sugar flavor; TestDiet, St. Louis, MO, USA) from a shelf that was attached to the outside of the box. The task was designed in such a way that the rats were encouraged to use their preferred hand only and could not reach the pellet with their nonpreferred hand (the latter was not evaluated in skilled reaching performance). The rats were trained to walk to the rear end of the box after each reach (successful or unsuccessful) before the next attempt to reach for a pellet. Thus, to initiate a new trial, the rats had to return from the rear of the box to the slit and reach for new pellet. Rats were tested until their success rates reached levels over 50% (each rat was presented with 20 pellets per test day). A successful reach was recorded if a rat obtained a food pellet at its first attempt (First Trial Success). Success after multiple attempts was expressed as Total Success (data not shown but available upon request).
Reaching behavior was filmed during baseline (2 days before lesion surgery), postLX behavior (6 weeks after lesion surgery), and postTX behavior (17 weeks after the transplantation; baseline and postLX data are not shown for the sake of brevity but available upon request). A miniDV video camera (Panasonic, NV-GS15, Osaka, Japan) set at a shutter speed of 1,000 s–1 was used to record reaching behavior. All First Trial Success reaches were scored for each animal. The video recording was analyzed frame by frame, and the reaching movements were scored according to a movement element rating scale as described previously (42). Briefly, a single reach was divided into five functional movement components (orient, transport, grasp, withdrawal, and release), which were split into 11 movement elements: orient, limb lift, digits close, aim, advance, digits open, pronation, grasp, supination 1, supination 2, and release. These 11 elements were subdivided into 34 subelements (Table 1) (42). Each subelement was scored as “normal” (1 point), “abnormal” (0.5 point), or “absent” (0 point). Data (total score and performance in each movement element) were averaged for each rat and group.
Movement Element Rating Scale

Spontaneous Behavior (Adjusting Step Test and Vibrissae-Evoked Hand-Placing Test)
During these tests, the experimenter allowed the rats to use first their impaired forelimb (restraining the healthy forelimb and both hindlimbs) and then their healthy forelimb (restraining the impaired hand from use and restraining both hindlimbs). The tests were repeated on 2 consecutive days during baseline, postLX, and postTX behavior.
Vibrissae-Evoked Hand-Placing Test
The rats were lifted upward toward the edge of a table so that only the vibrissae would touch the table edge (53, 54). A normal control rat will reflexively place its free hand onto the table surface as soon as the vibrissae contact the table surface. There was a 4-point scale for the evaluation of the rats' behavior: 0 = no movement of the hand, no contact with the table; 1 = little hand movement, no or very late contact with the table; 2 = slow and/or deficient reflexive movement, table contact; 3 = fast movement of hand, immediate contact with the table. This test was performed five times per side during each experimental day.
Adjusting Step Test
The rats were guided back and forth over a distance of 60 cm on a table surface, and the adjusting steps of each side were counted (48, 50). On each experimental day, the rats' performance was recorded twice in forehand stepping and twice in backhand stepping.
Histological Analyses
Perfusion and Staining
Eighteen weeks after transplantation surgery, all rats were terminally anesthetized and transcardially perfused with approximately 100 ml of 0.1 M phosphate-buffered saline (PBS; pH 7.4; Fisher) followed by 300–500 ml of 4% paraformaldehyde (PFA; Sigma-Aldrich). After postfixation in PFA overnight, the brains were cryoprotected in 30% sucrose (Fisher). The tissue was cut in a series of 12 coronal sections at 40-μm thickness using a freezing stage microtome (Leica, Solms, Germany). Parallel (one in six) series were stained for (i) Nissl with cresyl violet (Sigma-Aldrich) to visualize cell bodies; (ii) neuronal nuclei (NeuN), a marker of mature neurons (1:4,000; Sigma); (iii) DARPP-32, a dopamine-and adenosine 3′,5′-monophosphate-regulated phosphoprotein used to identify the MSNs (1:30,000; donated by Professor Hemmings, Cornell University, Ithaca, NY, USA); and (iv) Fluoro-Gold (Chemicon, Temecula, CA, USA; 1:16,000), enhancing the autofluorescent properties of this stereotaxically injected retrograde tracer.
Nissl staining was undertaken by pretreating the mounted sections in 50:50 chloroform/EtOH solution (Sigma-Aldrich). Then, the sections were hydrated (100% EtOH, 95% EtOH, dH2O) before they were dipped into cresyl violet solution (0.1 g of cresyl violet, 100 ml of dH2O, 10 drops of glacial acetic acid). Afterward, the sections were dehydrated (95% EtOH, 100% EtOH; 100% xylene) and coverslipped.
NeuN, DARPP-32, and Fluoro-Gold were visualized by immunohistochemical staining on free-floating sections by the streptavidin-biotin method (biotinylated, rat-adsorbed anti-mouse, secondary antibody from Vector, Burlingame, CA, USA; 1:200; 10% streptavidin-biotin-horseradish peroxidase solution from Dako, Glostrup, Denmark) with 3,3′-diaminobenzidine (DAB; Sigma-Aldrich) as chromogen (15). The stained sections were mounted on gelatinized slides [slides from Thermo-Scientific (Hemel Hempstead, UK) coated in-house with 0.5% gelatine (Sigma-Aldrich)], air dried, dehydrated in increasing concentrations of ethanol, and finally dipped into xylene prior to coverslipping.
Striatal Atrophy
Ventricle volume was used as an indirect measure to demonstrate striatal atrophy in lesion and in graft animals. Volume measurements were carried out bilaterally at four coordinates in the brain: AP +1.7, +1.2, −0.4, and −0.9. Images of NeuN-stained sections were used to calculate the area of the ventricle by outlining the ventricle circumference in ImageJ (National Institutes of Health, Bethesda, MD, USA). Results of all four sections were added, and ipsilateral (lesioned) ventricle volume was expressed as percent of contralateral (healthy) ventricle volume.
Graft Volume
Graft volume was measured on every section in which graft-derived NeuN-positive (NeuN+) cells were found. Photographs of NeuN-stained sections were taken under bright-field illumination and 1.6x magnification with a Leica DM-RBE microscope using Leica software (Leica, Heerbrugg, Switzerland). Digital images were analyzed with ImageJ. The borders of the graft core were highlighted, and the area of the graft core was calculated in μm2. The result of each section was multiplied by the original thickness of the section (40 μm) and by the number of series of coronal sections used (6). The results of all sections were added, and the total graft volume (μm3) was calculated.
Cell Counting
Somata of NeuN+ and DARPP32+ cells were stereologically counted within the grafts under bright-field illumination using a microscope with an X-Y motor stage (Olympus BX50, Ballerup, Denmark). Cell counts were performed by means of the C.A.S.T.-Grid software (Olympus). The region of interest (inclusion area) was drawn around the graft core according to the area that was immunoreactive for NeuN or DARPP32, respectively. The sampling grid size was set at 220 μm × 220 μm, and the counting frame size was 619 μm2. The total number estimated in full-thickness 2D sections was corrected using Abercrombie's formula (1): total number of cells = ∑c*[A/(∑n*a)]*f, where ∑c is cell count, A is inclusion area, ∑n is the number of samples, a is counting frame size, and f is frequency (i.e., six in the present study).
Statistics
The data were subjected to one-way or two-way analyses of variance (ANOVA) followed by Student-Newman-Keuls post hoc test (GraphPad Prism 5, GraphPad Software, Inc., San Diego, CA, USA), with a value of p < 0.05 set as the level of significance. Results are presented as means ± standard error of the mean (SEM) unless stated otherwise.
Results
Histology
Nissl, NeuN, and DARPP32 staining demonstrated substantial graft survival containing γ-aminobutyric acidergic (GABAergic) neuronal populations (Fig. 2A-C). Stereological counting of graft-derived NeuN+ and DARPP32+ cells revealed a ratio of 42.7 ± 4.1% DARPP32/NeuN (mean cell number: NeuN, 135,779 ± 16,037 cells; DARPP32, 56,816 ± 7,273 cells) (Fig. 2D).

Histological analyses. (A–C) Sections (40 μm thick) of transplanted rats (graft) stained for Nissl (A), neuronal nuclei (NeuN; B), and the dopamine- and adenosine 3′,5′-monophosphate-regulated phosphoprotein 32 (DARPP32; C) under 1.6× magnification. The dotted line surrounds the graft core “g.” The ipsilateral ventricle is marked with “V,” and its atrophy is expressed as percent of the healthy (contralateral) ventricle [see (F); note that despite substantial graft volume, there was no difference in striatal atrophy between shamTX and graft rats]. (A′–C′) Equivalent ipsilateral anatomical structures in shamTX rats. Graft volume is presented in a scatter plot in (D) (mean volume, 2.4 ± 0.4 mm3). Stereological counts of grafted cells are illustrated in (E). The mean number of all transplanted neurons as seen in NeuN staining was 135,779 cells (±48,110 SD). The mean number of DARPP32+ medium spiny neurons was 56,816 cells (±21,819 SD), resulting in an average percentage of 42.7 ± 4.1% DARPP32+ cells of NeuN+ cells.
Striatal atrophy was indirectly measured by calculating the size of the third ventricle for each hemisphere. Both lesion groups displayed striatal atrophy compared to the intact con rats [con 98.02 ± 3.75 mm3; shamTX 177.90 ± 9.51 mm3; graft 177.90 ± 15.07 mm3; F(2, 29) = 19.25, shamTX/graft vs. con, p < 0.001 post hoc]. Despite a graft volume of 2.43 ± 0.38 mm3 revealed by NeuN staining (Fig. 2E), there was no difference in ventricle volume between the shamTX and graft groups (Fig. 2F).
The injection of Fluoro-Gold into the GP, one of the targets of striatal MSNs, caused a retrograde transport of the fluorescent dye into striatal areas including the graft (Fig. 3), indicating that projections from the graft reach this region, and suggestive of homotopic rewiring of the basal ganglia circuitry.
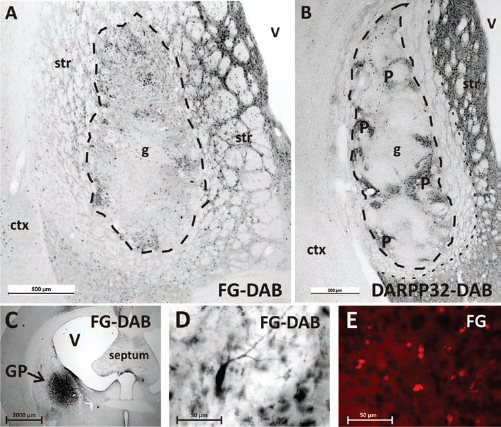
Fluoro-Gold tracing analysis. (A, C–E) Images of Fluoro-Gold (FG) injection into the globus pallidus (GP), one of the main projection areas of striatal medium spiny neurons. The retrograde labeling resulted in FG+ neurons in the host striatum and in the grafted area, indicating a homotopic reinnervation of the striatal projections (A). The dotted line highlights the graft core (g). (B) An image of a DARPP32-stained section of the same animal as seen in the other panels. Areas of high DARPP32+ neurons (P-zones) correspond with high density of FG+ neurons as seen in (A). The outer dotted line highlights the border between lesioned DARPP32-negative and intact striatal tissue. Note the exact placement of the graft into the center of the γ-aminobutyric acid (GABA)-depleted striatum. The pallidal injection site is visualized in (C). An individual FG+ neuron is displayed in (D). The autofluorescent capacity of FG is presented in (E). Images in (A–D) show visually enhanced FG staining using the DAB method (see Materials and Methods). str, striatum; ctx, cortex; V, ventricle.
Behavior
All rats underwent an extensive behavioral test battery during baseline, postLX, and postTX test sessions. The stereotaxic injection of QA into the striatum caused widespread brain atrophy and led to obvious motor deficits. The grafted group (graft) displayed significant improvement in many of the observed behavioral parameters after having received an injection of fetal E15 wGE-derived cells. The sham-transplanted rats (shamTX) did not improve at all and continued to exhibit lesion-induced motor deficits.
Spontaneous Behavior
The lesions impaired the performance of both shamTX and graft groups in the vibrissae-evoked hand-placing test (Fig. 4A). On the second test day after the transplantation, there was a significant improvement in the ability of the graft group to respond on the side contralateral to the graft; however, it did not reach the levels of the intact con rats [on T2: con 2.96 ± 0.03; shamTX 1.66 ± 0.17; graft 2.50 ± 0.13; F(2, 912) = 88.67: shamTX vs. graft, p < 0.001 post hoc; graft vs. con, p < 0.05 post hoc; shamTX vs. con, p < 0.001 post hoc]. There was no deficit in any of the groups on the ipsilateral side (data not shown).

Spontaneous behavior analysis. (A) Results of the vibrissae-evoked hand-placing test. The graft rats showed a significant improvement on T2 compared to the shamTX rats and compared to their performance on L2 (€p < 0.05). The con rats, however, remained on a higher performance level than the other groups after the lesion and transplantation [on T2: con 2.96 ± 0.03; shamTX 1.66 ± 0.17; graft 2.50 ± 0.13; F(2, 912) = 88.67: #shamTX vs. graft, p < 0.001 post hoc; *graft vs. con, p < 0.05 post hoc; *shamTX vs. con, p < 0.001 post hoc]. No graft but only a lesion effect was detected in forehand stepping as seen in (B) [*shamTX/graft vs. con, F(2, 354) = 302.2, p < 0.001].
The adjusting step test provided a sensitive measure of lesion deficits on the contralateral side induced by the injection of QA (Fig. 4B). After lesion, the shamTX and graft rats were significantly impaired in the forehand stepping parameter and displayed less adjusting steps than the con animals [shamTX/graft vs. con, F(2, 354) = 302.2, p < 0.001]. There was no improvement following transplantation. No lesion- or graft-related effects were observed on the ipsilateral side. The backhand stepping parameter did not show any deficits after the lesion or any change after transplantation (data not shown).
Single Pellet Reaching Task
First Trial Success
Data from the last four testing days during each test interval (baseline, postLX, and postTX) were pooled and are presented in Figure 5. The injection of QA caused severe motor deficits in the shamTX and graft rats; however, this deficit could be partially restored by the transplantation of wGE-derived cells [Group: F(2, 362) = 160.30, p < 0.0001; Time: F(2, 362) = 164.80, p < 0.0001; Group * Time: F(4, 362) = 44.46, p < 0.0001].

Single pellet reaching task—quantitative analysis. This figure displays the percentage of First Trial Success out of 20 trials per day (success to reach for a food pellet at first attempt). The data were pooled over the last 4 days of testing during baseline, postLX, and postTX. Reaching performance was reduced by 68% (shamTX) and 65% (graft) after the lesion (*shamTX/graft rats significantly different from con rats). The graft rats displayed an improved reaching performance during postTX (#shamTX significantly different from graft rats), whereas the con and shamTX rats remained on the same levels as before the transplantation [€within-group comparison: con −3% n.s., shamTX +3% n.s., graft +59% F(2, 362) = 164.8, p < 0.001 post hoc].
The number of successful reaches at first attempt dropped by 68% (shamTX) and 65% (graft) after the lesion. The con rats remained on the same performance level comparing baseline and postLX data [reaching success (%) during baseline: con = 64.77 ± 1.75, shamTX = 63.13 ± 1.95, graft = 67.13 ± 2.29; reaching success (%) during postLX: con = 64.77 ± 2.05, shamTX = 20.38 ± 2.38, graft = 23.25 ± 2.22; post hoc analysis postLX shamTX/ graft vs. con F(2, 362) = 164.8, p < 0.001]. Comparing the first (L1) and the last (L13) experimental days during the postLX training period, all QA-infused rats (shamTX and graft) developed a training effect by improving their performance moderately to the same degree over time (data not shown but available upon request). Their motor performance, however, remained below the baseline and con rats' levels and reached a plateau performance, beyond which no further improvement was observed.
Thirteen weeks after the transplantation surgery, all rats were tested again, and the graft rats showed a significant improvement in reaching success compared to the shamTX rats [reaching success (%) during postTX testing: con = 62.61 ± 1.91, shamTX = 21.00 ± 1.49, graft = 39.23 ± 2.63; shamTX vs. graft F(2, 362) = 160.30, p < 0.001 post hoc]. The graft group remained below the levels of the con rats [con vs. graft F(2, 362) = 160.30, p < 0.001 post hoc]. The comparison of postLX and postTX data within each group revealed that the con and shamTX rats' reaching success remained unchanged, whereas the graft rats' performance improved significantly: con −3% n.s., shamTX +3% n.s., graft +59% F(2, 362) = 164.8, p < 0.001 post hoc (Fig. 5, see €).
For the parameter Total Success, a similar pattern of lesion- and graft-related effects was detected in all groups (data not shown but available upon request).
Qualitative Reaching Pattern Analysis
The QA lesion induced significant impairment in reaching performance in the shamTX and graft rats (postLX), which was fully restored by the transplantation of wGE cells (postTX; Figs. 6A, B and 7). The total score analysis (Fig. 6B) revealed no difference between con and graft rats after grafting, whereas the shamTX animals displayed obvious motor deficits [Group: F(2, 84) = 15.92, p < 0.001; Time: F(2, 84) = 14.00, p < 0.001; Group * Time: F(4, 84) = 3.83, p = 0.007; post hoc analysis on postTX: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.01; graft vs. con n.s.].

Single pellet reaching task—qualitative analysis. (A) Results of the scoring of all First Trial Success reaches during the last day of filming (postTX). A frame-by-frame video analysis revealed severe impact of QA on posture and reaching patterns in 9 out of 11 movement elements; the transplantation of wGE-derived cells, however, could fully restore all deficits (*shamTX/graft rats significantly different from con rats; #shamTX significantly different from graft rats). (B) Development of the total reaching score over the course of the experiment. Although the performance within the shamTX and graft rats did not change significantly between postLX and postTX, the subtle changes were sufficient to result in a group difference on postTX [Group: F(2, 84) = 15.92, p < 0.001; Time: F(2, 84) = 14.00, p < 0.001; Group * Time: F(4, 84) = 3.83, p = 0.007; post hoc analysis on postTX: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.01; graft vs. con n.s.].

Skilled reaching images. This figure shows three different rats in the single pellet reaching task box during the movement component transport. (B, B′ and C, C′) Reaching behavior of the same rat before and after the transplantation, respectively. Note the con rat's posture in (A) the body weight is shifted laterally (head of rat is shifted to the healthy side and away from the dotted line, i.e., the body midline), the elbow “E” is turned to the outside, and the rat smoothly puts its hand “P” down into an arpeggio movement to grasp the food pellet. (B, B′, and C) Reaching behavior of lesioned rats. Note the variety of motor deficits: the arm can be overextended and overshoot the target (B). Furthermore, the rat's trunk can rise too high and might not shift far enough to the healthy side (B) or even shifts to the wrong side (B′). (B′, C) Rats do not extend their arm at all, but keep it flexed and their elbow in a lower position. Because of the flexed elbow position, some rats could not reach far enough without knocking off the pellet and needed multiple attempts to successfully grasp the target (C). However, the same rat seen in (C) significantly improved its reaching behavior after the transplantation (C′). Its weight is shifted further to the healthy side and its arm is fully extended to reach for the food pellet.
The evaluation of the individual movement components and elements after the transplantation (postTX) is illustrated in Figure 6A. For the sake of brevity, baseline and postLX reaching scores and full analysis of movement subelements during those two time points are not shown (but available upon request).
Orient
The component orient was affected neither by the lesion nor by the transplantation procedure; all rats showed normal head movements and sniffing behavior.
Transport
The movement element limb lift was severely affected by the injection of QA. However, the graft rats returned to normal behavior 17 weeks after the transplantation, whereas the shamTX rats remained impaired [Group: F(2, 84) = 36.09, p < 0.0001; Time: F(2, 84) = 37.27, p < 0.0001; Group * Time: F(4, 84) = 9.77, p < 0.0001; post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.]. Particularly, the postural balance represented by the subelements body weight shift back and hindlimbs aligned with body was impaired after the lesion, but returned to normal in the graft rats (post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.).
The movement element digits close was mildly impaired by the lesion; however, this moderate deficit could be reversed by the graft [Group: F(2, 84) = 5.63, p < 0.01; Time: F(2, 84) = 6.13, p < 0.01; Group * Time: F(4, 84) = 5.08, p < 0.01; post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.]. The subelement palm supinated, semi-in, describing a rotatory movement of the hand, continuously deteriorated in the shamTX rats over time, whereas the graft rats did not develop deficits in this parameter and remained on the same performance level as the con rats (post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.).
The element aim revealed an “improved-to-normal” elbow movement and keeping the palm on midline for the graft rats [Group: F(2, 84) = 13.14, p < 0.0001; Time: F(2, 84) = 6.98, p < 0.01; Group * Time: F(4, 84) = 4.92, p < 0.01; post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.].
During the movement, element advance postural stability was impaired [Group: F(2, 84) = 13.14, p < 0.0001; Time: F(2, 84) = 6.98, p < 0.01; Group * Time: F(4, 84) = 4.92, p < 0.01; post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.]. The subelements body weight shift front and body weight shift lateral suffered most from the lesion, but were fully restored after the transplantation (post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.). Interestingly, the smooth forward motion of the reaching forelimb showed only a transient impairment, as the motor performance of the shamTX and graft rats leveled with the con rats' motor performance (post hoc analysis: shamTX vs. con vs. graft n.s.). Directing the limb toward the food pellet (subelement limb directed to the target) deteriorated in the shamTX rats over time; however, the graft rats' hand trajectory could be partially restored (post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.). Similarly, shamTX rats developed motor deficits in elbow movements (elbow in) over time, whereas graft rats were prevented from such development (post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.05; graft vs. con p < 0.05).
The QA-induced motor deficits in the element digits open [Group: F(2, 84) = 14.10, p < 0.0001; Time: F(2, 84) = 20.16, p < 0.0001; Group * Time: F(4, 84) = 4.92, p < 0.001] were mainly driven by the abnormal behavior in the subelement discrete limb movement that describes adjustments of distal parts of the limb in order to prepare a successful grasp. The transplantation of wGE-derived cells reversed those deficits, normalizing the graft rats' performance compared to the con rats (post hoc analysis: shamTX vs. con p < 0.01; shamTX vs. graft p < 0.05; graft vs. con n.s.). The actual opening of the digits (subelement digits open) was not affected either by the QA injection or by the graft (post hoc analysis: shamTX vs. con vs. graft n.s.).
Grasp
The QA injection had a severe impact on the movement element grasp, which could be fully rescued by the transplantation of wGE cells [Group: F(2, 84) = 15.92, p < 0.001; Time: F(2, 84) = 14.00, p < 0.001; Group * Time: F(4, 84) = 3.83, p = 0.007; post hoc analysis on postTX: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.01; graft vs. con n.s.]. The actual grasping movement (i.e., closing the digits as seen in the movement subelement digits close) was not affected, neither by the QA injection nor by the graft; however, the ability to hold the arm still while carrying out the grasp (arm still) was severely impaired in the shamTX rats, but could be restored in the graft rats (post hoc analysis on postTX: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.).
Withdrawal
This movement component comprises the elements supination 1 and supination 2, which were both severely impaired after the injection of QA, but could be restored after grafting [supination 1: Group: F(2, 84) = 30.06, p < 0.0001; Time: F(2, 84) = 23.27, p < 0.0001; Group * Time: F(4, 84) = 7.27, p < 0.0001; supination 2: Group: F(2, 84) = 34.75, p < 0.0001; Time: F(2, 84) = 21.61, p < 0.0001; Group * Time: F(4, 84) = 7.33, p < 0.0001]. Particularly, the rotatory movement elements during supination 1 were impaired: the rats overextended their arms to reach for a pellet rather than “flexing and bringing the elbow in” toward the midline of the body; this was partially restored in the graft rats, allowing them to flex their arm to a greater degree (elbow in post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.). The shamTX rats also displayed a worsening motor deficit in keeping their palm medially in the slot before retracting their arm. Graft rats returned to baseline behavior after the transplantation and were able to keep their arm medially (palm medially post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.). Furthermore, graft rats fully recovered in the subelements palm straight up and distal limb movements, indicating an improvement in rotatory movements of distal parts of the rat's forelimb (palm straight up post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.; distal limb movements post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.).
Release
The graft rats greatly benefited from the transplantation of wGE-derived cells during the movement component release [Group: F(2, 84) = 33.40, p < 0.0001; Time: F(2, 84) = 24.77, p < 0.0001; Group * Time: F(4, 84) = 5.67, p < 0.001]. Particularly, opening the digits for pellet release into the mouth and body posture as seen in the subelements open digits and head and upper body lowered were affected by the grafts (post hoc analysis: shamTX vs. con p < 0.001; shamTX vs. graft p < 0.001; graft vs. con n.s.). In contrast, the shamTX had difficulties in opening their digits to release the pellet, and their body was often not lowered but rather elevated during the eating of the pellet.
Discussion
This study analyzes for the first time the effects of GABAergic grafts in a rat model of HD on qualitative aspects of skilled reaching movements in a reach-for-food paradigm. Nonoperated (con), sham-transplanted (shamTX), and transplanted (graft) rats were tested in the adjusting step test, the vibrissae-evoked hand-placing test, and the single pellet reaching task before and after the intrastriatal injection of QA and 13 weeks after the transplantation of wGE-derived cells. The infusion of QA caused severe motor deficits in forelimb use in all behavioral tests. The sensorimotor response in the vibrissae-evoked hand-placing test and reaching success in the single pellet reaching task were partially restored by the transplantation. Interestingly, the rats' reaching patterns and posture fully recovered in nearly all movement elements in the qualitative reaching analysis (Fig. 7). Histological analyses revealed good graft survival and graft integration as seen by homotopic connections between the graft and the globus pallidus.
Lesion
The QA lesion model has been found to be a valid model to reproduce (at least in part) the core striatal pathology seen in HD (19, 46, 66). The present study reflected typical outcomes as the injection of QA resulted in huge loss of striatal cells and an enlargement of the lateral ventricle with consequent impairments to motor (and cognitive) behavior (19, 52). Deficits occur in the initiation and direction of skilled motor actions and a more general disinhibition of spontaneous motor behaviors.
Skilled reaching in rats is considered homologous to human skilled forelimb use and offers a powerful comparative metric in both rodent and human studies (10, 13, 22, 31, 36, 37, 45, 51, 56, 60–64). The injection of QA and the resulting lesion cause severe, chronic deficits in reaching success as seen in the staircase test and the single pellet reaching task (13, 14, 22, 44, 66). We report similar deficits in the single pellet reaching task with a reduced success rate (–66.5%) and lowered reaching score (the qualitative analysis of reaching performance dropped by −4.5% after the lesion). Reaching success in the parameter First Trial Success remained on this low level in the shamTX group over time, whereas the movement score analysis revealed a progressive deterioration of reaching performance (–23%) in the same group after 23 weeks.
Whishaw and colleagues (66), interestingly, reported a transient impairment in HD rats in Total Success, which was completely restored after 3 weeks of reaching training. We did not observe this effect. We can only speculate that this difference might be due to the fact that the authors restarted training the rats in the single pellet reaching task 1 day postsurgery (we started 4 weeks postsurgery), thereby promoting recovery through self-repair, for example, increasing intrinsic brain-derived neurotrophic factor (BDNF) levels by daily motor training during the period of maximal neuroinflammation and in that way protecting neurons from dying (12), or simply through repeated testing, presumed to involve motor learning (13). Furthermore, their period of assessment ended 3 weeks after the lesion, whereas in the present study we report stable lesion effects up to 23 weeks after the injection of QA. In the same study, Whishaw et al. (66) also described a chronic impairment in First Trial Success rate and movement element score. In line with their observations, we observed motor deficits in all movement components except for orient and grasp. Particularly, during withdrawal of the hand, the rats had difficulties rotating the distal parts of their limbs. It is noteworthy that transport of the reaching hand toward the pellet, although still significant, was less affected. This might be due to the fact that transport is guided by olfaction (66). Olfaction per se, as seen in the movement element orient including the subelements head orientation and sniffing, was not impaired by the QA injection at all, suggesting a more or less normal initiation of this component of the reaching movement. Nevertheless, the rats displayed significant difficulties in abducting their elbows during transport, and often, the rats would over extend their arm (overshoot the target), showing similarities to HD subjects tested in a similar real-world reach-to-eat paradigm (36). In contrast, the movement component withdrawal may be under the control of propioceptive cues, which are severely impaired after QA lesion, hence the greater impairments during supination of the hand toward the end of an individual reach (66). Reaching has been described as a composite action of different movement patterns that needs training to build a smooth goal-oriented movement. Gharbawie and Whishaw (23) introduced this interesting concept, proving that the different components underlie different neuronal networks and striatal areas responsible for carrying out such movements. Therefore, it is understandable that causing striatal lesions impinges on those areas differently such that movement components are variously affected (leaving some unaltered, e.g., orient and grasp, but impairing others severely).
Although no choreic symptoms are observed in the QA lesion model (52), the postural balance of lesioned rats was impaired, with deficits in aligning their hindlimbs and shifting their body weight during transport and being unable to lift their ipsilateral hand to assist putting the pellet into the mouth during release (the limb was used for body support instead). Postural impairments are also obvious in human HD subjects (36); these impairments, however, might be exaggerated in humans by the choreic movements, which are absent in rats.
The analysis of spontaneous (unskilled) behavior revealed a unilateral and sustained deficit on the rats' forelimb performance in stepping (adjusting steps down by −84%) and vibrissae-evoked hand placing (sensorimotor-induced limb movement was down by −39%). Limited previous studies have analyzed forelimb akinesia in QA-lesioned rats; Döbrössy and Dunnett (13) describe a similar almost full ablation of adjusting steps, while Rossignol et al. (50), using a different HD rat model, report a slightly smaller impairment. One reason for the observed forelimb akinesia could be that the QA injection interferes with and disturbs corticostriatal connections. Although these connections seem to be spared by the toxin, the functional integrity of these passageways of (motor) information might be compromised (66). As these tight links between cortex and striatum share similar topographic organization, damage on either structure can cause deficits in both; therefore, a malfunction of these connections and motor maps leads to deficits in simple motor behavior as seen in the adjusting step test or in the vibrissae-evoked hand-placing test (26, 49, 66).
Transplantation
The capacity of wGE-derived striatal grafts to integrate well into the host tissue and to restore motor function is well established (7, 9, 15, 16, 22, 31, 32, 34, 41, 58, 67). In particular, motor and cognitive tests of corticostriatal function and skilled reaching in the staircase test have been found to reveal significant improvements after transplantation (7, 28, 29, 44). The functionality of striatal grafts has often been interpreted in terms of restoration of corticostriatal connectivity. Striatal grafts need to be homotopically placed in the striatum, where they form appropriate connections to corticostriatal and striatopallidal pathways, and are fully functional as seen by synapse formation, electrophysiological recordings, and behavioral measures (17, 40, 67). Here, we also report somatotypical connections as observed by FG immunohistochemistry. Particularly, striatal-like P-zones within the grafts were rich in FG+ fibers, indicating correctly targeted fiber outgrowth to the globus pallidus where FG was injected into (25, 47). In contrast to a previously published study (8), we did not observe compromised graft integration (and functionality) due to “delayed” grafting 42 days postlesion compared to the commonly used 10-day interval; graft volume and histological analyses were in line with previously published data using the short interval between lesion and transplantation. Brasted and colleagues (8) compared 9-day and 70-day intervals, reporting good graft survival in both groups, but with the absence of functional restoration in the 70-day interval group. They argue that striatal QA lesions seem to initiate a late bout of degeneration of corticostriatal afferents 2 months postsurgery, beyond which transplanting is simply too late and grafts are unable to reconstruct these pathways. Our study would suggest that 42 days postlesion still falls within the early period in QA-induced neurodegeneration, during which striatal afferents can be rescued and motor function restored. Graft volume and survival were of average level compared to the literature (14, 22). Noteworthy is that although all grafts were correctly placed into the atrophied striatal tissue, they were unable to prevent the collapse of striatal tissue, and a pathologically enlarged ventricle was seen in both graft and shamTX animals.
Behaviorally, striatal grafts have mostly been evaluated in simple motor tests (e.g., drug-induced rotation or cylinder test) or in skilled reaching success in the staircase test in a purely quantitative manner (13, 22, 28). Yet, a detailed qualitative analysis of changes in skilled reaching after grafting in a real-world reaching task that is translatable to the human condition has hitherto been lacking. One of the notable key features of restoration of motor function with striatal wGE-derived grafts is that the presence of a new (and well-integrated) GABA-releasing source (i.e., the graft) is not enough to replace lost MSNs and restore (motor) function: the rats have to learn how to use the graft; improvement of behavior (motor and cognitive) occurs only with repeated training over time (8, 15, 39). Endpoint measurements in the present study reveal an improvement by roughly 60%; yet, there was no difference in reaching success between the shamTX and graft groups during the initial retraining period 3 months after the transplantation (data not shown). Only with repeated reaching training did the graft group develop greater success in reaching for food while the shamTX group remained impaired.
Most strikingly in the present study was the fact that the wGE grafts fully restored posture and qualitative aspects of reaching patterns during the evaluated reaches: movement scores of grafted rats did not differ from reaching performance of intact animals and were distinctly different from those of rats that underwent sham graft surgery. This is somewhat surprising as the success rate did not improve to the same degree and remained below the control rats' level. It is important to remember that the reaching score was recorded on all reaches during which the rats grasped the pellet at first attempt. All failures or multiple attempts were not scored. So, in other words, when a rat manages to reach for a pellet on its first attempt, it always does it in a manner equal to con rats (i.e., normal). The graft seems to offer a higher rate of First Trail Success during which the skilled reaching is normal. This normal behavior, though, cannot be maintained for long, and QA-lesion deficits take over again, causing failure in reaching for the pellets at first attempt and making the rats' performance oscillate between good and bad reaches. Looking at the Total Reaching Score in Figure 6B, it is noteworthy that the shamTX animals' score decreased over time (although not significantly after the lesion, but compared to baseline values), whereas the performance of grafted animals improved slightly posttransplantation (although not significantly). These mild changes, nevertheless, were sufficient to see a significant difference between those groups 3 months after the transplantation and 2 weeks of final training. Interestingly, there were quite a few subelements that showed similar patterns of development: they were not or only mildly impaired after the lesion, but 3 months later (postTX) some subelements had deteriorated in lesioned rats but not in those with grafts; that is, the transplant enabled the maintenance of normal behaviors or the stabilization of impaired behaviors. These findings imply long-term effects of QA-mediated lesions on motor behavior resulting in a progressive loss of motor function with time. It remains speculative whether this loss of function has pathophysiological causes [i.e., progressive on-going plastic and/or neurodegenerative processes in the lesioned (nongrafted) brain] or whether the rats acquired learned helplessness or nonuse (i.e., they behaved as if they had been learning that they had a motor disability, leading to decreased performance) (20, 38). However, the effects in our study were small, and further investigations are needed, as most of the literature addresses learned nonuse in models of stroke or depression-like behavior.
In contrast to an earlier study from our lab (13), we could not reproduce graft-mediated improvement in forelimb akinesia: the extinction-like loss of adjusting steps could not be rescued by the grafts. We can only speculate that this might be due to subtle differences in how the stepping was carried out (different handling by the experimenter) or in graft placement. Interestingly, hand placing after stimulating the rats' vibrissae did benefit from the grafts. Based on the results from a modified version of the vibrissae-evoked hand-placing test, Schallert and colleagues suggest that sensory information is processed in striatal areas separately from motor programs (68); the authors also conclude from their results that deficits on the contralateral side in this test are “primarily motor, not requiring sensory neglect of vibrissae stimulation,” as the (“bad”) vibrissae of the lesioned side can still elicit motor responses cross-midline (53). Considering the complexity of topographical organization of the striatum, it could well be that not only sensory integration but also different types of motor behavior respond differently to grafts in the lesioned striatum, improving hand placing but not stepping.
Summary
This study investigated graft-mediated recovery on skilled reaching in a model of HD. We report that the intrastriatal transplantation of E15-wGE-derived fetal cells integrate well, form appropriate neuronal connections in their homotopic environment, and develop a striatal-like phenotype as seen in DARPP32+ cells. Functionally, the grafts were able to partially restore skilled reaching success by up to 60%. Surprisingly, the grafts had the potential to restore reaching performance fully (posture, proximal and distal movements) when the pellet was grasped at first attempt. Furthermore, sensorimotor deficits could also be partially repaired, whereas forelimb akinesia remained impaired with no graft effects whatsoever. We conclude that placing the graft into the lateral portion of the striatum provides partial somatotopic restoration of skilled reaching movements and sensorimotor stimulation.
Skilled reaching is a valuable measure of motor function in neurodegenerative diseases and its therapies. Given the homology between rodent and human reaching, the analysis of skilled forelimb use serves a very useful tool in research, readily translatable from rodent to human and vice versa. With the present model, we can further unravel the restorative potential of striatal grafts, which have been a promising new approach in clinical studies already (5, 18).
Footnotes
Acknowledgments
The authors would like to thank the Medical Research Council UK and the EU FP7 Replaces Programme for financial support. The authors thank Jane Heath for excellent technical support. The authors declare no conflict of interest.
