Abstract
When expanded through passage, chondrocytes lose their ability to produce high-quality cartilaginous matrix. This study attempts to improve the properties of constructs formed with expanded chondrocytes through alterations in the expansion protocol and the ratio of primary to expanded chondrocytes used to form cartilage constructs. A chondrogenically tuned expansion protocol provided similar monolayer growth rates as those obtained using serum-containing medium and enhanced cartilaginous properties of resultant constructs. Various ratios of primary to chondrogenically expanded chondrocytes were then self-assembled to form neocartilage. Biochemical analysis showed that constructs formed with only expanded cells had twice the GAG per wet weight and collagen II/collagen I ratio compared to constructs formed with primary chondrocytes. Biomechanically, compressive properties of constructs formed with only passaged cells matched the instantaneous modulus and exceeded the relaxation modulus of constructs formed with only primary cells. These counterintuitive results show that, by applying proper expansion and three-dimensional culture techniques, the cartilage-forming potential of adult chondrocytes expanded through passage can be enhanced over that of primary cells.
Introduction
Degenerative changes associated with osteoarthritis can result in severe pain and loss of joint function due to articular cartilage's poor intrinsic healing ability (27). The fibrocartilage that forms as a result of osteochondral injuries possesses inferior frictional and compressive properties (27). Thus, the tissue degrades via fibrillation, chondrocyte death and proliferation are induced, and deep fissures develop (6). Tissue engineering has the potential to mitigate the effects of this degenerative condition by providing tissue of the necessary functional properties to repair articular cartilage defects.
While many cartilage tissue engineering modalities employ exogenous scaffolds, the scaffold-less tissue engineering technique of a self-assembling process shows great promise in its ability to form cartilaginous constructs with properties approaching native tissue (25, 44, 46). Other scaffold-free methods include pellets culture, which forms small (~2 mm) constructs on the timescale of minutes, using 100–500 × g (22, 53, 56). Forming constructs under 1 × g and on the timescale of hours, the self-assembling process gives ample time for cells to adhere and organize differently from pellet culture. Self-assembly is guided by the Differential Adhesion Hypothesis; by depriving the cells of a surface to bind to, cells must bind to each other via N-cadherins in order to minimize free energy (19, 44). This method of construct formation circumvents concerns associated with scaffolding materials, for example, stress shielding and toxic degradation products (25) and can generate constructs with clinically relevant dimensions and mechanical properties on par with native tissue (17, 25, 26).
Much of the work using the self-assembly process and many other tissue engineering studies utilize primary, nonexpanded chondrocytes from skeletally immature animals (25, 39, 41, 44, 46). However, the applicability of the self-assembly process to cells that represent a population that could more readily be translated into a clinical setting, that is, adult, expanded chondrocytes (12), must be determined. Similarly, the use of adult, expanded chondrocytes are of concern in other cartilage tissue engineering efforts. The number of cells required for construct formation varies widely among studies. Normalized by construct volume, these methods (including self-assembly) can range from 10 million cells/ml to over 100 million cells/ml (39, 41, 46). In many cases, functional properties obtained using high cell densities cannot be replicated when cell numbers are reduced below a critical mass (46). This common limitation in using primary chondrocytes as starting material represents a bottleneck that requires improvement.
An abundance of reports have shown deviations from the chondrocyte phenotype during monolayer expansion, as evidenced by the upregulation of versican and collagen types I, III, and V, with concomitant down-regulation of collagen types II, IX, and XI and aggrecan (10, 16, 33). To improve the cartilaginous properties of constructs formed from expanded cells, chondrocyte-specific, in vitro expansion parameters, for example, high seeding and passaging densities and modulation of the culture medium, have been investigated. Insulin has been shown to increase collagen type II, aggrecan, and sulfated glycosaminoglycan (GAG) synthesis (14, 18). Dexamethasone, a glucosteroid, has been shown to enhance sex-determining region Y box 9 (SOX9) expression and the production of cartilage-specific extracellular matrix (ECM) (45, 49). Basic fibroblast growth factor (bFGF) has been shown to enhance chondrocyte proliferation and reexpression of chondrocyte characteristics in 3D culture (28, 37, 39, 43, 45). Elimination of serum from chondrocyte expansion medium results in increased SOX9 and aggrecan expression and collagen II per collagen I expression ratio (21, 36, 37). High-density seeding of chondrocytes increases cell-to-cell contact during expansion and has been shown to result in less phenotypic drift and enhanced potential for recovering chondrocytic characteristics lost during passaging (23, 37, 58).
Following expansion, 3D construct formation via scaffold-less culture techniques, such as aggregate suspension culture and pellet culture, have been employed to rescue the chondrocyte phenotype with encouraging results. In 3D culture, cells begin to reexpress chondrocyte markers (GAG and collagen type II) while decreasing collagen type I expression (21, 28, 36, 37). This 3D-induced chondrogenic effect may be further enhanced by coculturing with primary cells. Most commonly, this technique is employed to differentiate stem cells (7, 13, 15, 30, 47, 56); however, a study with passaged chondrocytes has also shown success (20). These studies indicate that the juxtacrine and paracrine signals associated with primary cells have a profound effect on cells exhibiting plastic phenotypic characteristics.
These studies on chondrocyte phenotypic changes provide abundant guidance regarding methods to enhance chondrogenesis during both monolayer cell expansion and 3D construct formation. As a preliminary study identified a chondrogenically tuned monolayer cell expansion protocol, to enhance construct cartilaginous properties, this study focused on manipulating the 3D construct formation phase. The hypothesis of this study is that, by incorporating a small number of primary chondrocytes into expanded chondrocyte constructs, the detrimental effects traditionally associated with the use of expanded chondrocytes for cartilage engineering can be partially mitigated.
Materials and Methods
Chondrocyte Isolation
Male New Zealand White rabbits (Heaton Rabbitry, Huntsville, TX, USA) were obtained within 8 h following sacrifice. The Institutional Animal Care and Use Committee (IACUC) exempted the protocol from full review and approved the use of rabbit tissue for cell procurement. Chondrocytes were isolated from both the tibial and femoral articular cartilage surfaces of skeletally mature (18- to 24-month-old) rabbits using 0.2% collagenase type II (Worthington Biochemical, Lakeland, NJ, USA) in chemically defined culture medium (CM) [Dulbecco's modified Eagle's medium (DMEM) with 4.5 g/l-glucose and GlutaMAX (Invitrogen, Carlsbad, CA, USA), 100 nM dexamethasone (Sigma, St. Louis, MO, USA), 1% fungizone, 1% penicillin/streptomycin (BD Biosciences, San Jose, CA, USA), 1% insulin transferrin selenium premix (ITS+) (BD Biosciences), 50 mg/ml ascorbate-2-phosphate (Sigma), 40 mg/ml l-proline (Sigma), and 100 mg/ml sodium pyruvate (Fisher Scientific, Pittsburgh, PA, USA)]. After overnight digestion, chondrocytes from 10 rabbits were pooled and frozen at −80°C in culture medium supplemented with 20% fetal bovine serum (FBS) (Gemini Bio-Products, West Sacramento, CA, USA) and 10% dimethyl sulfoxide (DMSO; Sigma). Prior to cryopreservation, the chondrocyte viability as determined by trypan blue exclusion was >5%. After freezing at −80°C, cells were placed in liquid nitrogen cryostorage until they were needed for expansion. Prior to expansion through passage, cells were designated as “primary” chondrocytes.
Chondrocyte Expansion
Primary chondrocytes were rapidly thawed and seeded on T-225 flasks for expansion. Following thawing of cryopreserved chondrocytes, cell viability was approximately 85%. A preliminary study compared the expansion characteristics and resultant construct properties following chondrogenically tuned expansion or the standard protocol for chondrocyte expansion. The standard protocol employed CM with 10% FBS instead of ITS+ and dexamethasone, a seeding density of 1.1 × 104 cells/cm2, and passaging at 90% confluence. The chondrogenically tuned protocol employed CM supplemented with 5 ng/ml basic fibroblast growth factor (Peprotech, Rocky Hill, NJ, USA), a seeding density of 2.5 × 104 cells/cm2, and passaging 4 days after 95% confluence was met. To allow adequate cell adhesion for chondrogenically tuned passaging, it was necessary to add 10% FBS for the first 24 h of monolayer seeding. Passaging was performed using 0.25% (w/v) trypsin/0.05% (w/v) Ethylenediaminetetraacetic acid (EDTA; Gibco/ Invitrogen) at 37°C. Since both trypsin digestion and seeding can alter cellular characteristics, passage number in this study refers to the number of trypsin/EDTA exposures, that is, cells expanded to passage 3 (P3) under these conditions and three trypsin/EDTA treatments. As the preliminary study identified chondrogenically tuned expansion as superior, this method was used for the current study.
Construct Seeding
Self-assembly wells were created by filling a well of a six-well plate with approximately 17 ml molten 2% agarose (Fisher Scientific) and placing a silicone cap on top of the well. The silicone cap was cylindrical with a diameter of 35 mm and a height of 10 mm and had 15 silicone posts (3 mm diameter and 10 mm long) projecting from its underside into the agarose. After the agarose had set, the silicone cap was removed leaving negative impressions of the 3-mm diameter and 10-mm long posts in the hardened agarose. These negative impressions were the self-assembly wells. The agarose cylinders containing the self-assembly wells were removed from the six-well plate, placed into a 100-mm petri dish with 15 ml CM, and allowed to become infiltrated with CM for 1 week prior to seeding. Suspensions of primary and expanded cells were combined to obtain a range of primary to passaged chondrocyte (P0/P3) ratios (100:0, 50:50, 25:75, 10:90, 2:98, 0:100). A 50 μl aliquot of a 40 million cells/ml cell solution was seeded into a self-assembly well to form a construct containing 2 million chondrocytes. Time of seeding was designated as t = 0. Constructs remained in the 3-mm diameter agarose wells until 4 days after they grew to come in contact with the well edges, at which time they were transferred to 5-mm-diameter agarose wells. Throughout the study, all constructs were cultured in the same volume of media and received media changes every other day for 4 weeks of 3D culture. At this time, constructs were subjected to gross morphological, histological, and biochemical assessments.
Construct Weight and Gross Morphology
Prior to destructive assays, six constructs from each experimental group were blotted dry, weighed for wet weight, and photographed for gross morphology. Construct dimensions (diameter and height) were measured via image analysis with ImageJ (NIH, Bethesda, MD, USA). Constructs were then divided into sections for histological, biochemical, and biomechanical testing.
Histology
Two samples from each treatment were frozen and sectioned at 14 μm and fixed in 10% neutral buffered formalin. Safranin-O/fast green staining was used to examine GAG distribution. Picrosirius red staining was used for qualitative examination of collagen content and distribution.
Quantitative Biochemistry
Biochemical analysis included Blyscan GAG assay (Biocolor, Carrickfergus, County Antrim, UK) based on dimethylmethylene blue (DMMB) binding, a modified colorometric hydroxyproline assay (60), PicoGreen (Invitrogen) for DNA content, and enzyme-linked immunosorbent assays (ELISAs) for collagens I and II. Samples were lyophilized for 48 h and then digested in pepsin for 4 days at 4°C followed by a 1-day elastase digest. For collagen II ELISA, Chondrex reagents (Chondrex, Inc., Redmond, WA) and protocols were used. For collagen I ELISA, a similar protocol was employed with antibodies from US Biological (Swampscott, MA, USA). Both ELISAs employed bovine collagen standards that have been previously validated to accurately quantify rabbit collagen types I and II. DNA content data from the PicoGreen assay was converted to cell number using a conversion factor of 7.7 pg DNA per cell (31). Six constructs per group were digested independently and used for biochemical testing.
Unconfined Compression
A 2-mm diameter punch was taken from each construct for analysis of compressive properties. Calipers were used to measure of the diameter of the compression sample to verify a 2-mm diameter sample was consistently obtained. The platens of an Instron 5565 (Instron, Norwood, MA, USA) were placed into contact, and the platen-to-platen displacement was zeroed. Compression samples were loaded to 0.2 N to determine sample height and compressed such that deformation occurred along the height of the cylinder, at 10% strain per second, to 10%, 20%, and 30% strain. Testing was conducted in a PBS bath (preload 0.2% strain, increments held for 10 min). The curve fitting tools in Matlab (MathWorks, Natick, MA, USA) were used to determine viscoelastic compressive properties (relaxation modulus, instantaneous modulus, coefficient of viscosity) as previously described by fitting the experimental data curves to the theoretical solution of the viscoelastic Kelvin model (3). Biomechanical properties were assessed using six constructs from each group.
Real-Time Polymerase Chain Reaction
Total RNA was extracted from primary and passaged chondrocytes after expansion and prior to self-assembly (i.e., before t = 0) using the protocol associated with the RNAqueous kit (Ambion). RNA concentration was determined using a Nanodrop™ spectrophotometer (Thermoscientific, Fisher), and a consistent amount of RNA across all samples was reverse-transcribed using the Super Script III kit (Invitrogen) to obtain cDNA. Real-time polymerase chain reaction (PCR) was performed on the cDNA using a FAM-conjugated primer and probe mixture from Applied Biosystems (Foster City, CA, USA), for β-actin (assay ID: Oc03824857_g1) and SOX9 (assay ID: Oc04096872_m) on a Rotor-gene system (Corbett Research, Qiagen, Valencia, CA, USA), following the protocol provided by Applied Biosystems. Three independent samples from each group were analyzed in triplicate. Data from the PCR assessment were analyzed using the delta-delta Ct (take-off cycle) method (34). Four samples per group were analyzed using real-time PCR.
Statistics
The study was performed three times (triplicates) with cells from three different 10-rabbit harvests. A single-factor ANOVA was used to examine the results obtained from biochemical and biomechanical testing. A two-factor ANOVA was used to analyze the real-time PCR data. Tukey's post hoc test was used, when warranted, to determine significance among groups. Significance for all tests was defined as p < 0.05. In the figures and table, significance between experimental groups or levels of particular factors exists when a common letter or character is not shared.
Results
Expansion
The preliminary study comparing standard and chondrogenically tuned expansion protocols found similar time durations to reach passage 3 (P3; 20 vs. 16 days, respectively), an approximately 64-fold increase in cell number at P3, and significant increases to the collagen II/collagen I ratio and the GAG per wet weight (ww) with chondrogenically tuned expansion (Fig. 1). Cellular morphological changes were noted throughout the expansion procedure. Regardless of the expansion protocol employed, shifts from a rounded cell shape to a more elongated shape with greater numbers of cellular projections were observed.
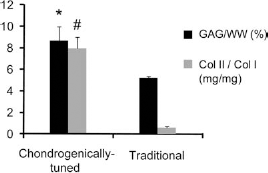
Preliminary expansion media study total glycosaminoglycan (GAG) normalized to wet weight (WW black bars) and collagen II (Col II) normalized to collagen I (gray bars). Data derived from n = 6 samples per group are displayed as mean ± SD. Constructs formed with cells expanded with the chondrogenically tuned protocol had significantly higher GAG/WW and Col II/Col I than constructs formed with cells expanded with the traditional protocol (denoted by ∗ and #, respectively). Differences were considered significant if p < 0.05.
Gross Morphology and Histology
Morphological differences were noted among the six experimental groups within the first week following seeding. The diameter and the growth kinetics of the constructs both varied directly with respect to the ratio of expanded chondrocytes; that is, the higher the percentage of expanded chondrocytes, the faster the construct grew and reached the well edge compared to those with lower percentages of expanded cells. Figure 2 and Table 1 illustrate the differences in the gross morphological properties of constructs at t = 4 weeks of culture.

(a–l) Gross morphological images from the top and side, respectively. White scale bar: 3 mm (in a, and applies to a–l). (m–r) Safranin O/fast green staining. Black scale bar: 0.5 mm (in m, and applies to m–r). Images are labeled by their corresponding primary to passaged cell ratio. There was an increasing trend in construct diameter and thickness when increasing the amount of P3 cells in the construct. Safranin O/fast green straining showed that by incorporating any amount of expanded cells, staining intensity increased.
Biochemical Results After 4 Weeks in Culture

Values shown as mean ± SD.
Significant differences (p < 0.05). PO, primary cells (passage 0); P3, passage 3; GAG, glycosaminoglycan; Col 1, collagen 1.
Histological examination with picrosirius red showed uniform collagen staining throughout all constructs (data not shown) and a small recess at the center of the construct. Safranin-O/fast green staining to identify GAGs revealed similar, intense staining of all groups except the 100:0 group, which stained faintly only at the center of the constructs (Fig. 2).
Quantitative Biochemistry
Quantitative biochemical values for collagen, GAG, and collagen types I and II are shown in Table 1. Collagen content ranged between 0.7 and 1.0 mg per construct. The 100:0 group was shown to contain significantly less collagen than the 2:98 and 25:75 groups. GAG content ranged from 0.3 to 1.6 mg per construct. All groups containing expanded cells had significantly higher GAG contents than the pure primary cell group. Further, GAG content increased 2-fold from pure primary cell to pure expanded cell constructs (black bars, Fig. 3). Collagen types I and II content per construct ranged between 33–57 μg and 195–329 μg, respectively. Notably, collagen type I content for pure primary cells was two times greater than the pure passaged cell group. All groups containing passaged cells possessed a significantly greater amount of collagen type II than the 100:0 group, specifically, a 50% increase was observed when comparing 0:100 to 100:0. A trend of increasing collagen type II to collagen type I ratio was observed with an increased amount of passaged cells per construct (gray bars, Fig. 3). The PicoGreen assay showed significant differences in construct cellularity among groups, with a range of 1.0–1.5 million cells. The 100:0 group possessed a significantly lower number of cells than all other groups.

Total GAG normalized to wet weight (black bars) and collagen II normalized to collagen I (gray bars). Data derived from n = 6 samples per group are displayed as mean ± SD. Significant differences for GAG/WW and Col II/Col I are denoted with capital and lower case letters, respectively. The horizontal axis corresponds to the primary to passaged cell ratio. Significant differences (p < 0.05) exist between groups not sharing the same letter. All groups containing P3 cells had significantly higher GAG/WW than P0 constructs. This trend is mostly replicated in Col II/Col I with the exception of the 50:50 group.
Unconfined Compression
For the 20% strain level, significant differences were observed in all three independent parameters of the viscoelastic model. The relaxation moduli ranged from 193 ± 37 to 443 ± 68 kPa with significantly higher values associated with all groups containing passaged cells (black bars, Fig. 4). Instantaneous moduli varied from 801 ± 55 to 1157 ± 128 kPa with the 50:50 group having a significantly greater value than all other groups (gray bars, Fig. 4). Finally, the coefficient of viscosity ranged from 14 ± 2 to 26 ± 5 MPa with a decreasing trend associated with a decrease in the amount of primary cells. Similar trends were obtained at the 10% and 30% strain levels.
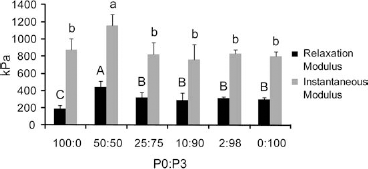
Relaxation (black bars) and instantaneous (gray bars) moduli of P0:P3 constructs at 20% strain. Data derived from n = 6 samples per group are displayed as mean ± SD. Significant differences for relaxation and instantaneous modulus values are denoted with capital and lower case letters, respectively. The horizontal axis corresponds to the primary to passaged cell ratio. Significant differences (p < 0.05) exist between groups not sharing the same letter. For both metrics the 50:50 group significantly outperformed all other groups. The 100:0 group had a significantly lower relaxation modulus than all other groups but there were no other significant differences for the instantaneous modulus.
Real-Time PCR
Prior to self-assembly, primary and P3 chondrogenically tuned chondrocytes showed no significant differences in terms of SOX9 expression. These levels of SOX9 expression did not significantly increase by t = 2 days. By t = 7 days, SOX9 expression had significantly increased in both primary and P3 chondrocytes compared to prior time points and was significantly higher in passaged cells compared to primary cells (Fig. 5). Compared to primary chondrocytes before seeding, primary chondrocytes at t = 7 days had approximately twice the SOX9 expression, while passaged chondrocytes at t = 7 days had approximately 3 times the SOX9 expression. Additionally, the two-factor ANOVA showed that overall P3 chondrocytes had higher SOX9 expression than primary chondrocytes.

Relative SOX9 gene expression. Primary (black bars) and passaged (gray bars) chondrocytes were assessed for the sex-determining region Y box 9 (SOX9) gene expression relative to β-actin and normalized to the relative SOX9 gene expression of primary chondrocytes at day 0 (prior to self-assembly). Gene expression was assessed at day 0, day 2, and day 7 of self-assembly culture. Data derived from four samples per group are presented as mean ± SD. Two-factor ANOVA was performed at each time point, and significant differences between the primary and passaged chondrocytes are designated by different letters.
Discussion
The central hypothesis of this study, that is, that the cartilaginous quality of constructs formed with expanded cells would be enhanced by including primary chondrocytes, was disproven. As previous literature has shown expansion through passage to be detrimental to chondrocytic phenotype (10, 16, 20, 33), it was expected that the cartilage-forming potential of primary chondrocyte would exceed those of P3 chondrocytes. Thus, through coculturing chondrogenic factors produced by primary chondrocytes were expected to stimulate expanded chondrocytes, therefore increasing the production of cartilage matrix in P3 chondrocyte constructs. Instead, it was shown that chondrocytes expanded using a chondrogenically tuned protocol formed constructs with improved cartilaginous properties compared to constructs formed with primary chondrocytes (e.g., twofold increase in GAG/WW, 57% increase in collagen II/collagen I ratio, 34% increase in relaxation modulus, and 60% increase in SOX9 expression).
It has been observed repeatedly that chondrocytes expanded in monolayer exhibit phenotypic changes, resulting in lowered collagen type II and GAG synthesis and increased collagen type I synthesis (10, 16, 33). In light of this extensive body of work, it may seem counterintuitive that the chondrogenically tuned expansion protocol can yield, after 3D self-assembly, cells that produce more collagen type II and express higher levels of SOX9 than primary, nonpassaged chondrocytes. However, the skeletal maturity of the rabbits from which chondrocytes were isolated and the factors associated with chondrogenically tuned expansion must also be considered. Compared to chondrocytes from juveniles, adult chondrocytes not only display lowered metabolic and mitotic activity, increased apoptosis, and decreased responsiveness to growth factors (1, 8, 21, 40), but also decreased ability to produce cartilaginous matrix (2, 8). Even though all chondrocytes used in this study were adult cells, the conditions applied in the chondrogenically tuned expansion protocol [i.e., insulin (14, 35), dexamethasone (29, 49), bFGF (38, 39, 43), and cell–cell interactions (23, 37, 48, 58)] have previously been shown to enhance the ability of expanded chondrocytes to generate a cartilaginous matrix. Thus, when comparing the results of this study to others, with regards to the effects of chondrocyte expansion, one must recognize that the use of cells with lower basal chondrogenic ability, adult cells, allows more potential for the enhancements stemming from chondrogenically tuned expansion conditions.
SOX9 is a transcription factor that has been shown to have a critical role in both chondrogenesis and the maintenance of the chondrocyte phenotype and as such likely influenced the results of this study (9, 11, 50, 55). The SOX9 protein binds to the first intron of both the collagen2a1 and aggrecan genes enabling transcriptional upregulation of these genes (9, 50). Furthermore, researchers have linked upregulation in SOX9 expression to increased chondroitin sulfate synthesis (55). Immediately prior to self assembly, the relative SOX9 expression of primary and passaged cells were not significantly different. This may be due to the low level of SOX9 expression that is expected with adult chondrocytes (24) or the ability of the chondrogenically tuned expansion protocol to maintain SOX9 expression throughout monolayer culture (36, 43, 49). After 7 days of construct culture, a significant increase in SOX9 expression was observed for chondrogenically expanded cells compared to primary cells. This increase in SOX9 correlates well with the GAG/WW and collagen II/collagen I ratio metrics. With SOX9's important role in chondrocyte differentiation (11, 50, 55), it is conceivable that its comparatively higher expression in passaged versus primary cells played a role in the higher quality of constructs formed by passaged cells. This is the first account that shows monolayer cell expansion is capable of maintaining the SOX9 expression of adult chondrocytes throughout monolayer culture.
By forming cocultures of primary and passaged chondrocytes, it was expected that chondrogenic factors produced by primary cells would stimulate the expanded cells and enhance the cartilaginous quality of the matrix. This hypothesis was based on a significant body of preliminary work that has shown primary chondrocytes produce factors that promote the chondrogenic differentiation of stem cells or passaged chondrocytes (15, 20, 51, 56). While the exact factor or factors produced by chondrocytes to enhance the chondrogenic differentiation of themselves and other cells is currently unknown, the literature provides guidance regarding the setup of cocultures to maximize chondrogenic differentiation. Chondrocyte-conditioned media have shown to be inferior to transwell cultures in terms of inducing chondrogenic differentiation (7, 30). Thus, it is assumed that the continuous exchange of factors between the two cell populations is critical. Additionally, physical interaction is known to play a critical role in the chondrogenesis of condensing mesenchyme (6). This is illustrated through the finding that chondrocyte-like cells from intervertebral discs require direct physical contact to exert a chondrogenic effect on mesenchymal stem cells (47). Although the current study employed a culture system allowing continuous chemical and physical signaling, a positive effect of coculturing was not observed. This disagrees with a number of other studies involving similar systems and cell types (15, 20, 56). However, none of these studies have employed adult chondrocytes. This study suggests that in addition to aging being detrimental to the cartilage-forming ability of chondrocytes, it also decreases the ability of primary chondrocytes to aid in chondrogenic differentiation.
When comparing the properties of the tissue constructs obtained in this study to the properties of native articular cartilage, it is clear that passaged, adult chondrocytes are a feasible cell source for tissue engineering via the self-assembly process. Paramount to a tissue replacement's functionality is mechanical properties, and, in the case of cartilaginous tissue, compression functionality is key. At 20% strain, adult rabbit articular cartilage has been shown to have instantaneous and relaxation moduli of approximately 1.39 and 0.74 MPa, respectively. This study produced constructs with instantaneous moduli up to 83% of native values and relaxation moduli of up to 60% of native values (59). Biochemical properties are also important as they may shed light upon how mechanical properties can further be increased. Histological features including cell and matrix distribution resemble native cartilage and appeared similar to constructs formed in other studies using passaged chondrocytes (4, 22, 52, 53). While few of these studies correlated histological features with the mechanical data, the histological and mechanical data in this study were in agreement; all groups except the 100:0 group showed both intense GAG staining and higher relaxation moduli as per the structure–function relationship between GAG and compressive properties (32). Native GAG/WW values (~5%) (6) were exceeded by all groups containing expanded cells. Collagen content was lower than native values (~25%) (6) for all groups.
The results of the biochemical analysis of the cartilage constructs warrant future investigations as to the different signals that may be present to affect the different responses seen in GAG versus collagen production, especially with respect to their temporal response. Previous work examining matrix production in self-assembled constructs using primary cells has shown that GAG production ramps up immediately after self-assembly. Collagen II production does not come into prominence until after 10 days when collagen VI synthesis begins to decline (44). These features mimic those seen in native cartilage development (5, 42). Since this earlier work was done using primary chondrocytes, future studies using passaged cells should be conducted.
Future tissue engineering attempts should focus on increasing collagen content of expanded cell constructs as this appears to be the main drawback of this cell source. Researchers employing the self-assembly process to bovine primary chondrocytes have identified a number of stimuli capable of increasing the mechanical and biochemical properties of constructs including transforming growth factor (TGF)-β1, hydrostatic pressure chondroitinase-ABC, and direct compression (17, 26). Besides initiating mechanotransduction pathways, an additional benefit associated with direct compression stimulation is enhanced nutrient/waste transport. While self-assembled constructs up to 3 mm in thickness can be engineered in static culture without diffusional concerns, dynamic compression may be able to increase nutrient/ waste transport to facilitate engineering thicker or denser neocartilage. Through the application of these chemical and mechanical stimuli, the properties of tissue constructs formed from expanded, adult cells may be enhanced even closer to native values.
Current processes such as osteochondral autograft transfer system (OATS) or mosiacplasty require a one-to-one match of defective to donor tissue, and the ability to engineer mechanically robust cartilage using passaged cells will address the common issue of limited donor tissue. Current use of autografts and allografts consist of implanting mature, hyaline tissue adjacent to the same. This leads to three major obstacles in obtaining cartilage-to-cartilage integration: (1) the densely crosslinked collagen matrix of both the native and implant tissues prevent integration, (2) negatively charged GAG in both native and implant tissues repel each other, and (3) cell death at the wound edge of the surgically prepared defect site and draft tissue (54, 57). Cartilage engineered per the methods described in this study may address these integration issues because (1) in ongoing studies, self-assembled tissues have been found to contain fewer collagen cross-links—this allows for the implant's collagen to crosslink with the native tissue; (2) previous studies have shown that chondroitinase-ABC can be successfully applied to momentarily remove GAG in engineered constructs without long-term detriment to GAG content or construct mechanical properties (26), and conceivably, constructs produced in this study can be similarly treated with chondroitinase-ABC prior to implantation to remove the hindrance of negatively charged GAGs to integration; and (3) the engineered tissue contains higher cell density than the native tissue and can deliver live cells to the wound edge, facilitating integration.
This study shows the current view that cellular expansion can only be detrimental to the chondrocyte phenotype may need to be revised. Chondrocyte-specific expansion techniques may allow for smaller biopsies to be taken for chondrocyte implantation procedures due to the higher chondrogenic potential of the resultant cells. These benefits will also translate to cartilage engineering where primary cell availability can also be limited. Overall, this study shows expanded chondrocytes may be a more useful cell source than previously believed and provides an excellent starting point for the use of adult, leporine cells in the self-assembly modality due to construct biomechanical and biochemical properties approaching those of native cartilage.
Footnotes
Acknowledgments
We gratefully acknowledge NIAMS R01 AR 47839 for funding this work. The authors declare no conflicts of interests.
