Abstract
The aim of this study was to evaluate the potential of culture-expanded human auricular and nasoseptal chondrocytes as cell source for regeneration of stable cartilage and to analyze the differences in gene expression profile of expanded chondrocytes from these specific locations. Auricular chondrocytes in monolayer proliferated less and more slowly (two passages took 26.7 ± 2.1 days and were reached in 4.37 ± 0.30 population doublings) than nasoseptal chondrocytes (19.3 ± 2.5 days; 5.45 ± 0.20 population doublings). However, auricular chondrocytes produced larger pellets with more cartilage-like matrix than nasoseptal chondrocytes (2.2 ± 0.71 vs. 1.7 ± 0.13 mm in diameter after 35 days of culture). Although the matrix formed by auricular and nasoseptal chondrocytes contained collagen X, it did not mineralize in an in vitro model or after in vivo subcutaneous implantation. A DNA microarray study on expanded auricular and nasoseptal chondrocytes from the same donors revealed 1,090 differentially expressed genes. No difference was observed in the expression of known markers of chondrogenic capacity (e.g., collagen II, FGFR3, BMP2, and ALK1). The most striking differences were that the auricular chondrocytes had a higher expression of anabolic growth factors BMP5 and IGF1, while matrix-degrading enzymes MMP13 and ADAMTS5 were higher expressed in nasoseptal chondrocytes. This might offer a possible explanation for the observed higher matrix production by auricular chondrocytes. Moreover, chondrocytes isolated from auricular or nasoseptal cartilage had specific gene expression profiles even after expansion. These differently expressed genes were not restricted to known characterization of donor site subtype (e.g., elastic), but were also related to developmental processes.
Introduction
Developments in regenerative medicine offer promise for the reconstruction of damaged tissues and organs, including cartilage (40). In orthopedics, cartilage regenerative medicine has been used clinically to treat focal joint defects with autologous chondrocyte implantation (ACI) since the first study in patients in 1994 (2). For this procedure, expanded articular chondrocytes are used. In otorhinolaryngology and head and neck surgery, cartilage regenerative medicine may offer a promising alternative to the current use of auricular or costal cartilage to reconstruct cartilage defects. Unfortunately, less research has been performed on cartilage regenerative medicine in this specialty (34,43). However, recent developments such as the first patient receiving a tissue-engineered trachea (23) and the clinical application of cultured autologous auricular chondrocytes to treat craniofacial or nasal augmentation (46) and microtia (45) underline the future possibilities of cartilage regenerative medicine. Naturally, in otorhinolaryngology and head and neck surgery chondrocyte sources other than articular cartilage might be a better choice.
To select a cartilage donor source for cell therapy, one should consider at least three important things: the amount of cartilage that can be harvested without doing harm to the function of the cartilage structure at the donor site, the invasiveness of the biopsy procedure, and the difference in chondrogenic phenotype (e.g., hyaline vs. elastic) of the original tissue. From the first two standpoints nasoseptal and especially auricular cartilage should be considered as favorable chondrocyte sources. Although articular chondrocytes have been extensively characterized and studied for their chondrogenic regeneration potential, overall fewer studies using alternative cell sources have been published (1,14,18,41).
Auricular cartilage is elastic from origin and expanded auricular chondrocytes may therefore produce a different cartilage matrix than expanded articular chondrocytes. Although harvesting nasoseptal cartilage would not be as minimally invasive as auricular cartilage, it does have the potential benefit to originate from hyaline cartilage. However, both donor cell sources differ from articular cartilage in one other major aspect: the presence of a perichondrium.
An important requirement for tissue-engineered cartilage is that it is stable and will not mineralize upon implantation for reconstruction. Therefore, recent studies comparing different cell sources or culture systems did not only evaluate chondrogenic capacity, but took the stability of the tissue-engineered cartilage in consideration; while articular chondrocytes produced stable hyaline cartilage that did not mineralize (25,33), bone marrow-derived mesenchymal stem cells (BMSCs) displayed signs of chondrocyte hypertrophy (i.e., production of collagen X) (11,13,17,27,33) and mineralized in vivo (5,33). In these comparisons auricular and nasoseptal chondrocytes were not taken into account.
Results from studies on expanded articular chondrocytes cannot be translated to expanded auricular or nasoseptal chondrocytes as auricular cartilage is elastic and auricular and nasoseptal cartilage differ from the former by their perichondrium. While the outer fibrous perichondrium can be easily removed from cartilage, this is very difficult for the inner layer (i.e., cambium) of the perichondrium. Undifferentiated progenitor cells in this cambium layer are responsible for new cartilage formation (9,36,39,42), but it is possible that culturing progenitor cells from the cambium layer gives rise to rather unstable cartilage-like constructs, similar to those arising from BMSCs. Therefore, it is essential to evaluate the mineralization potential of auricular and nasoseptal chondrocytes when considering them for reconstructions. To our knowledge this has never been done.
As expanded articular chondrocytes at the moment are clinically used for ACI, recent studies focus on better characterization of the molecular phenotype of the cells (8,35). In fact, some markers are defined that are predictive of the capacity of these expanded chondrocytes to form stable cartilage (8,12). Characterization might lead to more insight into the capacity of the cell sources. Furthermore, these types of characterizations before use can be expected to become more important in regenerative medicine. If culture-expanded auricular or nasoseptal chondrocytes are to be used in the future clinically, they should also be better characterized.
In this study, we demonstrate the potential of human auricular and nasoseptal chondrocytes as cell source for cartilage regenerative medicine. We investigate the general performance of these cells, focusing on subtype and stability of tissue-engineered constructs. Finally, we performed a DNA microarray study on expanded auricular and nasoseptal chondrocytes from the same donors in a first attempt to characterize these cells in more detail to evaluate differences as well as potential similarities in phenotype after expansion.
Materials and Methods
Cell Sources
From four donors (age 18, 26, 28, 47) undergoing rhinoplasty using ear cartilage, both ear and nasoseptal cartilage were obtained (MEC-2005-359). Additionally, auricular cartilage was obtained from in total four donors (age 5, 7, 9, 13) undergoing protruding ear reconstruction (MEC-2006-186). Nasoseptal cartilage was obtained as leftover material from two donors (age 33, 65) undergoing rhinoplasty (MEC-2005-359).
The first aim of this study was to evaluate the potential of culture-expanded human auricular and nasoseptal chondrocytes as cell source for regeneration of stable cartilage. Because it is known from previous studies that articular chondrocytes form stable hyaline cartilage, without collagen X expression and mineralization (25,33), we used these cells as negative controls for hypertrophic differentiation. Healthy, articular cartilage was obtained as leftover material from two donors (age 3, 8) undergoing triple arthrodesis to treat clubfoot deformity (MEC-2007-032). Because BMSCs are known to become hypertrophically differentiated and mineralize in vivo (5,11,13,17,27,33) these cells were used as a positive control. BMSCs were isolated from a femoral shaft biopsy of four donors (age 30, 42, 51, 60) undergoing total hip replacement, after informed consent had been obtained in accordance with the local ethical committee (MEC-2004-142).
Cell Isolation and Expansion
For isolation of chondrocytes, cartilage slices were rinsed with saline after careful resection of the (fibrous) perichondrium, and subsequently digested through incubation for 2 h at 37°C with 2 mg/ml pronase E (Sigma-Aldrich, St. Louis, MO, USA), followed by overnight incubation with 1.5 mg/ml collagenase B (Boehringer Mannheim, Germany) in DMEM containing glutamax (Gibco, Carlsbad, CA, USA) and 10% fetal calf serum (FCS) (Gibco). Next, cell suspensions were filtered through a 100-μm filter, centrifuged, and washed with saline. For expansion, chondrocytes were seeded at a density of 7,500 cells/cm2 in “chondrocyte-expansion medium” [DMEM containing glutamax, with 10% FCS and 1.5 μg/ml fungizone (Gibco) and 50 μg/ml gentamicin (Gibco)]. At subconfluency cells were trypsinized and further expanded. Chondrocytes from passage (P) 2 were used for the experiments. Small pieces of auricular and nasoseptal cartilage were kept aside for immunohistochemical characterization.
For isolation of BMSCs the heparinized bone marrow aspirate was seeded at a density of 2–5 × 105 cells/cm2 in “BMSC expansion medium” [DMEM-LG (Gibco), supplemented with 10% FCS, 1 ng/ml FGF2 (AbD Serotec, Kidlington, UK), 25 μg/ml ascorbic acid-2-phosphate (Sigma-Aldrich), 1.5 μg/ml fungizone, and 50 μg/ml gentamicin]. After 24 h, nonadherent cells were washed off and adherent cells were further expanded. At subconfluency, BMSCs were trypsinized, seeded at a density of 2,300 cells/cm2, and further expanded. BMSCs passages 2–4 were used for the experiment.
Chondrogenic Differentiation
Pellet Culture
To study cartilage subtype differentiation and stability in vitro, auricular and nasoseptal chondrocytes, as well as articular chondrocytes and BMSCs as controls, were chondrogenically differentiated in pellet cultures. Cells were cultured in pellets by centrifuging aliquots of 2 × 105 cells in 0.5 ml medium at 200 × g for 8 min in a polypropylene tube. Chondrogenic differentiation was induced by “chondrogenic differentiation medium” based on previous publications (3,13,19), consisting of DMEM containing glutamax, ITS+1 (B&D Bioscience, Bedford, MA, USA), 40 μg/ml L-proline (Sigma-Aldrich), 1 mM sodium-pyruvate (Gibco), 5 μg/ml fungizone, 50 μg/ml gentamicin, 25 μg/ml ascorbic acid-2-phosphate, 10 ng/ml TGF-β2 (R&D Systems, Minneapolis, MN, USA), and 10–7 M dexamethasone (Sigma-Aldrich). The medium was changed two times a week.
Flat Constructs
To study in vivo stability of tissue-engineered cartilage from auricular and nasoseptal chondrocytes larger and flat constructs were used because these resemble the clinical need better than pellets. Auricular and nasoseptal chondrocytes from one donor (age 18) were used for this experiment. Aliquots of 5 × 105 auricular chondrocytes or nasoseptal chondrocytes (suspended in 100 μl “chondrogenic differentiation medium”) were pipetted onto a dry 6.5-mm diameter, 0.4-μm pore size polycarbonate Transwell® filters (Corning B.V. Life Sciences, Schiphol-Rijk, the Netherlands), precoated with collagen II (Sigma-Aldrich), and centrifuged in a 24-well plate for 5 min at 200 × g (28). Chondrocytes were cultured on “chondrogenic differentiation medium” with medium refreshment three times a week.
General Performance of Auricular Versus Nasoseptal Chondrocytes
To directly compare the performance of auricular and nasoseptal chondrocytes, ear and nasoseptal cartilage from the same donor were used, for a total of three donors (age 26, 28, 47). The performance was evaluated in monolayer expansion by number of population doublings until subconfluency and time to subconfluency. All chondrocytes were expanded and trypsinized when subconfluency was reached. Chondrocytes from passage 2 were chondrogenically differentiated for 35 days in pellet culture. To measure the size of the chondrogenic pellet at day 35, a picture was taken of the intact pellet together with a stage micrometer at 10× magnification.
Statistical analysis was performed using Microsoft Office Excel 2007. Values are represented as mean ± SD. To compare rate of expansion in auricular and nasoseptal chondrocytes from the same three donors, a paired t-test was performed. To compare size of pellets and GAG amount an unpaired t-test was performed. The level of significance was set at p < 0.05.
Cartilage Subtype Differentiation
Characterization of subtype differentiation was studied on pellet cultures at day 35 from auricular and nasoseptal chondrocytes from all donors. This was compared with pellet cultures from articular chondrocytes and BMSCs, which are known to produce hyaline and hypertrophic cartilage, respectively, as well as with sections of native auricular and nasoseptal cartilage. In the flat constructs the cartilage subtype differentiation was evaluated before and after in vivo implantation. Characterization of cartilage subtype differentiation was performed through immunohistochemistry of collagen II, collagen X, and elastin.
To study the effect of passage number on subtype differentiation, chondrocytes from two auricular, nasoseptal, and articular donors were not only differentiated at passage 2 but also at passage 4 and 6.
Stability of Tissue-Engineered Cartilage
To study the ability to mineralize the matrix as a measure of stability, 10 mM β-glycerophosphate (BGP) was added to the medium of pellets after cartilage formation had started. Based on previous studies, BGP was added to the medium of auricular and nasoseptal chondrocytes from day 14 on, and these pellets were harvested at day 35. This experiment was performed with auricular and nasoseptal chondrocytes from three donors (auricular donors aged 9, 13 and 26; nasoseptal donors aged 26, 33 and 65). As a positive control for this in vitro mineralization model, pellets of BMSCs of two donors (age 30, 42) were used. Because of a later onset of cartilage differentiation in BMSCs, BGP was added to medium of these pellets from day 35 on, and pellets were harvested at day 49.
To confirm the stable phenotype of auricular- and nasoseptal-derived tissue-engineered constructs that was seen in the previous in vitro model, a subsequent experiment was performed implanting tissue-engineered constructs in vivo. To directly compare the auricular and nasoseptal chondrocytes, cartilage from one single donor (age 18) was used for this experiment and cells were trypsinized simultaneously. Cells were cultured on a Transwell filter as flat constructs because these resemble the clinical need better than pellets. As in the previous experiment cells were chondrogenically differentiated for 14 days. At this time, six flat constructs were harvested for histology and biochemistry and eight flat constructs were implanted into the back of a nude mouse. Four 6-week-old male NMRI nu/nu mice (Taconic, Germantown, NY, USA) were used. Two incisions were made along the central line of the spine of the mouse, one at the shoulders and one at the hips. Two auricular-chondrocyte constructs and two nasoseptal-chondrocyte constructs were implanted per mouse. Animals were terminated 6 weeks later to harvest the cartilage constructs for histology. This procedure was carried out with approval from the animal ethical committee (EUR1428).
(Immuno)histochemistry
Constructs were fixed in 4% formalin in phosphate-buffered saline (PBS), set in 2% agar, and embedded in paraffin.
Immunohistochemistry for Collagen II, Collagen X, Elastin, and Vimentine
Antigen retrieval for collagen II was performed through incubation with 0.1% pronase (Sigma-Aldrich) in PBS for 30 min, while antigen retrieval for collagen X required 0.1% pepsin (Sigma-Aldrich) in 0.5 M acetic acid (pH 2.0) for 2 h. Both collagen II and collagen X stainings continued with incubation with 1% hyaluronidase (Sigma-Aldrich) in PBS for 30 min. Antigen retrieval for elastin was performed through incubation with 0.25% trypsin (Sigma) in PBS for 20 min.
Nonspecific binding sites were blocked with 10% goat serum in PBS and sections were stained overnight with primary antibodies against collagen II (II/II6B3, Developmental Studies Hybridoma Bank, 1:100), collagen X (Quartett, Berlin, Germany, 1:10), and elastin (BA4, Sigma-Aldrich, 1:1000). To allow the use of monoclonal mouse antibodies on tissue-engineered cartilage constructs implanted in a nude mouse, we used a method described by Hierck et al. (15). An alkaline-phosphatase-conjugated secondary antibody was used, followed by incubation with Neu Fuchsin substrate (Chroma, Köngen, Germany) to demonstrate alkaline-phosphatase activity with a red staining.
To study whether the flat constructs harvested after in vivo implantation were from human origin, a monoclonal mouse anti-human vimentin antibody (AMF-17b, Developmental Studies Hybridoma Bank, 1:40) was used. To exclude any unspecific binding of the vimentin antibody to mouse IgGs, the mouse-on-mouse HRP-Polymer Kit (Biocare Medical, Concord, CA, USA) was used, according to the manufacturer's instructions with some small modifications. In short, slides were incubated in an aqueous 3% solution of hydrogen peroxidase, to inhibit any endogenous peroxidase and allow for a peroxidase-antiperoxidase staining method. Antigen retrieval for vimentin was performed through incubation in Rodent Decloaker® for 60 min at 95°C. Nonspecific binding sites were blocked with Rodent Block M® and sections were stained overnight with vimentin (V6630, Sigma-Aldrich, 1:40). The MM-polymer-HRP® secondary antibody was used, followed by incubation with 3′-diaminobenzidine chromogen solution (Sigma-Aldrich) to demonstrate positivity for the antibody with a brown staining. A mouse monoclonal negative control antibody (Dako) was used as an isotype control. Slides were counterstained with hematoxylin.
Von Kossa/Thionin Staining
For evaluation of mineralization, slides were immersed in 5% silver nitrate solution (Sigma, St. Louis, MO, USA) for 10 min, rinsed in ultrapure water, and exposed to light for 10 min. Excess silver nitrate was removed with 5% sodiumthiosulphate (Sigma) and cells were rinsed in distilled water. Sections were counterstained with 0.4% thionin in 0.01 M aqueous sodium acetate, pH 4.5, for 5 min, which aside from staining the nucleus also demonstrates glycosaminoglycan (GAG) content.
Biochemistry
Flat constructs were digested overnight at 56°C in 1 mg/ml proteinase K (Sigma-Aldrich) in Tris/EDTA buffer (pH 7.6) containing 185 μg/ml iodoacetamide and 1 μg/ml pepstatin A (Sigma-Aldrich). GAG amount was quantified using dimethylmethylene blue (DMB) assay (10). The metachromatic reaction of GAG with DMB was monitored with a spectrophotometer, and the ratio A530:A590 was used to determine the GAG amount, using chondroitin sulfate C (Sigma) as a standard. To determine the amount of DNA, each proteinase K-digested sample (50 μl) was treated with 100 μl heparin (8.3 IU/ml in PBS) and 50 μl ribonuclease A (50 μg/ml in PBS) for 30 min at 37°C. This was followed by adding 50 μl ethidium bromide solution (25 μg/ml in PBS). Samples were analyzed on the Wallac 1420 victor2 (Perkin-Elmer, Wellesley, MA, USA) using an extinction filter of 340 nm and an emission filter of 590 nm. For standards, calf thymus DNA (Sigma-Aldrich) was used.
Statistical analysis was conducted with Microsoft Office Excel 2007. Values are represented as mean ± SD. An unpaired t-test was performed to compare GAG amount. The level of significance was set at a value of p < 0.05.
Microarray Study
Both auricular and nasoseptal cartilage were harvested from the same donor, two donors in total (age 28, 47). Cells were trypsinized at 80% confluency, as judged by the same observer. To avoid a bias in gene expression data by differences in culture procedures, all cells from P2, P4, and P6 were plated in 24-well plate at high cell density (50,000 NC/cm2) in “chondrocyte-expansion medium.” After 3 days the medium was replaced, followed by harvesting of the cells exactly 24 h later. Therefore, from each donor three samples per cell source were available, offering the possibility to statistically adjust for between-donor differences. Cells from P2, P4, and P6 were also chondrogenically differentiated in pellets to determine the chondrogenic capacity after expansion.
RNA Isolation and Expression Profiling
For total RNA isolation, cells were manually homogenized in RNA-BeeTM (TEL-TEST, Friendswood, TX, USA). RNA was extracted with chloroform and purified from the supernatant using the RNAeasy Micro Kit (Qiagen, Hilden, Germany), according to the manufacturer's guidelines with on-column DNA digestion. Gene expression profiling was performed using Affymetrix 1.0 Human Exon ST arrays, representing all known genes (Affymetrix Inc., Santa Clara, CA, USA) according to the manufacturer's instructions. The Affymetrix GeneChip Whole Transcript Sense Target Labeling Assay (kit 900652; 1.0 μg of total RNA) was used to generate amplified and biotinylated sense strands DNA targets from the entire expressed genome. The manufacturer's instruction (version 4, P/N 701880 Rev. 4) was followed for the hybridization, washing, and scanning steps. Arrays were hybridized by rotating them at 60 rpm in the Affymetrix GeneChip hybridization oven at 45°C for 17 h. After hybridization, the arrays were washed in the Affymetrix GeneChip Fluidics station FS 450. Arrays were scanned using the Affymetrix GeneChip scanner 3000 7G system.
Data Extraction and Statistical Analyses
For quality control, the Affymetrix CEL-files were first imported into Affymetrix Expression Console version 1.1 where control probes were extracted and normalized using the default RMA algorithm. The area under the curve (AUC) of the Receiver Operator Characteristic was calculated using positive and negative control probes. All arrays had an AUC score above the empirically defined threshold of 0.85, indicating a good separation of positive and negative controls. Pearson correlation coefficients were determined for each and every combination of arrays. For each of the two tissues a cluster of high correlation was revealed. One of the three auricular samples of one donor did not show higher correlation to the arrays in the cluster of its tissue and was therefore considered an outlier and excluded from further analysis (Fig. 1). Next, the CEL-files were imported into Partek® Genomic Suite software (version 6.4, Partek Inc., St. Louis, MO, USA) where only core probe sets were extracted and normalized using the RMA algorithm with GC background correction. Core transcript summaries were calculated using the mean intensities of the corresponding probe sets, representing the quantitative expression levels of 17,881 genes.

Pearson correlation matrix and sources of variation graph. (A) Pearson correlation coefficients for every combination of arrays. A high correlation (dark gray-black) was revealed for each of the two tissues. Note that one array on auricular chondrocytes (donor 1, P2) did not show higher correlation to the arrays in the cluster of auricular chondrocytes and was therefore considered an outlier. (B) Sources of variation graph demonstrating that the variation in gene expression in the microarray samples was primarily due to cell source with an average F ratio of 5.32, and not to donor variation with an F ratio of 1.76.
The correspondence of the replicate samples was confirmed using principle component analysis (PCA) and Pearson correlation analysis. p-Values and fold differences for the expression differences between auricular and nasoseptal chondrocytes were generated by applying an ANOVA model. p-Values were corrected for multiple testing using the Benjamini-Hochberg method. Genes with values of p < 0.05 and fold differences higher than 2 were functionally annotated and classified by using the functional annotation clustering tool of Database for Annotation, Visualization, and Integrated Discovery (DAVID) (http://david.abcc.ncifcrf.gov/) (16).
Quantitative Real-Ttime RT-PCR
To validate the results from the microarray analysis, the three genes with the highest fold difference between auricular and nasoseptal chondrocytes and the three genes with a fold difference closest to 2 were analyzed by RT-PCR.
Complementary DNA (cDNA) was made using RevertAid™ First Strand cDNA Synthesis Kit (MBI Fermentas, Germany) and polymerase chain reactions were performed using TaqMan® Universal PCR MasterMix (Applied Biosystems, Capelle a/d Ijssel, Netherlands) on an ABI PRISM® 7000 (Applied Biosystems, Foster City, CA, USA). Taqman gene-expression assays were purchased from Applied Biosystems for STEAP4 (Hs00226415_m1), ALX1 (Hs00232518_m1), IL31RA (Hs00371172_m1), GRK5 (Hs00992173_m1), and TMED3 (Hs00201251_m1). Primer sequences for GAPDH and PRG4 are described elsewhere (6).
Expression was normalized to GAPDH and expressed relatively using the 2-ΔCt formula (22). Subsequently, fold differences for each gene were calculated between auricular and nasoseptal chondrocytes.
Results
General Performance
To directly compare the performance of auricular and nasoseptal chondrocytes, ear and nasoseptal cartilage from the same donor were used for this experiment, for a total of three donors (age 26, 28, 47). After isolation, the auricular chondrocytes adhering to the flask varied tremendously in morphology and size while nasoseptal chondrcytes were all round and of similar size. Upon culturing the auricular chondrocytes became more and more uniform in their appearance.
Auricular chondrocytes had gone through 4.37 ± 0.30 population doublings in two passages, while nasoseptal chondrocytes had gone through 5.45 ± 0.20 population doublings (p = 0.026). It took 26.7 ± ± 2.1 days for auricular chondrocytes and 19.3 ± 2.5 days for nasoseptal chondrocytes to perform 2 passages (p = 0.014).
Chondrocytes isolated from auricular cartilage produced significantly larger (p = 0.023) cartilage-like constructs (Fig. 2A–F). The average diameter of an auricular–chondrocyte pellet was 2.2 ± 0.71 mm, the average diameter of a pellet of nasoseptal chondrocytes was 1.7 ± 0.13 mm.

Subtype differentiation and stability of tissue-engineered cartilage in pellet culture. After 35 days of culture on chondrogenic differentiation medium, auricular (A, B) and nasoseptal (D, E) chondrocytes had produced both collagen II and collagen X. Von Kossa/thionin staining demonstrates a proteoglycan-rich matrix at day 35 without mineralization when BGP was added to the medium for the last 3 weeks (C, F). At day 35 articular chondrocytes had produced collagen II (G), but not collagen X (H) and BMSCs had produced collagen II (I) as well as collagen X (J). Von Kossa/thionin staining demonstrates abundant mineralization in BMSC pellets when BGP was added to the medium from day 35 to 49 (K). Pellets of auricular and nasoseptal chondrocytes are from the same donor. Note that pellets of auricular chondrocytes (A–C) were larger and contained more cartilage-like matrix than nasoseptal chondrocytes (D–F).
Cartilage Subtype Differentiation
All pellets of expanded auricular and nasoseptal chondrocytes produced cartilage, as was evaluated with thionin staining showing proteoglycans and immunohistochemistry for collagen type II (Fig. 2A, D). Pellets of both auricular and nasoseptal chondrocytes expressed collagen X (Fig. 2B, E), independent of donor age or passage number, indicating hypertrophic differentiation. In comparison, articular chondrocytes produced abundant collagen type II but no collagen type X (Fig. 2G, H), as was also demonstrated in previous studies. BMSCs, known for their potential to generate terminally differentiated chondrogenic pellets and the ability to mineralize, did indeed express both collagen II and collagen X in our experiment (Fig. 2I, J). Pellets of chondrocytes of all sources produced less cartilage-like matrix with increasing passage number (data not shown).
No elastin was detected in pellets from any of the cell sources, including auricular chondrocytes. Even after prolonged culturing (70 days) or when primary auricular chondrocytes were used, no elastin was observed in the matrix. This suggests that either the in vitro model does not support elastin production of these cells, or that mature elastic chondrocytes are incapable of elastin synthesis.
In comparison, native nasoseptal cartilage stained positively for collagen II, but not for collagen X or elastin. Native auricular cartilage (independent of age), on the other hand, stained positively for elastin and collagen type II, as well as collagen X. Therefore, the tissue-engineered cartilage differed molecularly from the native cartilage the chondrocytes were isolated from, as in vitro nasoseptal chondrocytes produced collagen X and auricular chondrocytes did not produce any elastin.
Stability of Tissue-Engineered Cartilage
Because auricular and nasoseptal chondrocytes formed cartilage positive for collagen X, a marker for hypertrophic cartilage, we studied the ability to mineralize as a measure of stability of the tissue-engineered cartilage formed by auricular and nasoseptal chondrocytes. In this in vitro model chondrocytes from one common donor (age 26) as well as with two distinct donors (auricular donors aged 9 and 13, nasoseptal donors aged 33 and 65) were used. In auricular and nasoseptal chondrocyte pellets a collagen II-rich matrix had formed at day 14 and collagen X was present at this time in the pellets of all except one nasoseptal chondrocyte donor. At the end of the culture period both collagen II and collagen X were observed in all pellets. Addition of β-glycerophosphate was used to evaluate the capacity to mineralize, resulting in a positive Von Kossa staining in BMSC pellets (donor age 30, 42), which served as a positive mineralization control (Fig. 2K). No mineralization was observed in any of the pellets of either auricular or nasoseptal chondrocytes (Fig. 2C, F).
To compare the performance of auricular and nasoseptal chondrocytes in vivo in an application set-up, auricular and nasoseptal cells were harvested from the one donor (age 18) seeded in monolayer for expansion and trypsinized simultaneously. Nasoseptal chondrocytes had gone through 5.4 population doublings and auricular chondrocytes through 2.0 population doublings.
After expansion in monolayer, flat constructs were made by seeding the cells in a Transwell culture system and culturing for 14 days on chondrogenic differentiation medium. Both auricular chondrocytes and nasoseptal chondrocytes had produced a cartilage-like matrix consisting of abundant collagen II and scarce collagen X expression. Similar to what was seen in the pellet cultures, cartilage constructs from auricular chondrocytes had produced more collagen II and were larger than constructs from nasoseptal chondrocytes. Moreover, biochemical analysis demonstrated that the matrix of auricular constructs contained significantly (p < 0.0005) more GAG than matrix of nasoseptal constructs (20.8 ± 5.2 μg GAG/μg DNA vs. 7.8 ± 2.4 μg GAG/μg DNA). Therefore, the difference in size of the cartilage constructs was primarily due to matrix production and not proliferation. In agreement with these findings, flat constructs from auricular chondrocytes were more solid than flat constructs from nasoseptal chondrocytes. However, constructs from both cell sources were still very flexible and fragile after 14 days of in vitro differentiation.
After 6 weeks of implantation in a nude mouse all cartilage constructs, except one from nasoseptal chondrocytes, were recognizable and could be harvested. Constructs from auricular chondrocytes were larger and therefore better recognizable in vivo than those from nasoseptal chondrocytes. Moreover, these constructs were stiff and strong on handling, while constructs from nasoseptal chondrocytes were still fragile and also macroscopically did not resemble cartilage. Immunohistochemistry demonstrated a rich collagen II and collagen X containing matrix in all harvested constructs of both cell sources (Fig. 3A–D). Elastin was demonstrated in the matrix of auricular–chondrocyte constructs, but not in nasoseptal chondrocytes (Fig. 3E, F). Von Kossa/thionin staining demonstrated a proteoglycan-rich matrix in constructs from both cell sources, while no mineralization was present after 6 weeks of subcutaneous implantation (Fig. 3G, H). By means of a vimentin staining, the cartilage-like constructs were demonstrated to be of human origin, while surrounding fibrous tissue was of murine origin (Fig. 3I, J).

In vivo stability of tissue-engineered cartilage in flat constructs. Characterization of tissue-engineered constructs of auricular and nasoseptal chondrocytes after 14 days of in vitro culture, followed by 6 weeks of in vivo subcutaneous implantation. Constructs of both cell sources had produced abundant collagen II (A, B) and little collagen X (C, D). Constructs of auricular chondrocytes, but not nasoseptal chondrocytes, had produced elastin (E, F). Von Kossa/thionin staining demonstrated a proteoglycan-rich matrix in constructs of both cell sources, but no mineralizations were observed (G, H). A positive vimentin staining, sensitive for human vimentine only, demonstrates that the cartilage-like constructs are from human origin, while the surrounding fibrous tissue is from murine origin (I, J). Positivity for the antibody is demonstrated by a red (A–F) or brown (I, J) staining.
Therefore, after in vivo implantation flat constructs derived from auricular chondrocytes did take on their stable native cartilage subtype, including elastin and collagen X production. Flat constructs derived from nasoseptal chondrocytes, on the other hand, did not as collagen X was still present in the matrix. However, although collagen X was present in flat constructs from both cell sources indicating hypertrophy, a stable cartilage was produced that did not mineralize.
Microarray Data
In the two donors (age 28, 47) used for microarray study auricular chondrocytes had gone through 4.7 population doublings at P2 and nasoseptal chondrocytes had gone through 5.4 population doublings. At P6 cells from both origins had gone through 15.5 and 14.2 population doublings, respectively.
Variation in gene expression in the microarray samples was primarily due to cell source with an average F ratio of 5.32, and not to donor variation with an F ratio of 1.76 (Fig. 1). Further analysis demonstrated a group of 1,090 genes whose expression was significantly different in auricular and nasoseptal chondrocytes (out of 17,881 studied genes in total), of which 487 genes showed a fold difference of 2 or more. No significant difference was observed in the expression of collagen II, FGFR3, BMP2, and ALK1, which were previously reported as markers predicting the capacity of expanded articular chondrocytes to produce cartilage (8).
Of these significantly differentially expressed genes, 58 genes were more than fivefold higher expressed in auricular chondrocytes compared to nasoseptal chondrocytes (Table 1). This list is headed by proteoglycan 4 (88×), a matrix molecule with a lubrication function. Surprisingly, two growth factors with a known anabolic effect on cartilage production were observed in this list of genes expressed higher in auricular chondrocytes than in nasoseptal chondrocytes: BMP5 (37×) and IGF1 (9×).
Genes Expressed Over Fivefold More in Auricular Chondrocytes Compared to Nasoseptal Chondrocytes

As expected from the production of elastin in vivo in the previous experiment, eight times more elastin was expressed in auricular chondrocytes. Fibrillin 2, a glycoprotein essential for the formation of elastic fibers, was also expressed significantly higher (7×) in auricular chondrocytes (Table 2). Other components of the elastic fiber, such as fibrillin 1 and 3 or the EMILINs (elastin microfibril interface located protein), were not significantly differentially expressed.
Genes Expressed Over Fivefold More in Nasoseptal Chondrocytes Compared to Auricular Chondrocytes

Forty-nine genes were more than fivefold higher expressed in nasoseptal chondrocytes (Table 2). In contrast, no anabolic growth factors were prominent among the genes that were higher expressed in nasoseptal chondrocytes than in auricular chondrocytes. In this list matrix degrading enzymes MMP13 (39×) and ADAMTS5 (5×) were prominent.
The 487 genes that were significantly differentially expressed in auricular and nasoseptal chondrocytes with a fold difference of ≥ 2 were analyzed with Functional Annotation Clustering in DAVID (Table 3). This analysis indentified “development” as the most important process in which these genes are involved (enrichment score: 13.3). Second in line of importance were processes related to “glycoproteins” (enrichment score: 4.83).
Results of Functional Annotation Clustering
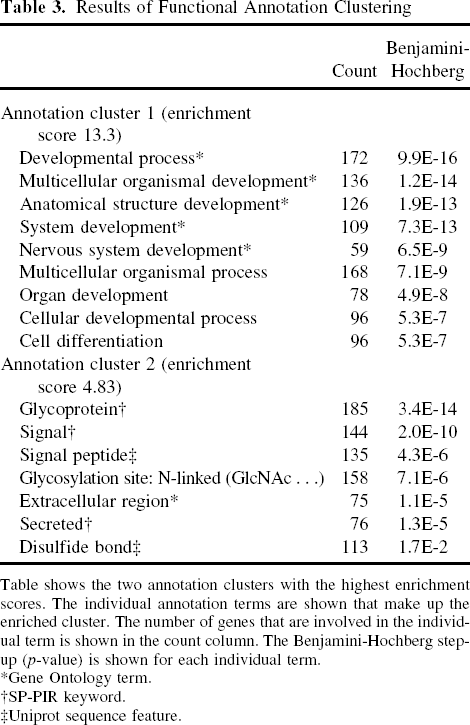
Table shows the two annotation clusters with the highest enrichment scores. The individual annotation terms are shown that make up the enriched cluster. The number of genes that are involved in the individual term is shown in the count column. The Benjamini-Hochberg step-up (p-value) is shown for each individual term.
Gene Ontology term.
SP-PIR keyword.
Uniprot sequence feature.
To validate the microarray results, RT-PCR was performed on the three genes with the highest fold difference (STEAP4, PRG4, ALX1), as well as the three genes with a fold difference closest to 2 (TMED3, IL31RA, GRK5). PCR confirmed the trend and order of magnitude in fold difference as observed in the microarray data, indicating the reliability of the data sets (Table 4).
RT-PCR Validation of Microarray

Fold differences in expression of STEAP4, PRG4, ALX1,TMED3, IL31RA, and GRK5 are shown for both RT-PCR and microarray analysis. + indicates higher expression in auricular chondrocytes, while – indicates higher expression in nasoseptal chondrocytes.
In conclusion, although no difference was observed in mRNA expression of known markers correlated with chondrogenic capacity, auricular chondrocytes do seem to be characterized by a higher expression of anabolic growth factors, while matrix degrading enzymes showed a higher expression in nasoseptal chondrocytes. Moreover, chondrocytes from different specific donor sites seem to preserve specific molecular characteristics even after expansion. These specific molecular signatures are not only indicative of cartilage subtype characteristics (e.g., elastic), but also reflect different developmental processes.
Discussion
This study demonstrates that both auricular and nasoseptal chondrocytes are promising candidate cell sources to generate cartilage for possible future reconstructions. Both cell sources were capable of producing cartilage. Although both produced collagen X, a marker for hypertrophic differentiation, tissue-engineered cartilage constructs (both pellets and flat constructs) of these cells were stable and did not mineralize. This was demonstrated in an in vitro model, as well as in vivo after subcutaneous implantation. A microarray study characterized these chondrocytes more profoundly, and revealed that chondrocytes from ear and nasal septum are different even after expansion and these differences provide possible explanations for the observed higher matrix production by auricular compared to nasoseptal chondrocytes.
This study was the first to examine subtype and stability of tissue-engineered constructs of human auricular and nasoseptal chondrocytes. Human cells were used in our study, while most other publications have studied chondrocytes from different locations in the body in rabbit (14), bovine (18), and porcine samples (4,30,44). Another strong point of this study was the elimination of donor differences, which usually contributes to a lot of noise in the data sets.
In our study, auricular chondrocytes seemed to produce more cartilage than nasoseptal chondrocytes, confirming a previous study (1,14). Although we cannot exclude that this was related to an increased proliferation rate of nasoseptal chondrocytes, a study performed by Tay et al. (38) indicated that the increased proliferation rate of nasoseptal chondrocytes did not influence matrix production. Our microarray study did offer a possible explanation for the observed higher matrix production by auricular chondrocytes. It did not show a different expression of predictive markers of chondrogenic capacity in auricular or nasoseptal chondrocytes (collagen II, BMP2, FGFR3 or ALK1) (8). However, it did demonstrate that two growth factors with a known anabolic effect on cartilage matrix production, BMP5 (24) and IGF1 (20), were significantly higher expressed in auricular chondrocytes. On the other hand, two enzymes involved in cartilage matrix degradation, MMP13 and ADAMTS5, were expressed significantly higher in nasoseptal chondrocytes. This is in line with recent results of Asawa et al. (1), who found a higher expression of MMP1, MMP2, MMP3, MMP13, and cathepsin B in nasal septum chondrocytes compared to auricular chondrocytes in 3D conditions. In this study they also demonstrate that the expression of MMP13 and cathepsin B was already higher in nasal chondrocytes compared to auricular chondrocytes in the monolayer condition. Moreover, in our study the cluster “glycoprotein” was identified by functional annotation clustering as a main target of the genes expressed differently in auricular and nasoseptal chondrocytes.
Cells isolated from auricular and nasoseptal cartilage produce collagen X as well as collagen II, confirming results of Naumann et al. (29). The subtype of the original donor site does not influence terminal differentiation, as expanded auricular (elastic) and nasoseptal (hyaline) chondrocytes both produced collagen X, while this molecule is only present in native auricular cartilage. In addition, collagen X production was not induced by expansion and dedifferentiation as even primary auricular chondrocytes produced abundant collagen X. The observed difference between chondrocytes from articular joints and the head and neck area could be caused by the presence of the perichondrium in both nasoseptal and auricular cartilage. The inner layer of the perichondrium is extremely difficult to separate from the underlying cartilage and is therefore likely contaminating cultures. Undifferentiated progenitor cells in this cambium layer are known for their capacity for cartilage regeneration (9,36,39,42). This may explain why culturing chondrocytes with these progenitor cells from the cambium leads to a cartilage matrix that contains collagen X. Because pellets of both auricular and nasoseptal chondrocytes demonstrated collagen X in the matrix, we studied their tendency to mineralize in an in vitro model. While BMSCs mineralized in this in vitro model, cartilage-like constructs of both auricular and nasoseptal chondrocytes did not and were considered stable. The stability of auricular and nasoseptal chondrocytes was further confirmed by implanting tissue-engineered constructs in vivo.
Although auricular chondrocytes had produced elastin after 6 weeks of in vivo implantation, preceded by 2 weeks of in vitro differentiation, it was not produced after 10 weeks of in vitro culture alone, even when primary chondrocytes were used. Other studies have demonstrated as well that elastin was produced by expanded auricular chondrocytes implanted directly in vivo (18,31,41), while it was not demonstrated in in vitro redifferentiated auricular chondrocytes (29). However, de Chalain et al. (7) demonstrated that elastin production was stimulated in chondrocyte aggregates embedded in hydrogels containing alginate, collagen and κ-elastin. Although this study was performed on primary and not expanded chondrocytes, this indicates that choosing the appropriate in vitro model and creating a microenvironment that sustains elastin production may be important to tissue engineer elastic cartilage, but the prerequisites for elastin formation are still largely unknown. The presence of elastin in auricular constructs implanted in vivo, but not in nasoseptal constructs, indicates that these expanded cells have retained some characteristics of their native donor site. This was supported by our microarray study. In fact, 1,090 genes were expressed on a significant different level in auricular and nasoseptal chondrocytes, of which 487 genes had a fold difference of 2 or more. Fibrillin 2 and elastin were expressed significantly higher in auricular chondrocytes, indicating that a subtype-specific phenotype was still present after expansion. More subtle differences, mostly in the field of developmental processes, were also observed between auricular and nasoseptal chondrocytes. Both auricular and nasoseptal cartilage are formed in the embryo by neural crest-derived cells, thereby differing from articular cartilage (32). But although the chondrocytes they derive from are similar origins, auricular chondrocytes produce elastic and nasoseptal chondrocytes hyaline cartilage. Despite this fundamental difference, only little is known about the distinct development processes that lead to these different cartilage subtypes. From embryonic development, it is known that while hyaline cartilage is present in the nose of murine embryos from day 14 on (32), elastic cartilage in the ear does not mature until late fetal and early neonatal periods (21), suggesting that developmental differences in timing and molecular signaling may attribute to cartilage subtype differentiation. More knowledge about these differential developmental processes may reveal clues for engineering specific subtypes of cartilage. For translation to clinical application the culture methods might be optimized, for example by using serum-free medium to expand the cells (37) or a lower oxygen tension (26). This might affect the gene expression profile as well as the amount and type of cartilage produced.
Our data indicate that using expanded chondrocytes from a donor site that is not directly related to the host tissue may result in cartilage regeneration of a different molecular character. Whether this may pose a clinical problem is not clear, as it is common practice in head and neck surgery to use auricular, elastic cartilage to repair hyaline cartilage defects of the nose. However, it does emphasize the importance to characterize auricular and nasoseptal chondrocytes and study them in more detail, as these cells seem to have great potential for cartilage regenerative medicine as they produce abundant, stable cartilage and can be harvested without doing harm to the donor site, in contrast to articular chondrocytes.
Footnotes
Acknowledgments
The authors would like to thank Suzanne Reneman for doing the in vivo experiments, Femke Verseijden for setting up a protocol for the vimentin staining, Marijn Rutgers for culturing articular chondrocytes, and Petra de Vries for performing the microarray study. This research was financially supported by the Dutch Program for Tissue Engineering.
