Abstract
As a result of less than optimal outcomes the use of islet allografts as a standard insulin replacement therapy is limited to adults with a history of extreme glucose dysregulation and hypoglycemia unawareness. In this study, we examined the use of prophylactic immunotherapy to prevent islet allograft rejection in the absence of antirejection drugs. Our protocol to achieve allograft acceptance used a negative vaccination strategy that is comprised of apoptotic donor cells delivered in Incomplete Freund's Adjuvant (IFA) 1 week prior to islet transplantation. The goal of this new protocol is to elicit hyporesponsiveness to alloantigen prior to islet transplantation. First, we examined our protocol without islet allograft transplants and determined that the negative vaccination was not globally immunosuppressive or immunostimulatory. Islet allograft experiments using fully MHC-mismatched islet donors and recipients demonstrated that the negative vaccination strategy induced long-term islet allograft acceptance. Upon rechallenge with alloantigen, the negative vaccination protocol successfully achieved hyporesponsiveness. In addition, the microenvironment at the site of the tolerant allograft revealed a decrease in proinflammatory mediators (IFN-γ, TNF-α) and an increase in the anti-inflammatory mediator IL-10, as well as increased expression of the master regulator of T-regulatory cells, FOXP3. Our data suggest that pretreating allograft recipients with apoptotic donor alloantigen delivered in IFA induced long-term islet allograft acceptance and glycemic control by introducing alloantigen to the recipient immune system in a nonimmunostimulatory manner prior to transplant.
Introduction
Only five of 36 subjects remained insulin independent 2 years posttransplant as reported in a trial of the Edmonton islet transplantation protocol (28). As a repercussion of the low percentage of islet transplant recipients that achieve long-term glycemic control, islet transplantation is limited to brittle diabetics (11,27). Although immunosuppressive drugs are efficient in inhibiting the T-cell-mediated destruction of the donor islet graft, they are also associated with islet toxicity, infection, and malignancy (1,17,36). These adverse conditions often require the transplant recipient to cease immunosuppressant therapy thereby triggering accelerated allograft rejection (28). The outlook of islet transplantation in its present form is stagnant. For our current study, we designed a strategy that would enable long-term islet graft survival without the use of immunosuppressive drugs. (1,5,6,24).
The goal of our transplant immunotherapy was to mask alloantigen as self-antigen using a negative vaccination as an islet-sparing approach performed in the absence of anti-rejection drugs. Instead of vaccinating to attain immunity, we aim to vaccinate to attain hyporesponsiveness to alloantigen as it is established that apoptotic cells induce tolerance and evade immunity (9,12). The literature reports that educating the recipient immune system in the absence of danger signals with apoptotic donor cells sustains dendritic cells (DCs) in the steady state (2,22,23). On this basis, we developed a negative vaccination protocol consisting of apoptotic donor splenocytes administered 7 days prior to allograft transplantation in Incomplete Freund's Adjuvant (IFA). Our protocol is tailored to slowly dispense apoptotic cells in the immunological steady state to induce prophylactic hyporesponsiveness to alloantigen prior to transplantation.
Subcutaneous administration of the negative vaccination in IFA allowed us to slowly deliver alloantigens to the immune system rather than in a bolus dose. This method of delivery has been reported to increase antigen exposure to and processing by antigen-presenting cells (APCs) (14). By exploiting this route of antigen delivery, the protocol elicited hyporesponsiveness to alloantigen prior to allograft transplantation. Other methods using donor cells to delay allograft rejection have been performed, but these strategies use live, chemically treated or manipulated cells, with most protocols requiring a form of immunosuppressant therapy in conjunction with the donor cells to achieve any delay in allograft rejection (13,18,21,30,32).
Our work demonstrates that the negative vaccination encourages extended presentation of apoptotic alloantigen in a steady state (14,23,30), thereby resulting in immune hyporesponsiveness to donor islets and long-term islet allograft acceptance.
Materials and Methods
Mice
Six- to 8-week-old male C57BL/6 (H2-b), BALB/c (H2-d), and FVB (H2-q) mice were purchased from Jackson Laboratories (Bar Harbor, ME, USA). All animal studies were approved by and trials were conducted in accordance with the University of Pittsburgh Animal Care and Use Committee.
Viability Assays
Spleens from BALB/c mice were harvested, homogenized, and lysed. Half of the splenocytes from each spleen remained untreated, and the remaining cells were exposed to 3,500 rad using an XRAD 160 X-ray machine (Precision X-Ray, Inc., North Branford, CT, USA). Both groups of splenocytes were incubated in Complete Splenocyte Medium (CM) [Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% heat-inactivated fetal bovine serum (FBS), 10 mM HEPES buffer, 4 mM l-glutamine, 200 μM nonessential amino acids, 1 mM sodium pyruvate, 70 μM 2-mercaptoethanol, and 100 μg/ml gentamicin (all from Invitrogen, Life Technologies, Carlsbad, CA, USA)] under normal conditions (37°C at 5% CO2 in a humid air incubator) for 18 h. After incubation, cells were harvested and stained with fluorescein isothiocyanate (FITC)-conjugated annexin V and propidium iodide according to the BD Annexin V-FITC Apoptosis Detection Kit I manufacturer's protocol (BD Biosciences, San Jose, CA, USA). Fluorescence was measured on a FACSCalibur (BD Biosciences).
Mixed Leukocyte Reactions (MLRs)
Unidirectional MLRs were performed by coculture of 106 BALB/c splenocyte stimulators (treated with 3,500 rad) with 106 C57BL/6 or 106 FVB splenocyte responders as described by Wolf et al. (34) with minor modification. All MLRs were performed in 96-well, round-bottomed plates (Greiner Bio One, Monroe, NC, USA) in a final volume of 200 μl of CM and incubated for 5 days under normal conditions.
In Vitro Analysis of the Th1 Response After Treatment with the Therapeutic Strategies
C57BL/6 mice were used as “recipients” and BALB/c mice were used as “donors” (transplants were not performed). To analyze the T helper 1 (Th1) response, mice remained naive and were treated with IFA (Difco/BD, Sparks, MD, USA) in a 1:1 ratio with Hanks balanced salt solution (HBSS; Sigma-Aldrich, St. Louis, MO, USA) or with a negative vaccination of 107 BALB/c splenocytes irradiated at 3,500 rad mixed 1:1 with IFA. Both injections were delivered subcutaneously at the base of the tail in a volume of 100 μl on day 1. Seven days later, on day 8, all subjects were sacrificed, and MLRs were performed as previously described.
Cytokine Measurements by ELISA
MLR assays were performed as described above, and supernatants were harvested on days 1 through 5 to measure interferon (IFN)-γ production by ELISA. IFN-γ production was measured using antibody pairs from BD Pharmingen (San Diego, CA, USA). ELISA plates were read on a Spectromax M2 microplate reader (Molecular Devices, Sunnyvale, CA, USA), and data were analyzed using Softmax Pro v5.0.1 (Molecular Devices).
Islet Allograft Transplants in Diabetic Recipients
Eight- to 12-week-old C57BL/6 males were used as recipients, and 8- to 12-week-old BALB/c mice were used as islet donors. Diabetes was induced with a single injection of 240 mg/kg streptozotocin (Sigma-Aldrich, St. Louis, MO, USA) on day 1. Animals were tested for diabetes using urine test strips (Bayer, Leverkusen, Germany) on day 3. All diabetic animals received 1 unit of insulin (Lantus; Sanofi-Aventis, Paris, France) and 500 μl of Ringers solution [20 mM HEPES (pH 7.4), 0.1 M NaCl, 5 mM KCl, 3.4 mM CaCl2, 1.2 mM KH2PO4, 1.2 mM MgSO4·7H2O, 5 mM NaHCO3, 5 mg/ml BSA, penicillin-streptomycin; all from Invitrogen] on days 3–5. On day 5, islets were isolated from naive donor BALB/c mice as described by Bertera et al. (4). Next, islets were simultaneously picked and counted into dishes with fresh islet media. Each dish of islets contained islet media [10% heat-inactivated FBS, 2% HEPES (1 M), 1% penicillin-streptomycin (10,000 mg/ml), 1% l-glutamine (200 mM), and 0.1% 2-b-mercaptoethanol (50 mM) in Roswell Park Memorial Institute (RPMI) sterile filtered (all Invitrogen)].
Islets were incubated up to 24 h under normal conditions. Nonfasting blood glucose levels were tested in recipient mice on day 6 by obtaining a small blood sample from the retro-orbital sinus and analyzing with Ascensia Breeze 2 Blood Gluose Strips (Bayer, Tarrytown, NY, USA). Only animals with blood glucose of 400 mg/dl and above were used as recipients in this study. Animals in the IFA-alone group were injected with a 1:1 mixture of IFA and HBSS, and negatively vaccinated mice were injected with a 1:1 mixture of 107 apoptotic BALB/c splenocytes to IFA 7 days before the islet transplant (day –1), subcutaneously at the base of the tail. Donor splenocytes were rendered apoptotic through exposure to 3,500 rad and then emulsified using a luer-lock syringe system (Hamilton; Hamilton Company, Reno, NV, USA) prior to subcutaneous injection at the base of the tail.
On day 6, 300 BALB/c islets were transplanted under the kidney capsule of diabetic C57BL/6 recipients as described in Bertera et al. (3). All diabetic recipients became euglycemic posttransplant. Blood glucose was checked every other day. Two consecutive blood glucose readings over 400 mg/dl were indicative of allograft rejection, upon which diabetic animals were sacrificed. Some recipients, however, were sacrificed 14 days posttransplant to use spleens and kidneys for further analysis of immune function.
Immunohistochemical Analysis
Murine kidney samples were fixed in 4% paraformaldehyde (USB Corporation, Cleveland, OH, USA) for 3 h, then transferred to 30% sucrose (Sigma) and stored at 4°C. After being imbedded in frozen section medium (Richard-Allan Scientific, Kalamazoo, MI, USA), cryosections (10 μm) were cut using a cryostat (Microm HM550, Microm International GmbH, Walldorf, Germany) and mounted onto gelatin coated or precleaned slides (Lab Scientific, Inc., Livingston, NJ, USA). Sections were fixed in 2% paraformaldehyde for 5 min on gelatin-coated slides and then blocked with 20% normal goat serum (Jackson ImmunoResearch Lab, Inc., West Grove, PA, USA) for 1 h at room temperature. Primary antibodies included rat anti-cluster of differentiation 4 (CD4) 1:20 and rat anti-CD8 1:20 (BD Biosciences, San Diego, CA, USA), rat anti-IFN-γ 1:50 and anti-tumor necrosis factor (TNF)-α 1:50 (Biolegend, San Diego, CA, USA), and rat anti-forkhead box P3 (FOXP3) 1:50 and anti-interleukin (IL)-10 1:50 (eBioscience, San Diego, CA). Polyclonal rabbit anti-insulin (1:100) (Santa Cruz Biotechnology, Inc. Dallas, TX, USA) was double stained on these sections. Slides were washed five times in PBS (Sigma) and incubated with an Alexa Fluor 488 goat anti-rabbit (Molecular Probes, Eugene, OR, USA), cyanine 3 (Cy3)-conjugated goat anti-rat or goat anti-hamster (Jackson ImmunoResearch Lab, Inc.) secondary antibody. Then, slides were washed three times in PBS, and nuclear staining was performed with Hoechst stain (Molecular Probes). Next, images were captured at 20× magnification using a Nikon confocal microscope (Nikon D-ECLIPSE C1, Tokyo, Japan).
Statistical Analysis
Mean data are expressed as standard error of the mean (SEM). A two-way ANOVA determined the difference between mean values with Bonferroni posttest. Kaplan-Meier survival plots were analyzed by the log-rank test. All statistical analysis was performed with the aid of PRISM (GraphPad, San Diego, CA, USA) or JMP statistical software from the SAS institute (Cary, NC, USA), using p < 0.05 to achieve significance.
Results
Treating Splenocytes with 3,500 Rad Induces Apoptosis
To demonstrate that exposing cells to radiation induces apoptosis, we performed an annexin V and propidium iodide viability assay on untreated splenocytes or splenocytes exposed to 3,500 rad (Fig. 1). After 18 h in culture, the majority of untreated cells were alive (74.86 ± 0.80), while the majority of cells exposed to radiation were late apoptotic (94.94 ± 0.39), staining double positive for annexin V and propidium iodide. These data indicate that radiation induces apoptosis in over 90% of cells, rendering cells prepared in this manner suitable for the prophylactic negative vaccination protocol.

Irradiation induces apoptosis. BALB/c splenocytes were untreated (white bars) or treated with 3,500 rad (black bars). Cells from both groups were plated at 106 cells/well in a 96-well plate. Eighteen hours later, cells were harvested for a viability assay using annexin V and propidium iodide. The cells were distributed based on classical annexin V and propidium iodide staining, that is, live: cells that express neither stain; early apoptotic: positive for annexin V; late apoptotic: double positive for annexin V and propidium iodide; necrotic: positive for propidium iodide. Results are presented as the mean (± SEM) of three independent experiments. ∗p < 0.05 between the respective treated (irradiated) and untreated (control) groups.
Analysis of the Th1 Response After Treatment with Negative Vaccination in the Absence of an Islet Allograft
The Th1 response is the dominant component of the T-cell-mediated alloimmune response that destroys donor islet allografts. For this reason, the effect of our therapeutic strategies on IFN-γ production was initially analyzed using nontransplanted animals. We tested our therapeutic approach by administering the negative vaccination or control injections to naive C57BL/6 mice. After 7 days, animals were sacrificed and MLRs were performed to measure IFN-γ production as an indicator of the Th1 response to alloantigen.
Animal treatment groups included (i) untreated, (ii) a subcutaneous negative vaccination of irradiated BALB/c splenocytes in IFA given 7 days prior to MLR, or (iii) IFA given subcutaneously at the base of the tail 7 days prior to MLR. Compared to untreated controls, mice receiving the negative vaccination demonstrated a marginally significant decrease in IFN-γ production on day 5 of the MLR (Fig. 2). However, when IFA was administered alone, we observed a substantial decrease in IFN-γ production in agreement with the classic skew toward a Th2 response historically observed with IFA treatment (10,35). These data suggest that the negative vaccination strategy is not immunogenic or immunosuppressive.

In vivo treatment with the negative vaccination is not immunogenic or immunosuppressive. C57BL/6 mice were injected with PBS daily for 7 days (n = 4) (white bars), given a single negative vaccination with apoptotic BALB/c splenocytes in Incomplete Freund's Adjuvant (IFA) on day 1 (n = 3) (checkered bars), or given a single injection of IFA on day 1 (n = 3) (gray bars). On the 8th day, all mice were sacrificed, and mixed leukocyte reaction (MLR) assays were performed with a 1:1 ratio of stimulators and responders using irradiated BALB/c splenocytes as stimulators. Supernatants were harvested on days 1–5 and interferon (IFN)-g ELISAs were performed. Results are presented as the mean (± SEM) of at least three independent experiments. Two-way ANOVA with Bonferroni posttest was conducted. ∗p < 0.05 between the respective treated and untreated (control) group.
The Negative Vaccination Strategy Induces Long-Term Hyporesponsiveness
Next, we performed allograft transplants using BALB/c (H-2d) donor islets and diabetic C57BL/6 (H-2b) untreated recipients, or diabetic recipients treated with either IFA alone or the negative vaccination in IFA 7 days prior to islet transplant (Fig. 3). On average, untreated control recipients rejected islet allografts in 22.0 ± 0.73 days (n = 6). Recipients treated with the negative vaccination reached the highest significance (log-rank, p < 0.0001) by maintaining functional islet allografts in excess of 60 days (n = 6), while recipients of IFA alone prior to transplant did not demonstrate any significant increase in allograft survival (29.17 ± 7.22 days, n = 6). After 60 days, functional grafts were removed from negatively vaccinated animals to ensure euglycemia was graft dependent (data not shown). These results demonstrate that a single negative vaccination induces long-term allograft hyporesponsiveness in the absence of nonspecific immunosuppressive agents.

A negative vaccination induces long-term allograft tolerance. Diabetic C57BL/6 mice were untreated (n = 6), given a single negative vaccination of apoptotic BALB/c splenocytes in IFA 7 days prior to transplant (n = 6), or given a single injection of IFA 7 days prior to transplant (n = 6). The log-rank test was used to determine statistical significance in allograft survival between treated and untreated groups.
Recipients of the BALB/c Negative Vaccination Demonstrate Hyporesponsiveness to BALB/c Alloantigen and Not FVB Alloantigen Posttransplant
In order to examine the effects of our therapeutic protocol on the Th1 immune response posttransplant, but before functional rejection ensued for untreated recipients, we used splenocytes from groups of transplant recipients 14 days posttransplant as responders and irradiated BALB/c splenocytes from naive donor mice as stimulators in recall MLRs. Upon secondary exposure to alloantigen in a recall MLR, untreated recipients produced significantly amplified levels of IFN-γ (Fig. 4A) compared to a primary MLR (Fig. 2). Recipients of the negative vaccination were first exposed to alloantigen 7 days before allograft transplant, and the allograft transplant itself provided the secondary exposure, followed by the in vitro MLR as a third exposure to alloantigen. Recipients of the negative vaccination produced IFN-γ levels comparable to those in a primary response in a tertiary response to alloantigen, indicative of hyporesponsive immune education induced by the prophylactic negative vaccination strategy.

The negative vaccination limits the Th1 response to alloantigen. C57BL/6 recipients were untreated (white bars) or given a negative vaccination of apoptotic BALB/c splenocytes in IFA (checkered bars) 7 days prior to transplant. (A) On posttransplant day 14, all mice were sacrificed, and recall MLR assays were performed with a 1:1 ratio of stimulators and responders using irradiated BALB/c splenocytes as stimulators (3 animals/group). (B) On posttransplant day 7, recipients of the BALB/c negative vaccination were sacrificed, and recall MLR assays were performed with a 1:1 ratio responders using irradiated BALB/c or FVB splenocytes as stimulators (3 animals/group). Supernatants were harvested on days 1–4 and IFN-γ ELISAs were performed. Results are presented as the mean (± SEM). ∗p < 0.05 between responders + BALB/c and responders + FVB.
To determine if our negative vaccination protocol is donor-specific, we harvested splenocytes from naive C57BL/6 (H2-b) mice that received a negative vaccination with apoptotic splenocytes 7 days prior and used them as responders in MLRs, using either naive BALB/c (H-2d) or FVB (H2-q) splenocytes as stimulators. As expected from previous experiments (Figs. 2 and 4A), C57BL/6 splenocytes from recipients of the BALB/c negative vaccination demonstrated hyporesponsiveness to BALB/c splenocytes; however, the recipient splenocytes demonstrated hyperresponsiveness to third-party, FVB, splenocytes (Fig. 4B). These findings suggest that our prophylactic immunotherapy induces donor-specific hyporesponsiveness to the alloantigen used in the negative vaccination and not to third-party alloantigen.
Immunohistochemical Analysis of Islet-Bearing Kidneys Posttransplant
To examine whether there was a cellular infiltrate at the site of the islet allografts, we performed immunohistochemical analysis on islet-bearing kidneys 14 days posttransplant. For all sections, insulin was stained green and nuclear (Hoechst) staining was blue. To assess the infiltrate of cells responsible for direct damage to islets, sections were stained with anti-CD8 and CD4 T-cell antibodies. We observed the most pronounced infiltrate of CD8 T-cells in untreated recipients, while the presence of CD8 T-cells at the site of the grafts of negatively vaccinated recipients was undetectable (Fig. 5). We observed, however, a CD4 T-cell infiltrate in untreated and negatively vaccinated recipients (Fig. 5). Comparatively, recipients of the negative vaccination showed a stronger presence of peri-islet CD4 T-cells. Through further immunohistochemical analysis of peri-islet CD4+ populations in the negatively vaccinated recipients, we determined that FOXP3 was elevated compared to untreated recipients (Fig. 5), suggesting that the peri-islet CD4 T-cells are FOXP3+ regulatory T-cells.
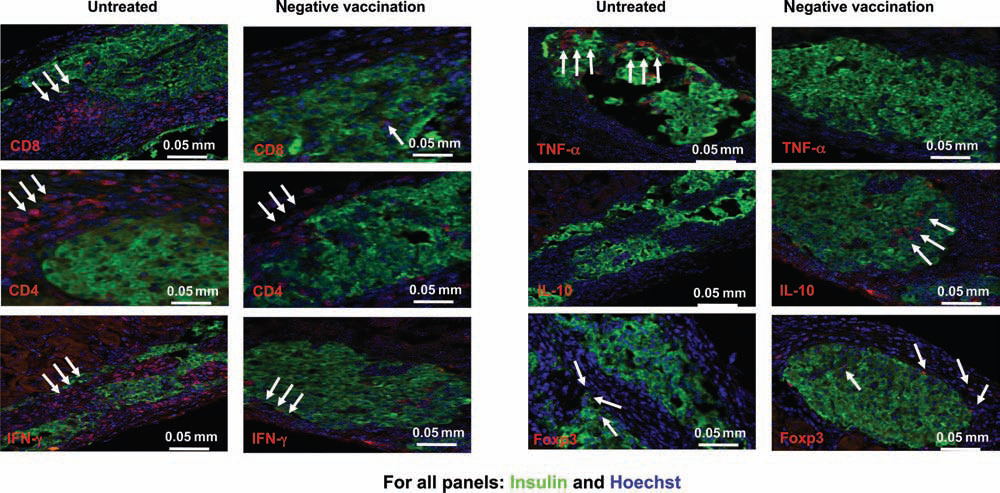
The negative vaccination protocol induces a regulatory environment within the islet allograft. C57BL/6 recipients were untreated or given a negative vaccination of apoptotic BALB/c splenocytes in IFA 7 days prior to transplant. For all slides: green, insulin; blue, Hoechst. Red staining is for the following: CD8, CD4, IFN-γ, forkhead box P3 (FOXP3), interleukin (IL)-10, and tumor necrosis factor (TNF)-α. Sections shown are from one animal from each group and are representative of the three sections examined from each of the respective groups.
To better define the role of FOXP3 at the site of the allograft, as there was a low level of FOXP3 expression in untreated animals as well, sections were stained for the anti-inflammatory cytokine IL-10. IL-10 was not detected in untreated recipients; however, peri-islet and intraislet IL-10 was expressed in allografts of negatively vaccinated recipients (Fig. 5). Additionally, sections from untreated recipients showed increased IFN-γ and TNF-α expression compared to negatively vaccinated recipients (Fig. 5). Taken together, the profile of the islet allograft microenvironment in untreated recipients appeared to have an increase in proinflammatory proteins (IFN-γ and TNF-α) and a decrease in the anti-inflammatory protein IL-10 and the regulatory marker FOXP3, while recipients of the negative vaccination display a profile marked by a decrease in proinflammatory mediators, IFN-γ and TNF-α, and an increase in anti-inflammatory mediators, IL-10 and FOXP3. Additionally, we performed immunohistochemical analysis of the negatively vaccinated group 60 days posttransplant. We observed strong insulin staining and a lack of an immune infiltrate in the islet-bearing kidneys of these long-term graft recipients (data not shown). Taken together, the negative vaccination strategy induced an environment fostering allograft acceptance as indicated by increased IL-10 and FOXP3 expression accompanied by decreased IFN-γ and TNF-α expression at the site of the islet allograft, which correlates to long-term allograft function.
Discussion
Poor outcomes in islet transplantation force the clinic to reserve the procedure for brittle diabetics with severe hypoglycemia unawareness, because transplant recipients often suffer posttransplant complications in excess of those attributed to type 1 diabetes (28). Current literature cites the shortcomings of antirejection regimens and calls for antigen-specific therapies to improve clinical outcomes (28). A donor-specific therapy may allow islet transplantation to generate more consistent outcomes of long-term insulin independence by reducing the use of toxic nonspecific immunosuppressive drugs. The aim of our study was to introduce a modified islet-sparing approach to islet transplantation in an attempt to resolve the need for indefinite use of immunosuppressive agents that result in counterproductive side effects. The safety of our therapeutic protocol was demonstrated in the absence of a transplant (Fig. 2) since subcutaneous delivery of apoptotic donor cells (Fig. 1) in IFA is not severely immunosuppressive, suggesting that this protocol will likely not induce chronic infection or malignancy. Typically, a secondary exposure to an antigen produces a substantially elevated response compared to a primary response. In our case, the MLR assay represents the secondary exposure to alloantigen for the negatively vaccinated animals, as the first exposure is the negative vaccination containing apoptotic allo-mismatched donor cells. IFN-γ levels in the MLR assays from the negatively vaccinated mice (no transplant) were remarkable as they mirrored the levels of IFN-γ achieved in naive MLR (Fig. 2). These results are evidence that our negative vaccination strategy is not immunostimulatory or immunosuppressive, though capable of controlling Th1 immunity to donor alloantigen. The negative vaccination immunotherapy proved to be a successful strategy as all recipients maintained long-term allograft acceptance (>60 days), compared to untreated and IFA-treated controls (Fig. 3). To examine the effects of the in vivo treatments prior to graft failure, we performed recall MLRs and analyzed the Th1 response in a cohort of transplant recipients 14 days posttransplant. Remarkably, for recipients of the negative vaccination, the IFN-γ levels observed in a primary MLR (naive) (Fig. 2), in a secondary MLR (negative vaccination + MLR) (Fig. 2), and in an MLR representing a tertiary response to alloantigen (negative vaccination + islet allograft + MLR) (Fig. 4A) are comparable. Since recipients of the negative vaccination maintained long-term allograft function, perhaps IFN-γ levels held to those observed in a primary response can serve as a predictor of long-term allograft hyporesponsiveness, as low levels of IFN-γ, similar to the levels in a naive MLR (Fig. 2), were detected in islet allografts of negatively vaccinated recipients (Fig. 5). Repeated exposure to alloantigen without a heightened memory response demonstrates that allograft acceptance can be achieved using the prophylactic negative vaccination strategy to induce hyporesponsiveness to alloantigen.
Our therapeutic protocol of negative vaccination is unique in that IFA alone does not confer allograft acceptance as seen by our group and others (Fig. 3) (8,10,14,31,35), thus indicating that alloantigen-expressing apoptotic BALB/c splenocytes are the crucial component of the negative vaccination therapy. The addition of irradiated apoptotic donor cells is critical as it prevents the decrease in IFN-γ production observed when mice are treated with IFA alone (Fig. 2). In vivo transplantation with IFA treatment alone does not result in any statistically significant increase in allograft survival (Fig. 3), confirming the importance of an optimal level of IFN-γ for allograft acceptance (15,19,25). This concept is also supported by the work of Konieczny et al. and Markees et al., which reveal that IFN-γ is required for graft acceptance, signifying that our negative vaccination allows for levels of IFN-γ sufficient for allograft acceptance (16,20), while immunosuppressive treatments tend to flatline IFN-γ levels.
Taken together, the subcutaneous delivery of apoptotic cells in IFA may be able to educate an increased number of T-cells to treat apoptotic alloantigen like self-antigen. Kearney et al. demonstrate that the total number of antigen-specific T-cells in the draining lymph nodes following a subcutaneous injection were two to threefold higher compared to IV or IP routes of delivery (14). Furthermore, unlike other reports that introduce apoptotic or apoptotic-like cells into recipients, depot delivery of alloantigen in IFA imparts hyporesponsiveness to alloantigen in the absence of chemical immunosuppression: it is introduced in a noninflammatory manner in an immunological steady state.
More insight into the mechanisms responsible for the prevention of allograft rejection using a donor-specific negative vaccination was presented with posttransplant immunohistochemical analyses (Fig. 5). A CD4 T-cell infiltrate was present in hyporesponsive negatively vaccinated recipients and in rejecting untreated recipients, although a regulatory role for the CD4 T-cells was only confirmed for recipients of the negative vaccination with the presence of FOXP3 and IL-10 in their islet allografts. In untreated recipients, minimal levels of FOXP3 expression were detectable in the untreated recipients, although the increased production of pro-inflammatory IFN-γ and TNF-α and the absence of anti-inflammatory IL-10 in these recipients confirmed that any anti-inflammatory or regulatory capacity of the FOXP3-expressing cells was overcome by the inflammatory environment and lacked impact on allograft survival (7). The dual presence of FOXP3 and IL-10 in negatively vaccinated allograft recipients is evidence that the CD4 T-cells are protective and likely regulatory in function. Additionally, direct effectors of islet cell death, the CD8 T-cell population, are prevalent in untreated recipients and scarce in recipients of the negative vaccination (Fig. 5), which corroborates with allograft survival (Fig. 3) and a regulatory environment (FOXP3, IL-10) at the site of the allograft as described by others (26,33). Figure 4B suggests that our negative vaccination protocol is donor specific. In future studies, we plan to more thoroughly examine the mechanism of donor-specific hyporesponsiveness resulting in long-term allograft acceptance.
The subcutaneous negative vaccination succeeds in attaining islet allograft acceptance (Fig. 3), although it does not bolster the function of the islet allograft or impair autoimmunity. Our experiments are not inclusive of an autoimmune state that is hallmark of type 1 diabetes and only represent a diabetic recipient of allogeneic islets. Ideally, recipients of islet transplants should receive islets that are functionally more robust in the absence of nonspecific immunosuppression in conjunction with a therapy to avert autoimmune destruction of the transplanted allograft (1,5,6,29). Presently, a clinically relevant application of the prophylactic negative vaccination strategy can be adapted using islets from brain-dead donors or in live donor kidney transplantation. In these transplant scenarios, donor spleen cells can be harvested from donors prior to transplant, irradiated to induce apoptosis, and delivered in alum adjuvant prior to transplantation. The efficacy of this approach can be tested pretransplant using peripheral blood leukocytes (PBLs) from donors and recipients in in vitro MLRs to assess hyporesponsiveness to alloantigen. The negative vaccination approach appears to be one that may limit the use of immunosuppressive drugs, thus reducing unwanted side effects, as the generation of hyporesponsiveness may be enough to ensure long-term graft function in a clinical setting or useful to reduce anti-rejection therapies. Studies that attempt to limit immunosuppression such as prophylactic negative vaccination immunotherapy hold potential to increase the probability of long-term islet allograft function and improve the quality of life for transplant recipients.
Footnotes
Acknowledgment
The authors declare no conflict of interest.
