Abstract
Acute paraquat (PQ) poisoning induces redox cycle and leads to fatal injury of lung. Clinical management is supportive in nature due to lack of effective antidote, and the mortality is very high. Mesenchymal stem cells (MSCs) process the properties of immunomodulation, anti-inflammatory, and antifibrotic effects and oxidative stress resistance. MSC transplantation may theoretically serve as an antidote in PQ intoxication. In this study, we examined the potential therapeutic effects of MSCs in PQ-induced lung injury. The degree of PQ toxicity in the rat type II pneumocyte cell line, L2, and MSCs was evaluated by examining cell viability, ultrastructural changes, and gene expression. L2 cells treated with 0.5 mM PQ were cocultured in the absence or presence of MSCs. For the in vivo study, adult male SD rats were administered an intraperitoneal injection of PQ (24 mg/kg body weight) and were divided into three groups: group I, control; group II, cyclophosphamide and methylprednisolone; group III, MSC transplantation 6 h after PQ exposure. MSCs were relatively resistant to PQ toxicity. Coculture with MSCs significantly inhibited PQ accumulation in L2 cells and upregulated the expression of antioxidative heme oxygenase 1 and metallothionein 1a genes, reversed epithelial-to-mesenchymal transition, and increased the viability of PQ-exposed L2 cells. Treatment with MSCs resulted in a significant reduction in severity of liver and renal function deterioration, alleviated lung injury, and prolonged the life span of rats. Altogether, our results suggest that MSCs possess antidote-like effect through multifactorial protection mechanism. The results of this preclinical study demonstrate that transplantation of MSCs may be a promising therapy and should be further validated clinically.
Introduction
Exposure to paraquat (PQ), a highly poisonous herbicide, may result in fulminating pulmonary fibrosis. Thousands of deaths due to intentional or accidental ingestion of PQ have been reported. The mechanisms mediating PQ-induced toxicity involve the inhibition of NADP (nicotinamide adenine dinucleotide phosphate) to reduced nicotinamide adenine dinucleotide phosphate (NADPH) and the formation of superoxide radicals, hydrogen peroxide, and hydroxyl radicals (22,29,47). This sustained redox-cycling effect leads to oxidative stress-related cell death, the recruitment of inflammatory cells, and subsequently, irreversible pulmonary fibrosis.
PQ primarily targets pneumocytes. Toxicity is the consequence of its accumulation against a concentration gradient through the highly developed polyamine uptake system (16). In acute stages, the most reliable predictor of outcome is the concentration of PQ in plasma and urine (27,60). Thus, the initial treatment of choice is hemodialysis, hemofiltration, and the administration of absorbents; the results of those treatments, however, are often disappointing and the mortality rate remains very high (24,32,72). Although other treatments such as antioxidants (46,65), glucocorticoids, and cytotoxic drugs (15,40,42) have shown some beneficial effects, acute PQ poisoning continues to carry a dismal prognosis (32,33). To date, there is no effective antidote.
Mesenchymal stem cells (MSCs) are multipotent cells capable of differentiating into a variety of mesenchymal lineages including cartilage, bone, muscle, tendon, ligament, and adipose tissue. More importantly, MSCs have been shown to process potent immunomodulatory (3,75), anti-inflammatory (31) and antifibrotic capacities (39,48) by secreting large quantities of cytokines and growth factors. Recently, MSCs have been demonstrated to be highly resistant to oxidative damage and to be potent scavengers of free radicals (71). A growing body of evidence suggests that MSCs have therapeutic potential in a wide range of lung diseases including bleomycin-induced lung fibrosis and idiopathic pulmonary fibrosis through either differentiating into alveolar epithelial cells or through a paracrine effect (49,70). Furthermore, the majority of intravenously injected MSCs are usually sequestered in the lungs after systemic circulation (20). Direct delivery of MSCs to the lung appears to confer effective protection against PQ-induced pulmonary injury. Given the unique characteristics of MSCs, we hypothesized that these cells may be an effective antidote for PQ poisoning. We first assessed the kinetics of PQ concentration and the ultrastructural changes in PQ-treated pneumocytes and MSCs. The effects of MSCs on PQ accumulation, cell death, and oxidative stress in cultured rat pneumocytes were studied. Subsequently, we investigated the effect of MSCs on PQ-induced lung toxicity in rats.
Materials and Methods
PQ Chemicals
PQ dichloride (1,1′-dimethyl-4,4′-bipyridinium dichloride) was obtained from Sigma-Aldrich (St. Louis, MO, USA).
Mesenchymal Stem Cell Culture
The isolation and characterization of MSCs from bone marrow were performed as previously reported (35,36). Bone marrow (BM) aspirates were obtained during fracture surgeries from two normal donors aged 22 years (male) and 56 years (female) with informed consent. Approval by the Institutional Review Board of the Taipei Veterans General Hospital was obtained prior to the commencement of this study. The MSCs used in this study were clonally derived and their surface immune phenotype and multilineage differentiation potentials into osteoblasts, adipocytes, chondrocytes, and hepatocytes were previously characterized as previously reported (35,36). Each experiment was repeated at least three times with MSCs from these two donors.
Culture of Rat L2 Pneumocytes
Normal rat lung alveolar type II cells (L2 cells) were purchased from the Bioresource Collection and Research Center of Food Industry Research and Development Institute, Hsin-Chu, Taiwan and maintained in F-12K medium (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Invitrogen) at 37°C in 5% CO2. Cells were passaged routinely and the media were changed every 2–3 days.
Time Course of Dynamic Change of PQ Concentration in Supernatants of L2 Cells and MSCs
MSCs and L2 cells were separately plated on 12-well culture plates (Falcon, Becton Dickinson, Franklin Lakes, NJ, USA), and the cell numbers were counted before seeding. The basal F-12K medium was replaced with culture medium containing PQ (0.5 mM) before the cells adhered to the plate. Every 6 h for the first 12 h and then every 24 h in the subsequent 5 days, the supernatant was collected and the PQ concentration was assayed using ultravioletvisible spectroscopy. In brief, samples were mixed with NaCl (Merck, Darmstadt, Germany) and alkaline dithionite working solutions (Merck), which produce a blue cation in the presence of PQ; absorbance was read at 396 nm by a Beckman DU 800 Spectrophotometer (Beckman Coulter, Brea, CA, USA). In the meantime, the cells on the bottom were detached by trypsinization (Life Technologies, Grand Island, NY, USA), incubated with trypan blue [Sigma-Aldrich; 0.04% in phosphate-buffered saline (PBS; Gibco BRL, Gaithersburg, MD, USA)] at room temperature, and counted with a hemocytometer 2 min later to obtain viable cell numbers.
Ultrastructural Damage to L2 Cells and MSCs After PQ Exposure
To study the antioxidant capacity of MSCs and L2 cells. MSCs and L2 cells were respectively cultured with culture medium containing PQ at a concentration of 0.5 mM for 48 h. After washing in prewarmed PBS, cells were fixed with 2.5% glutaraldehyde (Sigma-Aldrich) in 0.05 M cacodylate buffer (Sigma-Aldrich), with 0.1 M NaCl (Sigma-Aldrich), pH 7.5 at 37°C for 2 h and postfixed in 2% osmium tetroxide (Sigma-Aldrich) in the same buffer for 2 h and subsequently embedded in Spurr's Epon (Electron Microscopy Sciences, Hatfield, PA, USA). The semithin sections (1 μm) were used for evaluation and selection of tissue blocks for thin sections (90 nm). Semithin sections were stained with toluidine blue (Merck), and thin sections were stained with uranyl acetate (Riedel-de Haën; Seelze, Germany) and lead citrate (Merck). Cells were then examined using a transmission electron microscope (JEM-1230, JEOL, Tokyo, Japan).
Intracellular PQ Particle Assay
L2 cells were cultured in basal medium containing PQ (0.5 mM) throughout the experiment. After PQ exposure, MSC transwell coculture (see below) started at 6, 12, and 24 h and intracellular PQ particles were examined at 48, 60, and 72 h. Cells were then fixed for 10 min with 3.7% paraformaldehyde (Sigma-Aldrich) in PBS, pH 7.4, at room temperature, and permeabilized for 4 min in 0.1% Triton X-100 (Sigma-Aldrich) (in PBS). PQ particles were visualized in cells by staining for 10 min with solutions of sodium dithionite (Sigma-Aldrich) in 0.5 M of sodium hydroxide (Sigma-Aldrich), pH 10. The amount of PQ particles in the L2 cells was determined by counting the number of stained spots in each cell. A total of at least 500–1,000 cells in each group were counted under a high power (400×) microscope (Nikon, Tokyo, Japan).
Coculture of L2 Cells and MSCs
To analyze the mechanisms governing the protective effect of MSCs against PQ in L2 cells, L2 cells were pretreated with 0.5 mM PQ for 6 h followed by replacement with fresh PQ-free culture medium. The cells were then cocultured with or without MSCs at 2:1 ratios using a transwell coculture system (Falcon). MSCs were plated on the permeable membrane of transwell insert containing culture medium consisting of Iscove modified Dulbecco medium (IMDM; Gibco) supplemented with 10% FBS, 100 U penicillin, 1,000 U streptomycin, 2 mM l-glutamine (Gibco). Basic fibroblast growth factor (bFGF; 10 ng/ml; R&D Systems, Inc., Minneapolis, MN, USA) and 10 ng/ml epidermal growth factor (EGF; R&D Systems, Inc.). After overnight incubation, PQ-pretreated L2 cells were cocultured with MSCs in F-12K medium supplemented with 10% FBS for 3 or 5 days. The cell lysates were used for Western blot analysis. The extraction of total RNA followed by reverse transcription of total RNA into cDNA was used for quantitative polymerase chain reaction (PCR).
Western Blot Analysis of α-Smooth Muscle Actin Expression (α-SMA)
Expression of α-SMA protein in PQ-exposed L2 cell lysates was analyzed by Western blotting. L2 cells were pretreated with PQ for 6 h and then underwent transwell coculture with MSCs. After coculture with MSCs for 3 days, the L2 cells at the bottom of basal chamber were washed once with ice-cold PBS. Cells were lysed by the addition of 100 μl of CelLytic Reagent (Sigma-Aldrich) containing protease inhibitor cocktail (Sigma-Aldrich) for 15 min at 4°C, and the cells were collected using a disposable scraper (Falcon). Lysates were cleared by centrifugation for 10 min at 12,000×g at 4°C (Eppendorf, Hamburg, Germany). Protein concentration was determined using a Bio-Rad Dye Reagent (Bio-Rad Laboratories, Inc., Hercules, CA, USA). Protein (20 μg) was mixed with sample buffer (Bionovus Biotechnology, Cherrybrook, New South Wales, Australia), boiled for 5 min, electrophoresed on 8% sodium dodecylsulfate (SDS) polyacrylamide gel (Bio-Rad Laboratories, Inc.), and electroblotted onto nitrocellulose membranes (Millipore, Bedford, MA, USA). The membranes were blocked by PBS containing 5% skimmed milk powder (Anchor, Kowloon, Hong Kong), 1% FBS, and 0.02% Tween 20 (Sigma-Aldrich) for 1 h at room temperature. The membranes were incubated with 1A4 anti-α-SMA mAb (Sigma-Aldrich). After washing, the membranes were incubated with a secondary antibody (R&D Systems, Inc.) in PBS containing 1% normal goat serum (Sigma-Aldrich) and 1% FBS. The membranes were washed with PBS buffer, and the signals were visualized using a chemiluminescence (ECL) system with an ECL-detecting reagent (Thermo Scientific, Rockford, IL, USA). β-Tubulin (Sigma-Aldrich) was used as a loading control.
RNA Isolation and Quantitative Polymerase Chain Reaction (qPCR) for Heme Oxygenase-1 (HO-1) and Metallothionein 1A (MT1a)
Total RNA was harvested using Trizol reagents (Invitrogen) according to the manufacturer's instructions. Total RNA (2 μg) was then reverse transcribed using a cDNA synthesis kit (MMLV; Epicentre; Madison, WI, USA). TaqMan technology (Applied Biosystems, Foster City, CA, USA) was used for quantitative PCR reactions. Specific primers for HO-1, MT1a, and housekeeping gene β-actin were designed by Universal Probe Library (http://www.roche-applied-science.com) (Table 1). Amplification was performed in a thermal cycler (Eppendorf). The thermal cycling condition was 1 cycle for 30 s at 95°C, 40 cycles at 95°C for 1 s, and 40 cycles at 60°C for 15 s. The levels of HO-1 and MT1a mRNA expression were normalized to levels of β-actin for each sample.
Primers Used in qPCR Analysis

HO-1, heme oxygenase-1; MT1a, metallothionein 1a.
MTS Assay for Cell Viability of PQ-Treated L2 Cells
To assay cell viability of PQ-treated L2 cells in the presence of MSCs, L2 cells were pretreated with 0.5 mM PQ for 6 h, and then the media were replaced with PQ-free culture medium and the cells were then cocultured with or without MSCs at 2:1 ratios for 3 days. They were then pulsed with 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS; Promega, Madison, WI, USA) (5 μg/ml per well) for 2 h. Readings were measured at 490 nm on a Bio-Rad Model 680 microplate reader.
Animals and Experimental Protocol
All animal experiments were reviewed and approved by the Animal Care Committee of the Taipei Veterans General Hospital. Male Sprague–Dawley rats (n = 22) were obtained from the National Yang-Ming University Laboratory Animal Center (Taipei, Taiwan). All rats received an intraperitoneal injection of PQ (24 mg/kg). The experimental animals were randomly divided into three groups. The three groups were treated as follows. Group I (n = 7) was administered PQ only. Group II (n = 8) was administered PQ and then treated with intravenous injection of methylprednisolone (MP, Pharmacia and Upjohn Company, Kalamazoo, MI, USA; 25 mg/kg) and cyclophosphamide (CP, Baxter Oncology GmbH, Halle, Germany; 15 mg/kg) 6 and 24 h after PQ administration. Group III (n = 7) was treated with a single dose of MSCs (5×106 in cell dose) intravenously 6 h after administration of PQ. The rats were sacrificed 30 days after PQ exposure to evaluate the therapeutic effectiveness of MSC transplantation.
Measurement of Biochemical Parameters
Blood samples were collected at 0, 12, and 24 h after PQ administration. All samples were processed in the Division of Clinical Biochemistry, Taipei Veterans General Hospital, Taipei, Taiwan. Levels of blood urea nitrogen (BUN), creatinine, alanine transaminase (ALT), asparate transaminase (AST), and lactate dehydrogenase (LDH) were analyzed with Hitachi 7600 clinical analyzer (Taipei, Taiwan).
Tissue Processing for Histological Analysis
Lungs were removed within 3 h after rats had been sacrificed. Paraffin-embedded sections (3 μm thick) were stained with a hematoxylin and eosin kit (Sigma-Aldrich) and a Masson's trichrome stain kit (Dako, Glostrup, Denmark). The severity of fibrosis was evaluated semiquantitatively on a scale from 0 to 8 according to the method proposed by Ashcroft et al. (5), in which 0 represented no lung abnormality; 1 indicated the presence of inflammation and fibrosis involving <25% of the lung parenchyma; 2 represented lesions involving 25–50% of the lung; and 3 denoted lesions involving >50% of the lung. Histopathologic evidence of acute lung injury was quantified according to a previously described method (9,15). In brief, the severity of tissue disorganization was scored according to the percentage of the affected tissue: 0 = normal structure; 1 = less than one third of tissue affected; 2 = greater than one third but less than two thirds of tissue affected; 3 = greater than two thirds of tissue affected. The severity of inflammatory reaction was scored as follows: 0 = no cellular infiltration; 1 = mild leukocyte infiltration (one to three cells by visual field); 2 = moderate infiltration (four to six leukocytes by visual field); and 3 = heavy infiltration by neutrophils. The severity of necrosis was scored as follows: 0 = no necrosis; 1 = dispersed necrotic foci; 2 = confluent necrotic areas; 3 = massive necrosis.
PCR Experiment for Engraftment of MSCs
Genomic DNA from rats that had received MSCs was analyzed using PCR with primer sets specific for rat and human β2-microglobulin to evaluate engraftment of human MSCs to the lung. The primers for human β2-microglobulin and rat β2-microglobulin were as follows: human: forward, GTGTCTGGGTTTCATCCATC and reverse, GGCAGGCATACTCATCTTTT; rat: forward, CCAGTTTAACTCCAGATCCGG, and reverse, TTCTGATCAAAACACTCATTGAAGC. Amplification of β2-microglobulin was performed with initial denaturation at 95°C for 1 min, followed by 35 cycles of 30 s at 95°C, 1 min at 57.1°C, and 30 s at 72°C, with a final extension at 70°C for 5 min. Hep3B cells (Bioresource Collection and Research Center of the Food Industry Research Institute, Hsin-Chu, Taiwan), a human hepatocellular carcinoma line, were used as a positive control.
Statistical Analysis
Data are presented as mean ± SD. We used the chi-square test for linear-by-linear association to examine PQ concentration change over time. We used a two-tailed Student's t test for two groups and one-way ANOVA test for more than two groups. If the ANOVA analyses were significant, Tukey's post hoc analyses were performed. Animal survival was analyzed by the Kaplan–Meier method, and log-rank tests were performed for statistical comparisons of the curves. Data analysis was performed using SPSS software (Version 18.0, Chicago, IL, USA). A value of p < 0.05 was considered statistically significant.
Results
MSCs Were PQ Resistant
Changes in cell number of PQ-treated L2 cells and MSCs as well as PQ concentration kinetics are illustrated in Figure 1. PQ induced cell death in L2 cells and in MSCs. The results showed that L2 cells were more sensitive to PQ than MSCs. PQ induced a significant decrease in L2 cell number at all time points (p < 0.05). The majority of L2 cells were destroyed after 2 days. The dynamic change in cell numbers revealed that MSCs were more resistant to oxidative stress than L2 cells. The cell number in the MSCs group remained stable until the fourth day. In the L2 cells group, the decrease in PQ concentration was much sharper than that in the MSCs group during the first 2 days (p < 0.05). The apparent efflux of PQ from lysed L2 cells at day 3 caused an increase in PQ concentration in L2 cell supernatant. There was a significant change in PQ concentration in L2 supernatant over time (p = 0.014, based on linear-by-linear association). In the MSC group, the PQ concentration dropped quickly on the first day and remained steady.
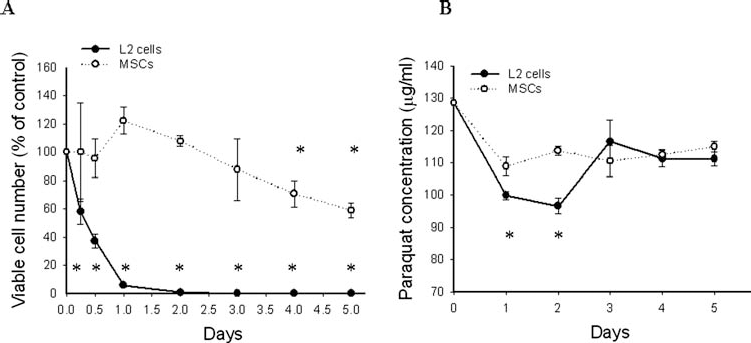
Dynamic change of PQ concentration in supernatant and cell numbers. (A) Viable L2 cell and mesenchymal stem cell numbers. (B) Paraquat (PQ) concentration in L2 cells and MSC supernatant. *p < 0.05 compared with control.
Ultrastructural Damage in L2 Cells 48 h After Treatment with PQ
Figure 2 shows the electron micrograph of PQ-treated L2 cells and MSCs. After 48 h of PQ exposure, there was marked cell damage in L2 cells with evidence of faint nuclear envelopes, swollen and tortured mitochondria, vacuole changes in cytoplasm, the appearance of dark granules in the cytoplasm, and loss of cell-to-cell contact. In contrast, the structural integrity of organelles in MSCs remained relatively normal.

Electron micrograph of PQ-treated L2 cells and MSCs. For exposure to oxidants, PQ at concentration of 0.5 mM was added to the culture medium. (A) L2 cells alone, (B) L2 cells after PQ exposure for 48 h, (C) MSCs alone, (D) MSCs after PQ exposure for 48 h (scale bar: 1 μm).
MSCs Inhibited PQ Accumulation in L2 Cells
Figures 3 and 4 show the results of the intracellular PQ particle assay. We examined the accumulation of PQ particles into L2 cells at 48, 60, and 72 h after exposure to 0.5 mM PQ alone and in L2 cells that had been exposed to MSCs from 6, 12, or 24 h. Reduction of PQ by sodium dithionite in alkaline solution formed a stable blue radical ion, indicating the presence of PQ. The PQ particles accumulated with time into cultured L2 cells in the absence of MSCs and in cells exposed to MSC treatment. The accumulation of PQ into L2 cells was reduced by coculture with MSCs. The number of PQ particle accumulation in L2 cells at 48 h was 21.57 ± 9.99 in the control group, 8.32 ± 5.38 in cells treated with MSCs at 6 h, 19.73 ± 10.43 in the group treated with MSCs at 12 h, and 15.00 ± 8.36 in cells treated with MSCs at 24 h after exposure to PQ. Treatment with MSCs 6 and 24 h after exposure to PQ significantly inhibited the accumulation of PQ in L2 cells at 48, 60, and 72 h (p < 0.05). In addition, there was a significant reduction in the number of PQ particles at 60 and 72 h in L2 cells that had been treated with MSCs at 12 h after exposure to PQ (p < 0.05). Early coculture with MSCs significantly decreased PQ accumulation in L2 cells.
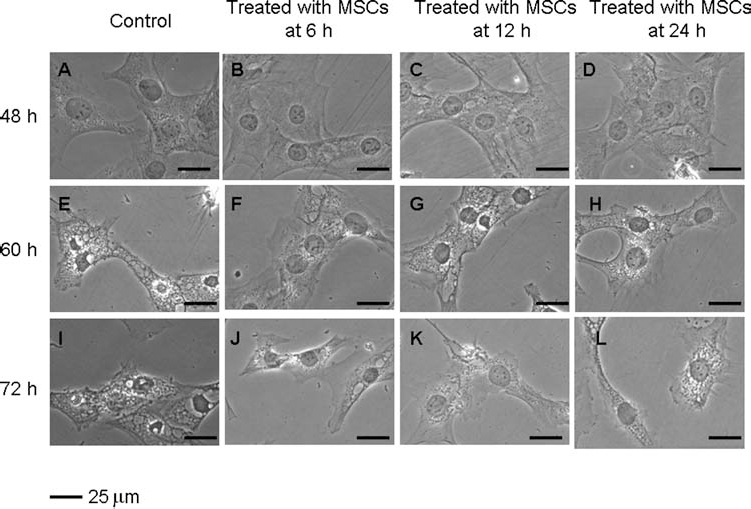
The effect of MSCs on PQ accumulation in L2 cells. L2 cells treated with 0.5 mM PQ were cocultured without or in the presence of MSCs for various times (6, 12, and 24 h after exposure to PQ), and respresentative images are shown at 48, 60, and 72 h. (A–D) PQ-exposed cells with or without treatment with MSCs after 48 h. (E–H) PQ-exposed cells with or without treatment with MSCs after 60 h. (I–L) PQ-exposed cells with or without treatment with MSCs after 72 h. *p < 0.05 as compared with control (scale bar: 25 μm).

Quantitative analysis of intracellular PQ accumulation in L2 cells. L2 cells incubated with 0.5 mM PQ were treated with MSCs 6, 12, and 24 h after PQ exposure. Accumulation of PQ into L2 cells at 48, 60, and 72 h was analyzed by sodium dithionite staining. *p < 0.05 as compared with normal controls; **p < 0.05 versus cells treated with MSCs at 6 h; ***p < 0.05 versus cells treated with MSCs at 12 h.
MSCs Induced Early Gene Expression of HO-1 and MT1a
As shown in Figures 5 and 6, we analyzed the expression levels of HO-1 and MT1a mRNA in PQ-exposed L2 cells in the absence or presence of MSCs using qPCR. Relative to the control group on day 3, HO-1 mRNA expression was 0.78 ± 0.10-fold lower in L2 cells that had been exposed to PQ only and 1.10 ± 0.07-fold higher in PQ-exposed L2 cells that received MSC treatment (p < 0.05) (Fig. 5A). The expression of MT1a mRNA (1.51 ± 0.12) in the MSC-treated group was also significantly higher than that in the control and PQ groups on day 3 (p < 0.05) (Fig. 6A). Both HO-1 and MT1a gene expression in L2 cells was significantly elevated in the presence of MSCs relative to that in the control group and the group of cells exposed to PQ alone on day 5 (p < 0.05) (Figs. 5B and 6B).
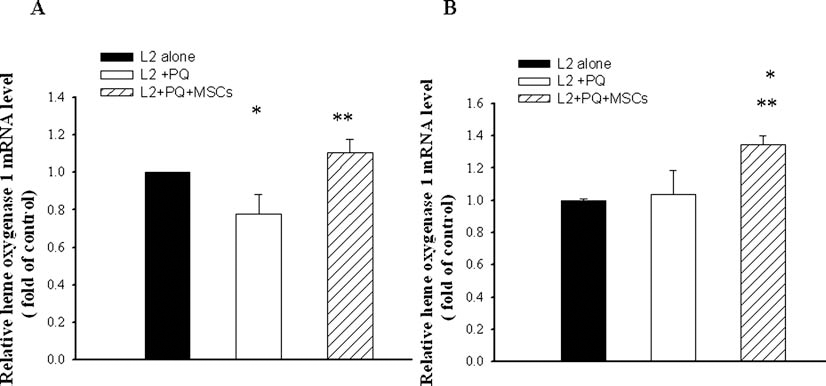
Effects of MSCs on mRNA levels of HO-1 in L2 cells. Bar graph of quantitative PCR analysis of heme oxygenase-1 (HO-1) expression relative to β-actin normalized to controls after coculture with MSCs for (A) 3 days and (B) 5 days. *p < 0.05 as compared with normal controls; **p < 0.05 versus PQ-exposed cells.
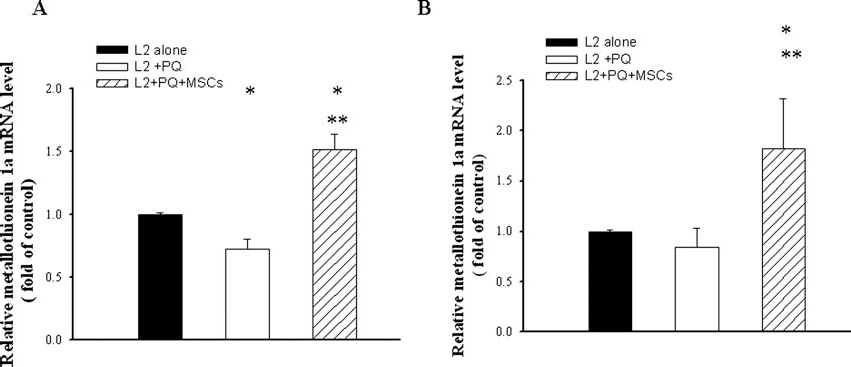
Effects of MSCs on mRNA level of MT1a in L2 cells. Bar graph of quantitative PCR analysis of metallothionein 1a (MT1a) expression relative to β-actin normalized to controls after coculture with MSCs for (A) 3 days and (B) 5 days. *p < 0.05 as compared with controls; **p < 0.05 versus PQ-exposed cells.
MSCs Prevented PQ-Induced Epithelial–Mesenchymal Transition (EMT) Marker α-SMA
As shown in Figure 7, Western blot analysis revealed that PQ resulted in an increase in the expression of α-SMA protein in L2 cells to 2.21 ± 0.31-fold compared to that of normal L2 group (p < 0.05); however, MSCs suppressed PQ induced α-SMA protein level to 1.41 ± 0.30-fold. Exposure to MSCs significantly protected against PQ induced increase expression of α-SMA protein (p < 0.05).
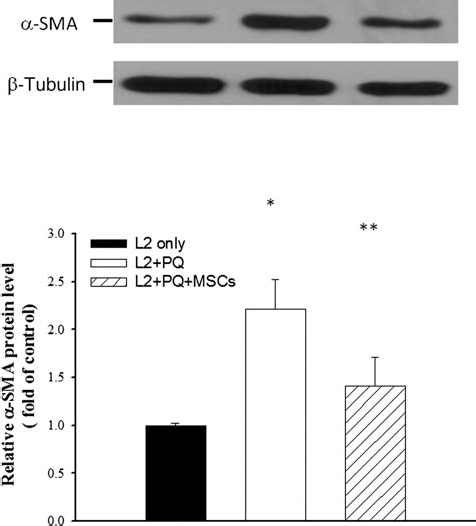
Effects of MSCs on protein expression of α-SMA in L2 cells. Representative Western blot analysis and relative bar graph analysis of α-smooth muscle actin (α-SMA expression) after coculture with MSCs for 3 days. Quantitative data are presented as mean ± SD. *p < 0.05 versus normal controls; **p < 0.05 versus PQ-exposed cells.

Effect of MSCs on PQ-induced cell death. PQ-exposed L2 cells were treated with MSCs for 3 days. Cell viability was determined by MTS assay. Data are expressed as mean ± SD. *p < 0.05 versus controls; **p < 0.05 versus PQ-treated cells.
MSCs Protected L2 Cells Against PQ-Induced Cell Death
To compare the effects of MSCs on cell death induced by PQ, PQ-treated L2 cells were cocultured in the absence or presence of MSCs. Results of the MTS assay revealed that exposure to PQ caused a significant decrease in cell viability of L2 cells (Fig. 8). Treatment of PQ-exposed L2 cells with MSCs resulted in a significant reduction in the number of apoptotic pneumocytes (p < 0.05).
MSC Transplantation Reduced the Severity of Liver and Renal Function Deterioration
Figure 9 shows the effects of MSCs on serial levels of AST, ALT, LDH, BUN, and creatinine in rats with PQ poisoning. Following PQ administration, rats developed acute liver and renal dysfunction as evidenced by elevated AST (Fig. 9A), ALT (Fig. 9B), serum BUN (Fig. 9D), and creatinine (Fig. 9E) levels. ALT and AST levels were significantly lower in MSC-treated rats than in groups I and II at 12 and 24 h post PQ exposure (p < 0.05). Peak LDH levels were noted 12 h after exposure and then declined progressively (Fig. 9C). Rats in group III had lower LDH levels than rats in group I. In addition, rats in group III had significantly lower serum creatinine levels at 24 h than rats in the control group (p < 0.05).
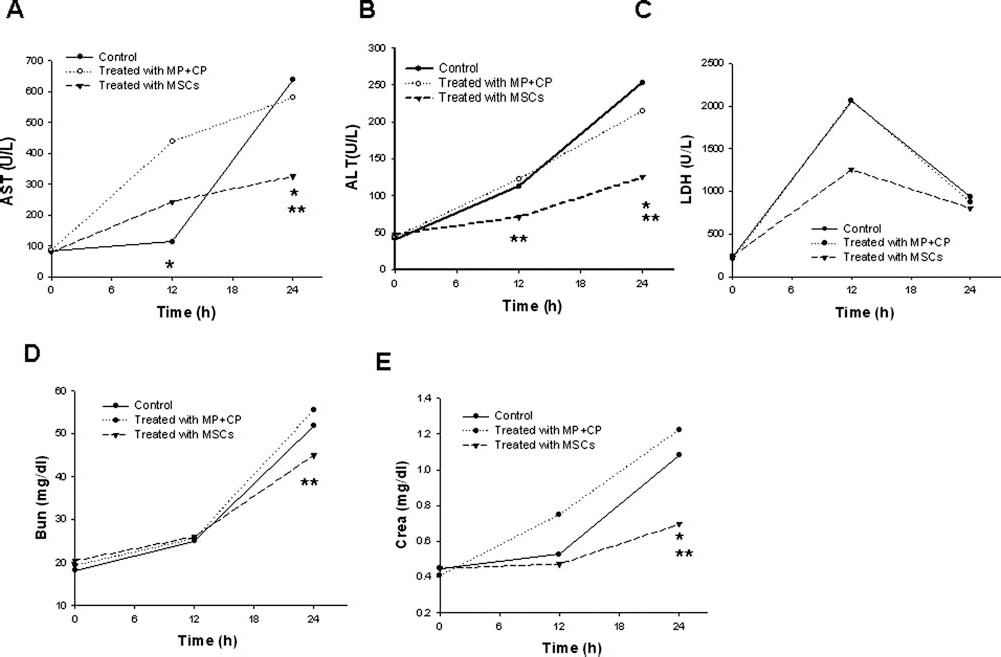
Comparisons of liver function, LDH, and renal function between groups. (A) Blood urea nitrogen (BUN), creatinine, (ALT), asparate transaminase (AST), and (LDH) AST. (B) Alanine transaminase (ALT). (C) Lactate dehydrogenase (LDH). (D) Plasma (blood) urea nitrogen concentration (BUN). (E) Plasma creatinine concentration (crea). *p < 0.05 versus controls; **p < 0.05 versus treated with methylprednisolone and cyclophosphamide (MP + CP).
MSC Transplantation Ameliorated PQ-Induced Pulmonary Injury and Fibrosis
Alterations in lung structure are depicted in Figure 10. In group I, rats had evidence of alveolar collapse, pulmonary hemorrhage, inflammatory cell infiltration, massive necrotic zones, tissue disorganization, and fibrosis. In group II, rats treated with MP + CP had similar pathologic changes. Lungs from the MSC-treated group exhibited normal pulmonary architecture and low levels of collagen content as revealed by Masson's trichrome stain. In addition, rats in the MSC-treated group showed significantly lower semiquantitative histopathological scores for tissue disorganization, cellular infiltration, necrosis, and pulmonary fibrosis than animals in group I and group II (Fig. 11).

Histopathological micrographs of lungs. Lung sections were stained with haematoxylin and eosin (left column, 100×), and Masson's trichrome stain (right column, 100×) from group I (A, B), group II (C, D), and group III (E, F). (A, C) A marked alveolar collapse with vascular congestion and severe inflammatory cells infiltration are visible. (B, D) Interstitial areas were filled with collagen fibers. On the other hand, rats treated with MSCs showed preserved pulmonary structure (E) and mild pulmonary fibrosis (F). Scale bar: 50 μm.

Semiquantitative histologic analysis of lung injury scores Three groups: control rats (group I), rats treated with methylprednisolone combined with cyclophosphamide (MP + CP, group II), and rats treated with MSCs (group III). (A) Fibrosis score, (B) inflammation score, (C) tissue disorganization, and (D) necrosis score. *p < 0.05 versus control group. **p < 0.05 versus treated with MP + CP.
MSC Transplantation Increased the Survival Rate of PQ-Intoxicated Rats
Survival rates of rats are shown in Figure 12. The 30-day survival rate was 14.3% in group I, 37.5% in group II, and 85.7% in group III. Treatment with MSCs resulted in significant improvement in overall survival of rats with PQ poisoning (p = 0.028).
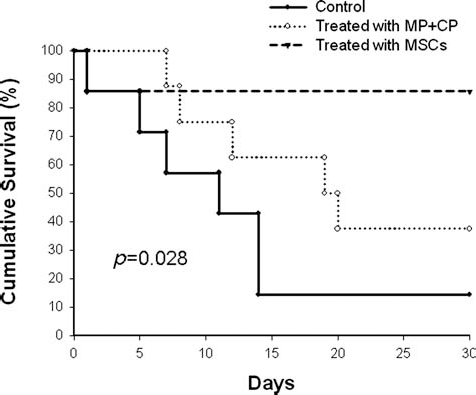
Kaplan–Meier survival analyses of treatment groups. Kaplan–Meier survival analysis demonstrated that cumulative survival was significantly higher in the MSC-treated group (p = 0.028).
Human MSCs Did Not Engraft Into Rat Lung Tissue
To assess engraftment of transplanted human MSCs to rat lungs, recipient rat received bilateral pneumonectomy at the end of the study. PCR analysis for human- and ratspecific β2-micorglobulin revealed that human cells did not exist in lung tissue at this time point (Fig. 13).

Human MSCs did not engraft into lung tissue. Human β2-microglobulin DNA was undetectable in the murine lung at 30 days after transplantation. DNA extracted from human Hep3B cell lines were used as positive controls for the human β2-microglobulin gene.
Discussion
Cytotoxic effects of PQ are attributed to the generation of superoxide radicals after reduction of PQ by intracellular oxidases. Amplified generation of reactive oxygen species further resulted in a neutrophil-mediated inflammatory response in the lung. After ingestion of PQ, peak plasma concentrations generally occur within 2 h. Previous reports have documented that type II pneumocytes actively take up PQ by an active polyamine uptake process (21,25). PQ concentration in the lung parenchyma is 10–20 times greater than in plasma. PQ toxicity correlates with PQ concentrations in lung tissue (27,63). Currently, several methods are used for early removal of PQ, namely hemodialysis, hemofiltration, and the administration of absorbents that bind the poison in blood and tissue (18,23,32,67). Unfortunately, the results of those treatments are often disappointing. Recent studies have reported that MSCs exhibit a complete host profile in bone marrow transplantation patients that received total body irradiation (14,55). That implicated MSCs are likely to possess better reactive oxygen species-scavenging capacity when exposed to ionizing radiation (10). Furthermore, in animal experiments, it has been shown that human bone marrow (BM)-derived MSCs administered intravenously are able to engraft in animals because MSCs are hypoimmunogenic and can evade the host immune elimination (4). Based on these facts, we used human BM-derived MSCs to treat immune-competent SD rats.
Data from our study also revealed that MSCs were more resistant to PQ-induced oxidative stress in vitro. The cell number in the MSCs group remained in a steady state until the fourth day. Additionally, the PQ concentrations in the supernatants of MSCs decreased but then did not change with time. In contrast, PQ concentrations in supernatants of L2 cells were lower than those in MSCs group initially for the highly developed polyamine uptake system in L2 cells. Rapid influx of PQ led to a cascade effect and uncontrollable cell death.
Subsequently, PQ was released into environment and revert the PQ concentrations that made a vicious circle till all cells destroyed. After coculture with MSCs, there was a significant reduction in accumulation of PQ in pneumocytes. Based on our findings, it appears that PQ is sequestered upon entrance into MSCs, resulting in lower concentrations of PQ in the culture system to release the environmental stress directly. Sequestration of PQ, therefore, prevents the accumulation of the toxin in the lungs, thereby resulting in decreased severity. Importantly, this study is the first time to report PQ particles stained in cytoplasm and the visible dynamic change of PQ influx can reveal strong evidence to elucidate the therapeutic effects of MSCs. We also observed that earlier intervention with MSC coculture could obtain better protective effects by decreasing PQ accumulation in L2 cells.
Several antioxidants such as vitamin C (46), vitamin E (66), N-acetylcysteine (77), and salicylic acid (17) have been tested as potential antidotes for PQ poisoning. However, there is no convincing evidence of the effectiveness of these antioxidants in PQ toxicity (19). Three HO isoforms have been identified, namely HO-1, HO-2, and HO-3. Among them, the inducible isoform HO-1 is a stress-responsive protein upregulated by ischemia, hypoxia, and oxidative stress (51). HO-1 catalyzes the rate-limiting step that breaks down heme to biliverdin, iron ions, and carbon monoxide (56). HO-1 provides protection against oxidative stress by the antioxidant activities of biliverdin and its metabolite bilirubin. Interleukin-10 (IL-10) is a cytokine with potent anti-inflammatory properties that inhibits the expression of inflammatory cytokines that are secreted by activated macrophages, namely tumor necrosis factor (TNF)-α, IL-6, and IL-1. Carbon monoxide, a by-product of heme metabolism, mediates the anti-inflammatory actions of IL-10 (37,57). Thus, these metabolites of heme exhibit cytoprotective effects through their antioxidant, anti-inflammatory, and antiapoptotic actions (50). HO-1 has been shown to protect against ischemia/reperfusion injury (8) as well as against inflammation and fibrosis in liver tissue in experimental animals (6). Another antioxidant enzyme, MT1a, is a cysteine-rich intracellular protein that serves as a regulator of metal homeostasis (zinc, copper, cadmium, and mercury). It has also been shown to be an efficient free radical scavenger. In PQ intoxication, MT1a is reported to possess protective effect in the lung (58,59). Evidence from previous in vitro and in vivo studies demonstrated homozygous HO-1 and MT knockout mice are markedly sensitive to oxidative stress and acute lung injury (54,68). In this study, we found that MSC coculture significantly increased the expression of HO-1 and MT1a mRNA on days 3 and 5 after exposure to PQ (all p < 0.05). This may represent a strengthened protective mechanism and a good indicator of this response to the environmental stress induced by PQ, and therefore, MSCs may have a role in modulating inflammation. Accordingly, induced cytoprotective genes HO-1 and MT1a by MSCs work synergistically on the maintenance of intracellular antioxidant capacity. Network of antioxidative mechanisms to afford effective protection is complex (11,12). Therefore, further studies investigate the role of MSCs in regulating the upstream signaling cascade and other antioxidant genes are warranted.
EMT is the transdifferentiation of epithelial cells into mesenchymal cells (26,44). At the molecular level, EMT is characterized by loss of epithelial marker E-cadherin and de novo expression of activated fibroblast marker α-SMA. The hallmark of pulmonary fibrosis is the myofibroblast. Emerging evidence suggests that EMT of alveolar epithelial cells represents one important source of myofibroblast formation in pulmonary fibrosis (73,74). Our results show that exposure of L2 cells to PQ results in increased expression of α-SMA, which was attenuated in the presence of MSCs.
Type I alveolar cells, which make up a majority of the alveolar surface area of pulmonary capillary, are responsible for gas exchange. Type II alveolar cells play an important role in maintaining alveolar stability by synthesis, storage, and secretion of surfactant. In the event of damage, type II alveolar cells reepithelize the alveolar surface though proliferation and transformation into type I alveolar cells within a few hours (28). Many studies have suggested that the development of pulmonary fibrosis lesions result from ineffective repair of damaged alveolar epithelium (1,62). Alveolar epithelial cell death was found in patients with idiopathic pulmonary fibrosis and in murine models of bleomycin-induced pulmonary fibrosis (38). Thus, loss of type II epithelial cells might play a significant role in pulmonary injury (64). Our data indicate that treatment with MSCs protects against cell death in L2 cells that have been exposed to PQ. Based on our findings, we conclude that treatment with MSCs can preserve or restore the alveolar epithelium following PQ injury.
Reactive oxygen species produced by PQ result in severe inflammation by activation of neutrophils and macrophages (45). A variety of inflammatory cytokines are implicated in the pathogenesis of PQ-induced lung injury (7). The immune system plays an important role in exacerbating PQ-induced lung fibrosis. Immunosuppressive treatments including CP, MP (2,40,43), and dexamethasone (41,52) are being developed and studied. Despite the beneficial effects observed in some studies, the role that those treatments play in protecting against PQ poisoning remains controversial. In the present study, the survival rate of rats treated with MP combined with CP at 1 month post PQ exposure was 37.5%, whereas the rate in rats in the control group was 14.3%. However, the survival rate did not differ significantly between rats in the control group and rats treated with MP combined with CP.
MSCs possess potential for repair and regeneration of solid tissues (34), potent immunomodulatory capacity (3,75) as well as potent anti-inflammatory (31) and antifibrotic effects (39,48), thus enabling these cells to efficiently treat a variety of diseases in animal models. Another feature that may make MSCs particularly appealing for clinical use is their resistance to oxidative stress (30,35). MSCs have been shown to be useful in the treatment of oxidative stress-related diseases such as acute myocardial infarction (13) as well as cerebral and renal ischemia (69). PQ-induced lung injury involves oxidative stress, a marked inflammatory response, and the development of pulmonary fibrosis (16). In addition, bone marrow-derived MSCs can engraft into the damaged lungs and differentiate into alveolar epithelial cells in bleomycin-induced acute lung injury model. Therefore, cell-based therapies for PQ-induced lung injury, such as the administration of MSCs, which target inflammatory pathways and act as antioxidants, are clinically relevant. In the present study, we demonstrate human BM derived MSCs treatment markedly alleviated pathologic changes in lung as well as liver and renal impairment and most importantly, increased survival rate significantly. However, lack of human-specific β2-microglobulin in lung indicated that there was no permanent engraftment of MSCs to the lungs. It is consistent with the previous findings that the allogenic MSC transplantation can inhibit the release of inflammatory cytokines, decrease lipid peroxidation, and suppress inflammatory reactions and cellular damage induced by PQ poisoning (76). The clearance of transplanted MSCs may result from either the host's innate immunity to xenogenic cells (53,60) or PQ-induced injuries to MSCs.
In conclusion, we have demonstrated that MSCs have potent antioxidant activity and protect L2 cells from oxidative injury, possibly by sequestering PQ in MSCs and by inducing the expression of HO-1 and MT1a. Therapeutic effects manifested in this study warrant further exploration of the possibility of using MSCs for treatment of patients poisoned with PQ as well as other oxidative stress-related diseases.
Footnotes
Acknowledgments
This work was supported in part by the UST-UCSD International Center of Excellence in Advanced Bioengineering sponsored by the Taiwan National Science Council I-RiCE Program under Grant Number NSC-99-2911-I-009-101. The authors also acknowledge financial support from the Taipei Veterans General Hospital (VGHV99F-007, VGH100E1-010, VGH100C-056, VN100-05, and VGH100D-003-2), the National Science Council, Taiwan (NSC-100-2314-B-075-064, NSC100-2120-M-010-001, NSC100-2314-B-010-030-MY3, NSC100-2321-B-010-019, NSC99-3111-B-010-002, NSC98-2314-B-010-001-MY3, NSC99-2911-I-010-501, and NSC99-3114-B-002-005). This study was also supported by a grant from the Ministry of Education, Aim for the Top University Plan. The authors declare no conflict of interest. The authors alone are responsible for the content and the writing of this article.
